Introduction
Prostate cancer is the second most frequently diagnosed cancer and the fifth leading cause of cancer death in men worldwide [1, 2]. It is relatively common in Europe and the US, but is uncommon in Asia [3]. In China, the incidence of prostate cancer has been increasing due to the increase in life expectancy; the incidence rose from 4.62 cases per 100,000 men in 2000 to 21.62 cases per 100,000 men in 2004 [4, 5]. Because prostate-specific antigen (PSA) screening for early detection of prostate cancer is not routinely performed in China, patients with prostate cancer tend to be relatively older at initial diagnosis and to have adverse prognostic features such as advanced-stage disease and high Gleason score and PSA level [6–8].
External beam radiotherapy is a well-established curative modality for patients with localized prostate cancer [9–12]. Important progress in external beam radiotherapy over the past decade include induction with highly conformal doses to the clinical target volume (CTV) through intensity-modulated beams (intensity-modulated radiotherapy; IMRT) or volumetric arcs (volumetric-modulated arc therapy; VMAT) [13]; stereotactic body radiotherapy (SBRT) [14]; proton therapy [13]; and precise imaging guidance [15]. These advances have led to successful dose escalations and hypofractionated schedules, and significantly reduced the dose delivered to adjacent normal tissues such as the rectum and bladder, reduced side effects, and improved treatment outcomes in patients with localized prostate cancer [9–15].
In standard external beam radiotherapy, daily fractions of 1.8-2.0 Gy are delivered up to a total radical dose of 74-80 Gy over 7-8 weeks. Efficacy and toxicity of moderately hypofractionated radiotherapy (HFRT), delivered at 2.4-3.4 Gy per fraction over 19-26 fractions in 4-6 weeks, has been shown to be equivalent to that with conventionally fractionated radiotherapy (CFRT) [16–25]. The goal of HFRT is to reduce overall treatment time, without compromising outcomes; thus, it saves resources and decreases healthcare costs, and is also convenient for patients. Based on the evidence from previous landmark studies, the use of HFRT in patients with localized prostate cancer has increased in Western countries [26]; however, only a very small proportion of patients have received this treatment in China. The slow adoption of HFRT in China may be due to concerns about the toxicity in patients with a more advanced-stage disease that may require large CTV, or the lack of validation data in Asian populations. In addition, previous randomized controlled trials comparing the efficiency of HFRT and CFRT did not always include careful image-guided (IG) radiotherapy (IGRT) or used it during the latter stages of recently published Conventional or Hypofractionated High-Dose Intensity Modulated Radiotherapy for Prostate Cancer (CHHiP) trial [27]; other studies restricted enrollment of low-risk or intermediate-risk patients with a favorable prognosis [16–18, 22–25]. More recently, studies have focused on the toxicity of ultra-hypofractionated radiotherapy to further shorten the treatment duration for low-risk patients in the IGRT setting [28–30]. It should also be noted that the mean age of patients with prostate cancer at initial diagnosis is around 72 years in China, almost 5-7 years more than that of patients in the US and Europe [5, 6]. No studies have specifically examined the efficacy and safety of HFRT and CFRT in Asian populations.
We hypothesized that HFRT for localized prostate cancer would be as effective and safe as CFRT in Chinese populations in the modern radiotherapy era. In 2016, we initiated this randomized, pilot, phase II trial to determine whether a hypofractionated 5.5-week schedule of VMAT is as safe as a standard 8-week schedule in Chinese patients treated with IGRT.
Results
Patient characteristics
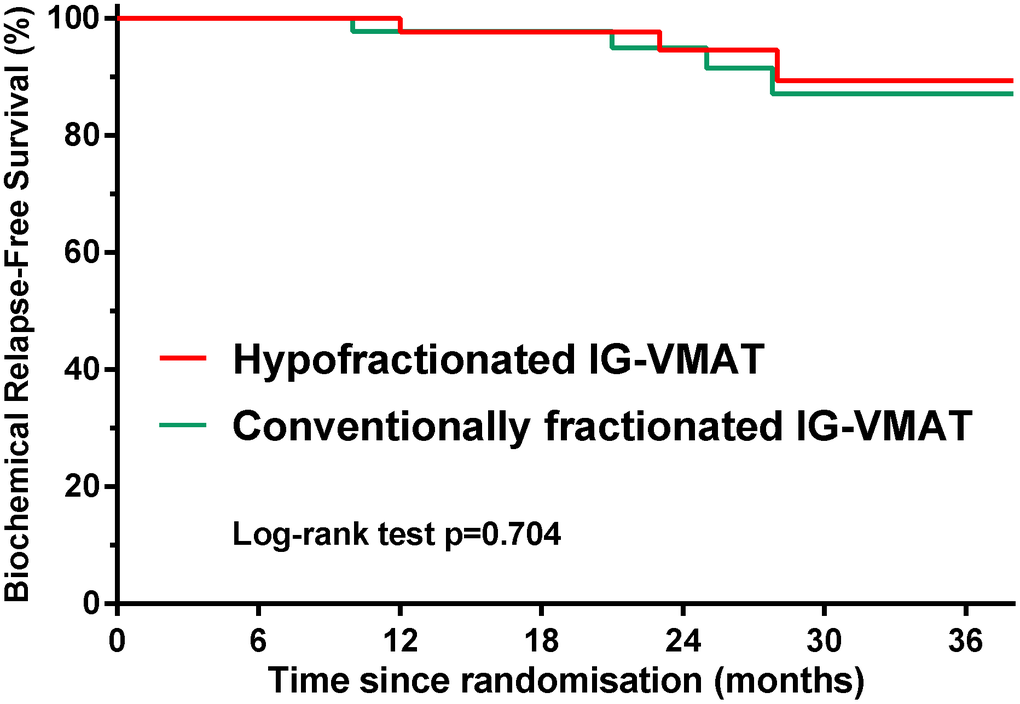
A total of 92 patients were enrolled between January 2016 and November 2018 and randomly assigned to either hypofractionated IG-VMAT (HFRT, 70 Gy/2.5 Gy/28 fractions; n = 46) or conventionally fractionated IG-VMAT (CFRT, 80 Gy/2 Gy/40 fractions; n = 46). All patients completed radiotherapy without interruption. Table 1 summarizes the clinical characteristics of patients in the two treatment arms. The median pre-therapy PSA level was 13 ng/mL. The dose coverage of PTV was not significantly different between the two groups (P = 0.639). Figure 1 shows a typical treatment planning of dose distribution and dose–volume histogram (DVH) with hypofractionated VMAT.
