Introduction
Osteoarthritis (OA) is the most common joint disease. It is the leading cause of pain and disability in the elderly population. It is estimated that at least 300 million people worldwide suffer from OA [1]. OA is a chronic disease characterized by cartilage degeneration, osteophyte formation, and synovial inflammation. The most common joints affected are the knee, hip, and hand. The pain and subsequent physical dysfunction caused by OA are associated with increased mortality risk [2]. In addition, because of the high prevalence of the disease, treatment presents an economic burden to society [3]. To treat the pain and other symptoms, most guidelines recommend the use of non-steroidal anti-inflammatory drugs (NSAIDs) and opioids [1]. However, the use of these drugs is limited by tolerability and safety concerns [4].
In the 1950s Levi-Montalcini et al. [5] discovered nerve growth factor (NGF), which was the first molecule in the class now known as the neurotrophins. Subsequent studies confirmed the important role of NGF in the development of sensory neurons responsible for nociception and temperature sensation. Studies showed that the withdrawal or inhibition of NGF decreases the sensitivity of peripheral nociceptors and down-regulates expression of neuropeptide transmitters [6]. Clinically this can result in significant pain relief. Based on these observations, numerous monoclonal NGF antibodies have been developed as potential alternative analgesics to NSAIDs and opioids in conditions with chronic severe pain. Three monoclonal NGF antibodies have been tested in clinical trials in OA, tanezumab, fulranumab and fasinumab. All trials have shown substantial and significant efficacy [7–15].
Numerous systematic reviews and meta-analyses have been conducted to investigate the efficacy and safety of NSAIDs and/or opioids for treatment of OA pain. The goal of our current network meta-analysis was to include the NGF antibodies in this comparison. Based on a recent network meta-analysis that showed NSAIDs and opioids are efficacious in pain relief in OA, we included 13 drugs in our network meta-analysis. These drugs were divided into 5 groups based on activity and mechanism of action: anti-NGFs (tanezumab, fulranumab, fasinumab), potent opioids (oxycodone, hydromorphone, oxymorphone), weak opioids (tramadol), selective COX-2 inhibitors (celecoxib, etoricoxib, rofecoxib), and traditional NSAIDs (ibuprofen, naproxen, diclofenac, paracetamol/acetaminophen). In a Bayesian network meta-analysis of 41 trials in OA, we assessed drug efficacy, including pain reduction and physical function improvement, and safety.
Results
Study selection
This network meta-analysis was conducted based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [16]. A total of 38 articles covering 41 trials [17–45], were included. The selection criteria are shown in Supplementary Figure 1. Five treatment arms (anti-NGFs, potent opioids, weak opioids, selective COX-2 inhibitors, and NSAIDs) were included in the network of the main analysis, and eight treatment arms (celecoxib, etoricoxib, rofecoxib, ibuprofen, naproxen, diclofenac, paracetamol/acetaminophen and placebo) were included in the network of the subgroup analysis (Figure 1).
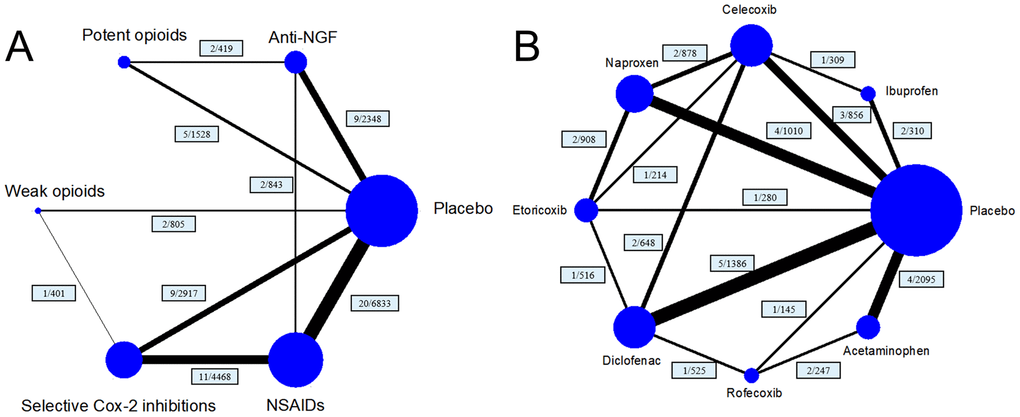
Figure 1. Structure of network formed by interventions. The lines between treatment nodes indicate the direct comparisons made within randomised controlled trials. Numbers (n/n) near the line indicate ‘number of trials/number of participants’ of the related comparisons. (A) the network plot of main network metanalysis. (B) the network plot of subgroup analysis comparing different selective COX-2 inhibitor and traditional NSAIDs.
Study characteristics
A total of 20489 patients were included in the studies. Among the 38 eligible articles, only one study with 385 patients was on hand-joint OA. Across all the trials, the mean age of patients was 61.25 years (range 57.41 to 70.00 years), the percentage of male patients was 32.26% (range 19.57% to 54.03%), and the median follow-up was 84 days (Interquartile range or IQR 42–84 days). The numbers of assessed patients for each treatment were NSAIDs 5408, selective COX-2 inhibitors 4131, anti-NGFs 3108, weak opioids 1405, and potent opioids 1274.
The baseline characteristics of the included studies are shown in Supplementary Table 1. The methodological quality and risk of bias were evaluated for all included trials (Supplementary Table 2). Based on these results, the main contributing factors to the risk of bias were performance bias, selection bias, and attrition bias. A funnel plot was used to assess evaluate effect of small sample size. The funnel plot was presented in Supplementary Figure 3. The detailed results of inconsistency of network comparison were presented in Supplementary Figures 4, 5.
Primary efficacy endpoint
Direct pair-wise meta-analysis
All the drugs except the potent opioids were superior to placebo for pain relief (see Table 1 for pairwise meta-analyses vs placebo). Notably, anti-NGFs showed a significant effect for pain relief (SMD 4.817, 95% Confidence Interval or CI 3.077 to 6.557).
