Introduction
According to Global Cancer Statistics, lung cancer has high incidence and mortality [1]. Most lung cancer patients are diagnosed at an advanced stage [2]. The reason is that most lung cancer patients have no obvious specific symptoms at the beginning of the disease, and many patients are at an advanced stage when they are definitely diagnosed [3]. Statistics show that the prognosis of lung cancer is closely associated with the clinical stage, which indicates that the early diagnosis can directly improve the prognosis of the patient [4]. Therefore, the selection of effective diagnosis and screening methods is quite crucial to improve the early diagnosis rate and prognosis of lung cancer.
Imaging examination and screening for lung cancer
Chest x-ray
It is not recommended to use chest x-ray to detect lung cancer at present, owing to the difficulty of displaying intrapulmonary lesions and small lesions. Furthermore, whether combined with sputum cytology examination or not, chest x-ray screening cannot reduce lung cancer disease-specific mortality [5, 6].
Low-dose CT (LDCT)
LDCT is recommended as an early screening method for high-risk groups [7]. The National Lung Screening Trial (NLST) [8] recommended LDCT scan during lung cancer screening. The results illustrated that compared with chest x-ray, LDCT screening could reduce mortality by 20.0%. Furthermore, Agency for Healthcare Research and Quality (AHRQ) summarized 8,149 papers published from 2000 to the last quarter of 2012, which supported lung cancer early screening. Based on these findings, USPSTF, the American Cancer Society (ACS) and many other medical organizations recommended that LDCT screening should be considered first for patients who meet the NLST inclusion criteria [9–11]. In addition, the NCCN guidelines also suggested that LDCT was used to screen high-risk groups, but it was not recommended for low and moderate-risk groups [11, 12].
The advantages of LDCT include low radiation dose, fast scanning speed, and sensitivity comparable to CT. However, it not only shows a false positive rate (FPR) of 23.3% in NLST, which is similar to the results of other studies [8, 13, 14], but also has several disadvantages such as over-diagnosis, radiation exposure and high cost [7, 15, 16]. Therefore, the application of LDCT for early screening of lung cancer still has many problems to be overcome. For example, further clarify the screening time interval to balance the benefits and potential harm; accurately classify the high-risk population of lung cancer to avoid over-diagnosis and treatment; and what kind of model should be established to evaluate which types of lung imaging features are prone to develop into lung cancer. These are still urgent problems to be solved in the future.
Positron emission tomography/CT (PET/CT)
PET/CT is an integrated fusion of PET and CT equipment and imaging, which can display accurate anatomical images and tissue metabolic function images. It has been widely used to identify the property and stage of tumors. Wang et al. [17] integrated 1330 patients with lung-occupying lesions in four clinical studies and found that PET/CT has higher sensitivity (98.7%) and higher specificity (58.2%) in distinguishing benign or malignant lung lesions. Yet, PET/CT also has a high FPR in lung cancer screening, and reducing FPR is still a dilemma in the early diagnosis of lung cancer through PET/CT [18]. In contrast with CT, PET/CT can enhance the accuracy of diagnosis of solitary pulmonary nodule (SPN) [19, 20]. The standard uptake value (SUV) of PET/CT reflects the metabolism and malignancy of diseased tissues under certain circumstances [21]. However, PET/CT also has its shortcomings. For instance, there are respiratory motion artifacts. FDG metabolism is not unique to tumors and the higher cost limits the application.
Bronchoscopy and diagnosis for lung cancer
Nowadays, pathological diagnosis has been regarded as gold standard for diagnosing cancer. There are several methods for obtaining histological specimens, including bronchoscopy, ultrasound or CT-guided percutaneous lung biopsy. Among them, bronchoscopy has been developed rapidly and widely recognized in recent years. It not only expands the field of vision for diagnosis, but also improves the efficiency of diagnosis.
White light bronchoscopy (WLB)
It is mainly used for early detection and diagnosis of central lung cancer, and the diagnosis rate can reach more than 95% in detecting high-grade dysplasia or worse. However, for some mucosal, submucosal early lesions and preneoplastic lesions, the diagnosis rate is very low (<30%) [22, 23].
Autofluorescence bronchoscopy (AFB)
The operating principle of AFB is that different spectrum emerge in normal tissues, dysplasia and carcinoma in situ [24]. As an important means for early detection of bronchial premalignant lesions, some studies have proposed that compared with sputum cytology, the sensitivity of AFB to detect hyperplasia and metaplasia was higher. Regardless of the results of sputum cytology, AFB can be recommended to high-risk patients. Moreover, when combined with spiral computed tomography (SCT), sputum examination or WLB, AFB can obviously enhance the diagnosis rate of premalignant lesions and carcinoma in situ [22, 25, 26]. Nevertheless, due to the high cost of equipment and inspection, it has not yet been widely accepted.
Narrow band imaging (NBI)
NBI is an imaging technique that can visualize vascular morphology and mucosal structure. A meta-analysis indicated that NBI has higher sensitivity (80%), specificity (84%) and diagnostic odds ratio (DOR 31.49%) than AFB in detecting premalignant airway lesions [27]. Another meta-analysis showed that NBI was also superior to WLB in detecting early and invasive lung cancer [28]. Besides, a prospective study suggested that dotted blood vessel visual pattern highly supported adenocarcinoma histology of lung cancer and tortuous vessels favored squamous cell cancer [29]. Therefore, when examining lung-occupying lesions with NBI, not only can the diagnosis rate be improved, but also the pathological type can be evaluated initially. So in the detection of early lung cancer, NBI can be used as an effective method.
Endo-bronchial ultrasound (EBUS)
EBUS combined with a special aspiration biopsy needle can be used for real-time ultrasound guided transbronchial needle aspiration biopsy, namely EBUS-TNAB [30]. In a randomized controlled trial [31], when EBUS-TBNA was used as the initial examination method after CT scanning in patients with suspected lung cancer, it could provide accurate diagnosis and lymph node staging. Compared with traditional diagnosis strategies, EBUS-TNAB can shorten the time for treatment decisions and may improve the survival of lung cancer patients without increasing costs.
Due to tumor heterogeneity, tissue biopsy just represents the state of the tumor at a certain moment and only a part of the malignant tumor, which cannot reflect the dynamic process of disease development [32]. Moreover, multiple biopsies are very invasive to patients, so new technologies need to be designed to improve this dilemma. Liquid biopsy can replace and complement this method [33, 34] (Figure 1).
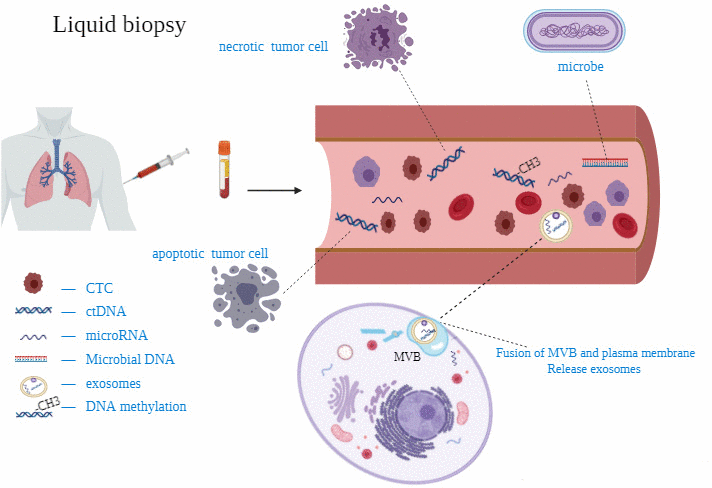
Figure 1. Some studies on circulating miRNAs for diagnosis of lung cancer.
