Introduction
Growing evidence has shown that cigarette smoking is a potential modifiable risk factor for Alzheimer’s Disease (AD) [1]. However, the neurobiological mechanism by which cigarette smoking increases the risk of AD remains unclear. To address this question, several studies have attempted to examine the effect of cigarette smoking on AD-related markers, such as global cognition, verbal memory, hippocampal volume, brain glucose metabolism, and AD pathologies in the brain [2–8].
Mild cognitive impairment (MCI) is regarded as a prodromal state for AD dementia [9]. MCI patients have a very high conversion rate to AD dementia, with 50% to 70% progressing to dementia within the next five to seven years after a diagnosis of MCI [10]. Surprisingly, the evidence on the effect of cigarette smoking on AD-related markers in MCI is scarce. In addition, no prior studies have attempted to examine the association of cigarette smoking with change in brain glucose metabolism or CSF AD pathologies over time in MCI patients. Therefore, in the present study, we aimed to systematically investigate the association of cigarette smoking with change in global cognition, verbal memory, functional performance, hippocampal volume, entorhinal cortex volume, brain glucose metabolism, and CSF AD pathologies over time in MCI subjects. Our findings may contribute to a better understanding of the underlying mechanism by which cigarette smoking increases the risk of developing AD.
Results
Demographical and clinical variables by smoking history
Table 1 demonstrates the demographical and clinical information by smoking history. At baseline, there were 870 subjects with MCI, including 618 non-smokers and 252 smokers. Compared to non-smokers, smokers were less likely to be females while more likely to be APOE4 carriers (p < 0.05). However, there was no difference in age or education between two groups (all p > 0.05). Smokers had lower MMSE score (lower score indicates more severe cognitive impairment) and RAVLT total score (lower score indicates more severe cognitive impairment) than non-smokers. Further, smokers showed more severe functional impairment as assesses by FAQ (higher score indicates more severe functional impairment) compared to non-smokers. In term of the difference in neuroimaging markers, smoking was associated with lower HpVR and EVR, but not FDG SUVRs. There was no significant difference in CSF AD pathologies between two group. In addition, the number of individuals with MMSE data at each follow-up visit is also presented in Table 1.
Table 1. Demographical and clinical variables by smoking history.
| Variables | Non-smokers (n = 618) | Smokers (n = 252) | P value | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age, years | 72.7 ± 7.66 | 73.8 ± 7.33 | 0.08 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Education, years | 16 ± 2.86 | 15.7 ± 2.79 | 0.22 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Female gender, n (%) | 270 (43.7) | 85 (33.7) | 0.007 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| APOE4 carriers, n (%) | 296 (47.9) | 142 (56.3) | 0.02 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MMSE | 27.7 ± 1.79 | 27.3 ± 1.83 | < 0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FAQ | 2.94 ± 3.88 | 3.8 ± 4.57 | 0.009 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| RAVLT total score | 34.8 ± 10.7 | 32.6 ± 10.6 | 0.002 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FDG SUVRs | 1.25 ± 0.14 | 1.23 ± 0.12 | 0.13 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HpVR | 4.52 ± 0.8 | 4.23 ± 0.75 | < 0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| EVR | 2.31 ± 0.49 | 2.21 ± 0.46 | 0.02 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CSF Aβ42, pg/ml | 171 ± 52 | 171 ± 52 | 0.9 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CSF total tau, pg/ml | 89.6 ± 51.6 | 93.3 ± 63.4 | 0.8 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CSF p-tau, pg/ml | 40.2 ± 23.4 | 37.1 ± 21.2 | 0.12 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sample size at each visita | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Baseline | 618 | 252 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 0.5 | 583 | 232 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1 | 573 | 221 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1.5 | 197 | 132 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2 | 485 | 194 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3 | 412 | 164 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 4 | 298 | 108 