Table 1. Characteristics of the included comparisons and the results of direct pair-wise meta-analysis (No. of patients, number of patients included; No. of trials, number of trials included into direct pair-wise meta-analysis; SMD, standardised mean difference).
| Comparison (compared with placebo) | No. of trials | No. of patients | Target joint | Mean age (Range) | Male,% | Heterogeneity for pain relief, I2 | SMD (95%CI) for pain relief | Heterogeneity for function improvement, I2 | SMD (95%CI) for function improvement |
| Anti-NGF | 9 | 2348 | Hip and Knee | 59.98 (57.41-62.32) | 36.67 | 99.30% | 4.182 (3.778,4.586) | 99.30% | 5.108 (3.165,7.051) |
| Potent opioids | 5 | 1528 | Hip and Knee | 61.1 (57.41-65.52) | 36.62 | 99.60% | 0.807 (-1.527,3.140) | 99.50% | 1.058 (-1.012,3.127) |
| Weak opioids | 2 | 805 | Hip and Knee | 59.09 (58.10-60.02) | 37.23 | 98.3%% | 3.451 (1.722,5.180) | 98.50% | 3.181 (1.466,4.896) |
| Selective cox-2 inhibition | 9 | 2917 | Hip and Knee | 62.81 (60.02-64.77) | 28.63 | 92.20% | 4.775 (2.836,6.714) | 92.20% | 4.528 (2.642,6.415) |
| NSAIDs | 20 | 6833 | Hand, Hip and Knee | 62.55 (58.66-70.00) | 29.90 | 99.60% | 4.775 2.573(1.789,3.357) | 99.50% | 2.677 (1.870,3.484) |
Network meta-analysis
A total of 38 trials were analyzed. No significant inconsistency was found in loop-inconsistency estimates, node-split tests, and global inconsistency tests. The consistency model was statistically significant compared with the inconsistency model.
Anti-NGFs were the most efficacious drugs for pain relief (SMD compared with placebo 4.25, 95% CI 2.87 to 5.63). The potent opioids had the lowest efficacy and no significant effect (SMD 0.90, 95% CI -1.04 to 2.84) (Figure 2 and Table 2). Based on the SUCRA value, anti-NGFs were the most efficacious drugs for pain relief (SUCRA=93.7%), followed by selective COX-2 inhibitors (SUCRA=69.0%), and lastly opioids (SUCRA=67.3%). Anti-NGF were not significantly different than selective COX-2 inhibitor (SMD 1.33, 95% CI [-0.55 to 3.21]) and weak opioid drug (SMD 1.22, 95% CI [-2.07 to 4.51]), while it demonstrated better pain relief than NSAIDs (SMD 2.33, 95% CI [0.69 to 3.96]) and potent opioid drug (SMD 3.35, 95% CI [1.16 to 5.53]). The details of the SURCA rank are shown in Supplementary Table 3.
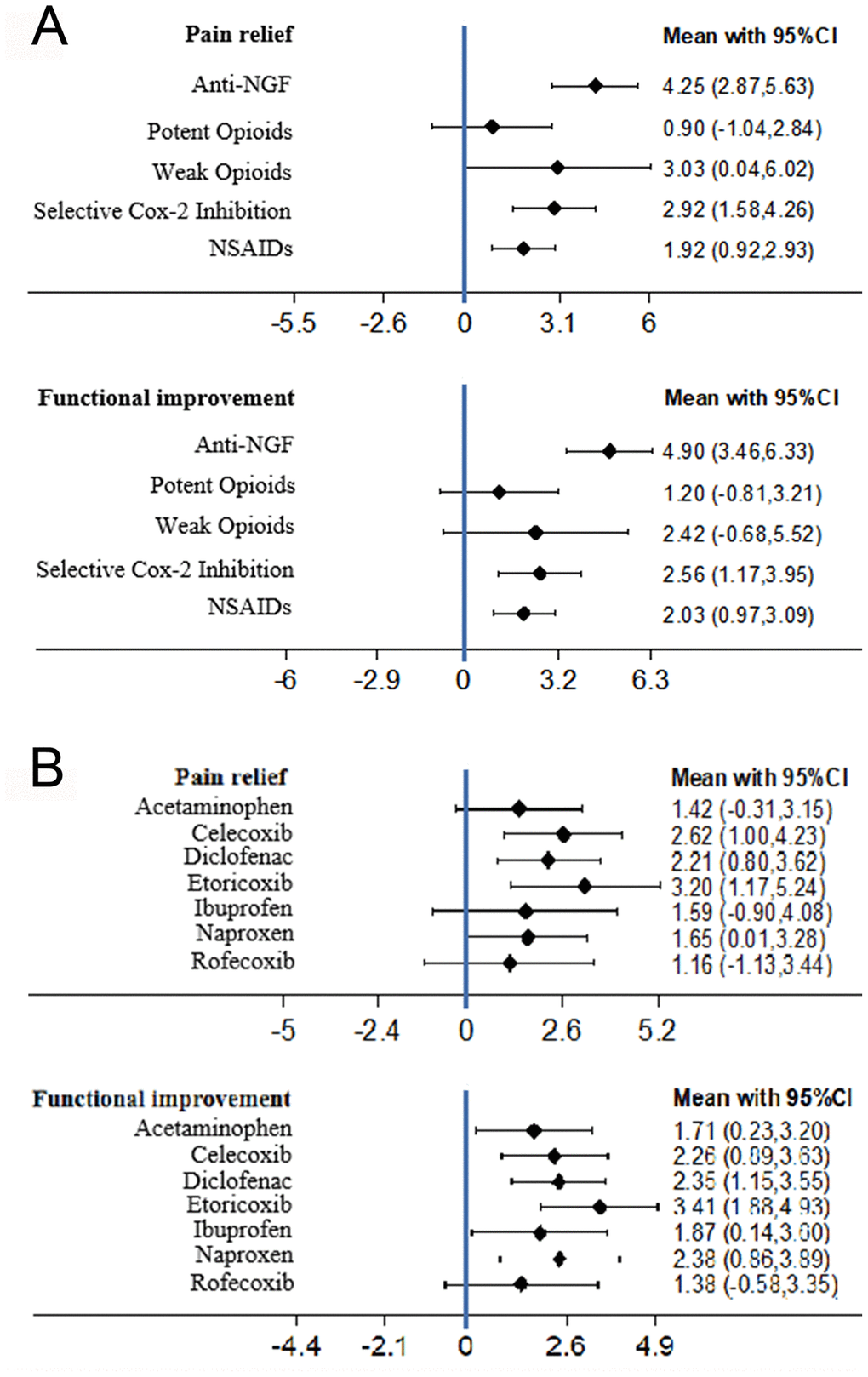
Figure 2. (A) The forest plots of pain relief and function improvement for main network meta-analysis. (B) The forest plots of pain relief and function improvement for subgroup analysis comparing different selective COX-2 inhibitor and traditional NSAIDs (SMD, standardised mean difference).