Liquid biopsy and screening for lung cancer
Circulating tumor cells (CTCs)
CTCs is released into the peripheral blood from a primary cancer or metastasis [33]. The capture of CTCs is of great significance for the early detection, diagnosis, prognosis and monitoring of cancer, as well as understanding the basic biology of the metastatic process [35]. The content of CTC is extremely low (1-10 CTCs/1ml peripheral blood), which makes CTC separation and enrichment technology difficult. The CellSearch system was approved as the first and only method by FDA for CTC detection [36]. The CellSearch CTCs detection kit contains ferrofluid-based capture reagents and fluorescent immunoreagents. The ferrofluid-based reagent is a particle with a magnetic core, the surface of which is coated with antibodies that recognize EpCAM antigen. EpCAM is a CTC-specific antigen. Therefore, the magnetic particles can capture CTC. After immunocapture and enrichment, fluorescent reagents are used to identify CTCs and CTCs counts. EpCAM (+) CK (+) DAPI (+) CD45 (-) cells are defined as CTCs. In addition, there are other common ways to detect CTCs (Table 1).
Table 1. Comparison of common CTCs isolation and enrichment techniques.
| Methods | Representative examples | Advantages | Disadvantages |
| Coated anti-EpCAM antibody | Cellsearch (FDA approved) | High repeatability and specificity | Due to EMT, epithelial specific antigens were lost, resulting in loss of capture efficiency |
| Size-Based selection | Isolation by size of epithelial tumor cells (ISET) [37] | Don’t rely on EpCAM antigen on the surface of CTCs | Don’t detect CTCs with a very small size |
| Density-Based selection | Oncoquick [38] | Preserve cell viability | Low sensitivity and unstable, Missing rare CTCs |
| Microfluidics | CTC-CHIP [35] | High sensitivity and high detection rate | Lack of clinical verification |
| immunomagnetic | Magnetic Activated Cell Sorting (MACS) [39] | Preserve cell viability and easy to count | Adsorption of non-specific cells containing target markers leads to decreased sensitivity |
Lou et al. [40] and Zhang et al. [41] suggested that CTCs could be detected in the lung cancer, especially stage I to IIIA. He et al. [42] evaluated that the CTC detection rate (CTC>1) of stage I and stage II lung cancer was as high as 62.5% by using CellCollector and there were 71.6% similarities between CTCs and tumor tissues in gene mutation site of TP53, FGFR1, HER2, PDGFRA and CFS1R. It is the first time to reveal the molecular and genetic variation characteristics of early lung cancer CTC, and provides new data support for the clinical application of CTC as a molecular diagnosis of early lung cancer.
In summary, as a sign of early diagnosis, CTCs also have certain limitations. Reasonable and effective enrichment methods are the most important and urgent problems which need to be overcome. The main challenge is to obtain a sufficient number of CTCs in optimal conditions for further evaluation. The technology for assessing the molecular characteristics of CTCs is still evolving and how to standardize them for clinical application need to be studied.
Circulating tumor DNA (ctDNA)
The DNA, released by necrotic or apoptotic cells into the blood, is called circulating free DNA (cfDNA). In the blood of tumor patients, part of the cfDNA from dead tumor cells is defined as ctDNA [43]. Normally, ctDNA accounts for only 0.01-1% of cfDNA and is released from multiple tumor regions, which can overcome tumor heterogeneity [44]. It is reported that plasma is a good source of ctDNA [45]. The ctDNA can be quantitatively detected by the technology based on the four main components of bead, emulsion, amplification and magnetic, while the CAPP-seq technology for personalized cancer analysis through deep sequencing has changed the diagnostic mode of ctDNA [46, 47].
So far, ctDNA is the best material to obtain the diagnosis, prognosis and predictive information of tumor-related changes in the blood of cancer patients [48].The study by Szpechcinski et al. [49] showed that the level of plasma ctDNA in patients with non-small cell lung cancer (NSCLC) were not only higher than that in healthy people, but also higher than those with chronic respiratory inflammatory diseases. Liang et al. [50] combined ctDNA and DNA methylation to distinguish lung cancer and benign nodules. The sensitivity and specificity of the method for distinguishing malignant tumors and benign lesions were 79.5% (63.5%~90.7%) and 85.2% (66.3%~95.8%). The method of identifying stage IB (sensitivity =85.7%) lung cancer was also better than stage IA (sensitivity =75.0%). Moreover, Chen et al. [51] measured ctDNA and tumor tissue DNA (tDNA) in patients with stage I to III NSCLC by targeted sequencing. They founded frequent driver mutations in ctDNA and tDNA, which confirmed the feasibility of peripheral blood ctDNA detection in early detection of NSCLC gene mutations. However, Sozzi et al. [52] reported that compared with cancer-free subjects, the DNA baseline levels of cohort that developed cancer during the 5-year period (n=38) was no significant difference (AUC =0.496), just slightly higher than the baseline at the time of diagnosis. But high levels of ctDNA were more likely to develop lung cancer in individuals who smoke heavily in longitudinal contrast.
Although ctDNA analysis provides a viable option for early diagnosis of lung cancer, the existing techniques still cannot conquer the difficulties of sensitivity analysis. In addition, there are many problems that need to be solved, including lack of reliable threshold value, the required purity of ctDNA, the low proportion of ctDNA reflecting all genetic mutations [33].
Microbial DNA (mbDNA)
Multiple studies have shown that microorganisms were closely implicated to tumor progression and primary drug resistance in some cancers [53–55]. Recently, Poore et al. [56] studied the microbial sequencing of 18,116 samples from 10,481 patients with 33 cancer types reported in the TCGA database. These researchers also analyzed the whole genome sequence of 4831 samples, as well as RNA sequencing data from 13,285 tumor samples, normal tumor-adjacent tissue, blood samples and matched tissues from non-cancer individuals. The sequences of microbial sources were quickly screened by Kraken software, and used machine learning methods (ML) for microbial reads. For stage Ia–IIc cancers whose genome variants cannot be detected by ctDNA, mbDNA has stronger predictive ability. In order to further verify the effectiveness of these results, patients with prostate (n=59), lung (n=25) or melanoma (n=16) and healthy volunteers (n=69) were selected as samples. In addition to the smallest melanoma cohort, a high degree of distinction was found in paired and multiclass comparisons between normal samples and cancer types. This means that the new type of microbiome-based cancer diagnostic tool can complement existing ctDNA detection methods to detect and monitor cancer.
Unique microbial DNA signatures are found in the tissues and blood samples of most cancer patients, which can identify the presence and type of cancer. Compared with ctDNA, one of the advantages of cancer detection based on circulating microbial DNA is its diversity among different body parts. This model performs well in differentiating stage I and IV tumors of colon adenocarcinoma, gastric adenocarcinoma, and renal clear cell carcinoma. However, there are still many problems to be solved in the field of mbDNA. For example, in the process of preparing DNA, microbial contamination is a problem we must pay attention to, how to identify whether mbDNA is a normal aging change or a tumor-related change [57].