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 5 | 196 | 66 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 6 | 153 | 60 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 7 | 109 | 36 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 8 | 55 | 24 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 9 | 30 | 12 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 10 | 24 | 4 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 11 | 14 | 2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 12 | 10 | 1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 13 | 2 | 0 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| aThe number of individuals with MMSE data at each follow-up visit. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: MMSE: Mini-mental state examination; FAQ: Functional Activities Questionnaire; RAVLT: Rey Auditory Verbal Learning Test; FDG SUVRs: fluorodeoxyglucose standardized uptake values ratios; HpVR: Hippocampal volume ratio; EVR: Entorhinal cortex volume ratio; Aβ: β-amyloid; t-tau: total tau; p-tau: phosphorylated tau. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Results of linear mixed models
To examine whether smoking was associated with changes in cognitive performance (MMSE, FAQ and RAVLT total score), neuroimaging markers (HpVR, EVR and FDG SUVRs) and CSF AD pathologies (CSF Aβ42, t-tau and p-tau levels), we fitted several linear mixed models for each outcome (Tables 2–4 and Figures 1–3). In MCI, we found that smoking was associated with changes in FAQ (estimate: 0.13, p = 0.03) and EVR (estimate: -0.02, p = 0.006), but not MMSE (estimate: -0.03, p = 0.42), RAVLT total score (estimate: 0.01, p = 0.89), HpVR (estimate: 0.0002, p = 0.96), FDG SUVRs (estimate: 0.003, p = 0.13), CSF Aβ42 (estimate: -0.72, p = 0.37), t-tau (estimate: 0.89, p = 0.4) or p-tau levels(estimate: -0.07, p = 0.93). Specifically, smoking was associated with faster decline in functional performance as assessed by FAQ (Table 2 and Figure 1). In addition, compared to non-smokers, smokers also showed steeper decline in EVR (Table 3 and Figure 2).
Table 2. Results of linear mixed models with cognitive and functional performance as dependent variable.
| Predictor | MMSE | FAQ | RAVLT total score | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Estimate (SE) | P value | Estimate (SE) | P value | Estimate (SE) | P value | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smoker × time | -0.03(0.04) | 0.42 | 0.13 (0.06) | 0.03 | 0.01 (0.09) | 0.89 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age × time | -0.01(0.002) | < 0.001 | 0.04 (0.004) | < 0.001 | -0.06 (0.006) | < 0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Female × time | -0.18 (0.04) | < 0.001 | 0.22 (0.06) | < 0.001 | -0.43 (0.09) | < 0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| APOE4 carriers × time | -0.61 (0.03) | < 0.001 | 1.13 (0.05) | < 0.001 | -0.88 (0.08) | < 0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Education × time | -0.01(0.006) | 0.2 | 0.009 (0.01) | 0.38 | -0.01 (0.015) | 0.48 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: MMSE: Mini-mental state examination; FAQ: Functional Activities Questionnaire; RAVLT: Rey Auditory Verbal Learning Test. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 3. Results of linear mixed models with neuroimaging markers as dependent variable.
| Predictor | FDG SUVRs | HpVR | EVR | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Estimate (SE) | P value | Estimate (SE) | P value | Estimate (SE) | P value | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smoker × time | 0.003 (0.002) | 0.13 | 0.0002 (0.004) | 0.96 | -0.02 (0.006) | 0.006 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age × time | -0.0002 (0.0001) | 0.1 | -0.002 (0.0003) | < 0.001 | -0.001 (0.0004) | 0.002 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Female × time | -0.01 (0.002) | < 0.001 | -0.04 (0.004) | < 0.001 | -0.02 (0.006) | < 0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| APOE4 carriers × time | -0.01 (0.002) | < 0.001 | -0.06 (0.004) | < 0.001 | -0.04 (0.005) | < 0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Education × time | -0.001 (0.0003) | 0.01 | -0.0004 (0.0006) | 0.54 | -0.002 (0.0009) | 0.006 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: FDG SUVRs: fluorodeoxyglucose standardized uptake values ratios; HpVR: Hippocampal volume ratio; EVR: Entorhinal cortex volume ratio. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 4. Results of linear mixed models with CSF AD pathologies as dependent variable.
