Identification of long non-coding RNA signatures for squamous cell carcinomas and adenocarcinomas
Abstract
Studies have demonstrated that both squamous cell carcinomas (SCCs) and adenocarcinomas (ACs) possess some common molecular characteristics. Evidence has accumulated to support the theory that long non-coding RNAs (lncRNAs) serve as novel biomarkers and therapeutic targets in complex diseases such as cancer.
In this study, we aimed to identify pan lncRNA signatures that are common to squamous cell carcinomas or adenocarcinomas with different tissues of origin. With the aid of elastic-net regularized regression models, a 35-lncRNA pan discriminative signature and an 11-lncRNA pan prognostic signature were identified for squamous cell carcinomas, whereas a 6-lncRNA pan discriminative signature and a 5-lncRNA pan prognostic signature were identified for adenocarcinomas. Among them, many well-known cancer relevant genes such as MALAT1 and PVT1 were included.
The identified pan lncRNA lists can help experimental biologists generate research hypotheses and adopt existing treatments for less prevalent cancers. Therefore, these signatures warrant further investigation.
Introduction
Squamous cell carcinomas (SCCs) are neoplasms of the squamous cells that compose most of the skin’s upper layers (epidermis). They may also occur in other tissues, including mouth, esophagus, bladder, prostate, lung, vagina and cervix. Studies [1–3] have suggested that regardless of the tissue of origin, SCC patients share some common molecular characteristics, which thus, may be clustered together. The statistical analyses were carried out using either an integrative analysis including several omics data types or a single analysis on mRNA expression profiles alone. For example, using the cluster-of-cluster-assignments method [4], an integrated dataset including 6 types of omics data for 12 human solid cancer types was analyzed. Results showed that one of the clusters (the squamous-like category) was dominated by lung squamous cell carcinoma and head and neck squamous cell carcinoma [2], in spite of both originating in distinct organs. Naturally, it is anticipated that a pan-gene signature that is commonly applicable to all SCCs may exist.
On the other hand, adenocarcinoma (AC) is a type of cancer that starts in the mucous glands inside of organs, including lungs, colon, esophagus, prostate or even breasts. The Cancer Genomic Atlas (TCGA) network [3] has demonstrated that esophageal adenocarcinoma resembles gastric adenocarcinoma more than it resembles esophageal squamous cell carcinoma (ESCC).
Genome-wide transcriptome analysis has revealed that non-protein-coding genes, which once were regarded as evolutionary junk, account for about 98 % of the human transcripts. Long non-coding RNAs (lncRNAs) are a major class of non-coding RNAs that have a length of more than 200 nucleotides [5]. Nowadays, significant evidence has accumulated to support the theory that lncRNAs can serve as novel biomarkers and therapeutic targets in complex diseases such as cancer [6]. According to Ching et al. [7], lncRNAs are more tissue specific than mRNAs. Nevertheless, in the literature it is easy to find many lncRNAs that are associated with several cancer types that start at different sites/organs. For example, metastasis associated lung adenocarcinoma transcript 1 (MALAT1), a well-known oncogene, has been linked with a variety of cancers such as pancreatic cancer [8–10], prostate cancer [11, 12], hepatocellular carcinoma [13] and thyroid cancer [14]. Specifically for SCCs, MALAT1 has been verified experimentally to correlate with esophageal [15, 16] and oral SCC [17]. Similarly, HOTAIR has been validated by experiments to be correlated with lung adenocarcinoma [18, 19] and gastric carcinoma [20] in addition to other cancer types according to the lncRNAdisease2.0 knowledgebase [21]. Therefore, it is reasonable to speculate the existence of common lncRNAs that play essential roles in many cancer types including SCCs and ACs despite the fact that lncRNAs are more tissue specific than mRNAs. To the best of our knowledge, no study has yet explored the existence of lncRNA signatures for either a pan SCC type or a pan AC type.
Similar to other high-dimensional omics data, feature selection is usually exploited when constructing lncRNA signatures. The goals of feature selection are to eliminate the curse of dimensionality issue, speed up the learning process, avoid over-fitting and thus generate more reliable discriminative or prognostic gene lists/signatures. The selection of relevant lncRNA lists can be realized by using a penalized/regularized regression model, which belongs to the family of embedded feature selection methods. As opposed to filter methods, the embedded methods take the joint effects of covariates into account, and thus can model gene dependencies and concordance. On the other hand, such methods have better computational complexity than the wrappers methods. Therefore, a penalized regression model has harnessed increasing attention from many statisticians and computational biologists [22].
In this study, pan lncRNA signatures commonly applicable to SCCs or ACs with different tissues of origin were constructed with the aid of elastic-net regularized regression models. Specifically, an extensive investigation of potential discriminative and prognostic gene signatures was sought in TCGA, where both pieces of information are available. While a gene signature that can distinguish normal controls from tumors may provide insightful clues on initiation and development of the disease, a prognostic gene signature focuses more on the prediction of disease progression, thus facilitating more effective interventions for patients with poor prognosis to prolong their survival or cure the disease. Therefore, both types of signatures are of crucial importance.
Results
Discriminative lncRNA signatures
Squamous cell carcinomas
Separate logistic elastic-net regression models were fit for the LUSC and HNSC studies (the CESC study were excluded due to the non-availability of normal tissues). A 173-lncRNA discriminative signature for HNSC and a 277-lncRNA signature for the LUSC study were identified. The gene lists for these two studies resulted in 35 overlapped lncRNAs (Table 1). Of those, 10 genes have been reported in the literature to correlate to one specific cancer type or more using real experiments (rather than being predicted using a computational method) according to the lncRNADisease 2.0 database.
Table 1. Pan discriminative lncRNAs for the squamous cell carcinoma type.
| Symbols | Target mRNA | Cancer types (experimentally validated) | Recent publications recording associated cancer types |
| WDFY3-AS2 | WDFY3 | | Ovarian [23]; ESCC [24]; LUAD [25]; breast [26, 27]; HCC [28] |
| CFAP99 | | | |
| DUXAP8 | | Stomach | HCC [29–31]; colon [32, 33]; bladder [34, 35]; pancreatic [36]; ESCC [37]; esophageal [38]; renal cell carcinoma [39, 40]; NSCLC [41] |
| FIRRE | | | Diffuse large B-cell lymphoma [42]; colon [43] |
| HAGLR | EVX2, HOXD13, HOXD12, HOXD11, HOXD10, HOXD9, HOXD8, HOXD3, AC009336.2, HOXD4, HOXD1, MTX2 | Stomach, cervical, NSCLC, neuroblastoma, glioma, ovarian, urinary bladder, HCC, prostate, thyroid | Esophageal [44]; LUAD [45]; colon [46] |
| LOC101929331 | | | |
| DUXAP10 | | Ovarian, colorectal | Myeloid leukemia [47]; ESCC [48]; bladder [49] |
| ABHD11-AS1 | VPS37D, DNAJC30, BUD23, STX1A, ABHD11, CLDN3, CLDN4, METTL27 | Urinary bladder, stomach | Thyroid [50, 51]; pancreatic [52, 53]; endometrial carcinoma [54]; ovarian [55]; colon [56, 57] |
| LOC101928118 | | | |
| SLC16A1-AS1 | SLC16A1, LRIG2 | Cervical, lung, astrocytoma | NSCLC [58]; OSCC [59]; HCC [60] |
| MFI2-AS1 | MFI2 | | Colon [61]; glioma [62]; HCC [63] |
| LINC00443 | ARGLU1 | | Renal cell carcinoma [64] |
| DLEU2 | SPRYD7,
TRIM13,
KCNRG | Lymphoma, laryngeal, leukemia, pancreatic, astrocytoma | Gastric [65]; NSCLC [66, 67]; pancreatic [68]; HCC [69]; esophageal [70] |
| LOC101927596 | | | |
| LOC105375401 | | | |
| LOC100128164 | | | |
| ADAMTS9-AS1 | ADAMTS9 | Ovarian epithelial cancer, malignant glioma | Prostate [71]; COAD [72] |
| ADAMTS9-AS2 | ADAMTS9 | Malignant glioma, renal, NSCLC | Ovarian [73]; gastric [74–76]; glioblastoma [77]; breast [26, 78]; TSCC [79]; esophageal [80] |
| LINC00996 | LRRC61, RARRES2, REPIN1, AC073111.3, ZNF775, AC073111.5, GIMAP8, GIMAP7 | | |
| LINC01296 | | Prostate, stomach, urinary bladder, colorectal | Osteosarcoma [81]; neuroblastoma [82]; ESCC [83, 84]; NSCLC [85]; HCC [86]; breast [87]; ovarian ([88]) |
| LOC101929066 | | | |
| LOC101929595 | | | |
| LOC440028 | | | |
| LOC101929340 | | | |
| LINC00958 | | | Cervical [89, 90]; OSCC [91, 92]; NSCLC [93]; pancreatic [94]; nasopharyngeal [95]; HNSCC [96]; HCC [97]; gastric [98] |
| LOC101927392 | | | |
| RBPMS-AS1 | RBPMS | | |
| BBOX1-AS1 | FIBIN, BBOX1 | | Colon [99]; cervical [100] |
| TMPO-AS1 | TMPO, SLC25A3, IKBIP | Astrocytoma | |
| GS1-120K12.4 | | | |
| TTC39A-AS1 | RNF11, TTC39A, EPS15 | | |
| LOC400568 | | | |
| TMEM220-AS1 | MYH3, SCO1, ADPRM, TMEM220, TMEM238L, PIRT | | |
| LDLRAD4-AS1 | | | |
| LINC00551 | EFNB2, ARGLU1 | | |
| * Cancer types (with experimental validations) to be associated with that specific lncRNA according to the lncRNAdisease2.0 database. |
| ** Only a few examples are given since the number of relevant publications is large. |
| COAD: colon adenocarcinoma; ESCC: esophageal squamous cell carcinoma; HCC: Hepatocellular carcinoma; HNSCC: head and neck squamous cell carcinoma; LUAD: lung adenocarcinoma; NSCLC: non-small cell lung cancer; OSCC: oral squamous cell carcinoma; TSCC: tongue squamous cell carcinoma. |
Adenocarcinomas
Among the AC cohort, separate logistic elastic-net regression models identified a 185-lncRNA signature for the LUAD study, a 173-lncRNA signature for the STAD study and a 114-lncRNA signature for the PRAD study. The intersection of these three gene lists has 6 lncRNAs: UBXN10-AS1, SNHG20, ADAMTS9-AS1, ADAMTS9-AS2, PVT1 and VPS9D1-AS1 (Table 2). Of note, both ADAMTS9-AS1 and ADAMTS9-AS2 also belong to the 35-overlapped lncRNA list for SCC discriminative analysis.