MicroRNA(miRNA)
MiRNAs is a class of endogenous non-coding small single-stranded RNA (19~24 nucleotides), originally found in nematodes [58]. MiRNA is released into the blood through some mechanisms, including passive leakage from broken cells in damaged tissues, apoptotic cells and process of inflammation and metastasis; active secretion by microvesicles and microvesicle-free miRNAs through different stimuli [59]. It degrades the mRNA by binding to the 3'UTR of the target mRNA or mediating post-transcriptional silencing through protein translation inhibition [60]. Compared with mRNA, its high stability and repeatability are its unique features. The corresponding resistance is also shown after many freeze-thaw cycles [59, 61]. Thence, researchers have been devoted to building reliable diagnostic tools with miRNA.
Lawrie et al. [62] demonstrated that high expression of MIRN21 is correlated to relapse-free survival (P=0.05). Hereafter, Zen et al. [63] proposed that circulating miRNAs better reflected all aspects of disease than tissue miRNAs. To date, using a panel of miRNAs to detect early-stage lung cancer is more common than using single miRNA (Table 2).
Table 2. Some studies on circulating miRNAs for diagnosis of lung cancer.
| miRNA | TNM stage | Source | Detection method | Performance | Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| miR-429,miR-205, miR-200b,miR-203,miR-125b,miR-34b | I-IV | serum | qRT-PCR | Sen=88% Spe=71% AUC=0.88 (I/II) AUC=0.89 (I/V) Threshold value=0.37 | NSCLC (n=100) [64] non-cancer (n=58) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| miR-146a,miR-222,miR-223 | I-III | serum | qRT-PCR | AC vs HD: Total AUC=0.951 Sen=84.35% Spe=90.83% Acu=87.27% Cutoff value = 0.5015 | Training set [65] AC (n=40) HD (n=40) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| In AC AUC=0.942 (I) AUC=0.968 (II) AUC=0.954(III) | Validation set AC (n=120) HD (n=120) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| miR-21,miR-145, miR-155 | I-III | plasma | qRT-PCR | Training set LC vs HS Total AUC=0.847 Sen=69.4% Spe=78.3% Cutoff value: 1.310, 0.4950,0.01535 | Training set [66] LC (n=62) HS(n=60) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Validation set LC vs HS Total AUC=0.841 Sen=76.5% Spe=80.0% | Validation set LC (n=34) BPN (n=30) HS (n=32) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| miR-146b,miR-205,miR-29c, miR-30b | I-III | serum | qRT-PCR | Training set AUC=0.99 Sen=93.65% Spe=93.33% Acu=95.00% Cutoff value = 0.5606 | Training set [67] NSCLS (n=63) HS (n=15) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Validation set AUC=0.93 Sen=78.13% Spe=95.38% Acu=89.69% | Validation set NSCLS (n=65) Cancer-free (n=32) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| In NSCLC AUC=0.96(I) AUC=0.95(II-III) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| miR-1254 MALAT1 miR-485-5p miR-574-5p | I-IV | serum | qRT-PCR | AUC=0.864/0.848/ 0.878/0.732(I/II/III /IV) Sen=94.6%/89.7%/ 94.9%/66.7%(I/II/III /IV) Spe=75.7%(I/II/III/ IV) | Training set [68] NSCLS (n=36) HS (n=36) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Validation set NSCLS (n=120) HS (n=71) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviation: Sen: sensitivity; Spe: specificity; Acu: accuracy; AC: lung adenocarcinoma; HD: healthy donor; HS: healthy smoker; LC: lung cancer; BPNs: benign pulmonary nodules; SCC: squamous cell carcinoma. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
The World Conference on Lung Cancer 2019 reported the results of a clinical trial called BioMILD [69], which recruited 4119 participants. This trial combined LDCT scanning with miRNA testing to screen the high risk groups of lung cancer and implied that combining miRNA with LDCT examination could greatly enhance early diagnosis efficiency.
Non-invasiveness and stable nature of circulating miRNA make it a potential tool as a biomarker for diagnosing cancer. Nevertheless, the limitation of miRNA is reflected in the inconsistency that the selection of internal reference genes for quantitative detection. Moreover, miRNAs from different sources, such as tissues, plasma and serum, lack a standardization protocol during the separation and extraction process. Therefore, standardization is a problem that should be solved.
Circulating exosomes
Exosomes are intracellular vesicles with a diameter between 30 nm and 100 nm. Fusion of multivesicular bodies with plasma membrane induces the release of exosomes from multivesicular bodies into the extracellular space [70]. It can be detected from various body fluids, comprising plasma, saliva, urine, breast milk, pleural effusion, cerebrospinal fluid, semen, and carries biological information such as protein, microRNA, mRNA, DNA [33, 70, 71]. It may promote invasion, immune escape and chemotherapy resistance of lung cancer [72]. Researchers are gradually paying attention to the potentiality of exosomes as a diagnostic biomarker.
Among them, there are many studies on miRNAs or proteins derived from circulating exosomes. Exosome miRNA (let-7b/let-7e/miR-23a-3p/miR-486) profiles in plasma determined by NGS technology can identify patients with stage I NSCLC (sensitivity= 80.3%, specificity= 92.3%, AUC=0.899) [73]. Li et al. [74] proposed that Exo-Gas5 (growth arrest-specific transcript 5) also has a similar effect as above. Moreover, when jointly evaluated with CEA, AUC value up to 0.929. In addition, when Exo-Gas5 was used alone to identify patients with stage I NSCLC, AUC value even reaches 0.822. Apart from these, more studies have shown that exosomes might be a candidate to develop highly sensitive, non-invasive and effective biomarkers for early NSCLC diagnosis [75].
Due to its nano-size, efficient extraction and purification protocol are still a big problem [33]. Yet it also has the characteristics of vesicle coating and stable properties, which can be acted as a natural drug carrier to broaden the prospect of exosomes in the treatment of lung cancer after definite diagnosis [76]. In short, exosomes are an indispensable development trend not only in early diagnosis but also in clinical pharmaceutical transformation.
DNA methylation
DNA methylation refers to the methyl group covalently integrate to cytosine of the cytosine phosphate-guanine (CpG) dinucleotide in the DNA genome under the action of DNA methyltransferase (DNMT) [77]. Some areas with a length of 0.5 to 4kb have high CpG density, which are called CpG islands (CGIs), and most of them are located at the 5'end of the gene. Under normal circumstances, CpG dinucleotides in CGIs remain unmethylated [78]. In patients with malignancy, the CGIs of anti-oncogene promoter was abnormally hypermethylated, and the normal genome level showed hypomethylation [79]. Abnormal DNA methylation cause the increase of chromosome rotation, the silencing and loss of expression of anti-oncogene, which ultimately leads to tumorigenesis [80, 81]. Previous studies have proved that early recurrence of curative stage I lung cancer and early lung cancer have specific DNA methylation [82]. Thus, abnormal methylation of DNA can become a biomarker for diagnosis and monitoring of lung tumors.
Ma et al. [83] utilized quantum dots-based fluorescence resonance energy transfer (QDs-FRET) technique to quantitatively analyze the methylation levels of PCDHGB6, HOXA9 and RASSF1A in lung cancer tissues and specimens. It was found that compared with tumor tissues, bronchial brush specimens have a slightly weaker ability to detect early cancer. In 50 NSCLC subjects with stage I to II, the total sensitivity (92%) and specificity (100%) are higher than single gene (68%, 80%, 64%), but the sensitivity to identify stage II NSCLC is better than stage I NSCLC (100% vs 83%). Therefore, this method for detecting DNA methylation can act as a potential non-invasive clinical diagnosis tool for early stage cancer. Besides, peripheral blood and sputum are an ideal sample for detecting abnormal methylation in early lung cancer [84, 85]. Liu et al. [86] took subjects with suspicious nodules on CT images as the research object, and found that methylation detection of 6 genes (CDO1, TAC1, HOXA7, HOXA9, SOX17, ZFP42) in plasma and urine was significantly related to the diagnosis of NSCLC. Based on this research, it further confirmed the effectiveness of DNA methylation as an auxiliary test for lung cancer CT screening.