Table 2. Detailed results of network meta-analysis for pain (Red) and function (Blue) (Data are standardised mean difference, from the top left to the bottom right, higher comparator vs lower comparator, and their related 95% CI).
| Anti-NGF | 2.34 (0.38,4.29) | 2.48 (-0.93,5.89) | 2.87 (1.16,4.57) | 3.70 (1.43,5.96) | 4.90 (3.46,6.33) |
| 1.33 (-0.55,3.21) | Selective cox-2 inhibition | 0.15 (-3.09,3.38) | 0.53 (-0.77,1.84) | 1.36 (-1.07,3.80) | 2.56 (1.17,3.95) |
| 1.22 (-2.07,4.51) | -0.11 (-3.23,3.00) | Weak opioids | 0.39 (-2.83,3.60) | 1.22 (-2.47,4.91) | 2.42 (-0.68,5.52) |
| 2.33 (0.69,3.96) | 1.00 (-0.24,2.23) | 1.11 (-1.99,4.20) | NSAIDS | 0.83 (-1.42,3.09) | 2.03 (0.97,3.09) |
| 3.35 (1.16,5.53) | 2.02 (-0.33,4.36) | 2.13 (-1.44,5.69) | 1.02 (-1.15,3.19) | Potent opioids | 1.20 (-0.81,3.21) |
| 4.25 (2.87,5.63) | 2.92 (1.58,4.26) | 3.03 (0.04,6.02) | 1.92 (0.92,2.93) | 0.90 (-1.04,2.84) | Placebo |
Subgroup analysis did not reveal any substantial change after excluding the 10 trials that were not commercially funded. Anti-NGFs were still the most efficacious drugs for pain relief (SUCRA 92.4%, SMD=4.30, 95% CI 2.85 to 5.74) (Supplementary Table 5).
Secondary efficacy endpoint
Direct pair-wise meta-analysis
The anti-NGFs and selective COX-2 inhibitors significantly improved physical function compared to placebo while the potent opioids had no significant effect (see Table 1 for pairwise meta-analyses vs placebo). Anti-NGFs had the highest efficacy for functional improvement (SMD 5.108, 95% CI 3.165 to 7.051).
Network meta-analysis
A total of 38 trials were analyzed. No significant inconsistency was reported, and the consistency model was statistically significant compared to the inconsistency model. Anti-NGFs had the highest efficacy for functional improvement (SMD 4.90, 95% CI 3.46 to 6.33) (Figure 2). The potent opioids had the lowest efficacy and no significant effect (SMD 1.20, 95% CI -0.81 to 3.21) (Figure 2 and Table 2). Based on SUCRA, the anti-NGFs were the most efficacious drugs for functional improvement (SUCRA=98.3%), followed by selective COX-2 inhibitors (SUCRA=63.5%) and opioids (SUCRA=56.7%) (Supplementary Table 3). In the subgroup analysis there was no substantial change after excluding the 10 trials that were not commercially funded. Anti-NGFs still had the highest efficacy for functional improvement (SUCRA 97.6%, SMD 4.96, 95% CI 3.42 to 6.50). Apart from the selective COX-2 inhibitor group (SMD 2.34, 95%CI [-0.38 to 4.29]) and weak opioid group (SMD 2.48, 95%CI [-0.93 to, 5.89]) which were not significantly different than Anti-NGF drug, other groups all demonstrated less function improvement (Supplementary Table 3). There was no significant difference in the subgroup analysis of the trials with commercial funding. Anti-NGF still had the highest efficacy (SUCRA 97.6%, SMD 4.96, 95% CI [3.42 to 6.50]) (Supplementary Table 5).
Primary safety endpoint
Direct pair-wise meta-analysis
There were significant increases in withdrawal rates related to AEs with anti-NGFs and opioids compared to placebo, but not with selective COX-2 inhibitors (Odds Ratio or OR 0.742, 95% CI: 0.436 to 1.261) (Table 3).
Table 3. Adverse effects of different treatment compared with placebo according to direct pair-wise meta analysis and network meta-analysis (AE, adverse effect; PSA, peripheral sensation abnormality; SUCRA, surface under the cumulative ranking.).
| Treatment | Heterogeneity for direct comparison, I2(%) | OR(95%CI) | SURCA(%) | ||
| Direct comparison | Network comparison | ||||
| Withdrawal due to AEs | |||||
| Placebo | Reference | Reference | Reference | 82.3 | |
| Anti-NGF | 33.1 | 1.677(1.045,2.692) | 1.36 (0.82,2.27) | 53.5 | |
| Potent Opioids | 36.8 | 5.265(3.705,7.482) | 8.63 (5.42,13.77) | 0.1 | |
| Weak Opioids | 68.4 | 2.798(1.348,5.807) | 3.27(1.89,5.66) | 20.1 | |
| Selective cox-2 inhibition | 56.9 | 0.742(0.436,1.261) | 0.89 (0.64,1.24) | 93.5 | |
| NSAIDs | 29.6 | 1.272(1.028,1.573) | 1.36(1.04,1.77) | 50.5 | |
| Headache AEs | |||||
| Placebo | Reference | Reference | Reference | 53.3 | |
| Anti-NGF | 0.0 | 0.970(0.634,1.483) | 1.03 (0.68,1.57) | 49.3 | |
| Potent Opioids | 0.0 | 1.283(0.897,1.837) | 1.19 (0.78,1.82) | 29.2 | |
| Weak Opioids | 49.8 | 1.305(0.709,2.399) | 1.41 (0.88,2.25) | 12.4 | |
| Selective cox-2 inhibition | 0.0 | 0.748(0.528,1.060) | 0.93 (0.68,1.27) | 65.4 | |
| NSAIDs | 1.3 | 0.928(0.733,1.175) | 0.82 (0.63,1.06) | 90.3 | |
| Nausea AEs | |||||
| Placebo | Reference | Reference | Reference | 74.2 | |
| Anti-NGF | 33.1 | 0.962(0.504,1.837) | 0.79 (0.41,1.53) | 88.7 | |
| Potent Opioids | 36.8 | 4.519(3.212,6.358) | 6.33 (3.37,11.90) | 3.3 | |
| Weak Opioids | 68.4 | 3.131(2.054,4.775) | 3.90 (1.82,8.36) | 17.0 | |
| Selective cox-2 inhibition | 56.9 | 0.825(0.398,1.708) | 0.99 (0.64,1.52) | 75.8 | |
| NSAIDs | 29.6 | 1.432(0.947,2.165) | 1.48 (1.04,2.13) | 41.1 | |
| PSA AEs | |||||
| Placebo | 0.0 | Reference | Reference | 82.7 | |
| Anti-NGF | 0.0 | 4.184(2.010,8.707) | 3.64 (1.87,7.10) | 30.7 | |
| Potent Opioids | 0.0 | 5.331(2.731,10.407) | 5.39 (2.41,12.06) | 14.2 | |
| Weak Opioids | 0.0 | 8.371(2.935,23.870) | 5.25 (1.95,14.15) | 15.4 | |
| Selective cox-2 inhibition | 0.0 | 0.777(0.021,28.890) | 0.96 (0.39,2.39) | 82.6 | |
| NSAIDs | 82.8 | 0.966(0.633,1.473) | 1.13 (0.67,1.90) | 74.4 | |
Network meta-analysis
In the withdrawal related to AEs network, a total of 36 trials were assessed. No significant inconsistency was reported. Significantly higher withdrawal rates related to AEs were reported with potent opioids (OR 8.63, 95% CI 5.42 to 13.77), weak opioids (OR 3.27, 95% CI 1.89 to 5.66) and NSAIDs (OR 1.36, 95% CI 1.04 to 1.77) compared to placebo. Selective COX-2 inhibitors (SUCRA = 93.4%) were the safest, followed by anti-NGFs (SUCRA=53.5%) and NSAIDs (SUCRA=50.5%) (Table 3 and Supplementary Table 3). In the subgroup analysis there was no substantial change after excluding the trials that were not commercially funded (Supplementary Table 5). The cluster-rank plots of the primary efficacy and primary safety endpoints showed the selective COX-2 inhibitors to be overall the safest drugs (Supplementary Figure 2).