Table 2. Pan discriminative lncRNAs for the adenocarcinoma type.
| Symbols | Target mRNAs | Cancer types (experimentally validated)* | Recent publications recording associated cancer types |
| UBXN10-AS1 | PLA2G5, PLA2G2D, PLA2G2F, PLA2G2C, UBXN10 | | |
| SNHG20 | SEC14L1 | HCC, ovarian, colorectal, NSLCL, stomach | Breast [101]; cervical [102]; bladder [103]; prostate [104]; ESCC [105]; OSCC [106, 107]; nasopharyngeal [108] |
| ADAMTS9-AS1 | ADAMTS9 | Ovarian epithelial cancer, glioma | Prostate [32]; COAD [72] |
| ADAMTS9-AS2 | ADAMTS9 | Glioma, renal, NSCLC | Ovarian [73]; gastric [74–76]; clear cell renal cell carcinoma [109]; glioblastoma [77]; breast [26, 78]; TSCC [79]; esophageal [80]; NSCLC [110] |
| PVT1 | MYC | Colorectal, HCC, prostate, cervical, stomach, lung, esophageal and others | EAC [111] ** |
| VPS9D1-AS1 | DPEP1, CHMP1A, SPATA33, CDK10, SPATA2L, VPS9D1, ZNF276, FANCA | Stomach | NSCLC [112, 113]; prostate [114, 115] |
| *Cancer types (with experimental validations) to be associated with that specific lncRNA according to the lncRNAdisease2.0 database. |
| **Only one example is given since the number of relevant publications is large. |
| COAD: colon adenocarcinoma; EAC: esophageal adenocarcinoma; ESCC: esophageal squamous cell carcinoma; HCC: Hepatocellular carcinoma; NSCLC: non-small cell lung cancer; OSCC: oral squamous cell carcinoma; TSCC: tongue squamous cell carcinoma. |
Furthermore, the number of overlaps between LUAD and STAD is 34; 18 between LUAD and PRAD and 11 between STAD and PRAD. All these overlaps took substantial proportions of the identified lncRNA signatures.
Prognostic lncRNA signatures
Squamous cell carcinoma
Cox elastic-net regression models selected a 462-lncRNA list for the LUSC study, a 597-lncRNA list for the HNSC study and a 263-lncRNA list for the CESC study, respectively. Among the 3 lists were 11 overlaps (Table 3), and 5 of them (CFLAR-AS1, SLC16A1-AS1, SIRPG-AS1, LOC389641 and LINC00593) were experimentally validated as cancer related genes according to the lncRNADisease 2.0 database. In Table 3, the target mRNAs of these 11 lncRNAs are given.
Table 3. Pan prognostic lncRNAs for the squamous cell carcinoma type.
| Symbols | Target mRNAs | Cancer types (experimentally validated)* | Recent publications recording associated cancer types with experimental validations |
| SLC16A1-AS1 | SLC16A1, LRIG2 | Cervical, lung,
astrocytoma | NSCLC [58]; OSCC [59]; HCC [60] |
| CFLAR-AS1 | FAM126B, NDUFB3
CFLAR, CASP10, CASP8 | ESCC, astrocytoma | |
| SPATA13-AS1 | AL359736.1, SPATA13, C1QTNF9 | | |
| LINC00311 | GSE1 | | Thyroid [117] |
| LINC01305 | OLA1, SP9, CIR1, SCRN3 | | Cervical [118]; NSCLC [119] |
| LINC01399 | ISX, HMGXB4, TOM1 | | |
| FGF14-AS1 | FGF14 | | |
| SIRPG-AS1 | SIRPD, AL049634.2, SIRPB1, SIRPG | Astrocytoma | |
| LOC389641 | TNFRSF10D, TNFRSF10A, CHMP7, R3HCC1, LOXL2 | PADA | PDAC [120] |
| LINC00593 | | Astrocytoma | |
| MKNK1-AS1 | DMBX1, AL136373.1, KNCN, MKNK1, MOB3C, ATPAF1, TEX38 | | |
| *Cancer types (with experimental validations) to be associated with that specific lncRNA according to the lncRNAdisease2.0 database. |
| ESCC: esophageal squamous cell carcinoma; HCC: Hepatocellular carcinoma; NSCLC: non-small cell lung cancer; OSCC: oral squamous cell carcinoma; PDAC: pancreatic ductal adenocarcinoma. |
Adenocarcinoma
Cox elastic-net regression models identified a 53-lncRNA set for the STAD study and a 95-lncRNA list for the LUAD study. The PRAD study was excluded from the prognosis analysis since the number of events (deaths) was too small to guarantee a valid analysis. The intersection set of the two gene lists includes 5 lncRNAs (EIF1AX-AS1, LINC00115, LINC01237, MALAT1 and LINC00528), among which only LINC00115 and MALAT1 have been experimentally validated to correlate with cancers according to the lncRNADisease2.0 database. The associated cancer types of and the target mRNAs by these five lncRNAs given by the lncRNADisease 2.0 database are listed in Table 4.
Table 4. Pan prognostic lncRNAs for the adenocarcinoma type.
| Symbols | Target mRNAs | Cancer types (experimentally validated)* | Recent publications recording associated cancer types with experimental validations |
| EIF1AX-AS1 | MAP7D2, EIF1AX, RPS6KA3 | | |
| LINC00115 | SAMD11 | Astrocytoma, lung adenocarcinoma | Glioma [126]; breast [127] |
| LINC01237 | GAL3ST2, NEU4, PDCD1, RTP5, AC131097.2 | | |
| LINC00528 | BCL2L13, BID, MICAL3 | | LSCC [128] |
| MALAT1 | FRMD8, SCYL1, LTBP3, SSSCA1, FAM89B, EHBP1L1, KCNK7, MAP3K11 | Pancreatic, prostate, breast, gallbladder, OSCC, stomach, NSCLC, ESCC, and others | Breast [129]; ovarian [130]; glioblastoma [131] ** |
| *Cancer types (with experimental validations) to be associated with that specific lncRNA according to the lncRNAdisease2.0 database. |
| **Only a few examples are given since the number of relevant publications is large. |
| ESCC: esophageal squamous cell carcinoma; LSCC: laryngeal squamous cell carcinoma; LUAD: lung adenocarcinoma; OSCC: oral squamous cell carcinoma. |
Performance evaluation
Using ROC curves (Figure 1), the discriminative ability of identified pan-SCC and pan-AC lncRNA signatures was evaluated. The AUCs of these two signatures are 0.951 and 0.920 for the SCC type and the AC type, respectively. Overall, these two signatures perform well. According to the log-rank tests (Figure 2), the identified pan-SCC and pan-AC prognostic lncRNA signatures have good prognostic values as well.
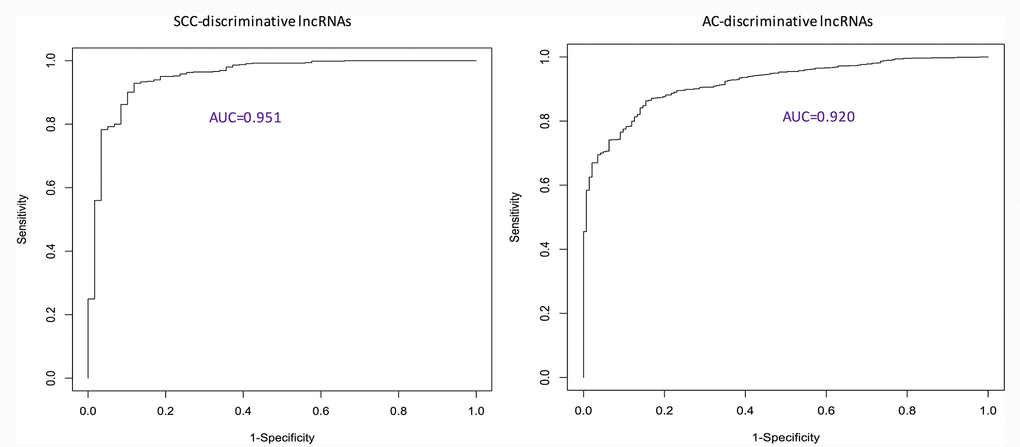
Figure 1. ROC curves showing the performance of identified discriminative lncRNA lists. AC: adenocarcinoma; AUC: area under the ROC curve; ROC: receiver characteristic operator; SCC: squamous cell carcinoma.
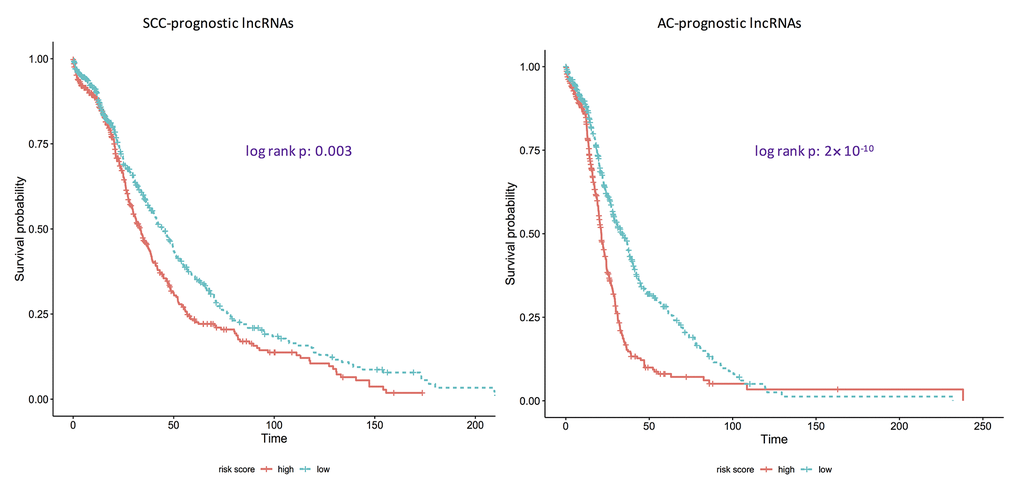
Figure 2. Kaplan-Meier curves showing if identified prognostic lncRNA lists are associated with survival rates for adenocarcinomas and squamous cell carcinomas. AC: adenocarcinoma; SCC: squamous cell carcinoma. p: the corresponding p-values of log-rank tests to test if the survival curves of high-risk group and low-risk group are same. Here, the ridge Cox regression models were used to estimate the coefficients of lncRNAs and then risk scores were calculated. Then the median value of risk scores was used as the cutoff to divide the AC/SCC patients into the high-risk group and the low-risk group.
Enriched pathways
Using String software, pathway enrichment analysis was conducted. The results showed that no pathway or GO biological process term was enriched by the AC prognostic signature. Enriched pathways/GO biological process terms for the SCC and AC discriminative signatures and the SCC prognostic signatures are given in Figure 3. As far as the enriched KEGG pathways are considered, there is one overlap – necroptosis between the AC discriminative category and the SCC prognostic category. Necroptosis is a programmed caspase-independent cell death. Studies have shown that tumor undertakes necroptosis in vivo and the process has pro- or anti- tumor effects in cancer development and progression [116]. At the level of pathways, both SCCs and ACs may share some common features.
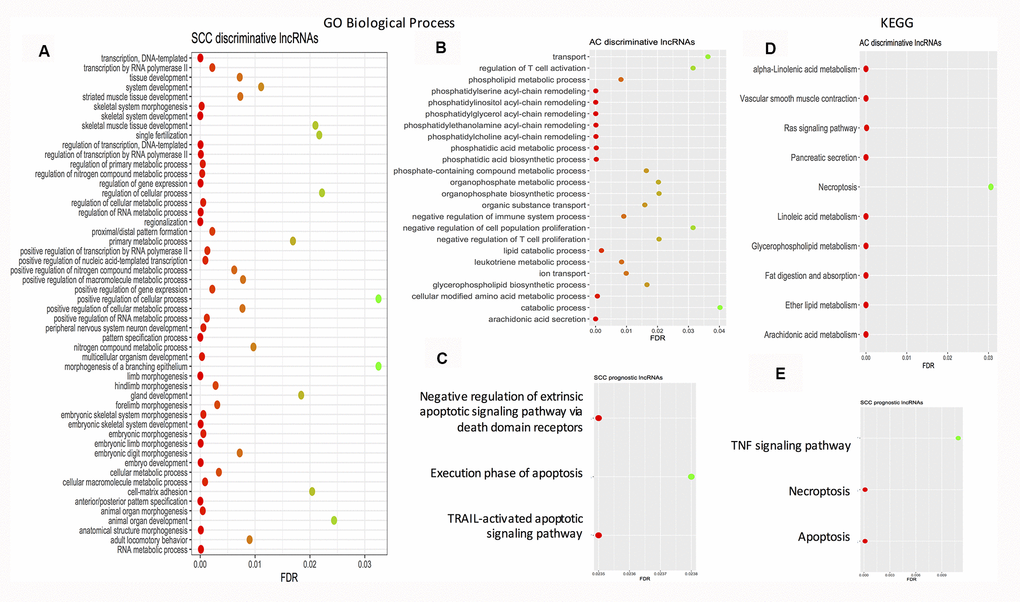
Figure 3. Dot plots showing the enriched gene ontology biological process terms and KEGG pathways. (A) Enriched GO biological process terms by SCC discriminative lncRNA signature. (B) Enriched GO biological process terms by AC discriminative lncRNA signature. (C) Enriched GO biological process terms by SCC prognostic lncRNA signature. (D) Enriched KEGG pathways by AC discriminative lncRNA signature. (E) Enriched KEGG pathways by SCC prognostic lncRNA signature. AC: adenocarcinoma; FDR: false discovery rate; GO: gene ontology; KEGG: Kyoto Encyclopedia of Genes and Genomes; FDR: false discovery rate; SCC: squamous cell carcinoma.