In conclusion, the research on DNA methylation-related issues in NSCLC helps to understand the pathogenesis at the molecular level, and also provides potential hope for the early diagnosis, outcome and treatment of NSCLC. However, there are still some problems in the application of DNA methylation as molecular marker in clinical diagnosis of lung cancer. First of all, there is a lack of uniform testing standards [79]. Secondly, as a tumor marker, the single gene positive rate is low. Last, the specificity is not high. All of these problems need to be ameliorated.
Volatile organic compounds (VOCS) and screening for lung cancer
In recent years, the detection of VOCs has the advantages of rapid, non-invasive, convenient, high sensitivity and good repeatability [87]. Exhaled breath contains a lot of substances, most of which are VOCs [88]. As a new diagnostic method, it has become a research hotspot. For example, Zhong et al. [89] constructed a disposable colorimetric array from diverse chemo-responsive colorants. Through chemical interaction with VOCs, these arrays could distinguish 20 different VOCs associated with lung cancer during exhalation with an accuracy of at least 90%. Li et al. [90] developed doping-assisted low-pressure photoionization mass spectrometry (DA-LPPI), proving the effectiveness of dichloromethane DA-LPPI technology in the detection of lung cancer-related polar VOCs.
Now, there are many types of VOCs and their sources are complex. The main difficulty is that many test techniques and methods used in sample collection, processing, storage and testing still lack uniform standards [91]. Therefore, although there are still great challenges in converting to clinical applications, the huge prospects for lung cancer detection are undeniable.
Autoantibodies and screening for lung cancer
Another clinical trial, ECLS, was also reported in World Conference on Lung Cancer 2019 and enrolled 12208 high-risk patients. EarlyCDT test is a novel blood test based on lung cancer autoantibodies. Patients receive EarlyCDT blood test. If there is a positive result, they will receive x-ray and CT scan, which is designated as the experimental group. Among the people who received EarlyCDT examination and continued to develop lung cancer in the next two years, 41.1% of patients were diagnosed in the early stages (stage I and II) of the disease, compared to 26.8% in the control group. After 2 years of follow-up in subjects randomized to participate in the EarlyCDT trial, it was found that the incidence of advanced lung cancer was reduced by 36%. The study also showed that the patients undergoing EarlyCDT testing had a subtle trend of lower lung cancer-related mortality within two years than the control group. This study proves that EarlyCDT has a strong application prospect in the clinical early diagnosis of lung cancer [92].
Summary
Among the above-mentioned early screening and diagnostic methods of lung cancer, x-ray screening is not recommended. LDCT has obvious advantages and is the most promising imaging method in early screening of lung cancer. Bronchoscopy has a greater advantage in direct vision of intraluminal lesions and can be used as a diagnostic tool. Liquid biopsy, VOCs and special tumor autoantibodies detection are simple and non-invasive. Especially liquid biopsy, it is a hot spot and the direction of future research in early diagnosis of lung cancer in recent years. However, there is currently no clear threshold and standard operating protocol for liquid biopsy, and tumor stage cannot be evaluated like CT. Therefore, the combination of LDCT and liquid biopsy to diagnose lung cancer or develop a reasonable diagnosis method may achieve early detection, early diagnosis and early treatment, thereby benefiting lung cancer patients. Besides, more large-scale research needs to be carried out.
Author Contributions
The corresponding author Yayi He provided the subject of the article. Jing Ning carried out the document collection and drafted the manuscript. Tao Ge was responsible for the revision of the article. Minlin Jiang, Keyi Jia, Lei Wang and Wei Li organized and optimized the document. Bin Chen, Yu Liu, Hao Wang, Sha Zhao were responsible for image beautification and the layout of article. All authors reviewed the manuscript and approved the content of the manuscript.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Funding
This study was supported in part by a grant of young talents in Shanghai, National Natural Science Foundation of China (81802255), Young Talents in Shanghai (2019QNBJ), ‘Dream Tutor’ Outstanding Young Talents Program (fkyq1901), Clinical Research Project of Shanghai Pulmonary Hospital (fk18005), Key Discipline in 2019 (oncology), Project of Shanghai Municipal Science and Technology Commission (Project of Municipal Science and Technology Commission), and Scientific research project of Shanghai Pulmonary Hospital (fkcx1903).
References
- 1. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality Worldwide for 36 cancers in 185 Countries. CA Cancer J Clin. 2018; 68:394–424. https://doi.org/10.3322/caac.21492 [PubMed]
- 2. Allemani C, Matsuda T, Di Carlo V, Harewood R, Matz M, Nikšić M, Bonaventure A, Valkov M, Johnson CJ, Estève J, Ogunbiyi OJ, Azevedo E Silva G, Chen WQ, et al, and CONCORD Working Group. Global surveillance of trends in cancer survival 2000-14 (CONCORD-3): analysis of individual records for 37 513 025 patients diagnosed with one of 18 cancers from 322 population-based registries in 71 Countries. Lancet. 2018; 391:1023–75. https://doi.org/10.1016/S0140-6736(17)33326-3 [PubMed]
- 3. Aberle DR, Berg CD, Black WC, Church TR, Fagerstrom RM, Galen B, Gareen IF, Gatsonis C, Goldin J, Gohagan JK, Hillman B, Jaffe C, Kramer BS, et al, and National Lung Screening Trial Research Team. The National lung screening trial: overview and study design. Radiology. 2011; 258:243–53. https://doi.org/10.1148/radiol.10091808 [PubMed]