Secondary safety endpoint
A total of 33 trials were selected for assessment. Based on incidence rates, the three most common AEs selected as the secondary safety endpoints were nausea, headache, and peripheral sensation abnormality (paresthesia and pruritus).
Direct pair-wise meta-analysis
There were no significant differences in the incidence rates of headache among the three drug classes from the pair-wise meta-analyses. However, potent opioids and weak opioids had significantly higher risks for nausea and peripheral sensation abnormality. In addition, anti-NGFs had significantly higher risk for peripheral sensation abnormality (Table 3).
Network meta-analysis
In the headache network, NSAIDs were the safest drugs (OR 0.82, SUCRA 90.3%, 95% CI 0.63 to 1.06). However, there were also no significant differences between the other treatments and placebo. NSAIDs (OR 1.48, 95 % CI 1.04 to 2.13), weak opioids (OR 3.90, 95 % CI 1.82 to 8.36) and potent opioids (OR 6.33, 95 % CI 3.37 to 11.90) had significantly higher risks for nausea. The incidence rate of peripheral sensation abnormality was significantly higher with anti-NGFs (OR 3.64, 95% CI 1.87 to 7.10), weak opioids (OR 5.25, 95% CI 1.95 to 14.15) and potent opioids (OR 5.39, 95% CI 2.41 to 12.06) (Table 3). No subgroup analysis was conducted for secondary safety endpoints because there was an insufficient number of trials.
Subgroup analysis comparing efficacy of NSAIDs and selective COX-2 inhibitors
A total of 24 trials assessing the efficacy of NSAIDs or selective COX-2 inhibitors were included. All drugs had significantly greater efficacy compared to placebo for both pain relief and function improvement (Figure 2, Supplementary Tables 4, 6) but there were no significant differences between drugs. Etoricoxib produced the highest values for both pain relief (SMD 3.20, 95% CI 1.17 to 5.24) and functional improvement (SMD 3.41, 95% CI 1.88 to 4.93), and based on the cluster-rank plot it was the most efficacious drug (Supplementary Figure 2) However, no significant difference was reported between drugs.
Discussion
There have been several systematic reviews and meta-analyses comparing the efficacy and safety of anti-NGF drugs with placebo in OA. Schnitzer TJ et al. [46] showed that in knee and hip OA, anti-NGF treatment can provide excellent and superior pain relief and improvement in joint function compared to placebo, and is generally well tolerated with acceptable AEs. Similarly, Chen J et al. [47] confirmed that anti-NGF treatment is superior to placebo in alleviating pain and improving function in knee OA. This current study is the first systematic review and network meta-analysis comparing the efficacy and safety of monoclonal NGF antibodies (anti-NGF drugs) with other drugs commonly used to treat pain associated with OA, including NSAIDs, opioids, and selective COX-2 inhibitors. The Bayesian method used in this study increases the number of studies within each comparison, which in turn increases the robustness and power of the results. Our results showed that in the treatment of OA, 1. monoclonal NGF antibody drugs have the highest overall efficacy, but are not significantly different from selective COX-2 inhibitors, NSAIDs, and opioids; 2. monoclonal NGF antibodies have the highest efficacy for both pain relief and function improvement, while selective COX-2 inhibitors are the safest (the lowest risk of withdrawal related to AEs); 3. among the COX-2 selective inhibitors and NSAIDs, etoricoxib is the most effective for both pain relief and functional improvement based on cluster-rank and SUCRA, but was not significantly different from other selective COX-2 inhibitors and NSAIDs; 4. potent opioids have the lowest efficacy and worst safety (highest risk of withdrawal related to AEs).
A previous network meta-analysis [48] reported that NSAIDs, weak opioids, and potent opioids have similar efficacy for pain relief in OA. However, the study only compared the drugs indirectly via effects versus placebo, and no direct comparisons were made between the drugs, including between the potent opioids and the other drugs. In the current analysis, we included more eligible studies and compared potent opioids directly and indirectly with other drugs. Additionally, we analyzed for functional improvement and safety as well as pain relief, which are all important outcomes in OA treatment. Based on our analyses, potent opioids have low safety and efficacy, and better options are available for treatment of OA.
The new finding from this study is that monoclonal NGF antibodies have the highest efficacy for pain relief and functional improvement, exceeding that of selective COX-2 inhibitors, NSAIDs, and weak opioids (tramadol), all of which are recommended in the 2019th American College of Rheumatology/Arthritis Foundation Guideline. However, monoclonal NGF antibodies have a higher risk of peripheral sensation abnormality (including paresthesia and pruritus), although overall withdrawal rate related to AEs is not significantly different compared to placebo. Our findings are consistent with the results of previous systematic reviews [47, 48] and support monoclonal NGF antibodies as the most effective treatment option for OA. The monoclonal NGF antibodies should be considered the first choice for patients with pain and/or disability related to OA. For patients who experience paresthesia and pruritus, selective COX-2 inhibitors and NSAIDs are preferred treatment options. The choice of a specific COX-2 inhibitor or NSAID can be made based on the mean rank order presented in Supplementary Table 5.
This study has several limitations. Firstly, only randomized, controlled trials (RCTs) were included to avoid uncontrolled confounding factors common in observational studies and other non-RCTs. However, observational studies and other non-RCTs can provide important information on drug efficacy and safety, and omitting these resulted in a small number of studies being included. Secondly, only high-quality studies were included in order to control the quality of the analysis and to minimize the impact of small-sized study effects. However, including small-sized studies can increase generalizability and robustness of the results. Publication bias may be a significant problem, but is difficult to control with a small number of studies. Thirdly, the length of follow-up was not long enough to assess long-term safety outcomes. The median follow-up of studies in this network-analysis of 84 days (IQR 42–84 days) is sufficient to assess AEs that develop relatively quickly but not for long-term safety outcomes such as cardiovascular changes, sensation abnormalities and joint damage. More high-quality RCTs, with long-term follow-up, are needed.
