Introduction
Coronavirus disease 2019 (COVID-19) is an infectious disease caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) [1–6]. Infection with SARS-CoV-2 is often asymptomatic but symptomatic cases most commonly present with fever, myalgia, fatigue, loss of taste or smell and upper respiratory symptoms including dry cough, dyspnea, and sore throat [1, 7]. COVID-19 has an extensive clinical course ranging from mild/moderate (MM) to severe/critical (SC) disease, in which the disease has progressed to pneumonia, acute respiratory distress syndrome, or multi-organ failure. Notably, in the largest cases series to date, approximately 10% of patients required intensive care admission [1, 6, 8]. SARS-CoV-2 has infected more than 88 million people worldwide, resulting in over 1.9 million deaths as of January 8, 2021 [9–12]. Due to its worldwide spread and severity, the World Health Organization has declared COVID-19 a pandemic [13–15]. Given no effective antiviral treatment and slow vaccine rollout, COVID-19 continues to be a serious threat to public health worldwide (see the Coronavirus Resource Centre or the latest worldwide data (https://coronavirus.jhu.edu/) [16].
As with other respiratory viruses, SARS-CoV-2 infects cells of the respiratory mucosa through the inhalation of viral particles or contact between contaminated surfaces and mucous membranes, such as those of the nose or eyes [7, 17–19]. Angiotensin converting enzyme 2 (ACE2) is one of the major entry receptors for SARS-CoV-2 [7, 20]. The host immune response to SARS-CoV-2 infection involves activation of both cellular and humoral arms. The innate immune system recognizes pathogens and induces production of proinflammatory cytokines and chemokines that trigger the adaptive immune system. This response involves activation of cytotoxic T lymphocytes (CTLs) which directly kill virus-infected cells and B cell differentiation into plasma cells that produce pathogen-specific antibodies in the serum and at mucosal surfaces [7]. SARS-CoV-2 infection in mucosal epithelial cells and macrophages activates several innate immune response pathways which are mediated by pattern recognition receptors (PRRs) including toll-like receptors (TLRs), NOD-like receptors (NLRs), and cytoplasmic DNA sensors such as RIG-I MAVS. These receptors recognize and bind viral DNA and RNA, activating downstream signaling pathways to induce the production of inflammatory cytokines including interferons (IFNs). IFN α and β mediate type-I interferon signaling that activates dendritic and natural killer cells as well as the adaptive immune response [21–24]. Activation of the IFN system subsequently stimulates antiviral gene expression in neighboring cells and recruits other immune cells involved in tissue repair and homeostasis.
Dysregulation of the innate immune response contributes to cytokine storm observed in severe COVID-19 patients [7, 19]. It has been reported that inborn errors of TLR3 and IRF7 dependent type-I IFN immunity may be associated with COVID-19 symptoms and outcomes [25, 26]. However, the molecular mechanisms that determine why some individuals suffer from severe illness whilst others are asymptomatic or exhibit mild/moderate symptoms are not well understood. Gene expression profiling technologies including PCR arrays and microarrays enable one to evaluate gene expression patterns on a specific or genome-wide scale [7, 19]. Human Antiviral Response PCR arrays contain receptors and signaling effectors for TLRs, NLRs and RIG-I-like receptors, as well as the genes regulated by these pathways and thus can be utilized to analyze type-I interferon signaling and downstream interferon-stimulated gene (ISG) expression in COVID-19 patients. We recently reported that COVID-19-induced liver dysfunction is associated with increasing age in 159 patients in a Chinese Fuyang City hospital [6]. In the present case-control study, we used PCR array and quantitative real time PCR (qPCR) to analyze the expression of a panel of genes that may be implicated in regulating COVID-19 progression in a subset of recovered patients from this cohort at the Second People's Hospital of Fuyang City in China. We sought to explore the relationship between host immune response gene expression and symptoms experienced by COVID-19 patients.
Results
Patient demographic and baseline characteristics
We recruited 46 recovered COVID-19 patients from the Second People's Hospital of Fuyang City. Of these patients, 30 had MM disease and 16 had SC disease, as in our previous report [6]. Demographic and clinical characteristics of the 46 recovered COVID-19 and 24 negative control patients are provided in Table 1.
Table 1. Comparison of demographics and clinical characteristics between SC, MM and control patients.