| Predictor | CSF Aβ42 | CSF t-tau | CSF p-tau | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Estimate (SE) | P value | Estimate (SE) | P value | Estimate (SE) | P value | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smoker × time | -0.72 (0.81) | 0.37 | 0.89 (1) | 0.4 | -0.07 (0.81) | 0.93 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age × time | -0.02 (0.06) | 0.66 | -0.05 (0.07) | 0.48 | -0.08 (0.06) | 0.17 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Female × time | 0.29 (0.79) | 0.71 | 1.3 (1) | 0.22 | -0.57 (0.79) | 0.47 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| APOE4 carriers × time | 1 (0.74) | 0.18 | -0.15 (0.96) | 0.87 | -0.72 (0.75) | 0.33 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Education × time | -0.14 (0.14) | 0.32 | 0.52 (0.18) | 0.004 | 0.04 (0.14) | 0.8 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: Aβ: β-amyloid; t-tau: total tau; p-tau: phosphorylated tau. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
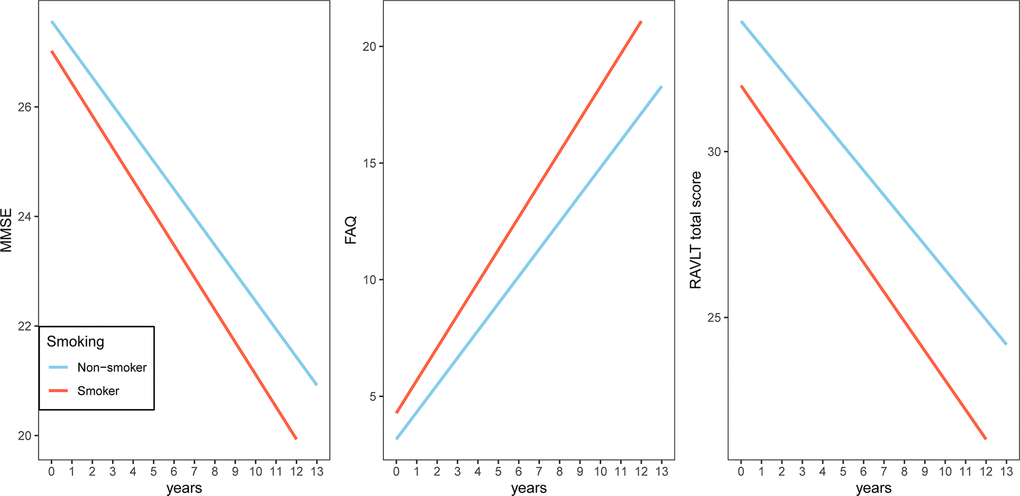
Figure 1. Association of a history of cigarette smoking with change in MMSE, FAQ and RAVLT total score over time in MCI subjects. A history of cigarette smoking was associated with change in FAQ, but not MMSE or RAVLT total score. Abbreviations: Abbreviations: MMSE: Mini-mental state examination; FAQ: Functional Activities Questionnaire; RAVLT: Rey Auditory Verbal Learning Test.
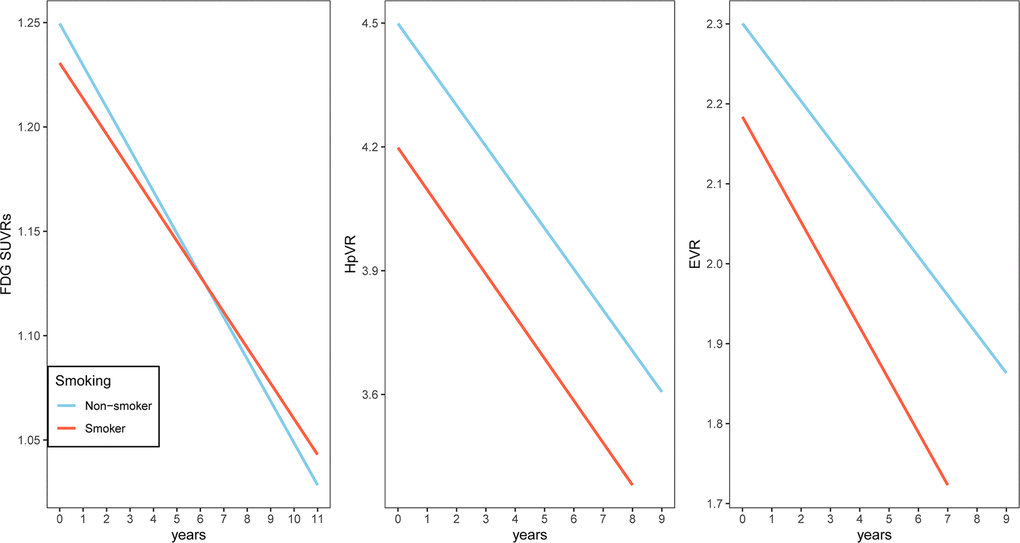
Figure 2. Association of a history of cigarette smoking with change in FDG SUVRs, HpVR and EVR over time in MCI subjects. A history of cigarette smoking was associated with change in EVR, but not FDG SUVRs or HpVR. Abbreviations: FDG SUVRs: fluorodeoxyglucose standardized uptake values ratios; HpVR: Hippocampal volume ratio; EVR: Entorhinal cortex volume ratio.