The overlapped proportions/ratios of these prognostic lncRNAs are substantially less than those taken by the discriminative lncRNAs, which may be explained by two reasons. First, many studies in the literature have pointed out that prognosis is more difficult than discrimination/diagnosis. For instance, for the LUAD patients at the same stage, distinct molecular subtypes with different prognoses exist. Correspondingly, prognostic gene signatures are anticipated to be more type-specific, while the sizes of these signatures are to be much larger. Indeed, several studies suggested that possibly dozens of genes can make a perfect segmentation of tumors and controls or of different subtypes, but this is not so for the segmentation of patients with good prognosis versus poor prognosis. Second, the overall survival time may not be a good surrogate for prognosis. Also, these AC and SCC cohorts may not be followed up for a period long enough to develop adequate events/deaths for an accurate survival analysis. With these two disadvantages, many false positives may be produced and included in the resulting gene lists.
Discussion
Discriminative lncRNAs with high biological relevance to cancer
Overlapped lncRNAs by both types
First, the focus is on the two overlapped lncRNAs – ADAMTS9-AS2 and ADAMTS9-AS1. According to the lncRNADisease 2.0 database [21], both lncRNAs are associated with several cancer types. In addition, a search of the PubMed database on recent investigation of the association between these two genes and cancer reveals that more studies concerned ADAMTS9-AS2. For example, a very recent study reported that the expression level of ADAMTS9-AS2 is lower in esophageal cancer tissues and over-expressing it can suppress the development of esophageal cancer via inducing CDHS promoter methylation [80]. The recently experimentally validated cancer types for this lncRNA included breast cancer [26] (using qRT-PCR), gastric cancer (by constructing a potential ceRNA network using bioinformatics tools and then validating the ADAMTS9-AS2/miR-372/CADM2 axis using qRT-PCR and dual luciferase reporter assay) [75], tongue squamous cell carcinoma (constructing ADAMTS9-AS2/miR-600/EZH2 ceRNA network) [79] and others. In contrast, only two recent studies on ADAMTS9-AS1 [71, 72] with experimental validations have been found.
Pan-AC lncRNAs
Of the pan-AC specific lncRNAs, all except UBXN10-AS1 and VPS9D1-AS1 have been experimentally validated to be associated with a variety of cancer types according to the lncRNADisease 2.0 knowledgebase [21]. For example, both SNHG20 and PVT1 are related to stomach cancer. PVT1 is a well-known oncogene and correlated with a variety of cancers according to both the lncRNADisease 2.0 knowledgebase and the GeneCards database [121]. In our previous study [122], PVT1 was identified as a subtype-specific prognostic gene for esophageal adenocarcinoma using a feature selection algorithm called the Cox-filter method [123]. In this study, it was identified as a pan AC discriminative gene, which leads us to anticipate that PVT1 may be more relevant to the AC type than the SCC type, even though PVT1 has also been verified in the literature to associate with the SCC cancer type such as esophageal squamous cell carcinoma [124]. Another feasible explanation is that PVT1 may be a pan-gene commonly applicable for both SCCs and ACs. Further investigation is warranted. Also, SNHG20 is regarded as vital in many cancers [125] and identified as a subtype-specific prognostic gene for laryngeal squamous cell carcinoma in our previous study [36] using a computational method. Recent studies have experimentally validated that this gene is related to esophageal squamous cell carcinoma [105], nasopharyngeal carcinoma [108] and oral squamous cell carcinoma [106–107]. Again, the existence of pan-genes for both AC and SCC types may explain this to some extent.
In addition to gastric cancer as indicated by the lncRNADisease 2.0 database, non-small cell lung cancer [112–115] and prostate cancer have been linked to VPS9D1-AS1 by several recent experimental studies.
Pan-SCC lncRNAs
DUXAP8 was recently identified as a pan-cancer gene using meta-analysis and TCGA pan-cancer data [30]. Focusing on hepatocellular carcinoma, the authors did a qRT-PCR experiment to verify the diagnostic and prognostic values of this gene for cancer patients. The results showed that the expression value of DUXAP8 increased in tumor tissues when compared with their paired normal tissues. In the meantime, high expression of this gene is related to a poor prognosis. In addition to this study, this lncRNA has been linked to several other cancer types, renal cell carcinoma [40] and colon cancer [33], for example. For SCCs specifically, it is linked to esophageal squamous cell carcinoma [37].
Additionally, HAGLR was listed as being highly relevant to cancer. Specific for the SCC or AC cancer type, based on the data analysis of MTT assay, qRT-PCR and western blot experiments, Lu et al. [132] showed that the expression levels of HAGLR were associated with non-small cell lung cancer tumor lymph node metastasis status, stage, and overall survival. With inhibition of HAGLR in non-small cell lung cancer cells, cell proliferation and invasion can be suppressed. Also, Yang et al. [133] showed that down-expression of HAGLR inhibited LAMP3 expression by sponging miR-143-5p and suppressed the progression of esophageal carcinoma.
Prognostic lncRNAs with high biological relevance to cancer
Pan-SCC lncRNAs
Interestingly, SLC16A1-AS1 is deemed as both a prognostic gene and a discriminative gene for the pan-SCC type. Some markers may play crucial roles in both diagnosis and prognosis of a disease, and in our opinion such markers deserve more attention since targeting them may not only prevent occurrence of the disease but also reverse the consequences of the disease after disease onset.
A search of the PubMed database revealed several articles that describe the association between this lncRNA and cancer. For example, Liu et al. [58] demonstrated that SLC16A1-AS1 was under-expressed in non-small cell lung cancer tissues and cell lines with an in situ hybridization experiment. They also showed that SLC16A1-AS1 overexpression could functionally inhibit the viability and proliferation of lung cancer cells, block the cell cycle and promote cell apoptosis in vitro.
It is worth pointing out that none of the 11 pan-SCC prognostic lncRNAs are under comprehensive investigation according to lncRNADisease 2.0. In the PubMed search for recent publications exploring the association of these lncRNAs with cancer, only several additional links were harnessed. For instance, one study indicated that LINC01305 was related to cervical cancer [118] and the other [119] associated it with non-small cell lung cancer.
Pan-AC lncRNAs
Of the pan-AC prognostic lncRNAs, MALAT1 is a well-known oncogene and has been linked to a variety of cancers so far, including non-small cell lung cancer, cervical cancer, tongue squamous cell carcinomas and gastric cancer. In our previous study [122], it was identified as a subtype-specific prognostic gene for laryngeal squamous cell carcinoma using the Cox-filter method [123], further implying its possibility of being a pan-cancer lncRNA.
As far as LINC00115 is concerned, the lncRNADisease 2.0 database linked it to astrocytoma and lung adenocarcinoma. Two very recent studies [126, 127] have added breast cancer and glioma to this list. On the other hand, LINC00528 has recently been demonstrated to relate to laryngeal squamous cell carcinoma [128]. The respective recent publications exploring the association of these lncRNAs with cancer in the PubMed database are summarized in Tables 1–4.
Given the promising results that a substantial proportion of pan-SCC/AC genes identified by the bioinformatics procedure are related to a variety of cancer types and thus have a good biological relevance, the overlapped discriminative- and prognostic- lncRNAs warrant further investigation.
Conclusions
In this study, discriminative and prognostic lncRNA lists for pan SCC and pan AC types were constructed using first elastic-net regression models to obtain individual lncRNA lists for each cancer study, and subsequently identifying the intersection of the resulting lists.
Given the fact that the research on lncRNAs has been a hot topic in the past several years, the shortage of lncRNA markers for complex diseases such as cancer, especially prognostic ones (since the outcome is survival time which needs a long period of follow-up to collect) is still common. The identified lncRNA lists in this article may help experimental biologists generate research hypotheses and design their own experiments correspondingly.
It is worth pointing out that pan cancer gene signatures are equal in importance to type-specific gene signatures. While with the type-specific signature a better prediction for progression and prognosis is possible, some existing drugs/therapies for other cancers may be adopted to treat a less prevalent cancer. Such adoption may help save resources and time for developing a brand-new drug for one specific cancer type, and therefore may increase the chance of survival for cancer patients.
Materials and Methods
Experimental data
The Atlas of ncRNA in Cancer (Tanric) database [134] included lncRNA expression profiles (RNA-Seq data) for 20 cancer types in the TCGA project. We identified 8 cancers that are either the SCC type or AC type with 100 % confidence: cervical squamous cell carcinoma (CESC), head and neck squamous cell carcinoma (HNSC) and lung squamous cell carcinoma (LUSC), lung adenocarcinoma (LUAD), prostate adenocarcinoma (PRAD), stomach adenocarcinoma (STAD), colon adenocarcinoma (COAD), and rectum adenocarcinoma (READ). For the colon and rectum cohorts, no normal tissues were provided and the number of deaths was also very small, rendering both the discrimination and prognosis analyses less trustworthy or even impossible. Consequently, these two cohorts were excluded, and the final SCC study included cervical, head and neck, and lung SCC cohorts. The AC cohorts included lung, prostate and stomach.
For the 6 cohorts, expression profiles were downloaded fromtheTanricwebpage(https://ibl.mdanderson.org/tanric/_design/basic/download.html). Corresponding clinical information such as overall survival time, American Joint Committee on Cancer staging status and age were downloaded from TCGA’s Genomic Data Commons (http://www.cbioportal.org/). Tanric [134] includes expression profiles of 12,727 lncRNAs quantified as the RPKM (fragments per kilo-bases per million) counts. Table 5 summarizes clinical characteristics of these six studies.
Table 5. Clinical characteristics of squamous cell carcinoma studies and adenocarcinoma studies.
| # of patients(stages I-IV) | # of deaths | Missing of survival time/Missing of stage information/ No. of all zero expression | # of normal tissues |
| Squamous Cell Carcinoma | | | | |
| Head and Neck | 426 (24/63/73/200) | 187 | 1/66/43 | 42 |
| Lung | 220 (116/55/43/4) | 105 | 4/2/17 | 17 |
| Cervical1 | 196 (--/--/--/--) | 42 | 0/--/220 | 3 |
| Adenocarcinoma | | | | |
| Lung | 488 (262/112/81/23) | 174 | 9/10/76 | 58 |
| Stomach | 285 (39/99/103/23) | 94 | 3/21/10 | 33 |
| Prostate2 | 374 (--/--/--/--) | 6 | 0/--/53 | 52 |
| --: not available. |
| 1Since no stage information is available and there are only 3 normal tissues, this cohort was excluded from and discriminative analysis. |
| 2The prostate cohort was not included in the cancer-type specific analysis for survival analysis since the number of events/deaths is small. In summary, 2 studies were used for SCC discriminative analysis and 3 for survival analysis, while for AC 3 studies were used for discriminative analysis and 2 for survival analysis. |
Next, ensemble IDs were mapped into gene symbols and lncRNAs without a valid gene symbol were deleted, leaving 3,173 lncRNAs retained for further analysis. Lastly, the RPKM were logarithm transformed (base 2) after being added 1s (in order to avoid having log transformation on zeros), to make the distribution of resulting lncRNA expression values approximate to a normal one.