| Characteristic | SC | MM | Control | P value | P1 value | P2 value | P3 value | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total number (n) | 16 | 30 | 24 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age (years) (median, IQR) | 54.0(49.3-65.5) | 48.0(37.0-59.3) | 36.0(25.8-54.3) | 0.002 | 0.110 | 0.001 | 0.189 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Male gender (n, %) | 13(81.3%) | 16(53.3%) | 10(41.7%) | 0.045 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Body mass index (kg/m2) (Median, IQR) | 26.2(24.5-28.5) | 24.5(23.1-26.2) | 23.1(19.2-25.9) | 0.049 | 0.763 | 0.05 | 0.261 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Temperature (° C) (Median, IQR) | 36.3(36.1-36.7) | 36.2(36.1-36.5) | 36.2(36.1-36.4) | 0.712 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Heart rate(beats / minute) (Median, IQR) | 72(65-80) | 77(68-86) | 69(68-76) | 0.335 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Blood Pressure (mmHg) (Median, IQR) | 140(113-150)/ | 127(118-139)/ | 122(117-136)/77(65-84) | 0.522/0.002 | 0.522/0.016 | 0.522/0.003 | 0.522/1.000 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 93(84-100) | 90(82-95) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Respiratory rate (breaths / minute) (Median, IQR) | 18(17-19) | 18(17-19) | 17(16-19) | 0.712 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| WBC | 5.2(4.5-5.8) | 5.4(4.5-5.9 | 6.7(5.4-7.5) | 0.008 | 0.041 | 0.011 | 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| RBC | 4.3(4.0-4.7)) | 4.4(3.8-4.7) | 4.5(4.1-4.8) | 0.446 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Oxygen saturation (%) (Median, IQR) | 98(97-98) | 98(97-99) | 99(98-99) | 0.002 | 0.020 | 0.003 | 0.973 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Comorbidities | SC | MM | Control | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chronic hepatitis B virus (HBV) | 2 | 3 | 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hypertension | 6 | 6 | 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diabetes | 2 | 0 | 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Fatty liver | 1 | 1 | 3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other | 4 | 5 | 3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| P1, P2, and P3 represent the statistical significance values calculated using T-tests for comparisons between Control-MM, Control-SC, and MM-SC groups, respectively. Values are expressed as median (range, 25-75%). Data were analyzed using SPSS Statistics v25.0 (Armonk, New York, USA). Continuous data are expressed as medians with interquartile range, and categorical data as frequencies. Groups were compared using analysis of variance and non-parametric tests, and correlations between clinical and laboratory parameters were evaluated using the two-tailed chi-squared test. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
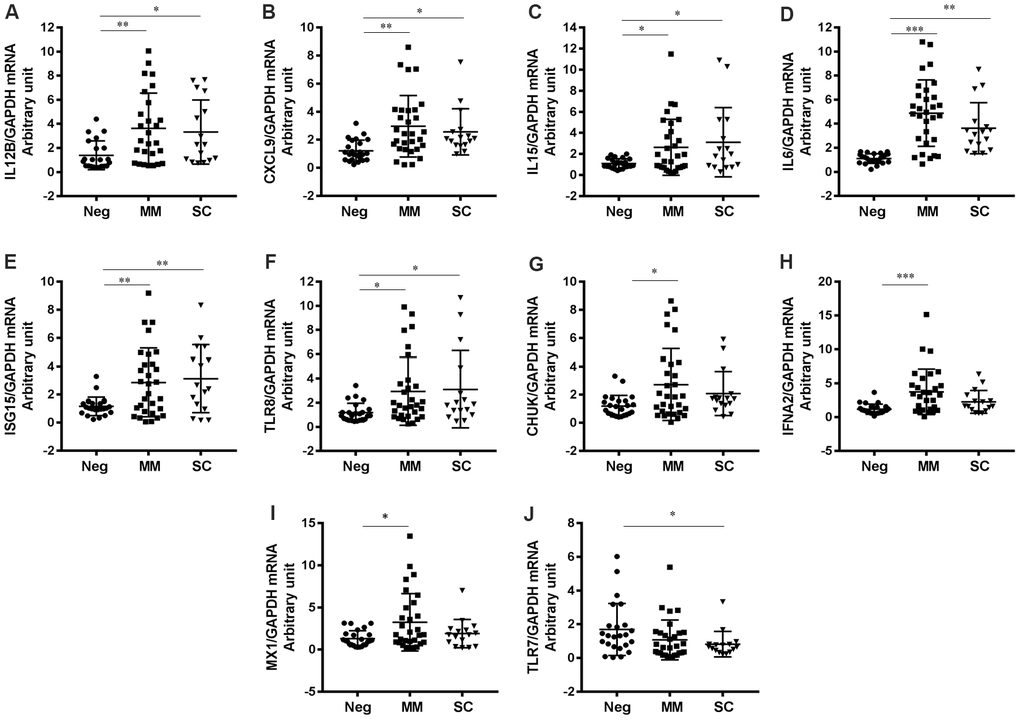
Interferon-stimulated genes were differentially expressed in patients with mild and severe disease
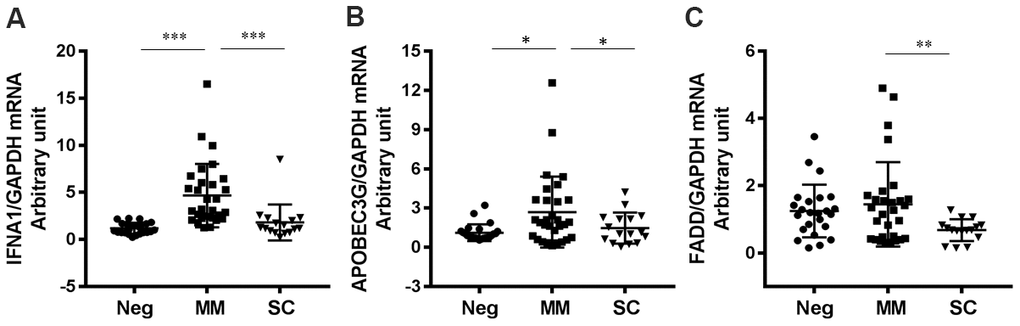
We found that expression levels of interferon alpha 1 (IFNA1), apolipoprotein B mRNA editing enzyme, catalytic polypeptide-like 3G (APOBEC3G), and Fas-associated protein with death domain (FADD) were significantly lower (P < 0.05) in the SC group than in the MM group (Figure 2). Notably, IFNA1 are APOBEC3G are interferon-stimulated genes (ISGs); IFNA1 encodes IFNα, which plays a critical role in antiviral activities and APOBEC3G (part of the APOBEC superfamily of proteins) is believed to function in innate antiviral immunity [27]. In addition, we found that two additional IFN pathway genes, IFNA2 and MX1, were slightly downregulated in the SC group compared to the MM group. These results suggest that the IFN system is important in regulating COVID-19 progression. FADD is a key adaptor molecule involved in numerous physiological processes including mediating the extrinsic apoptotic pathway and regulating type I interferon signaling [28]. Of note, it has previously been reported that FADD was significantly reduced in HBV-infected patients [29].