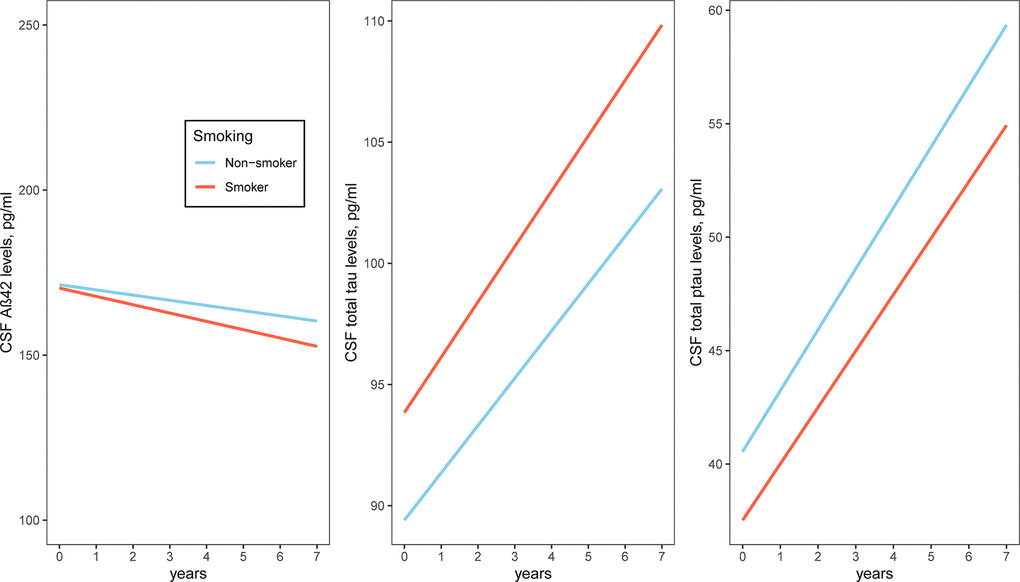
Figure 3. Association of a history of cigarette smoking with change in CSF AD pathologies over time in MCI subjects. A history of cigarette smoking was not associated with change in CSF AD pathologies. Abbreviations: Aβ: β-amyloid; t-tau: total tau; p-tau: phosphorylated tau.
Discussion
The primary findings of this longitudinal study are: (1) Among older people with MCI, smokers showed faster decline in functional performance compared to non-smokers; (2) Smokers demonstrated steeper decline in entorhinal cortex volume than non-smokers; (3) A history of cigarette smoking was not associated with change in CSF Aβ42, t-tau or p-tau levels over time at the prodromal stage of AD. Our findings may contribute to a better understanding of the underlying mechanism by which cigarette smoking increases the risk of developing AD.
The first finding that smokers had faster decline in functional performance compared to non-smokers is consistent with previous studies, which showed that smoking was associated with more impaired activities of daily living in middle-aged and older people [11, 12]. In addition, the English Longitudinal Study of Aging (ELSA) also demonstrated that among participants aged 50 or older, smoking was significantly associated with the impairment of instrumental activities of daily living [13], which is one of the key diagnostic items for dementia [14]. In the present study, we found that smoking history was associated with faster decline in functional performance in MCI subjects. To the best of our knowledge, this is the first longitudinal study to investigate the association of smoking history with change in the performance of instrumental activities of daily living at the prodromal stage of dementia.