Conclusion
A total of 38 studies comprising 20489 patients were included in this network meta-analysis. The results show that monoclonal NGF antibodies provide significantly greater pain relief and functional improvement in OA compared to selective COX-2 inhibitors, NSAIDs, and opioids. Monoclonal NGF antibodies are not associated with severe short-term AEs. More large scale RCTs are needed to confirm these findings.
Materials and Methods
Literature search
We searched the PubMed, CNKI, Web of Science, Scopus, Embase and Cochrane Library databases, from the start of January 1999 to end of January 2020. Our search terms were 'osteoarthritis', 'tanezumab', 'fulranumab', 'fasinumab', 'oxycodone', 'hydromorphone', 'oxymorphone', 'tramadol', 'celecoxib', 'etoricoxib', 'rofecoxib', 'ibuprofen', 'naproxen', 'diclofenac', and 'paracetamol/acetaminophen'. We also screened the reference lists of relevant systematic reviews and meta-analyses to identify additional eligible articles. All eligible articles were included irrespective of the language of publication.
Inclusion/exclusion criteria
The inclusion criteria for this network meta-analysis were: 1. RCTs; 2. studies comparing the target drugs with placebo, or with each other; 3. studies on patients with OA at any joint; 4. studies reporting the following endpoints: pain reduction, functional improvement, and withdrawal related to AEs. The exclusion criteria were: 1. dose-escalation studies of only one drug; 2. target drugs combined with other drugs; 3. studies for postoperative pain; 4. reviews, systematic reviews and meta-analyses, conference abstracts, letters, pharmacokinetic or pharmacodynamic studies, and studies with insufficient data.
For studies with insufficient data, the corresponding authors were contacted to see if the required information could be obtained. If no response was received, a reminder was sent, and if there was still no response the study was excluded. For studies that reported data visually but did not provide numeric values in text or tables, again the corresponding authors were contacted. If no response was received, two authors of the current analysis independently attempted to obtain the data by measurements from the graphs/figures. If that was not possible based on a clear scale and specific reference system then the study was excluded.
Quality assessment
The Cochrane risk of bias assessment tool was used to perform the methodological quality assessment of the RCTs [49]. The following indices were evaluated and ranked as low risk of bias, unclear risk of bias, or high risk of bias: sequence generation, allocation concealment, blinding, incomplete outcome data, selection outcome reporting, and other sources of bias. These assessments were performed by the authors independently, and all disputes were resolved by discussion among the authors.
Data extraction
Author, publication year, total sample size, mean age, gender ratio, affected joint, treatments, route of administration, intervention time, follow-up period, and endpoint data were collected and tabulated. To reduce the effect of withdrawal bias, we collected data from the intention-to-treat analysis.
Outcome measures
The primary efficacy endpoint was pain reduction, and the secondary efficacy endpoint was functional improvement. There were restrictions on the types of questionnaire used in pain evaluation. Functional improvement was evaluated using the function subscale of the Western Ontario and McMaster Universities Arthritis Index (WOMAC). If the WOMAC function score was not measured or reported, the Lequesne Index or other functional measurement scale was used. The change-from-baseline score (mean ± SD) at the last follow-up period was used to evaluate the extent of both pain relief and functional improvement. For studies involving multiple treatment groups with different doses of the same drug, we selected the most effective dose group based on the respective study’s recommendations [50]. We calculated the SMD since results from different scales were used in the same network.
Since patient compliance impacts the effect of treatment in clinical practice, we selected the withdrawal rates related to AEs as the primary safety endpoint. The most commonly related AEs were secondary safety endpoints. We calculated the OR with 95% CI for the safety of target drugs versus placebo or versus each other.
Statistical analysis
The efficacies of the target drugs were compared to placebo by direct pairwise meta-analyses using Review Manager Software (RevMan. Version 5.3, Copenhagen, the Nordic Cochrane Centre, the Cochrane Collaboration, 2014). Heterogeneity across studies were assessed from the Cochran Q statistic and the I2 statistic. When there was significant heterogeneity (P<0.05 or I2 >50%), we pooled data and used a random-effects model for analysis. When there was no significant heterogeneity we pooled data and used a fixed-effects model for analysis. The Bayesian network meta-analyses were conducted using Stata/MP (version 14.0, Stata Corp, College Station, Texas, USA) and GeMTC (version 0.14.3). This method increases the number of studies within each comparison and narrows the CI width, resulting in stable results [51–54]. In the Bayesian network meta-analysis, non-informative uniform and normal prior distributions were used. Subsequently, four different sets of starting values were set to fit the model to yield 40000 iterations (10000 per chain) and to obtain the posterior distributions of model parameters [55, 56]. The thinning interval was set at 10 and the burn-ins at 1000, for each chain. Convergence of iterations was assessed using the Gelman-Rubin-Brooks statistic [57]. Global inconsistency tests and local inconsistency tests (Loop-inconsistency tests and Node-split tests) were used to reconfirm the consistency of the network meta-analysis. Two subgroup analyses were conducted. The first was to determine the impact on the results of the network analyses based on whether the trials were commercially funded or not. The second was to determine the most effective drug (vs placebo) for combined pain relief and physical functional improvement (from the selective COX-2 inhibitors celecoxib, etoricoxib, rofecoxib, and the traditional NSAIDs ibuprofen, naproxen, diclofenac, paracetamol/acetaminophen).
All estimate outcomes (SMDs or ORs) with 95% CI were generated from the posterior distribution medians. Differences were considered significant if the 95% CI did not include 0 for SMD or 1 for OR. Differences were considered statistically significant at P<0.05. The minimum clinically important difference of -0.37 SD units was selected. This threshold of 0.37 SD units is based on the median minimum clinically important difference reported in studies in patients with osteoarthritis [58]. An effect size of 0.37 corresponds to a difference of 9 mm on a 100 mm visual analogue scale. The SUCRA was used to rank the efficacy and safety of the different drugs. An intervention with a SUCRA value of 100 is certain to be the best, whereas an intervention with 0 is certain to be the worst [59]. To select the most effective drug based on two or more endpoints, cluster-ranking plots were constructed.
Author Contributions
Jian Zhou conceived the study, participated in its design and coordination, and critically revised the manuscript. Ziqin Cao, Zeling Long and Yihan Li had full access to all of the data collection, analysis and interpretation. Ziqin Cao drafted the manuscript. Jian Zhou, Jingjing Sun, Yingquan Luo and Wanchun Wang were study investigators and contributed to the process of data collection. All authors read and approved the final manuscript.
Acknowledgments
The authors would like to express their gratitude to EditSprings (https://www.editsprings.com/) for the expert linguistic services provided. The authors would like to thank all co-investigators, and colleagues who made this study possible.
Conflicts of Interest
The authors confirmed that there were no conflicts of interest.