Statistical methods
Elastic-net regularized regression
A logistic elastic-net regularized regression model and a Cox elastic-net regularized regression model were fit for each cohort to identify the respective discriminative lncRNA signature and prognostic lncRNA signature for the specific cancer type. Briefly, elastic-net penalty is a linear combination of L1 (LASSO) and L2 (ridge) penalties. It is well known that the L1 penalty introduces sparseness into the model by offsetting the coefficients of insignificant genes to zeros. In contrast, the L2 penalty introduces a grouping effect by smoothing the coefficients of correlated genes to a common value. In this sense, it can also penalize large β coefficients. Therefore, the elastic-net penalty incorporates the advantages of both L1 and L2 penalties, i.e., being capable of feature selection and giving the grouping structure some consideration. The mathematical notation is,
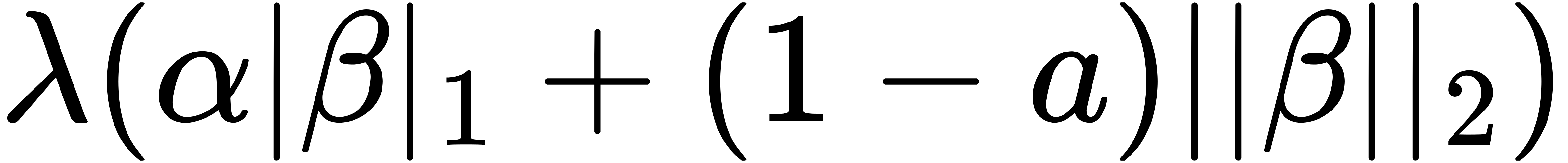
Here, the parameter controls the ratio of L1 penalty to L2 penalty. The tuning parameter λ determines the amount of regularization used, with a large value corresponding to a heavy penalization on β coefficients and a small value to a light one. Its optimal values were determined using 10-fold cross-validations.
Depending on the type of outcome, it may be combined with the corresponding objective function to frame into a variety of regularized regression models such as a logistic elastic-net regression for a binary outcome and a Cox elastic-net regression for survival time.
For individual cohorts, elastic-net regression models were fit. The respective intersections of identified lncRNA lists by the models were obtained and deemed as pan-AC discriminative lncRNAs, pan-SCC discriminative lncRNAs, pan-AC prognostic lncRNAs and pan-SCC prognostic lncRNAs. Their performance and biological relevance were investigated subsequently. Figure 4 shows how these four signatures were constructed.
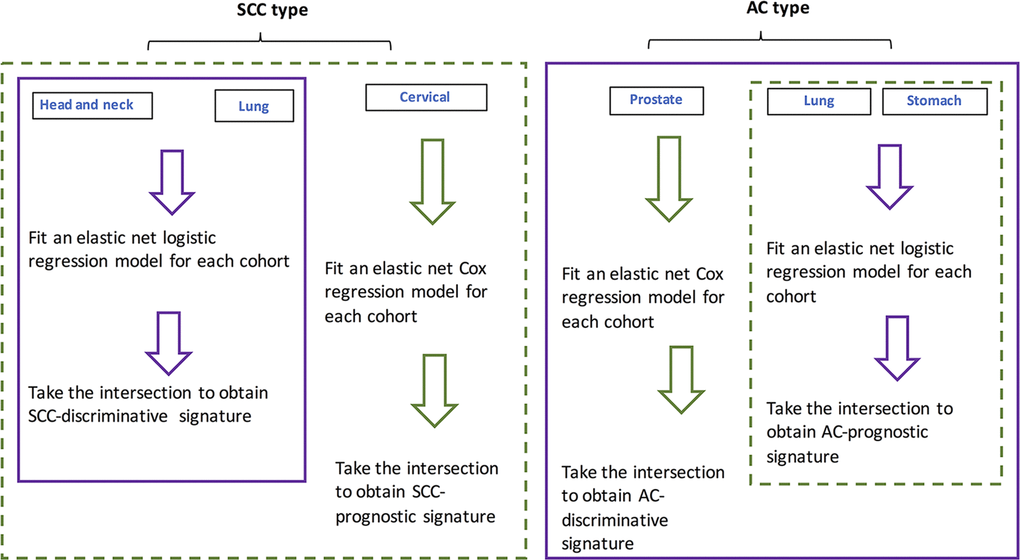
Figure 4. Flowchart showing the procedure for identifying SCC- and AC-discriminative, and SCC- and AC-prognostic lncRNAs.
Pathway enrichment analysis
Connectivity (gene-to-gene interaction) information was retrieved for the target mRNAs by identified lncRNAs from the lncRNADisease 2.0 database [21], and upon those target mRNAs Kyoto Encyclopedia of Genes and Genomes (KEGG) [135] pathway/gene ontology (GO) [136] enrichment analysis was conducted using the String software [137]. A false discovery rate (FDR) of <0.05 was deemed to be statistically significant.
Performance evaluation
For discriminative values of identified lncRNAs, ridge logistic regression models were fit to estimate the coefficients of identified genes and then probabilities of having tumors were calculated. The receiver characteristic operator (ROC) curves were made, and the area under the ROC curve (AUC) statistics were calculated to evaluate the performance of these signatures.
Multivariate Cox regression models with ridge penalty terms were fit to estimate the coefficients of identified prognostic lncRNAs and calculate the risk scores of death for all patients. The median expression value was used as a cutoff to divide the patients into high-risk group and the low-risk groups. Lastly, log-rank tests were carried out to test if the survival curves of the two groups were the same.
Statistical language and packages
All statistical analysis was carried out in R version 3.5 (https://www.r-project.org/). Specifically, the Bioconductor org.Hs.eg.db package was used to map ENSEMBL IDs to gene symbols. The glmnet package [138] was used to fit the elastic net and the ridge regression models. The survival and survminer packages were used for making Kaplan-Meier curves and carrying out log-rank tests. ROCR and ggplot2 were used for making ROC curves and calculating AUC statistics.
Author Contributions
Conceived and designed the study: ST CW WL. Analyzed the data: ST MT JL. Interpreted data analysis and results: ST WL MT JL CW. Wrote the paper: ST CW JL MT. All authors reviewed and approved the final manuscript.
Acknowledgments
We thank Donna Gilbreath in the Markey Cancer Center’s Research Communications Office for English editing.
Conflicts of Interest
No conflicts of interests have been declared.
Funding
This study was supported by the Education Department of Jilin Province (grant No. JJKH20190032KJ) and Jilin Province Department of Finance (grant No. 2018SCZWSZX-018).
References
-
1.
Akbani R, Ng PK, Werner HM, Shahmoradgoli M, Zhang F, Ju Z, Liu W, Yang JY, Yoshihara K, Li J, Ling S, Seviour EG, Ram PT, et al. A pan-cancer proteomic perspective on The Cancer Genome Atlas. Nat Commun. 2014; 5:3887. https://doi.org/10.1038/ncomms4887 [PubMed]
-
2.
Hoadley KA, Yau C, Wolf DM, Cherniack AD, Tamborero D, Ng S, Leiserson MD, Niu B, McLellan MD, Uzunangelov V, Zhang J, Kandoth C, Akbani R, et al, and Cancer Genome Atlas Research Network. Multiplatform analysis of 12 cancer types reveals molecular classification within and across tissues of origin. Cell. 2014; 158:929–44. https://doi.org/10.1016/j.cell.2014.06.049 [PubMed]
-
3.
Cancer Genome Atlas Research Network, Analysis Working Group: Asan University, BC Cancer Agency, Brigham and Women’s Hospital, Broad Institute, Brown University, Case Western Reserve University, Dana-Farber Cancer Institute, Duke University, Greater Poland Cancer Centre, Harvard Medical School, Institute for Systems Biology, KU Leuven, Mayo Clinic, Memorial Sloan Kettering Cancer Center, National Cancer Institute, Nationwide Children’s Hospital, Stanford University, University of Alabama, University of Michigan, University of North Carolina, University of Pittsburgh, University of Rochester, University of Southern California, University of Texas MD Anderson Cancer Center, University of Washington, Van Andel Research Institute, Vanderbilt University, Washington University, Genome Sequencing Center: Broad Institute, Washington University in St. Louis, Genome Characterization Centers: BC Cancer Agency, Broad Institute, Harvard Medical School, Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins University, University of North Carolina, University of Southern California Epigenome Center, University of Texas MD Anderson Cancer Center, Van Andel Research Institute, Genome Data Analysis Centers: Broad Institute, Brown University:, Harvard Medical School, Institute for Systems Biology, Memorial Sloan Kettering Cancer Center, University of California Santa Cruz, University of Texas MD Anderson Cancer Center, Biospecimen Core Resource: International Genomics Consortium, Research Institute at Nationwide Children’s Hospital, Tissue Source Sites: Analytic Biologic Services, Asan Medical Center, Asterand Bioscience, Barretos Cancer Hospital, BioreclamationIVT, Botkin Municipal Clinic, Chonnam National University Medical School, Christiana Care Health System, Cureline, Duke University, Emory University, Erasmus University, Indiana University School of Medicine, Institute of Oncology of Moldova, International Genomics Consortium, Invidumed, Israelitisches Krankenhaus Hamburg, Keimyung University School of Medicine, Memorial Sloan Kettering Cancer Center, National Cancer Center Goyang, Ontario Tumour Bank, Peter MacCallum Cancer Centre, Pusan National University Medical School, Ribeirão Preto Medical School, St. Joseph’s Hospital and Medical Center, St. Petersburg Academic University, Tayside Tissue Bank, University of Dundee, University of Kansas Medical Center, University of Michigan, University of North Carolina at Chapel Hill, University of Pittsburgh School of Medicine, University of Texas MD Anderson Cancer Center, Disease Working Group: Duke University, Memorial Sloan Kettering Cancer Center, National Cancer Institute, University of Texas MD Anderson Cancer Center, Yonsei University College of Medicine, Data Coordination Center: CSRA Inc., and Project Team: National Institutes of Health. Integrated genomic characterization of oesophageal carcinoma. Nature. 2017; 541:169–75. https://doi.org/10.1038/nature20805 [PubMed]
-
4.
Cancer Genome Atlas Network. Comprehensive molecular portraits of human breast tumours. Nature. 2012; 490:61–70. https://doi.org/10.1038/nature11412 [PubMed]
-
5.
St Laurent G, Wahlestedt C, Kapranov P. The landscape of long noncoding RNA classification. Trends Genet. 2015; 31:239–51. https://doi.org/10.1016/j.tig.2015.03.007 [PubMed]
-
6.
Chen X, Yan CC, Zhang X, You ZH. Long non-coding RNAs and complex diseases: from experimental results to computational models. Brief Bioinform. 2017; 18:558–76. https://doi.org/10.1093/bib/bbw060 [PubMed]
-
7.
Ching T, Peplowska K, Huang S, Zhu X, Shen Y, Molnar J, Yu H, Tiirikainen M, Fogelgren B, Fan R, Garmire LX. Pan-cancer analyses reveal long intergenic non-coding RNAs relevant to tumor diagnosis, subtyping and prognosis. EBioMedicine. 2016; 7:62–72. https://doi.org/10.1016/j.ebiom.2016.03.023 [PubMed]
-
8.