The second finding that smokers demonstrated steeper decline in entorhinal cortex volume than non-smokers in MCI subjects is novel. In the present study, we mainly focused on two medial temporal lobe regions (hippocampus and entorhinal cortex), which are affected in the very early stage of AD [15]. However, the finding of the present study that a history of cigarette smoking was not associated with hippocampal atrophy in MCI subjects is not consistent with previous studies showing that cigarette smoking was associated with smaller volume of hippocampus in a cohort consisting of young and middle-aged participants [7]. One possible explanation for this inconsistency is that the effect of cigarette smoking on decline in hippocampus volume may be masked by the effect of AD pathologies at the prodromal stage of AD dementia.
The third finding that a history of cigarette smoking was not associated with change in CSF Aβ42, t-tau or p-tau levels over time at the prodromal stage of AD is consistent with a previously published postmortem study showing that no significant difference in levels of plaques or tangles between non-smokers and smokers was found in the midfrontal cortex of patients with AD dementia [16]. However, Tyas and colleagues observed that compared to non-smokers, smokers showed higher levels of plagues rather than tangles in the hippocampal and cerebral cortex [17]. These discrepancies may be due to several factors, such as study design (longitudinal design vs cross-sectional design), sample size and the sample (CSF vs brain tissue).
There are several potential mechanisms by which cigarette smoking affects functional decline and reduction of entorhinal cortex volume in MCI. First, chronic cigarette smoking has been found to be associated with higher risk for other cerebrovascular and cardiovascular diseases [18–21], which may contribute to the compromise of brain function and neurobiology. Second, cigarette smoking is associated with increased levels of cerebral oxidative stress [22], which may lead to neuroinflammatory responses that play a crucial role in AD pathogenesis.
Several limitations should be noted. First, compared to cognitive assessments and structural MRI data (MMSE, FAQ, RAVLT total score, HpVR and EVR), FDG and CSF AD-related biomarkers (Aβ42, t-tau, and p-tau) data had shorter follow-up duration and more missing values at each visit. Therefore, in the linear mixed models with FDG and CSF AD-related biomarkers as outcomes, the statistical power was relatively low. Future studies with larger numbers of participants with FDG and CSF AD biomarkers data at each follow-up visit or longer follow-up duration will be needed to increase the statistical power to address this issue. Second, participants of the ADNI study were highly educated and Caucasian. Thus, it may be difficult to generalize our findings to other populations. Third, given the observational nature of the present study, we cannot provide any causal evidence on the association of cigarette smoking with functional decline and reduction of entorhinal cortex volume in MCI. Future interventional studies are warranted to clarify the nature of this relationship.
In conclusion, we found that a history of cigarette smoking was associated with faster decline in functional performance and entorhinal cortex volume over time at the prodromal stage of dementia.
Materials and Methods
Alzheimer’s Disease Neuroimaging Initiative
Longitudinal data were obtained from the Alzheimer’s Disease Neuroimaging Initiative (ADNI) database. The ADNI study is a longitudinal prospective study with the aim of identifying potential biomarkers of MCI and early AD (adni.loni.usc.edu). At each ADNI site across the United States and Canada, institutional review board approved the ADNI study, and informed consent was obtained from participants.
Participants
In the present study, we included 870 subjects with MCI at baseline. MCI subjects had a Clinical Dementia Rating (CDR) [23] score of 0.5, a Mini-Mental State Examination (MMSE) [24] score of 24 or above, an objective memory decline as assessed by the Wechsler Memory Scale Logical Memory II, and no evidence of dementia. MCI subjects were further classified into two groups according to their smoking history: 618 non-smokers and 252 smokers. It should be noted that the smoker group could be further categorized into two groups: 232 former smokers and 20 active smokers. However, we combined these two groups into one due to the fact that the sample size of the active smoker group is not sufficient to conduct a longitudinal analysis.
Cognitive assessments
The MMSE, Rey Auditory Verbal Learning Test (RAVLT) total score [25], and Functional Activities Questionnaire (FAQ) [26] were utilized to assess the global cognition, verbal memory and functional performance, respectively.