Expression levels of other genes implicated in SARS-CoV-2 infection and COVID-19 progression
We also evaluated genes that have been reported to regulate SARS-CoV-2 infection or host antiviral immunity. For example, expression of angiotensin-converting enzyme 2 (ACE2), which is the entry receptor for SARS-CoV-2, was substantially higher in both MM and SC groups than in the negative control group (Figure 4A). Furthermore, we found that IFNγ and PD-1 expression levels were significantly increased in all SARS-CoV-2-infected patients compared to negative controls (Figure 4B, 4C). Indoleamine 2,3-dioxygenase (IDO1) levels were higher in the MM group than in the control and severe groups (Figure 4D). The microRNAs, miR-155 and miR-130a have been reported to regulate viral infection by regulating the host immune response [30–32]. In this study, we found that both miR-155 and miR-130a were significantly upregulated in the MM disease group compared to the SC group (Figure 4E, 4F).
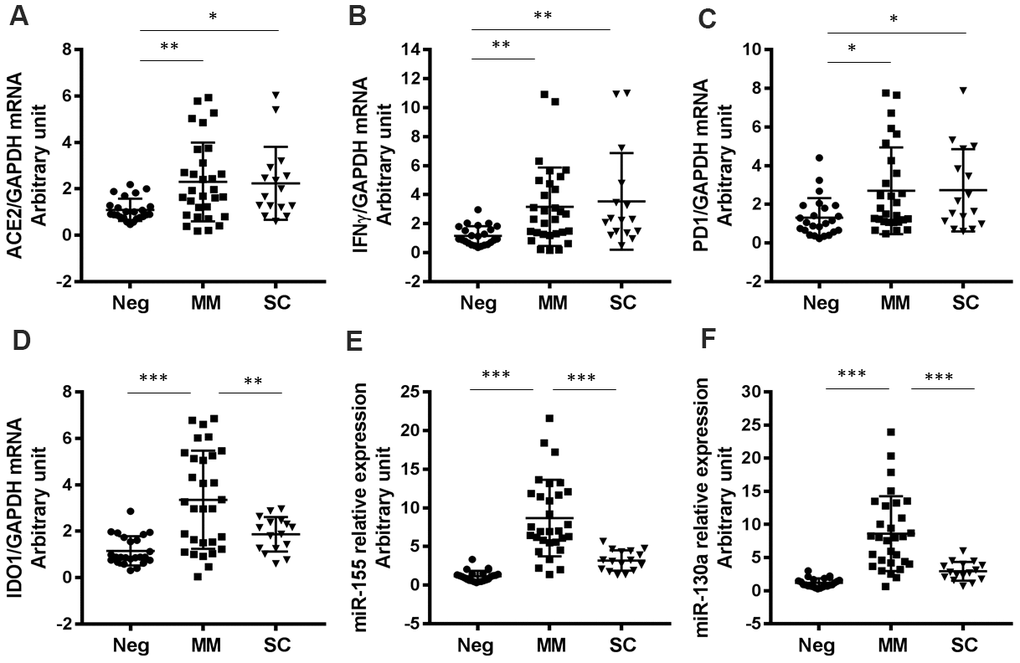
Figure 4. Expression levels of other COVID-19-related genes in patients with varying degrees of disease severity. (A) ACE2 (P=0.0057 for MM vs Neg; P=0.0313 for SC vs Neg), (B) IFNγ (P=0.0095 for MM vs Neg; P=0.0091 for SC vs Neg), (C) PD1 (P=0.0248 for MM vs Neg; P=0.0353 for SC vs Neg), (D) IDO1 (P<0.0001 for MM vs Neg; P=0.0052 for SC vs MM), (E) miR-155 (P<0.0001 for MM vs Neg; P<0.0001 for SC vs MM), (F) miR-130a (P<0.0001 for MM vs Neg; P<0.0001 for SC vs MM). *P<0.05, **P<0.01, ***P<0.001.