Jiao F, Hu H, Yuan C, Wang L, Jiang W, Jin Z, Guo Z, Wang L. Elevated expression level of long noncoding RNA MALAT-1 facilitates cell growth, migration and invasion in pancreatic cancer. Oncol Rep. 2014; 32:2485–92. https://doi.org/10.3892/or.2014.3518 [PubMed]
-
9.
Han T, Jiao F, Hu H, Yuan C, Wang L, Jin ZL, Song WF, Wang LW. EZH2 promotes cell migration and invasion but not alters cell proliferation by suppressing E-cadherin, partly through association with MALAT-1 in pancreatic cancer. Oncotarget. 2016; 7:11194–207. https://doi.org/10.18632/oncotarget.7156 [PubMed]
-
10.
Pang EJ, Yang R, Fu XB, Liu YF. Overexpression of long non-coding RNA MALAT1 is correlated with clinical progression and unfavorable prognosis in pancreatic cancer. Tumour Biol. 2015; 36:2403–07. https://doi.org/10.1007/s13277-014-2850-8 [PubMed]
-
11.
Ren S, Peng Z, Mao JH, Yu Y, Yin C, Gao X, Cui Z, Zhang J, Yi K, Xu W, Chen C, Wang F, Guo X, et al. RNA-seq analysis of prostate cancer in the Chinese population identifies recurrent gene fusions, cancer-associated long noncoding RNAs and aberrant alternative splicings. Cell Res. 2012; 22:806–21. https://doi.org/10.1038/cr.2012.30 [PubMed]
-
12.
Ren S, Wang F, Shen J, Sun Y, Xu W, Lu J, Wei M, Xu C, Wu C, Zhang Z, Gao X, Liu Z, Hou J, et al. Long non-coding RNA metastasis associated in lung adenocarcinoma transcript 1 derived miniRNA as a novel plasma-based biomarker for diagnosing prostate cancer. Eur J Cancer. 2013; 49:2949–59. https://doi.org/10.1016/j.ejca.2013.04.026 [PubMed]
-
13.
Malakar P, Shilo A, Mogilevsky A, Stein I, Pikarsky E, Nevo Y, Benyamini H, Elgavish S, Zong X, Prasanth KV, Karni R. Long noncoding RNA MALAT1 promotes hepatocellular carcinoma development by SRSF1 upregulation and mTOR activation. Cancer Res. 2017; 77:1155–67. https://doi.org/10.1158/0008-5472.CAN-16-1508 [PubMed]
-
14.
Huang JK, Ma L, Song WH, Lu BY, Huang YB, Dong HM, Ma XK, Zhu ZZ, Zhou R. MALAT1 promotes the proliferation and invasion of thyroid cancer cells via regulating the expression of IQGAP1. Biomed Pharmacother. 2016; 83:1–7. https://doi.org/10.1016/j.biopha.2016.05.039 [PubMed]
-
15.
Li Z, Zhou Y, Tu B, Bu Y, Liu A, Kong J. Long noncoding RNA MALAT1 affects the efficacy of radiotherapy for esophageal squamous cell carcinoma by regulating Cks1 expression. J Oral Pathol Med. 2017; 46:583–90. https://doi.org/10.1111/jop.12538 [PubMed]
-
16.
Li RQ, Ren Y, Liu W, Pan W, Xu FJ, Yang M. MicroRNA-mediated silence of onco-lncRNA MALAT1 in different ESCC cells via ligand-functionalized hydroxyl-rich nanovectors. Nanoscale. 2017; 9:2521–30. https://doi.org/10.1039/c6nr09668a [PubMed]
-
17.
Zhou X, Liu S, Cai G, Kong L, Zhang T, Ren Y, Wu Y, Mei M, Zhang L, Wang X. Long non coding RNA MALAT1 promotes tumor growth and metastasis by inducing epithelial-mesenchymal transition in oral squamous cell carcinoma. Sci Rep. 2015; 5:15972. https://doi.org/10.1038/srep15972 [PubMed]
-
18.
Liu Z, Sun M, Lu K, Liu J, Zhang M, Wu W, De W, Wang Z, Wang R. The long noncoding RNA HOTAIR contributes to cisplatin resistance of human lung adenocarcinoma cells via downregualtion of p21(WAF1/CIP1) expression. PLoS One. 2013; 8:e77293. https://doi.org/10.1371/journal.pone.0077293 [PubMed]
-
19.
Eades G, Zhang YS, Li QL, Xia JX, Yao Y, Zhou Q. Long non-coding RNAs in stem cells and cancer. World J Clin Oncol. 2014; 5:134–41. https://doi.org/10.5306/wjco.v5.i2.134 [PubMed]
-
20.
Guo W, Dong Z, Bai Y, Guo Y, Shen S, Kuang G, Xu J. Associations between polymorphisms of HOTAIR and risk of gastric cardia adenocarcinoma in a population of north China. Tumour Biol. 2015; 36:2845–54. https://doi.org/10.1007/s13277-014-2912-y [PubMed]
-
21.
Bao Z, Yang Z, Huang Z, Zhou Y, Cui Q, Dong D. LncRNADisease 2.0: an updated database of long non-coding RNA-associated diseases. Nucleic Acids Res. 2019; 47:D1034–37. https://doi.org/10.1093/nar/gky905 [PubMed]
-
22.
Ma S, Huang J. Penalized feature selection and classification in bioinformatics. Brief Bioinform. 2008; 9:392–403. https://doi.org/10.1093/bib/bbn027 [PubMed]
-
23.
Li W, Ma S, Bai X, Pan W, Ai L, Tan W. Long noncoding RNA WDFY3-AS2 suppresses tumor progression by acting as a competing endogenous RNA of microRNA-18a in ovarian cancer. J Cell Physiol. 2020; 235:1141–54. https://doi.org/10.1002/jcp.29028 [PubMed]
-
24.
Zhang Q, Guan F, Fan T, Li S, Ma S, Zhang Y, Guo W, Liu H. LncRNA WDFY3-AS2 suppresses proliferation and invasion in oesophageal squamous cell carcinoma by regulating miR-2355-5p/SOCS2 axis. J Cell Mol Med. 2020; 24:8206–20. https://doi.org/10.1111/jcmm.15488 [PubMed]
-
25.
Ren P, Hong X, Chang L, Xing L, Zhang H. USF1-induced overexpression of long noncoding RNA WDFY3-AS2 promotes lung adenocarcinoma progression via targeting miR-491-5p/ZNF703 axis. Mol Carcinog. 2020; 59:875–85. https://doi.org/10.1002/mc.23181 [PubMed]
-
26.
Deva Magendhra Rao AK, Patel K, Korivi Jyothiraj S, Meenakumari B, Sundersingh S, Sridevi V, Rajkumar T, Pandey A, Chatterjee A, Gowda H, Mani S. Identification of lncRNAs associated with early-stage breast cancer and their prognostic implications. Mol Oncol. 2019; 13:1342–55. https://doi.org/10.1002/1878-0261.12489 [PubMed]
-
27.
Rodrigues de Bastos D, Nagai MA. In silico analyses identify lncRNAs: WDFY3-AS2, BDNF-AS and AFAP1-AS1 as potential prognostic factors for patients with triple-negative breast tumors. PLoS One. 2020; 15:e0232284. https://doi.org/10.1371/journal.pone.0232284 [PubMed]
-
28.
Zhang Z, Wang S, Liu W. EMT-related long non-coding RNA in hepatocellular carcinoma: a study with TCGA database. Biochem Biophys Res Commun. 2018; 503:1530–36. https://doi.org/10.1016/j.bbrc.2018.07.075 [PubMed]
-
29.
Jiang H, Shi X, Ye G, Xu Y, Xu J, Lu J, Lu W. Up-regulated long non-coding RNA DUXAP8 promotes cell growth through repressing krüppel-like factor 2 expression in human hepatocellular carcinoma. Onco Targets Ther. 2019; 12:7429–36. https://doi.org/10.2147/OTT.S214336 [PubMed]
-
30.
Yue C, Liang C, Li P, Yan L, Zhang D, Xu Y, Wei Z, Wu J. DUXAP8 a pan-cancer prognostic marker involved in the molecular regulatory mechanism in hepatocellular carcinoma: a comprehensive study based on data mining, bioinformatics, and in vitro validation. Onco Targets Ther. 2019; 12:11637–50. https://doi.org/10.2147/OTT.S231750 [PubMed]
-
31.
Wei F, Yang L, Jiang D, Pan M, Tang G, Huang M, Zhang J. Long noncoding RNA DUXAP8 contributes to the progression of hepatocellular carcinoma via regulating miR-422a/PDK2 axis. Cancer Med. 2020; 9:2480–90. https://doi.org/10.1002/cam4.2861 [PubMed]
-
32.
Du C, Wang HX, Chen P, Chen CH. STAT3-induced upregulation of lncRNA DUXAP8 functions as ceRNA for miR-577 to promote the migration and invasion in colorectal cancer through the regulation of RAB14. Eur Rev Med Pharmacol Sci. 2019; 23:6105–18. https://doi.org/10.26355/eurrev_201907_18424 [PubMed]
-
33.
Gong A, Huang Z, Ge H, Cai Y, Yang C. The carcinogenic complex lncRNA DUXAP8/EZH2/LSD1 accelerates the proliferation, migration and invasion of colorectal cancer. J BUON. 2019; 24:1830–36. [PubMed]
-
34.
Lin MG, Hong YK, Zhang Y, Lin BB, He XJ. Mechanism of lncRNA DUXAP8 in promoting proliferation of bladder cancer cells by regulating PTEN. Eur Rev Med Pharmacol Sci. 2018; 22:3370–77. https://doi.org/10.26355/eurrev_201806_15158 [PubMed]
-
35.
Jiang B, Hailong S, Yuan J, Zhao H, Xia W, Zha Z, Bin W, Liu Z. Identification of oncogenic long noncoding RNA SNHG12 and DUXAP8 in human bladder cancer through a comprehensive profiling analysis. Biomed Pharmacother. 2018; 108:500–07. https://doi.org/10.1016/j.biopha.2018.09.025 [PubMed]
-
36.
Lian Y, Yang J, Lian Y, Xiao C, Hu X, Xu H. DUXAP8, a pseudogene derived lncRNA, promotes growth of pancreatic carcinoma cells by epigenetically silencing CDKN1A and KLF2. Cancer Commun (Lond). 2018; 38:64. https://doi.org/10.1186/s40880-018-0333-9 [PubMed]
-
37.
Xu LJ, Yu XJ, Wei B, Hui HX, Sun Y, Dai J, Chen XF. Long non-coding RNA DUXAP8 regulates proliferation and invasion of esophageal squamous cell cancer. Eur Rev Med Pharmacol Sci. 2018; 22:2646–52. https://doi.org/10.26355/eurrev_201805_14959 [PubMed]
-
38.
Liu W, Zhang Y, Chen M, Shi L, Xu L, Zou X. A genome-wide analysis of long noncoding RNA profile identifies differentially expressed lncRNAs associated with esophageal cancer. Cancer Med. 2018; 7:4181–89. https://doi.org/10.1002/cam4.1536 [PubMed]
-
39.
Huang T, Wang X, Yang X, Ji J, Wang Q, Yue X, Dong Z. Long non-coding RNA DUXAP8 enhances renal cell carcinoma progression via downregulating miR-126. Med Sci Monit. 2018; 24:7340–47. https://doi.org/10.12659/MSM.910054 [PubMed]
-
40.