Table 1. Clinical characteristics and treatments among the two treatment groups.
| HFRT | CFRT | Total | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| n (%) | n (%) | n (%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age (years) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Range | 54-84 | 61-86 | 54-96 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ≤ 70 | 4 (8.7) | 9 (19.6) | 13 (14.1) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| > 70 | 42 (91.3) | 37 (80.4) | 79 (85.9) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gleason score | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ≤ 6 | 17 (37.0) | 16 (34.8) | 33 (35.9) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 7 | 19 (41.3) | 16 (34.8) | 35 (38.0) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ≥ 8 | 10 (21.7) | 14 (30.4) | 24 (26.1) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Initial PSA level (ng/ml) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mean | 12.7 | 13.4 | 13.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Median (range) | 13 (5.8-41.7) | 14 (4.9-54.2) | 13 (4.9-54.2) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| <10 | 12 (26.1) | 14 (30.4) | 26 (28.3) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 10-20 | 24 (52.2) | 20 (43.5) | 44 (47.8) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| >20 | 10 (21.7) | 12 (26.1) | 22 (23.9) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T stage | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T1 | 7 (15.2) | 8 (17.4) | 15 (16.3) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T2 | 25 (54.3) | 26 (56.5) | 51 (55.4) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T3 | 14 (30.4) | 12 (26.1) | 26 (28.3) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| NCCN risk group | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Low risk | 16 (34.8) | 15 (32.6) | 31 (33.7) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Intermediate risk | 19 (41.3) | 17 (37.0) | 36 (39.1) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| High risk | 11 (23.9) | 14 (30.4) | 25 (27.2) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HFRT, hypofractionated radiotherapy; CFRT, conventionally fractionated radiotherapy; PSA, prostate-specific antigen; ADT, androgen-deprivation therapy. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| NCCN, National Comprehensive Cancer Network. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
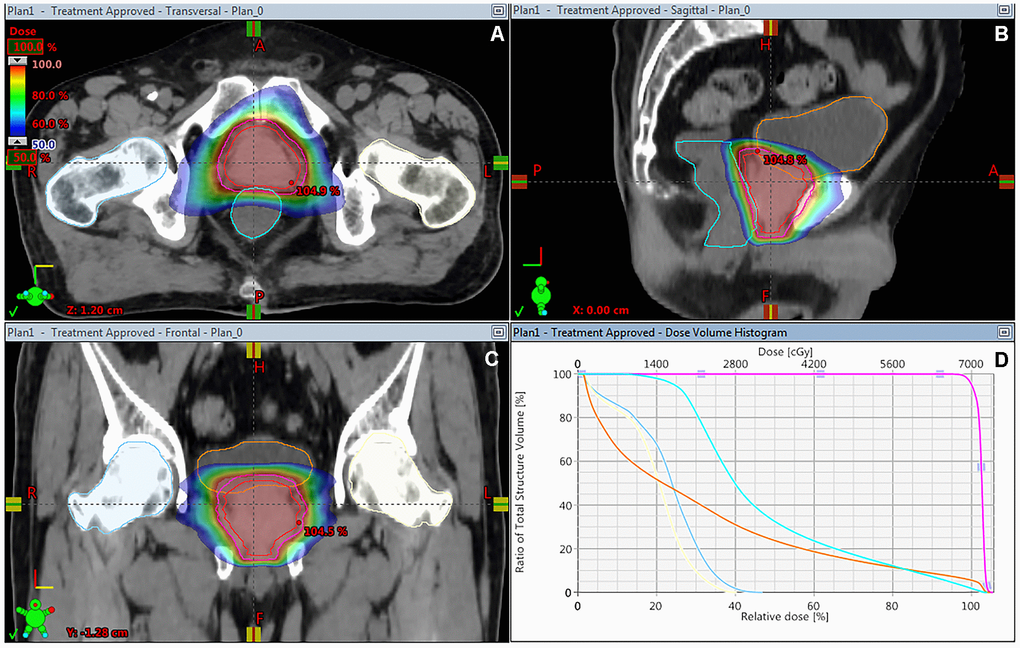
Figure 1. Dose distribution on the transverse (A), sagittal (B) and coronal (C) CT imaging, and DVH (D) of treatment planning of hypofractionated VMAT for patients with prostate cancer. DVH, dose–volume histogram; VMAT, volumetric modulated arc radiotherapy.