Discussion
ISGs are thought to be the primary effectors against many viral infections including human coronaviruses (HCoVs), hepatitis C virus (HCV) and HBV infections [7, 19, 23, 24, 26, 33, 34]. The host IFN signaling pathway and baseline ISG levels have been reported to be associated with treatment outcomes in HCV-infected patients [35]. TLR3 and IRF7 are vital mediators of type I IFN-induced immunity against SARS-CoV-2 infection [26]. Our findings illustrating increased IRF7 and IRF3 mRNA levels in COVID-19 patients with mild/moderate disease further confirm their role in the host antiviral response. APOBEC3 proteins bind to the coronavirus nucleoprotein that restricts HCoVs RNA replication and notably, APOBEC3-family gene expression is upregulated in HCoVs-infected cells [33]. Likewise, we observed significantly increased APOBEC3G mRNA levels in MM patients compared to SC patients. Our findings suggest that decreased APOBEC3G expression may serve as a biomarker for severe illness in SARS-CoV-2-infected patients.
Host cytokine perturbations have been observed in COVID-19 patients, particularly in SARS-CoV-2-induced cytokine storm syndrome (CSS), which is associated with disease severity [6, 7, 26]. The overactive immune response and nonspecific, disseminated cytokine release, including interleukin-6 (IL-6), in severe cases of COVID-19 may lead to organ failure [36]. Moreover, the combination of several cytokine levels has been used to predict COVID-19 severity and recovery [37]. IL-6 (proinflammatory) and IL-10 (anti-inflammatory) are two cytokines commonly dysregulated in COVID-19 patients and the ratio of IL6-to-IL10 may be used to predict outcomes of COVID-19 patients [37–39]. This new scoring system has been reported to help clinicians predict COVID-19 patients at risk for severe disease and those that require intervention [38]. We found that IL6 mRNA levels were significantly elevated in both MM and SC patients compared to negative controls. Our findings suggest that SARS-CoV-2 induces proinflammatory gene expression.
It has previously been reported that SARS-CoV-2 infection activates the host IFN system and upregulates expression of ACE2 (a critical entry receptor for SARS-CoV-2) [40]. We similarly found that expression of ACE2 was significantly induced in both MM and SC patient groups compared to negative controls. These findings suggest that SARS-CoV-2 infection induces ACE2 expression or that patients with elevated baseline ACE2 expression are more susceptible to severe COVID-19 illness. MicroRNAs have been reported to play an important role in host antiviral immunity. Notably, miR-155 is a multifunctional microRNA that has an indispensable capacity in immune responses [41]. We previously reported that miR-130a regulates HCV and HBV infection through the IFN pathway [32, 42]. In the present study, we also found that miR-155 and miR-130a were significantly upregulated in COVID-19 patients when compared to negative controls. It is possible that SARS-CoV-2-mediated regulation of ISGs may occur partially through miRNAs such as miR-155 and miR-130a. We previously reported that COVID-19-induced liver function abnormalities were relatively mild/moderate in this cohort [6]. This study also highlighted the importance of careful liver function management in these patients, particularly among older patients and those with underlying liver disease [6].
In summary, our study adds to the growing body of evidence that SARS-CoV-2 infection, recovery and disease severity are associated with deviations in host interferon responses. Specifically, we found that the expression of genes encoding proinflammatory cytokines (IL12B, IL15, IL6, IL12A and IL1B) and chemokines (CXCL9, CXCL11 and CXCL10) was broadly increased in COVID-19 patients and speculate that other viral infections may also induce expression of these genes. Activation of ISGs appear to be beneficial in controlling SARS-CoV-2, but over-activation of proinflammatory cytokines and downregulation of certain ISGs (IFNA1, APOBEC3G, and FADD) and microRNAs (miR-155 and miR-130a) may be associated with progression to severe/critical COVID-19. The present study has several limitations including a relatively small sample size and lack of blood samples from patients with current SARS-CoV-2 infection in Fuyang City, China. Furthermore, the age of the control group is significantly lower than that of the studied group. A more detailed understanding of the interactions between host ISGs, cytokines and SARS-CoV-2, as well as viral pathogenesis and antiviral responses in COVID-19 are therefore required in order to better predict disease severity and to optimize patient management.
Materials and Methods
Ethics approval and consent to participate
This study was registered at the Chinese Clinical Trial Registry (registration number: ChiCTR2000031620). The study protocol was approved by the Ethics Review Committee of the Second People's Hospital of Fuyang City (reference number: 2020006). This research was conducted in accordance with the ethical standards of the institutional and national research committees, and with the 1964 declaration of Helsinki.