- 4. Goldstraw P, Chansky K, Crowley J, Rami-Porta R, Asamura H, Eberhardt WE, Nicholson AG, Groome P, Mitchell A, Bolejack V, and International Association for the Study of Lung Cancer Staging and Prognostic Factors Committee, Advisory Boards, and Participating Institutions; International Association for the Study of Lung Cancer Staging and Prognostic Factors Committee Advisory Boards and Participating Institutions. The IASLC Lung Cancer Staging Project: Proposals for Revision of the TNM Stage Groupings in the Forthcoming (Eighth) Edition of the TNM Classification for Lung Cancer. J Thorac Oncol. 2016; 11:39–51. https://doi.org/10.1016/j.jtho.2015.09.009 [PubMed]
- 5. Hu J, Qian GS, Bai CX, and Lung Cancer Study Group of Chinese Thoracic Society and Chinese Alliance Against Lung Cancer Expert Group. Chinese consensus on early diagnosis of primary lung cancer (2014 version). Cancer. 2015 (Suppl 17); 121:3157–64. https://doi.org/10.1002/cncr.29571 [PubMed]
- 6. Xiang D, Zhang B, Doll D, Shen K, Kloecker G, Freter C. Lung cancer screening: from imaging to biomarker. Biomark Res. 2013; 1:4. https://doi.org/10.1186/2050-7771-1-4 [PubMed]
- 7. Shen H. Low-dose CT for lung cancer screening: opportunities and challenges. Front Med. 2018; 12:116–21. https://doi.org/10.1007/s11684-017-0600-1 [PubMed]
- 8. Aberle DR, Adams AM, Berg CD, Black WC, Clapp JD, Fagerstrom RM, Gareen IF, Gatsonis C, Marcus PM, Sicks JD, and National Lung Screening Trial Research Team. Reduced lung-cancer mortality with low-dose computed tomographic screening. N Engl J Med. 2011; 365:395–409. https://doi.org/10.1056/NEJMoa1102873 [PubMed]
- 9. Mulshine JL, D’Amico TA. Issues with implementing a high-quality lung cancer screening program. CA Cancer J Clin. 2014; 64:352–63. https://doi.org/10.3322/caac.21239 [PubMed]
- 10. Smith RA, Andrews KS, Brooks D, Fedewa SA, Manassaram-Baptiste D, Saslow D, Wender RC. Cancer screening in the United States, 2019: a review of current American Cancer Society guidelines and current issues in cancer screening. CA Cancer J Clin. 2019; 69:184–210. https://doi.org/10.3322/caac.21557 [PubMed]
- 11. Moyer VA, and U.S. Preventive Services Task Force. Screening for lung cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2014; 160:330–38. https://doi.org/10.7326/M13-2771 [PubMed]
- 12. Wood DE. National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines for Lung Cancer Screening. Thorac Surg Clin. 2015; 25:185–97. https://doi.org/10.1016/j.thorsurg.2014.12.003 [PubMed]
- 13. Becker N, Motsch E, Gross ML, Eigentopf A, Heussel CP, Dienemann H, Schnabel PA, Eichinger M, Optazaite DE, Puderbach M, Wielpütz M, Kauczor HU, Tremper J, Delorme S. Randomized study on early detection of lung cancer with MSCT in Germany: results of the first 3 years of follow-up after randomization. J Thorac Oncol. 2015; 10:890–96. https://doi.org/10.1097/JTO.0000000000000530 [PubMed]
- 14. Infante M, Cavuto S, Lutman FR, Passera E, Chiarenza M, Chiesa G, Brambilla G, Angeli E, Aranzulla G, Chiti A, Scorsetti M, Navarria P, Cavina R, et al, and DANTE Study Group. Long-term follow-up results of the DANTE trial, a randomized study of lung cancer screening with spiral computed tomography. Am J Respir Crit Care Med. 2015; 191:1166–75. https://doi.org/10.1164/rccm.201408-1475OC [PubMed]
- 15. Black WC, Gareen IF, Soneji SS, Sicks JD, Keeler EB, Aberle DR, Naeim A, Church TR, Silvestri GA, Gorelick J, Gatsonis C, and National Lung Screening Trial Research Team. Cost-effectiveness of CT screening in the National lung screening trial. N Engl J Med. 2014; 371:1793–802. https://doi.org/10.1056/NEJMoa1312547 [PubMed]
- 16. Patz EF
Jr , Pinsky P, Gatsonis C, Sicks JD, Kramer BS, Tammemägi MC, Chiles C, Black WC, Aberle DR, and NLST Overdiagnosis Manuscript Writing Team. Overdiagnosis in low-dose computed tomography screening for lung cancer. JAMA Intern Med. 2014; 174:269–74. https://doi.org/10.1001/jamainternmed.2013.12738 [PubMed] - 17. Wang HQ, Zhao L, Zhao J, Wang Q. Analysis on early detection of lung cancer by PET/CT scan. Asian Pac J Cancer Prev. 2015; 16:2215–17. https://doi.org/10.7314/apjcp.2015.16.6.2215 [PubMed]
- 18. Deppen SA, Blume JD, Kensinger CD, Morgan AM, Aldrich MC, Massion PP, Walker RC, McPheeters ML, Putnam JB
Jr , Grogan EL. Accuracy of FDG-PET to diagnose lung cancer in areas with infectious lung disease: a meta-analysis. JAMA. 2014; 312:1227–36. https://doi.org/10.1001/jama.2014.11488 [PubMed] - 19. Spadafora M, Pace L, Mansi L. Segmental 18F-FDG-PET/CT in a single pulmonary nodule: a better cost/effectiveness strategy. Eur J Nucl Med Mol Imaging. 2017; 44:1–4. https://doi.org/10.1007/s00259-016-3532-0 [PubMed]
- 20. Chang CY, Tzao C, Lee SC, Cheng CY, Liu CH, Huang WS, Ku CH, Lee JK, Oliver Wong CY. Incremental value of integrated FDG-PET/CT in evaluating indeterminate solitary pulmonary nodule for Malignancy. Mol Imaging Biol. 2010; 12:204–09. https://doi.org/10.1007/s11307-009-0241-0 [PubMed]
- 21. Hui Z, Wei F, Ren H, Xu W, Ren X. Primary tumor standardized uptake value (SUVmax) measured on 18F-FDG PET/CT and mixed NSCLC components predict survival in surgical-resected combined small-cell lung cancer. J Cancer Res Clin Oncol. 2020; 146:2595–605. https://doi.org/10.1007/s00432-020-03240-8 [PubMed]
- 22. Hirsch FR, Prindiville SA, Miller YE, Franklin WA, Dempsey EC, Murphy JR, Bunn PA
Jr , Kennedy TC. Fluorescence versus white-light bronchoscopy for detection of preneoplastic lesions: a randomized study. J Natl Cancer Inst. 2001; 93:1385–91. https://doi.org/10.1093/jnci/93.18.1385 [PubMed] - 23. Inage T, Nakajima T, Yoshino I, Yasufuku K. Early lung cancer detection. Clin Chest Med. 2018; 39:45–55. https://doi.org/10.1016/j.ccm.2017.10.003 [PubMed]
- 24. Hung J, Lam S, LeRiche JC, Palcic B. Autofluorescence of normal and Malignant bronchial tissue. Lasers Surg Med. 1991; 11:99–105. https://doi.org/10.1002/lsm.1900110203 [PubMed]
- 25. Loewen G, Natarajan N, Tan D, Nava E, Klippenstein D, Mahoney M, Cummings M, Reid M. Autofluorescence bronchoscopy for lung cancer surveillance based on risk assessment. Thorax. 2007; 62:335–40. https://doi.org/10.1136/thx.2006.068999 [PubMed]