Acute toxicity
Median follow-up was 26 months (range, 3–39 months). Table 2 lists the acute and late GI and GU toxicities in the two groups. No patient developed grade 3–5 toxicities in either group during or after radiotherapy. Grade 1 acute toxicities occurred in 74/92 (80.4%) patients and grade 2 acute toxicities in 27/92 (29.3%) patients. The most common GI and GU toxicities were diarrhea and urinary frequency and urgency. The incidence of grade 1 and higher acute GI and GU toxicities was not significantly different between the two groups (P = 0.063).
Table 2. Comparison of acute and late gastrointestinal and genitourinary toxicities between the HFRT and CFRT groups.
| Toxicity | HFRT | CFRT | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| No (%) | No (%) | P | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Acute toxicity | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Genitourinary (GU) | 0.132 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Grade 0 | 16 (34.8) | 20 (43.5) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Grade 1 | 22 (47.8) | 20 (43.5) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Grade 2 | 8 (17.4) | 6 (13.0) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gastrointestinal (GI) | 0.190 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Grade 0 | 21 (45.7) | 26 (56.5) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Grade 1 | 17 (37.0) | 15 (32.6) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Grade 2 | 8 (17.4) | 5 (10.9) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Late toxicity | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Genitourinary (GU) | 0.496 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Grade 0 | 40 (87.0) | 39 (84.8) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Grade 1 | 6 (13.0) | 5 (10.9) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Grade 2 | 0 (0.0) | 2 (4.3) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gastrointestinal (GI) | 0.915 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Grade 0 | 40 (87.0) | 41 (89.1) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Grade 1 | 3 (6.5) | 3 (6.5) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Grade 2 | 3 (6.5) | 2 (4.3) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HFRT, hypofractionated radiotherapy; CFRT, conventionally fractionated radiotherapy. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Late toxicity
The incidence of grade 2 and higher late GI/GU toxicity was low in both groups (Table 2). The 5-year cumulative incidence of RTOG grade 2 or higher GI/GU toxicity at 2 years was 7.6% in the HFRT group vs. 10.3% in the CFRT group (P = 0.707; Figure 2). The most common late radiation-related toxicity was mild (RTOG grade 0-1) rectal bleeding. No late grade 3 toxicity occurred.
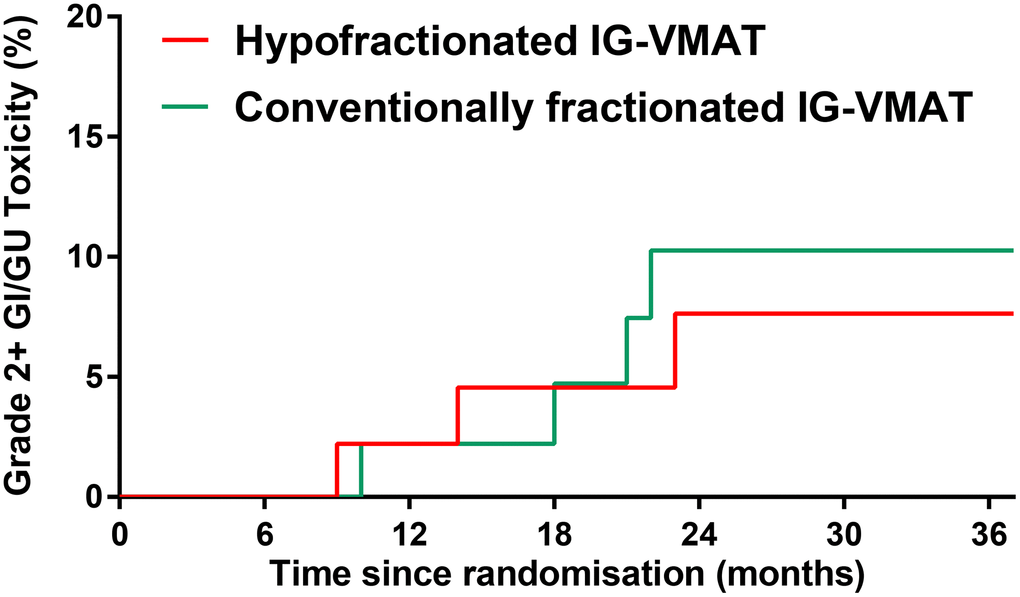
Figure 2. RTOG grade 2 or higher late GI/GU toxicity in patients treated with hypofractionated IG-VMAT versus conventionally fractionated IG-VMAT. RTOG, Radiation Therapy Oncology Group; GI, gastrointestinal; GU, genitourinary; IG, imaging-guided; volumetric-modulated arc therapy, VMAT.