Patients and study design
The Second People's Hospital of Fuyang City had originally admitted 159 patients with confirmed SARS-CoV-2 infection during the outbreak of the disease in Anhui Provence from January to March 2020 [6]. There were no SARS-CoV-2-related deaths in this hospital. COVID-19 diagnosis, laboratory testing, and treatment in this cohort have been previously described [6]. Patients with confirmed COVID-19 were classified as having had mild/moderate (MM) or severe/critical (SC) disease based on symptomatology [6]. We recruited 46 recovered COVID-19 patients (post-hospital discharge) from this cohort and 24 healthy subjects, age and gender matched, with no history of SARS-CoV-2 infection (negative control) from the Physical Examination Center in the Second People's Hospital of Fuyang City (Table 1). Blood samples were collected from each study subject for RNA extraction and PCR. We sought to assess changes in antiviral gene expression between patients with MM and SC disease and healthy controls.
RNA isolation
Total RNA from patient whole blood was extracted using TRIzol reagent (Invitrogen, CA, USA) as previously described [42].
RT2 Profiler™ PCR array
The RT2 Profiler™ PCR array (PAHS-122ZA) for human antiviral responses was purchased from Qiagen (Qiagen, MD, USA). The first strand cDNA was synthesized using ReverTra Ace® qPCR RT Master Mix with gDNA remover (Toyobo, Osaka, Japan). The cDNA was diluted with nuclease-free water and mixed with NovoStar SYBR qPCR SuperMix Plus with low ROX Premixed (Novoprotein, Shanghai, China) as previously described [32, 42]. Three samples from each group (SC, MM or negative control) were randomly selected and loaded into the 96-well RT2 Profiler™ PCR Array plate.
Quantitative real time PCR and gene quantification
Selected gene mRNA expression levels for all samples were quantified by qPCR using the ABI QuantStudio 3 system (Applied Biosystems Inc, MA, USA). Relative mRNA expression levels for each gene were calculated using the ∆∆Ct method and normalized to GAPDH to obtain an arbitrary unit as previously described [32, 42]. The qRT-PCR primer sequences are listed in Table 3.
Table 3. List of primer sequences used for qPCR.
| Gene name | Forward primer | Reverse primer |
| CXCL9 | CCAGTAGTGAGAAAGGGTCGC | AGGGCTTGGGGCAAATTGTT |
| IL12B | ACCCTGACCATCCAAGTCAAA | TTGGCCTCGCATCTTAGAAAG |
| IFNA1 | GCCTCGCCCTTTGCTTTACT | CTGTGGGTCTCAGGGAGATCA |
| IFNA2 | GCTTGGGATGAGACCCTCCTA | CCCACCCCCTGTATCACAC |
| IL15 | TTTCAGTGCAGGGCTTCCTAA | GGGTGAACATCACTTTCCGTAT |
| IFNAR1 | AACAGGAGCGATGAGTCTGTC | TGCGAAATGGTGTAAATGAGTCA |
| CHUK | GGCTTCGGGAACGTCTGTC | TTTGGTACTTAGCTCTAGGCGA |
| IL6 | CAGCCCTGAGAAAGGAGACAT | GGTTCAGGTTGTTTTCTGCCA |
| MX1 | GTTTCCGAAGTGGACATCGCA | GAAGGGCAACTCCTGACAGT |
| TLR7 | TCCTTGGGGCTAGATGGTTTC | TCCACGATCACATGGTTCTTTG |
| FADD | CCGCGCCTGGGGAAGAAG | CCAGCCTTCTCCAATCTTTCC |
| FOS | CCGGGGATAGCCTCTCTTACT | CCAGGTCCGTGCAGAAGTC |
| GAPDH | ACCTTCCCCATGGTGTCTGA | GCTCCTCCTGTTCGACAGTCA |
| SPP1 | TGGGAATAGCTTTGGGAAGTGG | CCGATGTCCAAAGGTGCAAT |
| TLR8 | ATGTTCCTTCAGTCGTCAATGC | TTGCTGCACTCTGCAATAACT |
| APOBEC3G | GCATCGTGACCAGGAGTATGA | GTCAGGGTAACCTTCGGGT |
| CASP1 | CAGCCCTGGTGTGGTGTG | AAAATCCTTCTCTATGTGGGCTTTC |
| MAP3K1 | TGATGTATGGAGTGTTGGCTG | AATGTGAAGGGATCGATGGAG |
| ACE2 | AACTGCTGCTCAGTCCACC | AAAAGGCAGACCATTTGTCCC |
| IDO1 | GCCAGCTTCGAGAAAGAGTTG | ATCCCAGAACTAGACGTGCAA |
Statistical analyses
Data analyses were performed using one-way ANOVA. Comparisons between negative control, MM and SC groups were analyzed by two-tailed T-tests as previously reported [6, 32, 42]. Data are expressed as the mean ± standard deviation of at least three biologic replicates, unless stated otherwise. Continuous data were expressed as medians with interquartile range, and categorical data as frequencies. Groups were compared using the Non-parametric test, and the correlations between clinical and laboratory parameters were evaluated using the two-tailed chi-squared test. In all analyses, * represents P < 0.05, ** represents P < 0.01 and *** represents P < 0.001 for indicated comparisons.