Chen J, Lou W, Ding B, Wang X. Overexpressed pseudogenes, DUXAP8 and DUXAP9, promote growth of renal cell carcinoma and serve as unfavorable prognostic biomarkers. Aging (Albany NY). 2019; 11:5666–88. https://doi.org/10.18632/aging.102152 [PubMed]
-
41.
Ji X, Tao R, Sun LY, Xu XL, Ling W. Down-regulation of long non-coding RNA DUXAP8 suppresses proliferation, metastasis and EMT by modulating miR-498 through TRIM44-mediated AKT/mTOR pathway in non-small-cell lung cancer. Eur Rev Med Pharmacol Sci. 2020; 24:3152–65. https://doi.org/10.26355/eurrev_202003_20682 [PubMed]
-
42.
Shi X, Cui Z, Liu X, Wu S, Wu Y, Fang F, Zhao H. LncRNA FIRRE is activated by MYC and promotes the development of diffuse large b-cell lymphoma via Wnt/β-catenin signaling pathway. Biochem Biophys Res Commun. 2019; 510:594–600. https://doi.org/10.1016/j.bbrc.2019.01.105 [PubMed]
-
43.
Li M, Zhao LM, Li SL, Li J, Gao B, Wang FF, Wang SP, Hu XH, Cao J, Wang GY. Differentially expressed lncRNAs and mRNAs identified by NGS analysis in colorectal cancer patients. Cancer Med. 2018; 7:4650–64. https://doi.org/10.1002/cam4.1696 [PubMed]
-
44.
Yang C, Shen S, Zheng X, Ye K, Sun Y, Lu Y, Ge H. Long noncoding RNA HAGLR acts as a microRNA-143-5p sponge to regulate epithelial-mesenchymal transition and metastatic potential in esophageal cancer by regulating LAMP3. FASEB J. 2019; 33:10490–504. https://doi.org/10.1096/fj.201802543RR [PubMed]
-
45.
Guo X, Chen Z, Zhao L, Cheng D, Song W, Zhang X. Long non-coding RNA-HAGLR suppressed tumor growth of lung adenocarcinoma through epigenetically silencing E2F1. Exp Cell Res. 2019; 382:111461. https://doi.org/10.1016/j.yexcr.2019.06.006 [PubMed]
-
46.
Sun W, Nie W, Wang Z, Zhang H, Li Y, Fang X. Lnc HAGLR promotes colon cancer progression through sponging miR-185-5p and activating CDK4 and CDK6 in vitro and in vivo. Onco Targets Ther. 2020; 13:5913–25. https://doi.org/10.2147/OTT.S246092 [PubMed]
-
47.
Yao R, Feng WT, Xu LJ, Zhong XM, Liu H, Sun Y, Zhou LL. DUXAP10 regulates proliferation and apoptosis of chronic myeloid leukemia via PTEN pathway. Eur Rev Med Pharmacol Sci. 2018; 22:4934–40. https://doi.org/10.26355/eurrev_201808_15632 [PubMed]
-
48.
Wang Z, Ren B, Huang J, Yin R, Jiang F, Zhang Q. LncRNA DUXAP10 modulates cell proliferation in esophageal squamous cell carcinoma through epigenetically silencing p21. Cancer Biol Ther. 2018; 19:998–1005. https://doi.org/10.1080/15384047.2018.1470723 [PubMed]
-
49.
Lv XY, Ma L, Chen JF, Yu R, Li Y, Yan ZJ, Cheng Y, Ma Q. Knockdown of DUXAP10 inhibits proliferation and promotes apoptosis in bladder cancer cells via PI3K/Akt/mTOR signaling pathway. Int J Oncol. 2018; 52:288–94. https://doi.org/10.3892/ijo.2017.4195 [PubMed]
-
50.
Zhuang X, Tong H, Ding Y, Wu L, Cai J, Si Y, Zhang H, Shen M. Long noncoding RNA ABHD11-AS1 functions as a competing endogenous RNA to regulate papillary thyroid cancer progression by miR-199a-5p/SLC1A5 axis. Cell Death Dis. 2019; 10:620. https://doi.org/10.1038/s41419-019-1850-4 [PubMed]
-
51.
Wen J, Wang H, Dong T, Gan P, Fang H, Wu S, Li J, Zhang Y, Du R, Zhu Q. STAT3-induced upregulation of lncRNA ABHD11-AS1 promotes tumour progression in papillary thyroid carcinoma by regulating miR-1301-3p/STAT3 axis and PI3K/AKT signalling pathway. Cell Prolif. 2019; 52:e12569. https://doi.org/10.1111/cpr.12569 [PubMed]
-
52.
Liu Y, Feng W, Liu W, Kong X, Li L, He J, Wang D, Zhang M, Zhou G, Xu W, Chen W, Gong A, Xu M. Circulating lncRNA ABHD11-AS1 serves as a biomarker for early pancreatic cancer diagnosis. J Cancer. 2019; 10:3746–56. https://doi.org/10.7150/jca.32052 [PubMed]
-
53.
Qiao X, Lv SX, Qiao Y, Li QP, Ye B, Wang CC, Miao L. Long noncoding RNA ABHD11-AS1 predicts the prognosis of pancreatic cancer patients and serves as a promoter by activating the PI3K-AKT pathway. Eur Rev Med Pharmacol Sci. 2018; 22:8630–39. https://doi.org/10.26355/eurrev_201812_16627 [PubMed]
-
54.
Liu Y, Wang LL, Chen S, Zong ZH, Guan X, Zhao Y. LncRNA ABHD11-AS1 promotes the development of endometrial carcinoma by targeting cyclin D1. J Cell Mol Med. 2018; 22:3955–64. https://doi.org/10.1111/jcmm.13675 [PubMed]
-
55.
Zeng XY, Jiang XY, Yong JH, Xie H, Yuan J, Zeng D, Dou YY, Xiao SS. lncRNA ABHD11-AS1, regulated by the EGFR pathway, contributes to the ovarian cancer tumorigenesis by epigenetically suppressing TIMP2. Cancer Med. 2019; 8:7074–85. https://doi.org/10.1002/cam4.2586 [PubMed]
-
56.
Lei X, Li L, Duan X. Long non-coding RNA ABHD11-AS1 promotes colorectal cancer development through regulation of miR-133a/SOX4 axis. Biosci Rep. 2018; 38:BSR20181386. https://doi.org/10.1042/BSR20181386 [PubMed]
-
57.
He D, Yue Z, Liu L, Fang X, Chen L, Han H. Long noncoding RNA ABHD11-AS1 promote cells proliferation and invasion of colorectal cancer via regulating the miR-1254-WNT11 pathway. J Cell Physiol. 2019; 234:12070–79. https://doi.org/10.1002/jcp.27877 [PubMed]
-
58.
Liu HY, Lu SR, Guo ZH, Zhang ZS, Ye X, Du Q, Li H, Wu Q, Yu B, Zhai Q, Liu JL. lncRNA SLC16A1-AS1 as a novel prognostic biomarker in non-small cell lung cancer. J Investig Med. 2020; 68:52–59. https://doi.org/10.1136/jim-2019-001080 [PubMed]
-
59.
Feng H, Zhang X, Lai W, Wang J. Long non-coding RNA SLC16A1-AS1: its multiple tumorigenesis features and regulatory role in cell cycle in oral squamous cell carcinoma. Cell Cycle. 2020; 19:1641–53. https://doi.org/10.1080/15384101.2020.1762048 [PubMed]
-
60.
Song M, Zhong A, Yang J, He J, Cheng S, Zeng J, Huang Y, Pan Q, Zhao J, Zhou Z, Zhu Q, Tang Y, Chen H, et al. Large-scale analyses identify a cluster of novel long noncoding RNAs as potential competitive endogenous RNAs in progression of hepatocellular carcinoma. Aging (Albany NY). 2019; 11:10422–53. https://doi.org/10.18632/aging.102468 [PubMed]
-
61.
Li C, Tan F, Pei Q, Zhou Z, Zhou Y, Zhang L, Wang D, Pei H. Non-coding RNA MFI2-AS1 promotes colorectal cancer cell proliferation, migration and invasion through miR-574-5p/MYCBP axis. Cell Prolif. 2019; 52:e12632. https://doi.org/10.1111/cpr.12632 [PubMed]
-
62.
Liu K, Liu J, Bo QF. MFI2-AS1 regulates the aggressive phenotypes in glioma by modulating MMP14 via a positive feedback loop. Eur Rev Med Pharmacol Sci. 2019; 23:5884–95. https://doi.org/10.26355/eurrev_201907_18333 [PubMed]
-
63.
Wei Y, Wang Z, Zong Y, Deng D, Chen P, Lu J. LncRNA MFI2-AS1 promotes HCC progression and metastasis by acting as a competing endogenous RNA of miR-134 to upregulate FOXM1 expression. Biomed Pharmacother. 2020; 125:109890. https://doi.org/10.1016/j.biopha.2020.109890 [PubMed]
-
64.
Yang K, Lu XF, Luo PC, Zhang J. Identification of six potentially long noncoding RNAs as biomarkers involved competitive endogenous RNA in clear cell renal cell carcinoma. Biomed Res Int. 2018; 2018:9303486. https://doi.org/10.1155/2018/9303486 [PubMed]
-
65.
Liu H, Zhang Z, Wu N, Guo H, Zhang H, Fan D, Nie Y, Liu Y. Integrative analysis of dysregulated lncRNA-associated ceRNA network reveals functional lncRNAs in gastric cancer. Genes (Basel). 2018; 9:303. https://doi.org/10.3390/genes9060303 [PubMed]
-
66.
Zhou Y, Shi H, Du Y, Zhao G, Wang X, Li Q, Liu J, Ye L, Shen Z, Guo Y, Huang Y. lncRNA DLEU2 modulates cell proliferation and invasion of non-small cell lung cancer by regulating miR-30c-5p/SOX9 axis. Aging (Albany NY). 2019; 11:7386–401. https://doi.org/10.18632/aging.102226 [PubMed]
-
67.
Wu W, Zhao Y, Gao E, Li Y, Guo X, Zhao T, He W, Zhang H. LncRNA DLEU2 accelerates the tumorigenesis and invasion of non-small cell lung cancer by sponging miR-30a-5p. J Cell Mol Med. 2020; 24:441–50. https://doi.org/10.1111/jcmm.14749 [PubMed]
-
68.
Xu B, Gong X, Zi L, Li G, Dong S, Chen X, Li Y. Silencing of DLEU2 suppresses pancreatic cancer cell proliferation and invasion by upregulating microRNA-455. Cancer Sci. 2019; 110:1676–85. https://doi.org/10.1111/cas.13987 [PubMed]
-
69.
Guo Y, Bai M, Lin L, Huang J, An Y, Liang L, Liu Y, Huang W. LncRNA DLEU2 aggravates the progression of hepatocellular carcinoma through binding to EZH2. Biomed Pharmacother. 2019; 118:109272. https://doi.org/10.1016/j.biopha.2019.109272 [PubMed]
-
70.
Lu T, Wang R, Cai H, Cui Y. Long non-coding RNA DLEU2 promotes the progression of esophageal cancer through miR-30e-5p/E2F7 axis. Biomed Pharmacother. 2020; 123:109650. https://doi.org/10.1016/j.biopha.2019.109650 [PubMed]
-
71.
Wan J, Jiang S, Jiang Y, Ma W, Wang X, He Z, Wang X, Cui R. Data Mining and Expression Analysis of Differential lncRNA ADAMTS9-AS1 in Prostate Cancer. Front Genet. 2020; 10:1377. https://doi.org/10.3389/fgene.2019.01377 [PubMed]
-
72.