Discussion
This phase II randomized, non-comparative trial aimed to determine the toxicity profile of hypofractionated IG-VMAT in patients with localized prostate cancer in China. The risk of GI/GU toxicities was found to be low in patients treated with either hypofractionated VMAT or conventionally fractionated VMAT. The bRFS was also comparable with the two treatment regimens. Although this was a single-center study on a small sample, the findings support the use of hypofractionated IG-VMAT for treatment of localized prostate cancer in China.
External beam radiotherapy is a definitive treatment option for localized prostate cancer. Moderate hypofractionation is defined as radiotherapy with a fraction size between 2.4 Gy and 3.4 Gy [16–25]. The radiobiological rationale that a slow proliferation rate of prostate cancer is reflected by an α/β ratio of 1.5, similar to that of adjacent toxic effect–limiting OAR, suggests that hypofractionated radiation regimens will provide comparable disease control without increasing toxic effects. Several randomized controlled studies have compared moderate HFRT with CFRT for prostate cancer and demonstrated equivalent efficacy and toxicity with the two modalities [16–25]. The largest randomized trials to date included the CHHiP trial [20], the Dutch Hypofractionated versus Conventionally Fractionated Radiotherapy for Patients with Prostate Cancer (HYPRO) trial [21], the RTOG-0415 trial [22], and the Prostate Fractionated Irradiation Trial (PROFIT) [24]. RTOG-0415 randomized 1115 men with low-risk prostate cancer to receive either 73.8 Gy in 41 fractions of 1.8 Gy over 8.2 weeks or 70 Gy in 28 fractions of 2.5 Gy over 5.6 weeks. After median follow-up of 5.8 years, disease-free survival was found to be comparable between the two treatment groups [7]. IGRT was introduced only during the latter stages of the recently published CHHiP trial [27], and was shown to provide dosimetric benefit with minimal toxicity [15]. IMRT, often in the form of VMAT, achieves excellent target coverage, dose conformity, normal tissue sparing, and reduced treatment delivery time in prostate cancer. In the present study, in patients treated with hypofractionated IG-VMAT or conventionally fractionated IG-VMAT, we confirmed that the moderate fractionation dose of 2.5 Gy per fraction provides favorable biochemical control with low toxicity.
Improvements in imaging guidance and the adaptation of IMRT or VMAT have increased confidence in the precision of targeting of the prostate gland and the ability to minimize radiation to surrounding normal tissue. This has allowed successful dose escalation and increased interest in hypofractionation for treatment of prostate cancer. In the present study, overall toxicity in the hypofractionated IG-VMAT group was similar to that in the conventionally fractionated IG-VMAT group. Consistent with previous studies [15, 31, 32], we found the cumulative incidence of late GI/GU toxicity to be low in both fractionation groups in the IGRT setting. Previous randomized controlled trials have demonstrated equivalent toxicities with HFRT and CFRT in prostate cancer patients in the modern radiotherapy era [21, 24, 28, 29]. Only one study from RTOG-0415 found higher incidence of late grade 2 GI/GU toxicity in patients treated with HFRT, but this was probably due to the use of three-dimensional conformal radiotherapy in 21% of patients [22]. In studies with longer follow-up, no statistically significant difference in toxicity and quality of life have been observed between the two treatments [23].
One of the strengths of this study was that IG-VMAT was delivered with the latest radiotherapeutic techniques. In addition, the enrollment of a heterogeneous population makes the findings generalizable. Thus, the findings provide high-level evidence affirming that HFRT could be a practice standard for Chinese patients with localized prostate cancer [16–18, 22–25].
The main limitation of this trial was that the small sample size and the low frequency of events made efficacy analysis difficult; however, the results are entirely consistent with other trials [16–25]. Second, only bRFS was used to assess effectiveness of IG-VMAT. The data for bRFS or overall survival is not yet mature due to the short follow-up time. Despite these limitations, the results of this study can be helpful when designing a large multicenter phase III randomized trial of imaging-guided moderately hypofractionated VMAT versus conventionally fractionated VMAT in the modern radiotherapy era.
In conclusion, radiation-related toxicities and biochemical control appear to be favorable with both hypofractionated (70 Gy/2.5Gy/28f) and conventionally fractionated (80 Gy/2Gy/40f) IG-VMAT in Chinese patients with localized prostate cancer. The use of HFRT can benefit both the healthcare system and the patient by decreasing treatment costs and treatment time; these benefits can be particularly important in countries with limited resources.
Materials and Methods
Patients and eligibility
This randomized non-comparative phase II trial recruited patients from a single academic hospital in China. Male patients were eligible if 1) they were aged ≥50 years, 2) had histologically confirmed prostate adenocarcinoma, 3) had good performance status (Eastern Cooperative Oncology Group [ECOG] score 0-1), and 4) had clinical stage T1-3 disease by the 2009 American Joint Committee on Cancer (AJCC) criteria. Exclusion criteria were 1) clinical stage T4, 2) evidence of nodal or distant metastases, 3) previous pelvic radiation therapy, or 4) previous malignancies. The clinical stage work-up included physical examination; biochemistry; PSA; computed tomography (CT) scans of the chest, abdomen, and pelvis; magnetic resonance imaging (MRI) of the pelvis and prostate; and whole-body bone scan.