- 26. Häussinger K, Becker H, Stanzel F, Kreuzer A, Schmidt B, Strausz J, Cavaliere S, Herth F, Kohlhäufl M, Müller KM, Huber RM, Pichlmeier U, Bolliger CT. Autofluorescence bronchoscopy with white light bronchoscopy compared with white light bronchoscopy alone for the detection of precancerous lesions: a European randomised controlled multicentre trial. Thorax. 2005; 60:496–503. https://doi.org/10.1136/thx.2005.041475 [PubMed]
- 27. Iftikhar IH, Musani AI. Narrow-band imaging bronchoscopy in the detection of premalignant airway lesions: a meta-analysis of diagnostic test accuracy. Ther Adv Respir Dis. 2015; 9:207–16. https://doi.org/10.1177/1753465815589698 [PubMed]
- 28. Zhu J, Li W, Zhou J, Chen Y, Zhao C, Zhang T, Peng W, Wang X. The diagnostic value of narrow-band imaging for early and invasive lung cancer: a meta-analysis. Clinics (São Paulo). 2017; 72:438–48. https://doi.org/10.6061/clinics/2017(07)09 [PubMed]
- 29. Zaric B, Perin B, Stojsic V, Carapic V, Eri Z, Panjkovic M, Andrijevic I, Matijasevic J. Relation between vascular patterns visualized by Narrow Band Imaging (NBI) videobronchoscopy and histological type of lung cancer. Med Oncol. 2013; 30:374. https://doi.org/10.1007/s12032-012-0374-x [PubMed]
- 30. Wahidi MM, Herth F, Yasufuku K, Shepherd RW, Yarmus L, Chawla M, Lamb C, Casey KR, Patel S, Silvestri GA, Feller-Kopman DJ. Technical aspects of endobronchial ultrasound-guided transbronchial needle aspiration: CHEST guideline and expert panel report. Chest. 2016; 149:816–35. https://doi.org/10.1378/chest.15-1216 [PubMed]
- 31. Navani N, Nankivell M, Lawrence DR, Lock S, Makker H, Baldwin DR, Stephens RJ, Parmar MK, Spiro SG, Morris S, Janes SM, and Lung-BOOST trial investigators. Lung cancer diagnosis and staging with endobronchial ultrasound-guided transbronchial needle aspiration compared with conventional approaches: an open-label, pragmatic, randomised controlled trial. Lancet Respir Med. 2015; 3:282–89. https://doi.org/10.1016/S2213-2600(15)00029-6
- 32. Tan DS, Camilleri-Broët S, Tan EH, Alifano M, Lim WT, Bobbio A, Zhang S, Ng QS, Ang MK, Iyer NG, Takano A, Lim KH, Régnard JF, et al. Intertumor heterogeneity of non-small-cell lung carcinomas revealed by multiplexed mutation profiling and integrative genomics. Int J Cancer. 2014; 135:1092–100. https://doi.org/10.1002/ijc.28750 [PubMed]
- 33. Rijavec E, Coco S, Genova C, Rossi G, Longo L, Grossi F. Liquid biopsy in non-small cell lung cancer: highlights and challenges. Cancers (Basel). 2019; 12:17. https://doi.org/10.3390/cancers12010017 [PubMed]
- 34. Strotman LN, Millner LM, Valdes R
Jr , Linder MW. Liquid biopsies in oncology and the current regulatory landscape. Mol Diagn Ther. 2016; 20:429–36. https://doi.org/10.1007/s40291-016-0220-5 [PubMed] - 35. Sequist LV, Nagrath S, Toner M, Haber DA, Lynch TJ. The CTC-chip: an exciting new tool to detect circulating tumor cells in lung cancer patients. J Thorac Oncol. 2009; 4:281–83. https://doi.org/10.1097/JTO.0b013e3181989565 [PubMed]
- 36. Andree KC, van Dalum G, Terstappen LW. Challenges in circulating tumor cell detection by the CellSearch system. Mol Oncol. 2016; 10:395–407. https://doi.org/10.1016/j.molonc.2015.12.002 [PubMed]
- 37. Alama A, Truini A, Coco S, Genova C, Grossi F. Prognostic and predictive relevance of circulating tumor cells in patients with non-small-cell lung cancer. Drug Discov Today. 2014; 19:1671–76. https://doi.org/10.1016/j.drudis.2014.06.001 [PubMed]
- 38. Kapeleris J, Kulasinghe A, Warkiani ME, Vela I, Kenny L, O’Byrne K, Punyadeera C. The prognostic role of circulating tumor cells (CTCs) in lung cancer. Front Oncol. 2018; 8:311. https://doi.org/10.3389/fonc.2018.00311 [PubMed]
- 39. Mayo C, Ortega FG, Giménez-Capitán A, Molina-Vila MA, Serrano MJ, Viteri S, Costa C, Gascó A, Bertran-Alamillo J, Karachaliou N, Lorente JA, Tarón M, Rosell R. CK-coated magnetic-based beads as a tool to isolate circulating tumor cells (CTCs) in human tumors. Transl Lung Cancer Res. 2013; 2:65–71. https://doi.org/10.3978/j.issn.2218-6751.2013.02.06 [PubMed]
- 40. Lou J, Ben S, Yang G, Liang X, Wang X, Ni S, Han B. Quantification of rare circulating tumor cells in non-small cell lung cancer by ligand-targeted PCR. PLoS One. 2013; 8:e80458. https://doi.org/10.1371/journal.pone.0080458 [PubMed]
- 41. Zhang Z, Shiratsuchi H, Lin J, Chen G, Reddy RM, Azizi E, Fouladdel S, Chang AC, Lin L, Jiang H, Waghray M, Luker G, Simeone DM, et al. Expansion of CTCs from early stage lung cancer patients using a microfluidic co-culture model. Oncotarget. 2014; 5:12383–97. https://doi.org/10.18632/oncotarget.2592 [PubMed]
- 42. He Y, Shi J, Schmidt B, Liu Q, Shi G, Xu X, Liu C, Gao Z, Guo T, Shan B. Circulating tumor cells as a biomarker to assist molecular diagnosis for early stage non-small cell lung cancer. Cancer Manag Res. 2020; 12:841–54. https://doi.org/10.2147/CMAR.S240773 [PubMed]
- 43. Spellman PT, Gray JW. Detecting cancer by monitoring circulating tumor DNA. Nat Med. 2014; 20:474–75. https://doi.org/10.1038/nm.3564 [PubMed]
- 44. Santarpia M, Liguori A, D’Aveni A, Karachaliou N, Gonzalez-Cao M, Daffinà MG, Lazzari C, Altavilla G, Rosell R. Liquid biopsy for lung cancer early detection. J Thorac Dis. 2018 (Suppl 7); 10:S882–97. https://doi.org/10.21037/jtd.2018.03.81 [PubMed]
- 45. Vallée A, Marcq M, Bizieux A, Kouri CE, Lacroix H, Bennouna J, Douillard JY, Denis MG. Plasma is a better source of tumor-derived circulating cell-free DNA than serum for the detection of EGFR alterations in lung tumor patients. Lung Cancer. 2013; 82:373–74. https://doi.org/10.1016/j.lungcan.2013.08.014 [PubMed]
- 46. Herbreteau G, Vallée A, Charpentier S, Normanno N, Hofman P, Denis MG. Circulating free tumor DNA in non-small cell lung cancer (NSCLC): clinical application and future perspectives. J Thorac Dis. 2019 (Suppl 1); 11:S113–26. https://doi.org/10.21037/jtd.2018.12.18 [PubMed]