References
- 1. Kolasinski SL, Neogi T, Hochberg MC, Oatis C, Guyatt G, Block J, Callahan L, Copenhaver C, Dodge C, Felson D, Gellar K, Harvey WF, Hawker G, et al. 2019 American college of rheumatology/arthritis foundation guideline for the management of osteoarthritis of the hand, hip, and knee. Arthritis Rheumatol. 2020; 72:220–33. https://doi.org/10.1002/art.41142 [PubMed]
- 2. Nüesch E, Dieppe P, Reichenbach S, Williams S, Iff S, Jüni P. All cause and disease specific mortality in patients with knee or hip osteoarthritis: population based cohort study. BMJ. 2011; 342:d1165. https://doi.org/10.1136/bmj.d1165 [PubMed]
- 3. Institute of Medicine (US) Committee on Advancing Pain Research, Care, and Education. Relieving Pain in America: A Blueprint for Transforming Prevention, Care, Education, and Research. Washington (DC): National Academies Press (US); 2011. [PubMed]
- 4. Glyn-Jones S, Palmer AJ, Agricola R, Price AJ, Vincent TL, Weinans H, Carr AJ. Osteoarthritis. Lancet. 2015; 386:376–87. https://doi.org/10.1016/S0140-6736(14)60802-3 [PubMed]
- 5. Levi-Montalcini R. The nerve growth factor 35 years later. Science. 1987; 237:1154–62. https://doi.org/10.1126/science.3306916 [PubMed]
- 6. Levi-Montalcini R, Skaper SD, Dal Toso R, Petrelli L, Leon A. Nerve growth factor: from neurotrophin to neurokine. Trends Neurosci. 1996; 19:514–20. https://doi.org/10.1016/S0166-2236(96)10058-8 [PubMed]
- 7. Spierings EL, Fidelholtz J, Wolfram G, Smith MD, Brown MT, West CR. A phase III placebo- and oxycodone-controlled study of tanezumab in adults with osteoarthritis pain of the hip or knee. Pain. 2013; 154:1603–12. https://doi.org/10.1016/j.pain.2013.04.035 [PubMed]
- 8. Schnitzer TJ, Easton R, Pang S, Levinson DJ, Pixton G, Viktrup L, Davignon I, Brown MT, West CR, Verburg KM. Effect of tanezumab on joint pain, physical function, and patient global assessment of osteoarthritis among patients with osteoarthritis of the hip or knee: a randomized clinical trial. JAMA. 2019; 322:37–48. https://doi.org/10.1001/jama.2019.8044 [PubMed]
- 9. Mayorga AJ, Wang S, Kelly KM, Thipphawong J. Efficacy and safety of fulranumab as monotherapy in patients with moderate to severe, chronic knee pain of primary osteoarthritis: a randomised, placebo- and active-controlled trial. Int J Clin Pract. 2016; 70:493–505. https://doi.org/10.1111/ijcp.12807 [PubMed]
- 10. Ekman EF, Gimbel JS, Bello AE, Smith MD, Keller DS, Annis KM, Brown MT, West CR, Verburg KM. Efficacy and safety of intravenous tanezumab for the symptomatic treatment of osteoarthritis: 2 randomized controlled trials versus naproxen. J Rheumatol. 2014; 41:2249–59. https://doi.org/10.3899/jrheum.131294 [PubMed]
- 11. Tiseo PJ, Kivitz AJ, Ervin JE, Ren H, Mellis SJ. Fasinumab (REGN475), an antibody against nerve growth factor for the treatment of pain: results from a double-blind, placebo-controlled exploratory study in osteoarthritis of the knee. Pain. 2014; 155:1245–52. https://doi.org/10.1016/j.pain.2014.03.018 [PubMed]
- 12. Nagashima H, Suzuki M, Araki S, Yamabe T, Muto C, and Tanezumab Investigators. Preliminary assessment of the safety and efficacy of tanezumab in Japanese patients with moderate to severe osteoarthritis of the knee: a randomized, double-blind, dose-escalation, placebo-controlled study. Osteoarthritis Cartilage. 2011; 19:1405–12. https://doi.org/10.1016/j.joca.2011.09.006 [PubMed]
- 13. Lane NE, Schnitzer TJ, Birbara CA, Mokhtarani M, Shelton DL, Smith MD, Brown MT. Tanezumab for the treatment of pain from osteoarthritis of the knee. N Engl J Med. 2010; 363:1521–31. https://doi.org/10.1056/NEJMoa0901510 [PubMed]
- 14. Brown MT, Murphy FT, Radin DM, Davignon I, Smith MD, West CR. Tanezumab reduces osteoarthritic hip pain: results of a randomized, double-blind, placebo-controlled phase III trial. Arthritis Rheum. 2013; 65:1795–803. https://doi.org/10.1002/art.37950 [PubMed]
- 15. Dakin P, DiMartino SJ, Gao H, Maloney J, Kivitz AJ, Schnitzer TJ, Stahl N, Yancopoulos GD, Geba GP. The efficacy, tolerability, and joint safety of fasinumab in osteoarthritis pain: a phase IIb/III double-blind, placebo-controlled, randomized clinical trial. Arthritis Rheumatol. 2019; 71:1824–34. https://doi.org/10.1002/art.41012 [PubMed]
- 16. Moher D, Liberati A, Tetzlaff J, Altman DG, and PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ. 2009; 339:b2535. https://doi.org/10.1136/bmj.b2535 [PubMed]
- 17. Kivitz A, Ma C, Ahdieh H, Galer BS. A 2-week, multicenter, randomized, double-blind, placebo-controlled, dose-ranging, phase III trial comparing the efficacy of oxymorphone extended release and placebo in adults with pain associated with osteoarthritis of the hip or knee. Clin Ther. 2006; 28:352–64. https://doi.org/10.1016/j.clinthera.2006.03.008 [PubMed]
- 18. Zacher J, Feldman D, Gerli R, Scott D, Hou SM, Uebelhart D, Rodger IW, Ozturk ZE, and etoricoxib OA study group. A comparison of the therapeutic efficacy and tolerability of etoricoxib and diclofenac in patients with osteoarthritis. Curr Med Res Opin. 2003; 19:725–36. https://doi.org/10.1185/030079903125002469 [PubMed]
- 19. Puopolo A, Boice JA, Fidelholtz JL, Littlejohn TW, Miranda P, Berrocal A, Ko A, Cichanowitz N, Reicin AS. A randomized placebo-controlled trial comparing the efficacy of etoricoxib 30 mg and ibuprofen 2400 mg for the treatment of patients with osteoarthritis. Osteoarthritis Cartilage. 2007; 15:1348–56. https://doi.org/10.1016/j.joca.2007.05.022 [PubMed]