Patients were categorized as low-risk, intermediate-risk, or high-risk according to the National Comprehensive Cancer Network (NCCN) guidelines [33]. Low-risk patients had cT1c–T2aN0M0, Gleason score ≤6, and PSA <10 ng/mL. Intermediate-risk patients had at least one of the following risk factors: T2c, Gleason score 7, or PSA 10-20 ng/mL. High-risk patients had at least one of the following risk factors: stage T3a or T3b, Gleason score ≥8, or PSA >20 ng/mL.
Study design and treatment regimens
A computer-generated central randomization schedule was used to assign eligible patients in a 1:1 ratio to receive either conventionally fractionated IG-VMAT at 80 Gy in 40 fractions (2.0 Gy/fraction) over 8 weeks (CFRT group) or hypofractionated IG-VMAT at 70 Gy in 28 fractions (2.5 Gy/fraction) over 5.6 weeks (HFRT group), The total dose of 70 Gy in 28 fractions is biologically equivalent to 80 Gy in 40 fractions according to the α/β ratio of 1.5 Gy for prostate cancer.
As per NCCN guidelines [33], intermediate-risk and high-risk patients received, respectively, 4-6 months and 24 months of neoadjuvant/concurrent androgen deprivation therapy.
Ethical compliance
All procedures were in conformance with the Helsinki declaration. The study protocol was approved by the local ethics committee and was registered at http://clinicaltrials.gov/ (no. NCT02934685). All patients were explained and signed written informed consent at enrollment.
Radiotherapy technique
All patients underwent CT simulation with 3-mm slice thickness. Patients were instructed to have a full bladder and empty rectum at CT simulation and during the period of irradiation. The CTV included the whole prostate with or without seminal vesicles, depending on whether the patient was classified as high-, intermediate- or low- risk by NCCN criteria. The planning target volume (PTV) was the CTV plus a surrounding 5-mm margin—except posteriorly, where only a 3-mm margin was included. The bladder, rectum, small bowel, and femoral heads were delineated as organs at risk (OAR). The rectum volume included the entire rectal wall and lumen from the anus to the rectosigmoid flexure. The bladder was contoured from its base to the dome.
IG-VMAT was delivered without placement of fiducial markers. All doses were prescribed to a minimum isodose line encompassing 95% of the PTV. Planning CT and the verification cone-beam computed tomography (CBCT) were registered first with automatic bony registration, followed by a manual registration based on the soft-tissue alignment and prostate position in the CBCT. CBCT was performed daily in the HFRT group whereas, in the CFRT group, CBCT was performed daily during the first five fractions and, thereafter, three times a week.
Dose constraints for the HFRT group were as follows: volume of bladder receiving 25 Gy and 50 Gy were <65% and <55%, respectively, and volume of rectum receiving 25 Gy and 50 Gy were <60% Gy and <50%, respectively. Dose constraints for the CFRT group were as follows: volume of bladder receiving 25 Gy and 50 Gy were <75% and <65%, respectively, and volume of rectum receiving 25 Gy and 50 Gy were <70% and <60%, respectively.
Evaluation and endpoints
Assessment of toxicity was performed every week during treatment. Patients were followed up at 1 month after completion of chemotherapy, then every 3 months for 2 years, every 6 months for the next 5 years, and yearly thereafter.
The primary endpoint was acute and late toxicity. Additional endpoints included biochemical relapse–free survival (bRFS), which was calculated from the date of randomization to the date of biochemical (PSA) failure, disease progression, relapse, death, or last follow-up. The Phoenix definition was used for PSA relapse (absolute nadir plus 2 ng/mL). Acute gastrointestinal (GI) and genitourinary (GU) toxicities were evaluated according to the Common Terminology Criteria for Adverse Events (CTCAE) version 3.0. Late radiation toxicity was assessed using the Radiation Therapy Oncology Group (RTOG)/the European Organization for Research and Treatment of Cancer (EORTC) late radiation morbidity scale [34].
Statistical analysis
This hypofractionated IG-VMAT study was non-comparative and powered to assess toxicity independently in each treatment technique group, using Simon single-stage design with exact p-values [35]. The primary endpoint was late RTOG GI/GU toxicity at 2 years from start of radiation therapy. The late RTOG grade ≥2 GI/GU toxicity-free rate at 2 years was set at 80%, with an expected rate of 92% [15]. With a 5% one-sided alpha and 80% power, 43 patients in the hypofractionated IG-VMAT group were required. Assuming that there would be a 5% non-compliance rate, we enrolled 46 patients per group. An equal number of patients in the conventionally fractionated IG-VMAT group was sought to obtain prospectively collected toxicity data for standard treatment, summing in a total of sample size of 92 patients.
The chi-square or Fisher exact test was used for testing relationships between variables. Time to first occurrence of late radiation-induced side effects was analyzed using the Kaplan–Meier method to calculate the cumulative proportion with events reported on 2-year assessment. The log-rank test was used to compare hypofractionated IG-VMAT and conventionally fractionated IG-VMAT. Statistical calculations were performed using SPSS 16.0 (SPSS Inc., Chicago, IL, USA).