Measurement of neuroimaging markers
The neuroimaging data, including FDG standardized uptake values ratios (SUVRs), hippocampal volume and entorhinal cortex volume, were extracted from the ADNI file “ADNIMERGE”. In the present study, to adjust for sex difference in head size, the structural MRI data were transformed based on formulas below:
Hippocampal volume ratio (HpVR) = hippocampal/intracranial volume × 103
Entorhinal cortex volume ratio (EVR) = entorhinal cortex volume/intracranial volume × 103
Measurement of CSF AD pathologies
Measurement of CSF Aβ42, t-tau and p-tau levels was conducted using the multiple xMAP Luminex platform (Luminex Corp) with Innogenetics (INNO-BIA AlzBio3) immunoassay kit-based reagents, details of which could be found elsewhere [27]. All values were given as pg/ml.
Statistical analysis
To examine the difference in continuous variables (Age, education, MMSE, FAQ, RAVLT total score, HpVR, EVR, FDG SUVRs, CSF Aβ42, t-tau and p-tau) between two smoking groups (non-smokers vs smokers), Wilcoxon test was used. To compare the frequency of categorical variables (gender and APOE4 genotype) between two smoking groups, x2 test was conducted. In order to examine the longitudinal relationship between smoking history and cognitive assessments, neuroimaging markers and CSF AD pathologies, several linear mixed models were performed for each outcome (MMSE, FAQ, RAVLT total score, HpVR, EVR, FDG SUVRs, CSF Aβ42, t-tau and p-tau). All linear mixed models included main effects of smoking status, age, education, gender, APOE4 genotype, and their interactions with time, as well as a random intercept for each subject.
Author Contributions
PW and HY: conceived and designed the study. MC, CH and HD performed the research; analyzed the data; wrote the manuscript. All authors read and approved the final manuscript
Acknowledgments
Data collection and sharing for this project was funded by the Alzheimer’s Disease Neuroimaging Initiative (ADNI) (National Institutes of Health Grant U01 AG024904) and DOD ADNI (Department of Defense award number W81XWH-12-2-0012). ADNI is funded by the National Institute on Aging, the National Institute of Biomedical Imaging and Bioengineering, and through generous contributions from the following: AbbVie, Alzheimer’s Association; Alzheimer’s Drug Discovery Foundation; Araclon Biotech; BioClinica, Inc.; Biogen; Bristol-Myers Squibb Company; CereSpir, Inc.; Cogstate; Eisai Inc.; Elan Pharmaceuticals, Inc.; Eli Lilly and Company; EuroImmun; F. Hoffmann-La Roche Ltd and its affiliated company Genentech, Inc.; Fujirebio; GE Healthcare; IXICO Ltd.; Janssen Alzheimer Immunotherapy Research and Development, LLC.; Johnson and Johnson Pharmaceutical Research and Development LLC.; Lumosity; Lundbeck; Merck and Co., Inc.; Meso Scale Diagnostics, LLC.; NeuroRx Research; Neurotrack Technologies; Novartis Pharmaceuticals Corporation; Pfizer Inc.; Piramal Imaging; Servier; Takeda Pharmaceutical Company; and Transition Therapeutics. The Canadian Institutes of Health Research is providing funds to support ADNI clinical sites in Canada. Private sector contributions are facilitated by the Foundation for the National Institutes of Health (https://fnih.org/). The grantee organization is the Northern California Institute for Research and Education, and the study is coordinated by the Alzheimer’s Therapeutic Research Institute at the University of Southern California. ADNI data are disseminated by the Laboratory for Neuro Imaging at the University of Southern California.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Funding
This study was supported by Zhejiang Provincial Natural Science Foundation of China (LQ19H010003), the Project of Health Commission of Zhejiang Province (2019RC047, 2020KY177) and the Science and Technology Project of Wenzhou (Y20180600).