- 47. Moding EJ, Diehn M, Wakelee HA. Circulating tumor DNA testing in advanced non-small cell lung cancer. Lung Cancer. 2018; 119:42–47. https://doi.org/10.1016/j.lungcan.2018.02.019 [PubMed]
- 48. Bettegowda C, Sausen M, Leary RJ, Kinde I, Wang Y, Agrawal N, Bartlett BR, Wang H, Luber B, Alani RM, Antonarakis ES, Azad NS, Bardelli A, et al. Detection of circulating tumor DNA in early- and late-stage human Malignancies. Sci Transl Med. 2014; 6:224ra24. https://doi.org/10.1126/scitranslmed.3007094 [PubMed]
- 49. Szpechcinski A, Chorostowska-Wynimko J, Struniawski R, Kupis W, Rudzinski P, Langfort R, Puscinska E, Bielen P, Sliwinski P, Orlowski T. Cell-free DNA levels in plasma of patients with non-small-cell lung cancer and inflammatory lung disease. Br J Cancer. 2015; 113:476–83. https://doi.org/10.1038/bjc.2015.225 [PubMed]
- 50. Liang W, Zhao Y, Huang W, Gao Y, Xu W, Tao J, Yang M, Li L, Ping W, Shen H, Fu X, Chen Z, Laird PW, et al. Non-invasive diagnosis of early-stage lung cancer using high-throughput targeted DNA methylation sequencing of circulating tumor DNA (ctDNA). Theranostics. 2019; 9:2056–70. https://doi.org/10.7150/thno.28119 [PubMed]
- 51. Chen KZ, Lou F, Yang F, Zhang JB, Ye H, Chen W, Guan T, Zhao MY, Su XX, Shi R, Jones L, Huang XF, Chen SY, Wang J. Circulating tumor DNA detection in early-stage non-small cell lung cancer patients by targeted sequencing. Sci Rep. 2016; 6:31985. https://doi.org/10.1038/srep31985 [PubMed]
- 52. Sozzi G, Roz L, Conte D, Mariani L, Andriani F, Lo Vullo S, Verri C, Pastorino U. Plasma DNA quantification in lung cancer computed tomography screening: five-year results of a prospective study. Am J Respir Crit Care Med. 2009; 179:69–74. https://doi.org/10.1164/rccm.200807-1068OC [PubMed]
- 53. Dejea CM, Fathi P, Craig JM, Boleij A, Taddese R, Geis AL, Wu X, DeStefano Shields CE, Hechenbleikner EM, Huso DL, Anders RA, Giardiello FM, Wick EC, et al. Patients with familial adenomatous polyposis harbor colonic biofilms containing tumorigenic bacteria. Science. 2018; 359:592–97. https://doi.org/10.1126/science.aah3648 [PubMed]
- 54. Jin C, Lagoudas GK, Zhao C, Bullman S, Bhutkar A, Hu B, Ameh S, Sandel D, Liang XS, Mazzilli S, Whary MT, Meyerson M, Germain R, et al. Commensal microbiota promote lung cancer development via γδ T cells. Cell. 2019; 176:998–1013.e16. https://doi.org/10.1016/j.cell.2018.12.040 [PubMed]
- 55. Routy B, Le Chatelier E, Derosa L, Duong CP, Alou MT, Daillère R, Fluckiger A, Messaoudene M, Rauber C, Roberti MP, Fidelle M, Flament C, Poirier-Colame V, et al. Gut microbiome influences efficacy of PD-1-based immunotherapy against epithelial tumors. Science. 2018; 359:91–97. https://doi.org/10.1126/science.aan3706 [PubMed]
- 56. Poore GD, Kopylova E, Zhu Q, Carpenter C, Fraraccio S, Wandro S, Kosciolek T, Janssen S, Metcalf J, Song SJ, Kanbar J, Miller-Montgomery S, Heaton R, et al. Microbiome analyses of blood and tissues suggest cancer diagnostic approach. Nature. 2020; 579:567–74. https://doi.org/10.1038/s41586-020-2095-1 [PubMed]
- 57. Dzutsev A, Trinchieri G. Microbial DNA signature in plasma enables cancer diagnosis. Nat Rev Clin Oncol. 2020; 17:453–54. https://doi.org/10.1038/s41571-020-0391-1 [PubMed]
- 58. Lee RC, Feinbaum RL, Ambros V. The C. Elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell. 1993; 75:843–54. https://doi.org/10.1016/0092-8674(93)90529-y [PubMed]
- 59. Ulivi P, Zoli W. miRNAs as non-invasive biomarkers for lung cancer diagnosis. Molecules. 2014; 19:8220–37. https://doi.org/10.3390/molecules19068220 [PubMed]
- 60. Hou J, Meng F, Chan LW, Cho WC, Wong SC. Circulating plasma MicroRNAs as diagnostic markers for NSCLC. Front Genet. 2016; 7:193. https://doi.org/10.3389/fgene.2016.00193 [PubMed]
- 61. Mitchell PS, Parkin RK, Kroh EM, Fritz BR, Wyman SK, Pogosova-Agadjanyan EL, Peterson A, Noteboom J, O’Briant KC, Allen A, Lin DW, Urban N, Drescher CW, et al. Circulating microRNAs as stable blood-based markers for cancer detection. Proc Natl Acad Sci USA. 2008; 105:10513–18. https://doi.org/10.1073/pnas.0804549105 [PubMed]
- 62. Lawrie CH, Gal S, Dunlop HM, Pushkaran B, Liggins AP, Pulford K, Banham AH, Pezzella F, Boultwood J, Wainscoat JS, Hatton CS, Harris AL. Detection of elevated levels of tumour-associated microRNAs in serum of patients with diffuse large B-cell lymphoma. Br J Haematol. 2008; 141:672–75. https://doi.org/10.1111/j.1365-2141.2008.07077.x [PubMed]
- 63. Zen K, Zhang CY. Circulating microRNAs: a novel class of biomarkers to diagnose and monitor human cancers. Med Res Rev. 2012; 32:326–48. https://doi.org/10.1002/med.20215 [PubMed]
- 64. Halvorsen AR, Bjaanæs M, LeBlanc M, Holm AM, Bolstad N, Rubio L, Peñalver JC, Cervera J, Mojarrieta JC, López-Guerrero JA, Brustugun OT, Helland Å. A unique set of 6 circulating microRNAs for early detection of non-small cell lung cancer. Oncotarget. 2016; 7:37250–59. https://doi.org/10.18632/oncotarget.9363 [PubMed]
- 65. Lv S, Xue J, Wu C, Wang L, Wu J, Xu S, Liang X, Lou J. Identification of a panel of serum microRNAs as biomarkers for early detection of lung adenocarcinoma. J Cancer. 2017; 8:48–56. https://doi.org/10.7150/jca.16644 [PubMed]
- 66. Tang D, Shen Y, Wang M, Yang R, Wang Z, Sui A, Jiao W, Wang Y. Identification of plasma microRNAs as novel noninvasive biomarkers for early detection of lung cancer. Eur J Cancer Prev. 2013; 22:540–48. https://doi.org/10.1097/CEJ.0b013e32835f3be9 [PubMed]
- 67. Yang X, Zhang Q, Zhang M, Su W, Wang Z, Li Y, Zhang J, Beer DG, Yang S, Chen G. Serum microRNA signature is capable of early diagnosis for non-small cell lung cancer. Int J Biol Sci. 2019; 15:1712–22. https://doi.org/10.7150/ijbs.33986 [PubMed]
- 68. Peng H, Wang J, Li J, Zhao M, Huang SK, Gu YY, Li Y, Sun XJ, Yang L, Luo Q, Huang CZ. A circulating non-coding RNA panel as an early detection predictor of non-small cell lung cancer. Life Sci. 2016; 151:235–42. https://doi.org/10.1016/j.lfs.2016.03.002 [PubMed]
- 69. Venkatesan P. IASLC 2019 World conference on lung cancer. Lancet Respir Med. 2019; 7:930–32. https://doi.org/10.1016/S2213-2600(19)30330-3 [PubMed]