Author Contributions
Qiu-Zi Zhong, Gao-Feng Li, and Xiu Xia: conception and design. Qiu-Zi Zhong, Hong Gao, Yong-Gang Xu, and Ting Zhao: collection and assembly of data, Qiu-Zi Zhong, Gao-Feng Li, Qin-Hong Wu, Dan Wang, Hai-Lei Lin, and Xiang-Yan Sha: data analysis and interpretation. All authors wrote the manuscript and final approval of manuscript.
Acknowledgment
The authors would like to thank Blot Info and Tech (Beijing) Co. Ltd. for technical support for this project.
Conflicts of Interest
Gao-Feng Li has received grants from the Varian Medical Systems, outside the submitted work. All other authors declare no conflicts of interest.
Funding
This work was supported by the National Natural Science Foundation of China (Grant nos. 81101694 and 81372415) and VARIAN Research Foundation. The funding sources had no influence on the design, performance, or reporting of this study.
References
- 1. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018; 68:394–424. https://doi.org/10.3322/caac.21492 [PubMed]
- 2. Center MM, Jemal A, Lortet-Tieulent J, Ward E, Ferlay J, Brawley O, Bray F. International variation in prostate cancer incidence and mortality rates. Eur Urol. 2012; 61:1079–92. https://doi.org/10.1016/j.eururo.2012.02.054 [PubMed]
- 3. Culp MB, Soerjomataram I, Efstathiou JA, Bray F, Jemal A. Recent global patterns in prostate cancer incidence and mortality rates. Eur Urol. 2020; 77:38–52. https://doi.org/10.1016/j.eururo.2019.08.005 [PubMed]
- 4. Chen W, Zheng R, Baade PD, Zhang S, Zeng H, Bray F, Jemal A, Yu XQ, He J. Cancer statistics in China, 2015. CA Cancer J Clin. 2016; 66:115–32. https://doi.org/10.3322/caac.21338 [PubMed]
- 5. Gu XY, Zheng RS, Zhang SW, Zeng HM, Sun KX, Zou XN, Xia CF, Yang ZX, Li H, Chen WQ, He J. [Analysis on the trend of prostate cancer incidence and age change in cancer registration areas of China, 2000 to 2014]. Zhonghua Yu Fang Yi Xue Za Zhi. 2018; 52:586–92. https://doi.org/10.3760/cma.j.issn.0253-9624.2018.06.006 [PubMed]
- 6. Chen R, Xie LP, Zhou LQ. Current status of prostate biopsy in Chinese Prostate Cancer Consortium member hospitals. Zhonghua Mi Niao Wai Ke Za Zhi. 2015; 36:342–45.
- 7. Chen R, Ren S, Yiu MK, Fai NC, Cheng WS, Ian LH, Naito S, Matsuda T, Kehinde E, Kural A, Chiu JY, Umbas R, Wei Q, et al, and Chinese Prostate Cancer Consortium. Prostate cancer in Asia: a collaborative report. Asian J Urol. 2014; 1:15–29. https://doi.org/10.1016/j.ajur.2014.08.007 [PubMed]
- 8. Huynh-Le MP, Myklebust TÅ, Feng CH, Karunamuni R, Johannesen TB, Dale AM, Andreassen OA, Seibert TM. Age dependence of modern clinical risk groups for localized prostate cancer-a population-based study. Cancer. 2020; 126:1691–99. https://doi.org/10.1002/cncr.32702 [PubMed]
- 9. Kuban DA, Tucker SL, Dong L, Starkschall G, Huang EH, Cheung MR, Lee AK, Pollack A. Long-term results of the M. D. Anderson randomized dose-escalation trial for prostate cancer. Int J Radiat Oncol Biol Phys. 2008; 70:67–74. https://doi.org/10.1016/j.ijrobp.2007.06.054 [PubMed]
- 10. Morgan SC, Hoffman K, Loblaw DA, Buyyounouski MK, Patton C, Barocas D, Bentzen S, Chang M, Efstathiou J, Greany P, Halvorsen P, Koontz BF, Lawton C, et al. Hypofractionated radiation therapy for localized prostate cancer: executive summary of an ASTRO, ASCO and AUA evidence-based guideline. J Urol. 2019; 201:528–34. https://doi.org/10.1097/JU.0000000000000071 [PubMed]
- 11. Michalski JM, Moughan J, Purdy J, Bosch W, Bruner DW, Bahary JP, Lau H, Duclos M, Parliament M, Morton G, Hamstra D, Seider M, Lock MI, et al. Effect of standard vs dose-escalated radiation therapy for patients with intermediate-risk prostate cancer: the NRG oncology RTOG 0126 randomized clinical trial. JAMA Oncol. 2018; 4:e180039. https://doi.org/10.1001/jamaoncol.2018.0039 [PubMed]
- 12. Zietman AL, DeSilvio ML, Slater JD, Rossi CJ