Xing Y, Zhao Z, Zhu Y, Zhao L, Zhu A, Piao D. Comprehensive analysis of differential expression profiles of mRNAs and lncRNAs and identification of a 14-lncRNA prognostic signature for patients with colon adenocarcinoma. Oncol Rep. 2018; 39:2365–75. https://doi.org/10.3892/or.2018.6324 [PubMed]
-
73.
Wang A, Jin C, Li H, Qin Q, Li L. LncRNA ADAMTS9-AS2 regulates ovarian cancer progression by targeting miR-182-5p/FOXF2 signaling pathway. Int J Biol Macromol. 2018; 120:1705–13. https://doi.org/10.1016/j.ijbiomac.2018.09.179 [PubMed]
-
74.
Cao B, Liu C, Yang G. Down-regulation of lncRNA ADAMTS9-AS2 contributes to gastric cancer development via activation of PI3K/Akt pathway. Biomed Pharmacother. 2018; 107:185–93. https://doi.org/10.1016/j.biopha.2018.06.146 [PubMed]
-
75.
Pan H, Guo C, Pan J, Guo D, Song S, Zhou Y, Xu D. Construction of a competitive endogenous RNA network and identification of potential regulatory axis in gastric cancer. Front Oncol. 2019; 9:912. https://doi.org/10.3389/fonc.2019.00912 [PubMed]
-
76.
Ren N, Jiang T, Wang C, Xie S, Xing Y, Piao D, Zhang T, Zhu Y. LncRNA ADAMTS9-AS2 inhibits gastric cancer (GC) development and sensitizes chemoresistant GC cells to cisplatin by regulating miR-223-3p/NLRP3 axis. Aging (Albany NY). 2020; 12:11025–41. https://doi.org/10.18632/aging.103314 [PubMed]
-
77.
Yan Y, Xu Z, Chen X, Wang X, Zeng S, Zhao Z, Qian L, Li Z, Wei J, Huo L, Li X, Gong Z, Sun L. Novel function of lncRNA ADAMTS9-AS2 in promoting temozolomide resistance in glioblastoma via upregulating the FUS/MDM2 ubiquitination axis. Front Cell Dev Biol. 2019; 7:217. https://doi.org/10.3389/fcell.2019.00217 [PubMed]
-
78.
Shi YF, Lu H, Wang HB. Downregulated lncRNA ADAMTS9-AS2 in breast cancer enhances tamoxifen resistance by activating microRNA-130a-5p. Eur Rev Med Pharmacol Sci. 2019; 23:1563–73. https://doi.org/10.26355/eurrev_201902_17115 [PubMed]
-
79.
Li Y, Wan Q, Wang W, Mai L, Sha L, Mashrah M, Lin Z, Pan C. LncRNA ADAMTS9-AS2 promotes tongue squamous cell carcinoma proliferation, migration and EMT via the miR-600/EZH2 axis. Biomed Pharmacother. 2019; 112:108719. https://doi.org/10.1016/j.biopha.2019.108719 [PubMed]
-
80.
Liu D, Wu K, Yang Y, Zhu D, Zhang C, Zhao S. Long noncoding RNA ADAMTS9-AS2 suppresses the progression of esophageal cancer by mediating CDH3 promoter methylation. Mol Carcinog. 2020; 59:32–44. https://doi.org/10.1002/mc.23126 [PubMed]
-
81.
Yu X, Pang L, Yang T, Liu P. lncRNA LINC01296 regulates the proliferation, metastasis and cell cycle of osteosarcoma through cyclin D1. Oncol Rep. 2018; 40:2507–14. https://doi.org/10.3892/or.2018.6674 [PubMed]
-
82.
Wang J, Wang Z, Yao W, Dong K, Zheng S, Li K. The association between lncRNA LINC01296 and the clinical characteristics in neuroblastoma. J Pediatr Surg. 2019; 54:2589–94. https://doi.org/10.1016/j.jpedsurg.2019.08.032 [PubMed]
-
83.
Wang L, Meng D, Wang Y, Hu J. Long non-coding RNA LINC01296 promotes esophageal squamous cell carcinoma cell proliferation and invasion by epigenetic suppression of KLF2. Am J Cancer Res. 2018; 8:2020–29. [PubMed]
-
84.
Wang B, Liang T, Li J. Long noncoding RNA LINC01296 is associated with poor prognosis in ESCC and promotes ESCC cell proliferation, migration and invasion. Eur Rev Med Pharmacol Sci. 2018; 22:4524–31. https://doi.org/10.26355/eurrev_201807_15507 [PubMed]
-
85.
Hu X, Duan L, Liu H, Zhang L. Long noncoding RNA LINC01296 induces non-small cell lung cancer growth and progression through sponging miR-5095. Am J Transl Res. 2019; 11:895–903. [PubMed]
-
86.
Wang ZC, Yang S, Chen MQ, Wu SS, Lv HH, Jin WX. LINC01296 promotes the proliferation and invasion by regulating microRNA-760 expression and predicts poor prognosis of hepatocellular carcinoma. Eur Rev Med Pharmacol Sci. 2019; 23:9848–56. https://doi.org/10.26355/eurrev_201911_19548 [PubMed]
-
87.
Jiang M, Xiao Y, Liu D, Luo N, Gao Q, Guan Y. Overexpression of long noncoding RNA LINC01296 indicates an unfavorable prognosis and promotes tumorigenesis in breast cancer. Gene. 2018; 675:217–24. https://doi.org/10.1016/j.gene.2018.07.004 [PubMed]
-
88.
Xu H, Mao HL, Zhao XR, Li Y, Liu PS. MiR-29c-3p, a target miRNA of LINC01296, accelerates tumor Malignancy: therapeutic potential of a LINC01296/miR-29c-3p axis in ovarian cancer. J Ovarian Res. 2020; 13:31. https://doi.org/10.1186/s13048-020-00631-w [PubMed]
-
89.
Zhao H, Zheng GH, Li GC, Xin L, Wang YS, Chen Y, Zheng XM. Long noncoding RNA LINC00958 regulates cell sensitivity to radiotherapy through RRM2 by binding to microRNA-5095 in cervical cancer. J Cell Physiol. 2019; 234:23349–59. https://doi.org/10.1002/jcp.28902 [PubMed]
-
90.
Wang L, Zhong Y, Yang B, Zhu Y, Zhu X, Xia Z, Xu J, Xu L. LINC00958 facilitates cervical cancer cell proliferation and metastasis by sponging miR-625-5p to upregulate LRRC8E expression. J Cell Biochem. 2020; 121:2500–09. https://doi.org/10.1002/jcb.29472 [PubMed]
-
91.
Chen F, Liu M, Yu Y, Sun Y, Li J, Hu W, Wang X, Tong D. LINC00958 regulated miR-627-5p/YBX2 axis to facilitate cell proliferation and migration in oral squamous cell carcinoma. Cancer Biol Ther. 2019; 20:1270–80. https://doi.org/10.1080/15384047.2019.1617571 [PubMed]
-
92.
Wang Z, Zhu X, Dong P, Cai J. Long noncoding RNA LINC00958 promotes the oral squamous cell carcinoma by sponging miR-185-5p/YWHAZ. Life Sci. 2020; 242:116782. https://doi.org/10.1016/j.lfs.2019.116782 [PubMed]
-
93.
Luo Z, Han Z, Shou F, Li Y, Chen Y. LINC00958 accelerates cell proliferation and migration in non-small cell lung cancer through JNK/c-JUN signaling. Hum Gene Ther Methods. 2019; 30:226–34. https://doi.org/10.1089/hgtb.2019.115 [PubMed]
-
94.
Chen S, Chen JZ, Zhang JQ, Chen HX, Qiu FN, Yan ML, Tian YF, Peng CH, Shen BY, Chen YL, Wang YD. Silencing of long noncoding RNA LINC00958 prevents tumor initiation of pancreatic cancer by acting as a sponge of microRNA-330-5p to down-regulate PAX8. Cancer Lett. 2019; 446:49–61. https://doi.org/10.1016/j.canlet.2018.12.017 [PubMed]
-
95.
Chen M, Xu Z, Zhang Y, Zhang X. LINC00958 promotes the Malignancy of nasopharyngeal carcinoma by sponging microRNA-625 and thus upregulating NUAK1. Onco Targets Ther. 2019; 12:9277–90. https://doi.org/10.2147/OTT.S216342 [PubMed]
-
96.
Huang S, Zhan Z, Li L, Guo H, Yao Y, Feng M, Deng J, Xiong J. LINC00958-MYC positive feedback loop modulates resistance of head and neck squamous cell carcinoma cells to chemo- and radiotherapy in vitro. Onco Targets Ther. 2019; 12:5989–6000. https://doi.org/10.2147/OTT.S208318 [PubMed]
-
97.
Zuo X, Chen Z, Gao W, Zhang Y, Wang J, Wang J, Cao M, Cai J, Wu J, Wang X. M6A-mediated upregulation of LINC00958 increases lipogenesis and acts as a nanotherapeutic target in hepatocellular carcinoma. J Hematol Oncol. 2020; 13:5. https://doi.org/10.1186/s13045-019-0839-x [PubMed]
-
98.
Wang W, Song ZJ, Wang Y, Zhong WF, Kang P, Yang Y. Elevated long non-coding RNA LINC00958 was associated with metastasis and unfavorable prognosis in gastric cancer. Eur Rev Med Pharmacol Sci. 2019; 23:598–603. https://doi.org/10.26355/eurrev_201901_16872 [PubMed]
-
99.
Liu J, Zhu J, Xiao Z, Wang X, Luo J. BBOX1-AS1 contributes to colorectal cancer progression by sponging hsa-miR-361-3p and targeting SH2B1. FEBS Open Bio. 2020. [Epub ahead of print]. https://doi.org/10.1002/2211-5463.12802 [PubMed]
-
100.
Xu J, Yang B, Wang L, Zhu Y, Zhu X, Xia Z, Zhao Z, Xu L. LncRNA BBOX1-AS1 upregulates HOXC6 expression through miR-361-3p and HuR to drive cervical cancer progression. Cell Prolif. 2020; 53:e12823. https://doi.org/10.1111/cpr.12823 [PubMed]
-
101.
Guan YX, Zhang MZ, Chen XZ, Zhang Q, Liu SZ, Zhang YL. Lnc RNA SNHG20 participated in proliferation, invasion, and migration of breast cancer cells via miR-495. J Cell Biochem. 2018; 119:7971–81. https://doi.org/10.1002/jcb.26588 [PubMed]
-
102.
Guo H, Yang S, Li S, Yan M, Li L, Zhang H. LncRNA SNHG20 promotes cell proliferation and invasion via miR-140-5p-ADAM10 axis in cervical cancer. Biomed Pharmacother. 2018; 102:749–57. https://doi.org/10.1016/j.biopha.2018.03.024 [PubMed]
-
103.
Zhao Q, Gao S, Du Q, Liu Y. Long non-coding RNA SNHG20 promotes bladder cancer via activating the Wnt/β-catenin signalling pathway. Int J Mol Med. 2018; 42:2839–48. https://doi.org/10.3892/ijmm.2018.3819 [PubMed]
-
104.
Wu X, Xiao Y, Zhou Y, Zhou Z, Yan W. lncRNA SNHG20 promotes prostate cancer migration and invasion via targeting the miR-6516-5p/SCGB2A1 axis. Am J Transl Res. 2019; 11:5162–69. [PubMed]
-
105.
Zhang C, Jiang F, Su C, Xie P, Xu L. Upregulation of long noncoding RNA SNHG20 promotes cell growth and metastasis in esophageal squamous cell carcinoma via modulating ATM-JAK-PD-L1 pathway. J Cell Biochem. 2019. [Epub ahead of print]. https://doi.org/10.1002/jcb.28444 [PubMed]
-
106.