Author Contributions
Shasha Li, Xiaoqiong Duan, Yujia Li, Shilin Li, Tuo Shao, Limin Chen, Mingfeng Han, Andre J. Jeyarajan, Wenyu Lin and Xiuyong Li designed the research study, analyzed and interpreted the data, and wrote the manuscript. Shasha Li, Yong Gao, Tuantuan Li, Lin Tan, Ming Li, Mingfeng Han and Xiuyong Li were involved in diagnosis and treatment of patients, recruiting patients and collection of clinical data. All authors were involved in critical appraisal of the manuscript and approved the final version of the manuscript.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Funding
This study was funded in part by grant of Natural Science Foundation of China (NSFC 81871661 to Wenyu Lin), National Institutes of Health (NIH) /NIAID 1R01AI155140 (RTC and Wenyu Lin), the COVID-19 Urgent Research Grant of Fuyang City Department of Science and Technology (FK20202801 to Xiuyong Li).
Editorial Note
This corresponding author has a verified history of publications using the personal email address for correspondence.
References
- 1. Chen N, Zhou M, Dong X, Qu J, Gong F, Han Y, Qiu Y, Wang J, Liu Y, Wei Y, Xia J, Yu T, Zhang X, Zhang L. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020; 395:507–13. https://doi.org/10.1016/S0140-6736(20)30211-7 [PubMed]
- 2. Menachery VD, Schäfer A, Burnum-Johnson KE, Mitchell HD, Eisfeld AJ, Walters KB, Nicora CD, Purvine SO, Casey CP, Monroe ME, Weitz KK, Stratton KG, Webb-Robertson BM, et al. MERS-CoV and H5N1 influenza virus antagonize antigen presentation by altering the epigenetic landscape. Proc Natl Acad Sci USA. 2018; 115:E1012–21. https://doi.org/10.1073/pnas.1706928115 [PubMed]
- 3. Song Z, Xu Y, Bao L, Zhang L, Yu P, Qu Y, Zhu H, Zhao W, Han Y, Qin C. From SARS to MERS, thrusting coronaviruses into the spotlight. Viruses. 2019; 11:59. https://doi.org/10.3390/v11010059 [PubMed]
- 4. The Lancet. Emerging understandings of 2019-nCoV. Lancet. 2020; 395:311. https://doi.org/10.1016/S0140-6736(20)30186-0 [PubMed]
- 5. Yin Y, Wunderink RG. MERS, SARS and other coronaviruses as causes of pneumonia. Respirology. 2018; 23:130–37. https://doi.org/10.1111/resp.13196 [PubMed]
- 6. Li S, Li J, Zhang Z, Tan L, Shao T, Li M, Li X, Holmes JA, Lin W, Han M. COVID-19 induced liver function abnormality associates with age. Aging (Albany NY). 2020; 12:13895–904. https://doi.org/10.18632/aging.103720 [PubMed]
- 7. Subbarao K, Mahanty S. Respiratory virus infections: understanding COVID-19. Immunity. 2020; 52:905–09. https://doi.org/10.1016/j.immuni.2020.05.004 [PubMed]
- 8. Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J, Wang B, Xiang H, Cheng Z, Xiong Y, Zhao Y, Li Y, Wang X, Peng Z. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA. 2020; 323:1061–69. https://doi.org/10.1001/jama.2020.1585 [PubMed]
- 9. Ki M, and Task Force for 2019-nCoV. Epidemiologic characteristics of early cases with 2019 novel coronavirus (2019-nCoV) disease in Korea. Epidemiol Health. 2020; 42:e2020007. https://doi.org/10.4178/epih.e2020007 [PubMed]
- 10. Giovanetti M, Benvenuto D, Angeletti S, Ciccozzi M. The first two cases of 2019-nCoV in Italy: where they come from? J Med Virol. 2020; 92:518–21. https://doi.org/10.1002/jmv.25699 [PubMed]
- 11. Chan JF, Yuan S, Kok KH, To KK, Chu H, Yang J, Xing F, Liu J, Yip CC, Poon RW, Tsoi HW, Lo SK, Chan KH, et al. A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: a study of a family cluster. Lancet. 2020; 395:514–23. https://doi.org/10.1016/S0140-6736(20)30154-9 [PubMed]
- 12. Lu H, Stratton CW, Tang YW. Outbreak of pneumonia of unknown etiology in Wuhan, China: the mystery and the miracle. J Med Virol. 2020; 92:401–02. https://doi.org/10.1002/jmv.25678 [PubMed]
- 13. Hui DS, Azhar EI, Madani TA, Ntoumi F, Kock R, Dar O, Ippolito G, Mchugh TD, Memish ZA, Drosten C, Zumla A, Petersen E. The continuing 2019-nCoV epidemic threat of novel coronaviruses to global health - the latest 2019 novel coronavirus outbreak in Wuhan, China. Int J Infect Dis. 2020; 91:264–66. https://doi.org/10.1016/j.ijid.2020.01.009 [PubMed]