- 20. Vojtaššák J, Vojtaššák J, Jacobs A, Rynn L, Waechter S, Richarz U. A phase IIIb, multicentre, randomised, parallel-group, placebo-controlled, double-blind study to investigate the efficacy and safety of OROS hydromorphone in subjects with moderate-to-severe chronic pain induced by osteoarthritis of the hip or the knee. Pain Res Treat. 2011; 2011:239501. https://doi.org/10.1155/2011/239501 [PubMed]
- 21. Prior MJ, Harrison DD, Frustaci ME. A randomized, double-blind, placebo-controlled 12 week trial of acetaminophen extended release for the treatment of signs and symptoms of osteoarthritis. Curr Med Res Opin. 2014; 30:2377–87. https://doi.org/10.1185/03007995.2014.949646 [PubMed]
- 22. Emery P, Koncz T, Pan S, Lowry S. Analgesic effectiveness of celecoxib and diclofenac in patients with osteoarthritis of the hip requiring joint replacement surgery: a 12-week, multicenter, randomized, double-blind, parallel-group, double-dummy, noninferiority study. Clin Ther. 2008; 30:70–83. https://doi.org/10.1016/j.clinthera.2008.01.016 [PubMed]
- 23. McKenna F, Borenstein D, Wendt H, Wallemark C, Lefkowith JB, Geis GS. Celecoxib versus diclofenac in the management of osteoarthritis of the knee. Scand J Rheumatol. 2001; 30:11–18. https://doi.org/10.1080/030097401750065265 [PubMed]
- 24. Altman RD, Dreiser RL, Fisher CL, Chase WF, Dreher DS, Zacher J. Diclofenac sodium gel in patients with primary hand osteoarthritis: a randomized, double-blind, placebo-controlled trial. J Rheumatol. 2009; 36:1991–99. https://doi.org/10.3899/jrheum.081316 [PubMed]
- 25. Bookman AA, Williams KS, Shainhouse JZ. Effect of a topical diclofenac solution for relieving symptoms of primary osteoarthritis of the knee: a randomized controlled trial. CMAJ. 2004; 171:333–38. https://doi.org/10.1503/cmaj.1031793 [PubMed]
- 26. Gibofsky A, Hochberg MC, Jaros MJ, Young CL. Efficacy and safety of low-dose submicron diclofenac for the treatment of osteoarthritis pain: a 12 week, phase 3 study. Curr Med Res Opin. 2014; 30:1883–93. https://doi.org/10.1185/03007995.2014.946123 [PubMed]
- 27. Essex MN, O’Connell MA, Behar R, Bao W. Efficacy and safety of nonsteroidal anti-inflammatory drugs in Asian patients with knee osteoarthritis: summary of a randomized, placebo-controlled study. Int J Rheum Dis. 2016; 19:262–70. https://doi.org/10.1111/1756-185X.12667 [PubMed]
- 28. Reed K, Collaku A, Moreira S. Efficacy and safety of twice daily sustained-release paracetamol formulation for osteoarthritis pain of the knee or hip: a randomized, double-blind, placebo-controlled, twelve-week study. Curr Med Res Opin. 2018; 34:689–99. https://doi.org/10.1080/03007995.2017.1417245 [PubMed]
- 29. Essex MN, Bhadra P, Sands GH. Efficacy and tolerability of celecoxib versus naproxen in patients with osteoarthritis of the knee: a randomized, double-blind, double-dummy trial. J Int Med Res. 2012; 40:1357–70. https://doi.org/10.1177/147323001204000414 [PubMed]
- 30. Leung AT, Malmstrom K, Gallacher AE, Sarembock B, Poor G, Beaulieu A, Castro R, Sanchez M, Detora LM, Ng J. Efficacy and tolerability profile of etoricoxib in patients with osteoarthritis: a randomized, double-blind, placebo and active-comparator controlled 12-week efficacy trial. Curr Med Res Opin. 2002; 18:49–58. https://doi.org/10.1185/030079902125000282 [PubMed]
- 31. Gordo AC, Walker C, Armada B, Zhou D. Efficacy of celecoxib versus ibuprofen for the treatment of patients with osteoarthritis of the knee: a randomized double-blind, non-inferiority trial. J Int Med Res. 2017; 45:59–74. https://doi.org/10.1177/0300060516673707 [PubMed]
- 32. Niethard FU, Gold MS, Solomon GS, Liu JM, Unkauf M, Albrecht HH, Elkik F. Efficacy of topical diclofenac diethylamine gel in osteoarthritis of the knee. J Rheumatol. 2005; 32:2384–92. [PubMed]
- 33. Yoo MC, Yoo WH, Kang SB, Park YW, Kim SS, Moon KH, Song YW, Min BW, Cho YJ, Moon SH, Bin SI, Baek HJ, Shim SC, et al. Etoricoxib in the treatment of Korean patients with osteoarthritis in a double-blind, randomized controlled trial. Curr Med Res Opin. 2014; 30:2399–408. https://doi.org/10.1185/03007995.2014.955169 [PubMed]
- 34. DeLemos BP, Xiang J, Benson C, Gana TJ, Pascual ML, Rosanna R, Fleming B. Tramadol hydrochloride extended-release once-daily in the treatment of osteoarthritis of the knee and/or hip: a double-blind, randomized, dose-ranging trial. Am J Ther. 2011; 18:216–26. https://doi.org/10.1097/MJT.0b013e3181cec307 [PubMed]
- 35. Reginster JY, Malmstrom K, Mehta A, Bergman G, Ko AT, Curtis SP, Reicin AS. Evaluation of the efficacy and safety of etoricoxib compared with naproxen in two, 138-week randomised studies of patients with osteoarthritis. Ann Rheum Dis. 2007; 66:945–51. https://doi.org/10.1136/ard.2006.059162 [PubMed]
- 36. Gana TJ, Pascual ML, Fleming RR, Schein JR, Janagap CC, Xiang J, Vorsanger GJ, and 023 Study Group. Extended-release tramadol in the treatment of osteoarthritis: a multicenter, randomized, double-blind, placebo-controlled clinical trial. Curr Med Res Opin. 2006; 22:1391–401. https://doi.org/10.1185/030079906X115595 [PubMed]
- 37. Svensson O, Malmenäs M, Fajutrao L, Roos EM, Lohmander LS. Greater reduction of knee than hip pain in osteoarthritis treated with naproxen, as evaluated by WOMAC and SF-36. Ann Rheum Dis. 2006; 65:781–84. https://doi.org/10.1136/ard.2005.040519 [PubMed]
- 38. Rauck R, Rapoport R, Thipphawong J. Results of a double-blind, placebo-controlled, fixed-dose assessment of once-daily OROS® hydromorphone ER in patients with moderate to severe pain associated with chronic osteoarthritis. Pain Pract. 2013; 13:18–29. https://doi.org/10.1111/j.1533-2500.2012.00555.x [PubMed]