Jr , Miller DW, Adams JA, Shipley WU. Comparison of conventional-dose vs high-dose conformal radiation therapy in clinically localized adenocarcinoma of the prostate: a randomized controlled trial. JAMA. 2005; 294:1233–39. https://doi.org/10.1001/jama.294.10.1233 [PubMed] - 13. Pan HY, Jiang J, Hoffman KE, Tang C, Choi SL, Nguyen QN, Frank SJ, Anscher MS, Shih YT, Smith BD. Comparative toxicities and cost of intensity-modulated radiotherapy, proton radiation, and stereotactic body radiotherapy among younger men with prostate cancer. J Clin Oncol. 2018; 36:1823–30. https://doi.org/10.1200/JCO.2017.75.5371 [PubMed]
- 14. Anwar M, Weinberg V, Chang AJ, Hsu IC, Roach M 3rd, Gottschalk A. Hypofractionated SBRT versus conventionally fractionated EBRT for prostate cancer: comparison of PSA slope and nadir. Radiat Oncol. 2014; 9:42. https://doi.org/10.1186/1748-717X-9-42 [PubMed]
- 15. Zelefsky MJ, Kollmeier M, Cox B, Fidaleo A, Sperling D, Pei X, Carver B, Coleman J, Lovelock M, Hunt M. Improved clinical outcomes with high-dose image guided radiotherapy compared with non-IGRT for the treatment of clinically localized prostate cancer. Int J Radiat Oncol Biol Phys. 2012; 84:125–29. https://doi.org/10.1016/j.ijrobp.2011.11.047 [PubMed]
- 16. Lukka H, Hayter C, Julian JA, Warde P, Morris WJ, Gospodarowicz M, Levine M, Sathya J, Choo R, Prichard H, Brundage M, Kwan W. Randomized trial comparing two fractionation schedules for patients with localized prostate cancer. J Clin Oncol. 2005; 23:6132–38. https://doi.org/10.1200/JCO.2005.06.153 [PubMed]
- 17. Arcangeli G, Saracino B, Arcangeli S, Gomellini S, Petrongari MG, Sanguineti G, Strigari L. Moderate hypofractionation in high-risk, organ-confined prostate cancer: final results of a phase III randomized trial. J Clin Oncol. 2017; 35:1891–97. https://doi.org/10.1200/JCO.2016.70.4189 [PubMed]
- 18. Yeoh EE, Botten RJ, Butters J, Di Matteo AC, Holloway RH, Fowler J. Hypofractionated versus conventionally fractionated radiotherapy for prostate carcinoma: final results of phase III randomized trial. Int J Radiat Oncol Biol Phys. 2011; 81:1271–78. https://doi.org/10.1016/j.ijrobp.2010.07.1984 [PubMed]
- 19. Pollack A, Walker G, Horwitz EM, Price R, Feigenberg S, Konski AA, Stoyanova R, Movsas B, Greenberg RE, Uzzo RG, Ma C, Buyyounouski MK. Randomized trial of hypofractionated external-beam radiotherapy for prostate cancer. J Clin Oncol. 2013; 31:3860–68. https://doi.org/10.1200/JCO.2013.51.1972 [PubMed]
- 20. Dearnaley D, Syndikus I, Mossop H, Khoo V, Birtle A, Bloomfield D, Graham J, Kirkbride P, Logue J, Malik Z, Money-Kyrle J, O’Sullivan JM, Panades M, et al, and CHHiP Investigators. Conventional versus hypofractionated high-dose intensity-modulated radiotherapy for prostate cancer: 5-year outcomes of the randomised, non-inferiority, phase 3 CHHiP trial. Lancet Oncol. 2016; 17:1047–60. https://doi.org/10.1016/S1470-2045(16)30102-4 [PubMed]
- 21. Incrocci L, Wortel RC, Alemayehu WG, Aluwini S, Schimmel E, Krol S, van der Toorn PP, Jager H, Heemsbergen W, Heijmen B, Pos F. Hypofractionated versus conventionally fractionated radiotherapy for patients with localised prostate cancer (HYPRO): final efficacy results from a randomised, multicentre, open-label, phase 3 trial. Lancet Oncol. 2016; 17:1061–69. https://doi.org/10.1016/S1470-2045(16)30070-5 [PubMed]
- 22. Lee WR, Dignam JJ, Amin MB, Bruner DW, Low D, Swanson GP, Shah AB, D’Souza DP, Michalski JM, Dayes IS, Seaward SA, Hall WA, Nguyen PL, et al. Randomized phase III noninferiority study comparing two radiotherapy fractionation schedules in patients with low-risk prostate cancer. J Clin Oncol. 2016; 34:2325–32. https://doi.org/10.1200/JCO.2016.67.0448 [PubMed]
- 23. Bruner DW, Pugh SL, Lee WR, Hall WA, Dignam JJ, Low D, Swanson GP, Shah AB, Malone S, Michalski JM, Dayes IS, Seaward SA, Nguyen PL, et al. Quality of life in patients with low-risk prostate cancer treated with hypofractionated vs conventional radiotherapy: a phase 3 randomized clinical trial. JAMA Oncol. 2019; 5:664–70. https://doi.org/10.1001/jamaoncol.2018.6752 [PubMed]