- 14. Li Q, Guan X, Wu P, Wang X, Zhou L, Tong Y, Ren R, Leung KS, Lau EH, Wong JY, Xing X, Xiang N, Wu Y, et al. Early transmission dynamics in Wuhan, China, of novel coronavirus-infected pneumonia. N Engl J Med. 2020; 382:1199–207. https://doi.org/10.1056/NEJMoa2001316 [PubMed]
- 15. Cai Q, Huang D, Yu H, Zhu Z, Xia Z, Su Y, Li Z, Zhou G, Gou J, Qu J, Sun Y, Liu Y, He Q, et al. COVID-19: abnormal liver function tests. J Hepatol. 2020; 73:566–74. https://doi.org/10.1016/j.jhep.2020.04.006 [PubMed]
- 16. Dong E, Du H, Gardner L. An interactive web-based dashboard to track COVID-19 in real time. Lancet Infect Dis. 2020; 20:533–34. https://doi.org/10.1016/S1473-3099(20)30120-1 [PubMed]
- 17. Li FF, Zhang Q, Wang GY, Liu SL. Comparative analysis of SARS-CoV-2 and its receptor ACE2 with evolutionarily related coronaviruses. Aging (Albany NY). 2020; 12:20938–45. https://doi.org/10.18632/aging.104024 [PubMed]
- 18. Li H, Xie L, Chen L, Zhang L, Han Y, Yan Z, Guo X. Genomic, epigenomic, and immune subtype analysis of CTSL/B and SARS-CoV-2 receptor ACE2 in pan-cancer. Aging (Albany NY). 2020; 12:22370–89. https://doi.org/10.18632/aging.104147 [PubMed]
- 19. Troy NM, Bosco A. Respiratory viral infections and host responses; insights from genomics. Respir Res. 2016; 17:156. https://doi.org/10.1186/s12931-016-0474-9 [PubMed]
- 20. Zhou P, Yang XL, Wang XG, Hu B, Zhang L, Zhang W, Si HR, Zhu Y, Li B, Huang CL, Chen HD, Chen J, Luo Y, et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020; 579:270–73. https://doi.org/10.1038/s41586-020-2012-7 [PubMed]
- 21. Song Y, Zhong H, Li L, Yin M, Yin Y, Guo X, Zhang B, Liu W. Dynamic monitoring of immune function indexes in COVID-19 patients. Aging (Albany NY). 2020; 12:24596–603. https://doi.org/10.18632/aging.202362 [PubMed]
- 22. Liao Y, Feng Y, Wang B, Wang H, Huang J, Wu Y, Wu Z, Chen X, Yang C, Fu X, Sun H. Clinical characteristics and prognostic factors of COVID-19 patients progression to severe: a retrospective, observational study. Aging (Albany NY). 2020; 12:18853–65. https://doi.org/10.18632/aging.103931 [PubMed]
- 23. Lin W, Zhu C, Hong J, Zhao L, Jilg N, Fusco DN, Schaefer EA, Brisac C, Liu X, Peng LF, Xu Q, Chung RT. The spliceosome factor SART1 exerts its anti-HCV action through mRNA splicing. J Hepatol. 2015; 62:1024–32. https://doi.org/10.1016/j.jhep.2014.11.038 [PubMed]
- 24. Jilg N, Lin W, Hong J, Schaefer EA, Wolski D, Meixong J, Goto K, Brisac C, Chusri P, Fusco DN, Chevaliez S, Luther J, Kumthip K, et al. Kinetic differences in the induction of interferon stimulated genes by interferon-α and interleukin 28B are altered by infection with hepatitis C virus. Hepatology. 2014; 59:1250–61. https://doi.org/10.1002/hep.26653 [PubMed]
- 25. Ebrahimi N, Aslani S, Babaie F, Hemmatzadeh M, Hosseinzadeh R, Joneidi Z, Mehdizadeh Tourzani Z, Pakravan N, Mohammadi H. Recent findings on the coronavirus disease 2019 (COVID-19); immunopathogenesis and immunotherapeutics. Int Immunopharmacol. 2020; 89:107082. https://doi.org/10.1016/j.intimp.2020.107082 [PubMed]
- 26. Zhang Q, Bastard P, Liu Z, Le Pen J, Moncada-Velez M, Chen J, Ogishi M, Sabli IK, Hodeib S, Korol C, Rosain J, Bilguvar K, Ye J, et al, and COVID-STORM Clinicians, COVID Clinicians, Imagine COVID Group, French COVID Cohort Study Group, CoV-Contact Cohort, Amsterdam UMC Covid-19 Biobank, COVID Human Genetic Effort, and NIAID-USUHS/TAGC COVID Immunity Group. Inborn errors of type I IFN immunity in patients with life-threatening COVID-19. Science. 2020; 370:eabd4570. https://doi.org/10.1126/science.abd4570 [PubMed]
- 27. Mohanram V, Sköld AE, Bächle SM, Pathak SK, Spetz AL. IFN-α induces APOBEC3G, F, and a in immature dendritic cells and limits HIV-1 spread to CD4+ T cells. J Immunol. 2013; 190:3346–53. https://doi.org/10.4049/jimmunol.1201184 [PubMed]