- 39. Miceli-Richard C, Le Bars M, Schmidely N, Dougados M. Paracetamol in osteoarthritis of the knee. Ann Rheum Dis. 2004; 63:923–30. https://doi.org/10.1136/ard.2003.017236 [PubMed]
- 40. Varadi G, Zhu Z, Blattler T, Hosle M, Loher A, Pokorny R, Frey D, Carter SG. Randomized clinical trial evaluating transdermal ibuprofen for moderate to severe knee osteoarthritis. Pain Physician. 2013; 16:E749–62. [PubMed]
- 41. Altman RD, Zinsenheim JR, Temple AR, Schweinle JE. Three-month efficacy and safety of acetaminophen extended-release for osteoarthritis pain of the hip or knee: a randomized, double-blind, placebo-controlled study. Osteoarthritis Cartilage. 2007; 15:454–61. https://doi.org/10.1016/j.joca.2006.10.008 [PubMed]
- 42. Schnitzer TJ, Tesser JR, Cooper KM, Altman RD. A 4-week randomized study of acetaminophen extended-release vs rofecoxib in knee osteoarthritis. Osteoarthritis Cartilage. 2009; 17:1–7. https://doi.org/10.1016/j.joca.2008.05.018 [PubMed]
- 43. Day R, Morrison B, Luza A, Castaneda O, Strusberg A, Nahir M, Helgetveit KB, Kress B, Daniels B, Bolognese J, Krupa D, Seidenberg B, Ehrich E. A randomized trial of the efficacy and tolerability of the COX-2 inhibitor rofecoxib vs ibuprofen in patients with osteoarthritis. Rofecoxib/ibuprofen comparator study group. Arch Intern Med. 2000; 160:1781–87. https://doi.org/10.1001/archinte.160.12.1781 [PubMed]
- 44. Ehrich EW, Schnitzer TJ, McIlwain H, Levy R, Wolfe F, Weisman M, Zeng Q, Morrison B, Bolognese J, Seidenberg B, Gertz BJ. Effect of specific COX-2 inhibition in osteoarthritis of the knee: a 6 week double blind, placebo controlled pilot study of rofecoxib. Rofecoxib osteoarthritis pilot study group. J Rheumatol. 1999; 26:2438–47. [PubMed]
- 45. Cannon GW, Caldwell JR, Holt P, McLean B, Seidenberg B, Bolognese J, Ehrich E, Mukhopadhyay S, Daniels B. Rofecoxib, a specific inhibitor of cyclooxygenase 2, with clinical efficacy comparable with that of diclofenac sodium: results of a one-year, randomized, clinical trial in patients with osteoarthritis of the knee and hip. Rofecoxib phase III protocol 035 study group. Arthritis Rheum. 2000; 43:978–87. https://doi.org/10.1002/1529-0131(200005)43:5<978::AID-ANR4>3.0.CO;2-0 [PubMed]
- 46. Schnitzer TJ, Marks JA. A systematic review of the efficacy and general safety of antibodies to NGF in the treatment of OA of the hip or knee. Osteoarthritis Cartilage. 2015 (Suppl 1); 23:S8–17. https://doi.org/10.1016/j.joca.2014.10.003 [PubMed]
- 47. Chen J, Li J, Li R, Wang H, Yang J, Xu J, Zha Z. Efficacy and safety of tanezumab on osteoarthritis knee and hip pains: a meta-analysis of randomized controlled trials. Pain Med. 2017; 18:374–85. https://doi.org/10.1093/pm/pnw262 [PubMed]
- 48. Smith SR, Deshpande BR, Collins JE, Katz JN, Losina E. Comparative pain reduction of oral non-steroidal anti-inflammatory drugs and opioids for knee osteoarthritis: systematic analytic review. Osteoarthritis Cartilage. 2016; 24:962–72. https://doi.org/10.1016/j.joca.2016.01.135 [PubMed]
- 49. Higgins JPT, Green S. Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration, 2011. https://training.cochrane.org/handbook/archive/v5.1/.
- 50. Higgins JPT, Deeks JJ, Altman DG. Chapter 16: Special topics in statistics. In: Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions. Version 5.1.0 [updated March 2011]. The Cochrane Collaboration, 2011. https://training.cochrane.org/handbook/archive/v5.1/.
- 51. Caldwell DM, Ades AE, Higgins JP. Simultaneous comparison of multiple treatments: combining direct and indirect evidence. BMJ. 2005; 331:897–900. https://doi.org/10.1136/bmj.331.7521.897 [PubMed]
- 52. Caldwell DM, Welton NJ, Ades AE. Mixed treatment comparison analysis provides internally coherent treatment effect estimates based on overviews of reviews and can reveal inconsistency. J Clin Epidemiol. 2010; 63:875–82. https://doi.org/10.1016/j.jclinepi.2009.08.025 [PubMed]
- 53. Lu G, Ades AE. Combination of direct and indirect evidence in mixed treatment comparisons. Stat Med. 2004; 23:3105–24. https://doi.org/10.1002/sim.1875 [PubMed]
- 54. Bucher HC, Guyatt GH, Griffith LE, Walter SD. The results of direct and indirect treatment comparisons in meta-analysis of randomized controlled trials. J Clin Epidemiol. 1997; 50:683–91. https://doi.org/10.1016/s0895-4356(97)00049-8 [PubMed]
- 55. Ades AE, Sculpher M, Sutton A, Abrams K, Cooper N, Welton N, Lu G. Bayesian methods for evidence synthesis in cost-effectiveness analysis. Pharmacoeconomics. 2006; 24:1–19. https://doi.org/10.2165/00019053-200624010-00001 [PubMed]
- 56. Sutton A, Ades AE, Cooper N, Abrams K. Use of indirect and mixed treatment comparisons for technology assessment. Pharmacoeconomics. 2008; 26:753–67. https://doi.org/10.2165/00019053-200826090-00006 [PubMed]
- 57. Brooks SP, Gelman A. General Methods for Monitoring Convergence of Iterative Simulations. Journal of Computational and Graphical Statistics. 1998; 7:434–455.
- 58. Wandel S, Jüni P, Tendal B, Nüesch E, Villiger PM, Welton NJ, Reichenbach S, Trelle S. Effects of glucosamine, chondroitin, or placebo in patients with osteoarthritis of hip or knee: network meta-analysis. BMJ. 2010; 341:c4675. https://doi.org/10.1136/bmj.c4675 [PubMed]
- 59. Salanti G, Ades AE, Ioannidis JP. Graphical methods and numerical summaries for presenting results from multiple-treatment meta-analysis: an overview and tutorial. J Clin Epidemiol. 2011; 64:163–71. https://doi.org/10.1016/j.jclinepi.2010.03.016 [PubMed]