- 70. Taverna S, Giallombardo M, Gil-Bazo I, Carreca AP, Castiglia M, Chacártegui J, Araujo A, Alessandro R, Pauwels P, Peeters M, Rolfo C. Exosomes isolation and characterization in serum is feasible in non-small cell lung cancer patients: critical analysis of evidence and potential role in clinical practice. Oncotarget. 2016; 7:28748–60. https://doi.org/10.18632/oncotarget.7638 [PubMed]
- 71. Vanni I, Alama A, Grossi F, Dal Bello MG, Coco S. Exosomes: a new horizon in lung cancer. Drug Discov Today. 2017; 22:927–36. https://doi.org/10.1016/j.drudis.2017.03.004 [PubMed]
- 72. Chen R, Xu X, Qian Z, Zhang C, Niu Y, Wang Z, Sun J, Zhang X, Yu Y. The biological functions and clinical applications of exosomes in lung cancer. Cell Mol Life Sci. 2019; 76:4613–33. https://doi.org/10.1007/s00018-019-03233-y [PubMed]
- 73. Jin X, Chen Y, Chen H, Fei S, Chen D, Cai X, Liu L, Lin B, Su H, Zhao L, Su M, Pan H, Shen L, et al. Evaluation of tumor-derived exosomal miRNA as potential diagnostic biomarkers for early-stage non-small cell lung cancer using next-generation sequencing. Clin Cancer Res. 2017; 23:5311–19. https://doi.org/10.1158/1078-0432.CCR-17-0577 [PubMed]
- 74. Li C, Lv Y, Shao C, Chen C, Zhang T, Wei Y, Fan H, Lv T, Liu H, Song Y. Tumor-derived exosomal lncRNA GAS5 as a biomarker for early-stage non-small-cell lung cancer diagnosis. J Cell Physiol. 2019; 234:20721–27. https://doi.org/10.1002/jcp.28678 [PubMed]
- 75. Fortunato O, Gasparini P, Boeri M, Sozzi G. exo-miRNAs as a new tool for liquid biopsy in lung cancer. Cancers (Basel). 2019; 11:888. https://doi.org/10.3390/cancers11060888 [PubMed]
- 76. Kim MS, Haney MJ, Zhao Y, Mahajan V, Deygen I, Klyachko NL, Inskoe E, Piroyan A, Sokolsky M, Okolie O, Hingtgen SD, Kabanov AV, Batrakova EV. Development of exosome-encapsulated paclitaxel to overcome MDR in cancer cells. Nanomedicine. 2016; 12:655–64. https://doi.org/10.1016/j.nano.2015.10.012 [PubMed]
- 77. Li E, Zhang Y. DNA methylation in mammals. Cold Spring Harb Perspect Biol. 2014; 6:a019133. https://doi.org/10.1101/cshperspect.a019133 [PubMed]
- 78. Esteller M. Epigenetics in cancer. N Engl J Med. 2008; 358:1148–59. https://doi.org/10.1056/NEJMra072067 [PubMed]
- 79. Pan Y, Liu G, Zhou F, Su B, Li Y. DNA methylation profiles in cancer diagnosis and therapeutics. Clin Exp Med. 2018; 18:1–14. https://doi.org/10.1007/s10238-017-0467-0 [PubMed]
- 80. Ikegami K, Ohgane J, Tanaka S, Yagi S, Shiota K. Interplay between DNA methylation, histone modification and chromatin remodeling in stem cells and during development. Int J Dev Biol. 2009; 53:203–14. https://doi.org/10.1387/ijdb.082741ki [PubMed]
- 81. Fraga MF, Ballestar E, Villar-Garea A, Boix-Chornet M, Espada J, Schotta G, Bonaldi T, Haydon C, Ropero S, Petrie K, Iyer NG, Pérez-Rosado A, Calvo E, et al. Loss of acetylation at Lys16 and trimethylation at Lys20 of histone H4 is a common hallmark of human cancer. Nat Genet. 2005; 37:391–400. https://doi.org/10.1038/ng1531 [PubMed]
- 82. Chen Z, Xiong S, Li J, Ou L, Li C, Tao J, Jiang Z, Fan J, He J, Liang W. DNA methylation markers that correlate with occult lymph node metastases of non-small cell lung cancer and a preliminary prediction model. Transl Lung Cancer Res. 2020; 9:280–87. https://doi.org/10.21037/tlcr.2020.03.13 [PubMed]
- 83. Ma Y, Bai Y, Mao H, Hong Q, Yang D, Zhang H, Liu F, Wu Z, Jin Q, Zhou H, Cao J, Zhao J, Zhong X, Mao H. A panel of promoter methylation markers for invasive and noninvasive early detection of NSCLC using a quantum dots-based FRET approach. Biosens Bioelectron. 2016; 85:641–48. https://doi.org/10.1016/j.bios.2016.05.067 [PubMed]
- 84. Shivapurkar N, Stastny V, Suzuki M, Wistuba II, Li L, Zheng Y, Feng Z, Hol B, Prinsen C, Thunnissen FB, Gazdar AF. Application of a methylation gene panel by quantitative PCR for lung cancers. Cancer Lett. 2007; 247:56–71. https://doi.org/10.1016/j.canlet.2006.03.020 [PubMed]
- 85. Dietrich D, Kneip C, Raji O, Liloglou T, Seegebarth A, Schlegel T, Flemming N, Rausch S, Distler J, Fleischhacker M, Schmidt B, Giles T, Walshaw M, et al. Performance evaluation of the DNA methylation biomarker SHOX2 for the aid in diagnosis of lung cancer based on the analysis of bronchial aspirates. Int J Oncol. 2012; 40:825–32. https://doi.org/10.3892/ijo.2011.1264 [PubMed]
- 86. Liu B, Ricarte Filho J, Mallisetty A, Villani C, Kottorou A, Rodgers K, Chen C, Ito T, Holmes K, Gastala N, Valyi-Nagy K, David O, Gaba RC, et al. Detection of promoter DNA methylation in urine and plasma aids the detection of non-small cell lung cancer. Clin Cancer Res. 2020; 26:4339–48. https://doi.org/10.1158/1078-0432.CCR-19-2896 [PubMed]
- 87. Hua Q, Zhu Y, Liu H. Detection of volatile organic compounds in exhaled breath to screen lung cancer: a systematic review. Future Oncol. 2018; 14:1647–62. https://doi.org/10.2217/fon-2017-0676 [PubMed]
- 88. Wang M, Sheng J, Wu Q, Zou Y, Hu Y, Ying K, Wan H, Wang P. Confounding effect of benign pulmonary diseases in selecting volatile organic compounds as markers of lung cancer. J Breath Res. 2018; 12:046013. https://doi.org/10.1088/1752-7163/aad9cc [PubMed]
- 89. Zhong X, Li D, Du W, Yan M, Wang Y, Huo D, Hou C. Rapid recognition of volatile organic compounds with colorimetric sensor arrays for lung cancer screening. Anal Bioanal Chem. 2018; 410:3671–81. https://doi.org/10.1007/s00216-018-0948-3 [PubMed]
- 90. Li Z, Xu C, Shu J, Yang B, Zou Y. Doping-assisted low-pressure photoionization mass spectrometry for the real-time detection of lung cancer-related volatile organic compounds. Talanta. 2017; 165:98–106. https://doi.org/10.1016/j.talanta.2016.12.039 [PubMed]
- 91. Zhou J, Huang ZA, Kumar U, Chen DD. Review of recent developments in determining volatile organic compounds in exhaled breath as biomarkers for lung cancer diagnosis. Anal Chim Acta. 2017; 996:1–9. https://doi.org/10.1016/j.aca.2017.09.021 [PubMed]
- 92. Leavitt J, Pangilinan J. 2019 World conference of lung cancer round-up. Oncology (Williston Park). 2019; 33:687519. [PubMed]