References
- 1. Ott A, Slooter AJ, Hofman A, van Harskamp F, Witteman JC, Van Broeckhoven C, van Duijn CM, Breteler MM. Smoking and risk of dementia and Alzheimer’s disease in a population-based cohort study: the Rotterdam study. Lancet. 1998; 351:1840–43. https://doi.org/10.1016/s0140-6736(97)07541-7 [PubMed]
- 2. Durazzo TC, Mattsson N, Weiner MW, and Alzheimer’s Disease Neuroimaging Initiative. Interaction of cigarette smoking history with APOE genotype and age on amyloid level, glucose metabolism, and neurocognition in cognitively normal elders. Nicotine Tob Res. 2016; 18:204–11. https://doi.org/10.1093/ntr/ntv075 [PubMed]
- 3. Wingbermühle R, Wen KX, Wolters FJ, Ikram MA, Bos D. Smoking, APOE genotype, and cognitive decline: the Rotterdam study. J Alzheimers Dis. 2017; 57:1191–95. https://doi.org/10.3233/JAD-170063 [PubMed]
- 4. Hawkins KA, Emadi N, Pearlson GD, Taylor B, Khadka S, King D, Blank K. The effect of age and smoking on the hippocampus and memory in late middle age. Hippocampus. 2018; 28:846–49. https://doi.org/10.1002/hipo.23014 [PubMed]
- 5. Durazzo TC, Meyerhoff DJ, Nixon SJ. Chronic cigarette smoking: implications for neurocognition and brain neurobiology. Int J Environ Res Public Health. 2010; 7:3760–91. https://doi.org/10.3390/ijerph7103760 [PubMed]
- 6. Durazzo TC, Meyerhoff DJ, Nixon SJ. A comprehensive assessment of neurocognition in middle-aged chronic cigarette smokers. Drug Alcohol Depend. 2012; 122:105–11. https://doi.org/10.1016/j.drugalcdep.2011.09.019 [PubMed]
- 7. Durazzo TC, Meyerhoff DJ, Nixon SJ. Interactive effects of chronic cigarette smoking and age on hippocampal volumes. Drug Alcohol Depend. 2013; 133:704–11. https://doi.org/10.1016/j.drugalcdep.2013.08.020 [PubMed]
- 8. Wu P, Li W, Cai X, Yan H, Chen M, and for Alzheimer’s Disease Neuroimaging Initiative. Associations of cigarette smoking with memory decline and neurodegeneration among cognitively normal older individuals. Neurosci Lett. 2020; 714:134563. https://doi.org/10.1016/j.neulet.2019.134563 [PubMed]
- 9. Petersen RC, Roberts RO, Knopman DS, Boeve BF, Geda YE, Ivnik RJ, Smith GE, Jack CR
Jr . Mild cognitive impairment: ten years later. Arch Neurol. 2009; 66:1447–55. https://doi.org/10.1001/archneurol.2009.266 [PubMed] - 10. Drago V, Babiloni C, Bartrés-Faz D, Caroli A, Bosch B, Hensch T, Didic M, Klafki HW, Pievani M, Jovicich J, Venturi L, Spitzer P, Vecchio F, et al. Disease tracking markers for Alzheimer’s disease at the prodromal (MCI) stage. J Alzheimers Dis. 2011 (Suppl 3); 26:159–99. https://doi.org/10.3233/JAD-2011-0043 [PubMed]
- 11. Takashima N, Miura K, Hozawa A, Okamura T, Hayakawa T, Okuda N, Kadowaki T, Murakami Y, Kita Y, Nakamura Y, Okayama A, Ueshima H, and NIPPON DATA80 Research Group. Cigarette smoking in middle age and a long-term risk of impaired activities of daily living: NIPPON DATA80. Nicotine Tob Res. 2010; 12:944–49. https://doi.org/10.1093/ntr/ntq121 [PubMed]
- 12. Kamiyama T, Muratani H, Kimura Y, Fukiyama K, Abe K, Fujii J, Kuwajima I, Ishii M, Shiomi T, Kawano Y, Mikami H, Ibayashi S, Omae T. Factors related to impairment of activities of daily living. Intern Med. 1999; 38:698–704. https://doi.org/10.2169/internalmedicine.38.698 [PubMed]