Gao P, Fan R, Ge T. SNHG20 serves as a predictor for prognosis and promotes cell growth in oral squamous cell carcinoma. Oncol Lett. 2019; 17:951–57. https://doi.org/10.3892/ol.2018.9709 [PubMed]
-
107.
Wu J, Zhao W, Wang Z, Xiang X, Zhang S, Liu L. Long non-coding RNA SNHG20 promotes the tumorigenesis of oral squamous cell carcinoma via targeting miR-197/LIN28 axis. J Cell Mol Med. 2019; 23:680–88. https://doi.org/10.1111/jcmm.13987 [PubMed]
-
108.
Sun C, Sun Y, Zhang E. Long non-coding RNA SNHG20 promotes nasopharyngeal carcinoma cell migration and invasion by upregulating TGF-β1. Exp Ther Med. 2018; 16:4967–74. https://doi.org/10.3892/etm.2018.6849 [PubMed]
-
109.
Song EL, Xing L, Wang L, Song WT, Li DB, Wang Y, Gu YW, Liu MM, Ni WJ, Zhang P, Ma X, Zhang X, Yao J, et al. LncRNA ADAMTS9-AS2 inhibits cell proliferation and decreases chemoresistance in clear cell renal cell carcinoma via the miR-27a-3p/FOXO1 axis. Aging (Albany NY). 2019; 11:5705–25. https://doi.org/10.18632/aging.102154 [PubMed]
-
110.
Acha-Sagredo A, Uko B, Pantazi P, Bediaga NG, Moschandrea C, Rainbow L, Marcus MW, Davies MP, Field JK, Liloglou T. Long non-coding RNA dysregulation is a frequent event in non-small cell lung carcinoma pathogenesis. Br J Cancer. 2020; 122:1050–58. https://doi.org/10.1038/s41416-020-0742-9 [PubMed]
-
111.
Xu Y, Li Y, Jin J, Han G, Sun C, Pizzi MP, Huo L, Scott A, Wang Y, Ma L, Lee JH, Bhutani MS, Weston B, et al. LncRNA PVT1 up-regulation is a poor prognosticator and serves as a therapeutic target in esophageal adenocarcinoma. Mol Cancer. 2019; 18:141. https://doi.org/10.1186/s12943-019-1064-5 [PubMed]
-
112.
Tan J, Yang L. Long noncoding RNA VPS9D1-AS1 overexpression predicts a poor prognosis in non-small cell lung cancer. Biomed Pharmacother. 2018; 106:1600–06. https://doi.org/10.1016/j.biopha.2018.07.113 [PubMed]
-
113.
Han X, Huang T, Han J. Long noncoding RNA VPS9D1-AS1 augments the Malignant phenotype of non-small cell lung cancer by sponging microRNA-532-3p and thereby enhancing HMGA2 expression. Aging (Albany NY). 2020; 12:370–86. https://doi.org/10.18632/aging.102628 [PubMed]
-
114.
Wang J, Yang X, Li R, Wang L, Gu Y, Zhao Y, Huang KH, Cheng T, Yuan Y, Gao S. Long non-coding RNA MYU promotes prostate cancer proliferation by mediating the miR-184/c-Myc axis. Oncol Rep. 2018; 40:2814–25. https://doi.org/10.3892/or.2018.6661 [PubMed]
-
115.
Wang X, Chen Q, Wang X, Li W, Yu G, Zhu Z, Zhang W. ZEB1 activated-VPS9D1-AS1 promotes the tumorigenesis and progression of prostate cancer by sponging miR-4739 to upregulate MEF2D. Biomed Pharmacother. 2020; 122:109557. https://doi.org/10.1016/j.biopha.2019.109557 [PubMed]
-
116.
Qin X, Ma D, Tan YX, Wang HY, Cai Z. The role of necroptosis in cancer: a double-edged sword? Biochim Biophys Acta Rev Cancer. 2019; 1871:259–66. https://doi.org/10.1016/j.bbcan.2019.01.006 [PubMed]
-
117.
Gao Y, Wang F, Zhang L, Kang M, Zhu L, Xu L, Liang W, Zhang W. LINC00311 promotes cancer stem-like properties by targeting miR-330-5p/TLR4 pathway in human papillary thyroid cancer. Cancer Med. 2020; 9:1515–28. https://doi.org/10.1002/cam4.2815 [PubMed]
-
118.
Yan SP, Chu DX, Qiu HF, Xie Y, Wang CF, Zhang JY, Li WC, Guo RX. LncRNA LINC01305 silencing inhibits cell epithelial-mesenchymal transition in cervical cancer by inhibiting TNXB-mediated PI3K/Akt signalling pathway. J Cell Mol Med. 2019; 23:2656–66. https://doi.org/10.1111/jcmm.14161 [PubMed]
-
119.
Yan F, Liu SW, Li XY, Li CC, Wu Y. Silencing LncRNA LINC01305 inhibits epithelial mesenchymal transition in lung cancer cells by regulating TNXB-mediated PI3K/Akt signaling pathway. J Biol Regul Homeost Agents. 2020; 34:499–508. https://doi.org/10.23812/20-73-A-33 [PubMed]
-
120.
Zhang Y, Yang H, Du Y, Liu P, Zhang J, Li Y, Shen H, Xing L, Xue X, Chen J, Zhang X. Long noncoding RNA TP53TG1 promotes pancreatic ductal adenocarcinoma development by acting as a molecular sponge of microRNA-96. Cancer Sci. 2019; 110:2760–72. https://doi.org/10.1111/cas.14136 [PubMed]
-
121.
Safran M, Dalah I, Alexander J, Rosen N, Iny Stein T, Shmoish M, Nativ N, Bahir I, Doniger T, Krug H, Sirota-Madi A, Olender T, Golan Y, et al. GeneCards version 3: the human gene integrator. Database (Oxford). 2010; 2010:baq020. https://doi.org/10.1093/database/baq020 [PubMed]
-
122.
Tian S, Wang C, Zhang J, Yu D. The cox-filter method identifies respective subtype-specific lncRNA prognostic signatures for two human cancers. BMC Med Genomics. 2020; 13:18. https://doi.org/10.1186/s12920-020-0691-4 [PubMed]
-
123.
Tian S, Wang C, An MW. Test on existence of histology subtype-specific prognostic signatures among early stage lung adenocarcinoma and squamous cell carcinoma patients using a cox-model based filter. Biol Direct. 2015; 10:15. https://doi.org/10.1186/s13062-015-0051-z [PubMed]
-
124.
Li PD, Hu JL, Ma C, Ma H, Yao J, Chen LL, Chen J, Cheng TT, Yang KY, Wu G, Zhang WJ, Cao RB. Upregulation of the long non-coding RNA PVT1 promotes esophageal squamous cell carcinoma progression by acting as a molecular sponge of miR-203 and LASP1. Oncotarget. 2017; 8:34164–76. https://doi.org/10.18632/oncotarget.15878 [PubMed]
-
125.
Zhao W, Ma X, Liu L, Chen Q, Liu Z, Zhang Z, Ma S, Wang Z, Li H, Wang Z, Wu J. SNHG20: a vital lncRNA in multiple human cancers. J Cell Physiol. 2019. [Epub ahead of print]. https://doi.org/10.1002/jcp.28143 [PubMed]
-
126.
Tang J, Yu B, Li Y, Zhang W, Alvarez AA, Hu B, Cheng SY, Feng H. TGF-β-activated lncRNA LINC00115 is a critical regulator of glioma stem-like cell tumorigenicity. EMBO Rep. 2019; 20:e48170. https://doi.org/10.15252/embr.201948170 [PubMed]
-
127.
Yuan C, Luo X, Duan S, Guo L. Long noncoding RNA LINC00115 promotes breast cancer metastasis by inhibiting miR-7. FEBS Open Bio. 2020; 10:1230–37. https://doi.org/10.1002/2211-5463.12842 [PubMed]
-
128.
Gong S, Xu M, Zhang Y, Shan Y, Zhang H. The prognostic signature and potential target genes of six long non-coding RNA in laryngeal squamous cell carcinoma. Front Genet. 2020; 11:413. https://doi.org/10.3389/fgene.2020.00413 [PubMed]
-
129.
Stone JK, Kim JH, Vukadin L, Richard A, Giannini HK, Lim SS, Tan M, Ahn EE. Hypoxia induces cancer cell-specific chromatin interactions and increases MALAT1 expression in breast cancer cells. J Biol Chem. 2019; 294:11213–24. https://doi.org/10.1074/jbc.RA118.006889 [PubMed]
-
130.
Gordon MA, Babbs B, Cochrane DR, Bitler BG, Richer JK. The long non-coding RNA MALAT1 promotes ovarian cancer progression by regulating RBFOX2-mediated alternative splicing. Mol Carcinog. 2019; 58:196–205. https://doi.org/10.1002/mc.22919 [PubMed]
-
131.
Voce DJ, Bernal GM, Wu L, Crawley CD, Zhang W, Mansour NM, Cahill KE, Szymura SJ, Uppal A, Raleigh DR, Spretz R, Nunez L, Larsen G, et al. Temozolomide treatment induces lncRNA MALAT1 in an NF-κB and p53 codependent manner in glioblastoma. Cancer Res. 2019; 79:2536–48. https://doi.org/10.1158/0008-5472.CAN-18-2170 [PubMed]
-
132.
Lu C, Ma J, Cai D. Increased HAGLR expression promotes non-small cell lung cancer proliferation and invasion via enhanced de novo lipogenesis. Tumour Biol. 2017; 39:1010428317697574. https://doi.org/10.1177/1010428317697574 [PubMed]
-
133.
Hu YC, Wang AM, Lu JK, Cen R, Liu LL. Long noncoding RNA HOXD-AS1 regulates proliferation of cervical cancer cells by activating Ras/ERK signaling pathway. Eur Rev Med Pharmacol Sci. 2017; 21:5049–55. https://doi.org/10.26355/eurrev_201711_13817 [PubMed]
-
134.
Li J, Han L, Roebuck P, Diao L, Liu L, Yuan Y, Weinstein JN, Liang H. TANRIC: an interactive open platform to explore the function of lncRNAs in cancer. Cancer Res. 2015; 75:3728–37. https://doi.org/10.1158/0008-5472.CAN-15-0273 [PubMed]
-
135.
Ogata H, Goto S, Sato K, Fujibuchi W, Bono H, Kanehisa M. KEGG: kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 1999; 27:29–34. https://doi.org/10.1093/nar/27.1.29 [PubMed]
-
136.
Ashburner M, Ball CA, Blake JA, Botstein D, Butler H, Cherry JM, Davis AP, Dolinski K, Dwight SS, Eppig JT, Harris MA, Hill DP, Issel-Tarver L, et al. Gene ontology: tool for the unification of biology. The gene ontology consortium. Nat Genet. 2000; 25:25–29. https://doi.org/10.1038/75556 [PubMed]
-
137.
Franceschini A, Szklarczyk D, Frankild S, Kuhn M, Simonovic M, Roth A, Lin J, Minguez P, Bork P, von Mering C, Jensen LJ. STRING v9.1: protein-protein interaction networks, with increased coverage and integration. Nucleic Acids Res. 2013; 41:D808–15. https://doi.org/10.1093/nar/gks1094 [PubMed]
-
138.
Friedman J, Hastie T, Tibshirani R. Regularization paths for generalized linear models via coordinate descent. J Stat Softw. 2010; 33:1–22. https://doi.org/10.18637/jss.v033.i01 [PubMed]