- 24. Catton CN, Lukka H, Gu CS, Martin JM, Supiot S, Chung PW, Bauman GS, Bahary JP, Ahmed S, Cheung P, Tai KH, Wu JS, Parliament MB, et al. Randomized trial of a hypofractionated radiation regimen for the treatment of localized prostate cancer. J Clin Oncol. 2017; 35:1884–90. https://doi.org/10.1200/JCO.2016.71.7397 [PubMed]
- 25. Hoffman KE, Voong KR, Levy LB, Allen PK, Choi S, Schlembach PJ, Lee AK, McGuire SE, Nguyen Q, Pugh TJ, Frank SJ, Kudchadker RJ, Du W, Kuban DA. Randomized Trial of Hypofractionated, Dose-Escalated, Intensity-Modulated Radiation Therapy (IMRT) Versus Conventionally Fractionated IMRT for Localized Prostate Cancer. J Clin Oncol. 2018; 36:2943–49. https://doi.org/10.1200/JCO.2018.77.9868 [PubMed]
- 26. Stokes WA, Kavanagh BD, Raben D, Pugh TJ. Implementation of hypofractionated prostate radiation therapy in the United States: a national cancer database analysis. Pract Radiat Oncol. 2017; 7:270–78. https://doi.org/10.1016/j.prro.2017.03.011 [PubMed]
- 27. Murray J, Griffin C, Gulliford S, Syndikus I, Staffurth J, Panades M, Scrase C, Parker C, Khoo V, Dean J, Mayles H, Mayles P, Thomas S, et al, and CHHiP Investigators. A randomised assessment of image guided radiotherapy within a phase 3 trial of conventional or hypofractionated high dose intensity modulated radiotherapy for prostate cancer. Radiother Oncol. 2020; 142:62–71. https://doi.org/10.1016/j.radonc.2019.10.017 [PubMed]
- 28. Widmark A, Gunnlaugsson A, Beckman L, Thellenberg-Karlsson C, Hoyer M, Lagerlund M, Kindblom J, Ginman C, Johansson B, Björnlinger K, Seke M, Agrup M, Fransson P, et al. Ultra-hypofractionated versus conventionally fractionated radiotherapy for prostate cancer: 5-year outcomes of the HYPO-RT-PC randomised, non-inferiority, phase 3 trial. Lancet. 2019; 394:385–95. https://doi.org/10.1016/S0140-6736(19)31131-6 [PubMed]
- 29. Brand DH, Tree AC, Ostler P, van der Voet H, Loblaw A, Chu W, Ford D, Tolan S, Jain S, Martin A, Staffurth J, Camilleri P, Kancherla K, et al, and PACE Trial Investigators. Intensity-modulated fractionated radiotherapy versus stereotactic body radiotherapy for prostate cancer (PACE-B): acute toxicity findings from an international, randomised, open-label, phase 3, non-inferiority trial. Lancet Oncol. 2019; 20:1531–43. https://doi.org/10.1016/S1470-2045(19)30569-8 [PubMed]
- 30. Greco C, Pares O, Pimentel N, Louro V, Morales J, Nunes B, Vasconcelos AL, Antunes I, Kociolek J, Stroom J, Viera S, Mateus D, Cardoso MJ, et al. Target motion mitigation promotes high-precision treatment planning and delivery of extreme hypofractionated prostate cancer radiotherapy: results from a phase II study. Radiother Oncol. 2020; 146:21–28. https://doi.org/10.1016/j.radonc.2020.01.029 [PubMed]
- 31. Wilkins A, Mossop H, Syndikus I, Khoo V, Bloomfield D, Parker C, Logue J, Scrase C, Patterson H, Birtle A, Staffurth J, Malik Z, Panades M, et al. Hypofractionated radiotherapy versus conventionally fractionated radiotherapy for patients with intermediate-risk localised prostate cancer: 2-year patient-reported outcomes of the randomised, non-inferiority, phase 3 CHHiP trial. Lancet Oncol. 2015; 16:1605–16. https://doi.org/10.1016/S1470-2045(15)00280-6 [PubMed]
- 32. Spratt DE, Pei X, Yamada J, Kollmeier MA, Cox B, Zelefsky MJ. Long-term survival and toxicity in patients treated with high-dose intensity modulated radiation therapy for localized prostate cancer. Int J Radiat Oncol Biol Phys. 2013; 85:686–92. https://doi.org/10.1016/j.ijrobp.2012.05.023 [PubMed]
- 33. National Comprehensive Cancer Network. NCCN Guidelines: prostate cancer version 1. https://www.nccn.org/professionals/physician_gls/default.aspx#prostate. 2020.
- 34. Cox JD, Stetz J, Pajak TF. Toxicity criteria of the radiation therapy oncology group (RTOG) and the European organization for research and treatment of cancer (EORTC). Int J Radiat Oncol Biol Phys. 1995; 31:1341–46. https://doi.org/10.1016/0360-3016(95)00060-C [PubMed]
- 35. A’Hern RP. Sample size tables for exact single-stage phase II designs. Stat Med. 2001; 20:859–66. https://doi.org/10.1002/sim.721 [PubMed]