- 13. d’Orsi E, Xavier AJ, Steptoe A, de Oliveira C, Ramos LR, Orrell M, Demakakos P, Marmot MG. Socioeconomic and lifestyle factors related to instrumental activity of daily living dynamics: results from the English longitudinal study of ageing. J Am Geriatr Soc. 2014; 62:1630–39. https://doi.org/10.1111/jgs.12990 [PubMed]
- 14. McKhann GM, Knopman DS, Chertkow H, Hyman BT, Jack CR
Jr , Kawas CH, Klunk WE, Koroshetz WJ, Manly JJ, Mayeux R, Mohs RC, Morris JC, Rossor MN, et al. The diagnosis of dementia due to Alzheimer’s disease: recommendations from the national institute on aging-Alzheimer’s association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement. 2011; 7:263–69. https://doi.org/10.1016/j.jalz.2011.03.005 [PubMed] - 15. Braak H, Braak E. Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol. 1991; 82:239–59. https://doi.org/10.1007/BF00308809 [PubMed]
- 16. Sabbagh MN, Tyas SL, Emery SC, Hansen LA, Alford MF, Reid RT, Tiraboschi P, Thal LJ. Smoking affects the phenotype of Alzheimer disease. Neurology. 2005; 64:1301–03. https://doi.org/10.1212/01.WNL.0000156912.54593.65 [PubMed]
- 17. Tyas SL, White LR, Petrovitch H, Webster Ross G, Foley DJ, Heimovitz HK, Launer LJ. Mid-life smoking and late-life dementia: the Honolulu-Asia aging study. Neurobiol Aging. 2003; 24:589–96. https://doi.org/10.1016/s0197-4580(02)00156-2 [PubMed]
- 18. Garey KW, Neuhauser MM, Robbins RA, Danziger LH, Rubinstein I. Markers of inflammation in exhaled breath condensate of young healthy smokers. Chest. 2004; 125:22–26. https://doi.org/10.1378/chest.125.1.22 [PubMed]
- 19. Tsushima Y, Tanizaki Y, Aoki J, Endo K. MR detection of microhemorrhages in neurologically healthy adults. Neuroradiology. 2002; 44:31–36. https://doi.org/10.1007/s002340100649 [PubMed]
- 20. Bolego C, Poli A, Paoletti R. Smoking and gender. Cardiovasc Res. 2002; 53:568–76. https://doi.org/10.1016/s0008-6363(01)00520-x [PubMed]
- 21. Hawkins BT, Brown RC, Davis TP. Smoking and ischemic stroke: a role for nicotine? Trends Pharmacol Sci. 2002; 23:78–82. https://doi.org/10.1016/s0165-6147(02)01893-x [PubMed]
- 22. Durazzo TC, Korecka M, Trojanowski JQ, Weiner MW, O’ Hara R, Ashford JW, Shaw LM, and Alzheimer’s Disease Neuroimaging Initiative. Active cigarette smoking in cognitively-normal elders and probable Alzheimer’s disease is associated with elevated cerebrospinal fluid oxidative stress biomarkers. J Alzheimers Dis. 2016; 54:99–107. https://doi.org/10.3233/JAD-160413 [PubMed]
- 23. Morris JC. The clinical dementia rating (CDR): current version and scoring rules. Neurology. 1993; 43:2412–14. https://doi.org/10.1212/wnl.43.11.2412-a [PubMed]
- 24. Folstein MF, Folstein SE, McHugh PR. ”mini-mental state“. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975; 12:189–98. https://doi.org/10.1016/0022-3956(75)90026-6 [PubMed]
- 25. Schmidt M. Rey auditory verbal learning test: A handbook: Western Psychological Services Los Angeles, CA. 1996.
- 26. Pfeffer RI, Kurosaki TT, Harrah CH
Jr , Chance JM, Filos S. Measurement of functional activities in older adults in the community. J Gerontol. 1982; 37:323–29. https://doi.org/10.1093/geronj/37.3.323 [PubMed] - 27. Shaw LM, Vanderstichele H, Knapik-Czajka M, Clark CM, Aisen PS, Petersen RC, Blennow K, Soares H, Simon A, Lewczuk P, Dean R, Siemers E, Potter W, et al, and Alzheimer’s Disease Neuroimaging Initiative. Cerebrospinal fluid biomarker signature in Alzheimer’s disease neuroimaging initiative subjects. Ann Neurol. 2009; 65:403–13. https://doi.org/10.1002/ana.21610 [PubMed]