- 28. Bolze A, Byun M, McDonald D, Morgan NV, Abhyankar A, Premkumar L, Puel A, Bacon CM, Rieux-Laucat F, Pang K, Britland A, Abel L, Cant A, et al. Whole-exome-sequencing-based discovery of human FADD deficiency. Am J Hum Genet. 2010; 87:873–81. https://doi.org/10.1016/j.ajhg.2010.10.028 [PubMed]
- 29. Abdirizak F, Lewis R, Chowell G. Evaluating the potential impact of targeted vaccination strategies against severe acute respiratory syndrome coronavirus (SARS-CoV) and Middle East respiratory syndrome coronavirus (MERS-CoV) outbreaks in the healthcare setting. Theor Biol Med Model. 2019; 16:16. https://doi.org/10.1186/s12976-019-0112-6 [PubMed]
- 30. Sarkar N, Panigrahi R, Pal A, Biswas A, Singh SP, Kar SK, Bandopadhyay M, Das D, Saha D, Kanda T, Sugiyama M, Chakrabarti S, Banerjee A, Chakravarty R. Expression of microRNA-155 correlates positively with the expression of toll-like receptor 7 and modulates hepatitis B virus via C/EBP-β in hepatocytes. J Viral Hepat. 2015; 22:817–27. https://doi.org/10.1111/jvh.12390 [PubMed]
- 31. Dudda JC, Salaun B, Ji Y, Palmer DC, Monnot GC, Merck E, Boudousquie C, Utzschneider DT, Escobar TM, Perret R, Muljo SA, Hebeisen M, Rufer N, et al. MicroRNA-155 is required for effector CD8+ T cell responses to virus infection and cancer. Immunity. 2013; 38:742–53. https://doi.org/10.1016/j.immuni.2012.12.006 [PubMed]
- 32. Duan X, Li S, Holmes JA, Tu Z, Li Y, Cai D, Liu X, Li W, Yang C, Jiao B, Schaefer EA, Fusco DN, Salloum S, et al. MicroRNA 130a regulates both hepatitis C virus and hepatitis B virus replication through a central metabolic pathway. J Virol. 2018; 92:e02009–17. https://doi.org/10.1128/JVI.02009-17 [PubMed]
- 33. Milewska A, Kindler E, Vkovski P, Zeglen S, Ochman M, Thiel V, Rajfur Z, Pyrc K. APOBEC3-mediated restriction of RNA virus replication. Sci Rep. 2018; 8:5960. https://doi.org/10.1038/s41598-018-24448-2 [PubMed]
- 34. Schoggins JW, Rice CM. Innate immune responses to hepatitis C virus. Curr Top Microbiol Immunol. 2013; 369:219–42. https://doi.org/10.1007/978-3-642-27340-7_9 [PubMed]
- 35. Alao H, Cam M, Keembiyehetty C, Zhang F, Serti E, Suarez D, Park H, Fourie NH, Wright EC, Henderson WA, Li Q, Liang TJ, Rehermann B, Ghany MG. Baseline intrahepatic and peripheral innate immunity are associated with hepatitis C virus clearance during direct-acting antiviral therapy. Hepatology. 2018; 68:2078–88. https://doi.org/10.1002/hep.29921 [PubMed]
- 36. Atal S, Fatima Z. IL-6 inhibitors in the treatment of serious COVID-19: a promising therapy? Pharmaceut Med. 2020; 34:223–31. https://doi.org/10.1007/s40290-020-00342-z [PubMed]
- 37. Nagant C, Ponthieux F, Smet J, Dauby N, Doyen V, Besse-Hammer T, De Bels D, Maillart E, Corazza F. A score combining early detection of cytokines accurately predicts COVID-19 severity and intensive care unit transfer. Int J Infect Dis. 2020; 101:342–45. https://doi.org/10.1016/j.ijid.2020.10.003 [PubMed]
- 38. McElvaney OJ, Hobbs BD, Qiao D, McElvaney OF, Moll M, McEvoy NL, Clarke J, O’Connor E, Walsh S, Cho MH, Curley GF, McElvaney NG. A linear prognostic score based on the ratio of interleukin-6 to interleukin-10 predicts outcomes in COVID-19. EBioMedicine. 2020; 61:103026. https://doi.org/10.1016/j.ebiom.2020.103026 [PubMed]
- 39. Zhang J, Yu M, Tong S, Liu LY, Tang LV. Predictive factors for disease progression in hospitalized patients with coronavirus disease 2019 in Wuhan, China. J Clin Virol. 2020; 127:104392. https://doi.org/10.1016/j.jcv.2020.104392 [PubMed]
- 40. Busnadiego I, Fernbach S, Pohl MO, Karakus U, Huber M, Trkola A, Stertz S, Hale BG. Antiviral activity of type I, II, and III interferons counterbalances ACE2 inducibility and restricts SARS-CoV-2. mBio. 2020; 11:e01928–20. https://doi.org/10.1128/mBio.01928-20 [PubMed]
- 41. Alivernini S, Gremese E, McSharry C, Tolusso B, Ferraccioli G, McInnes IB, Kurowska-Stolarska M. MicroRNA-155-at the critical interface of innate and adaptive immunity in arthritis. Front Immunol. 2018; 8:1932. https://doi.org/10.3389/fimmu.2017.01932 [PubMed]
- 42. Duan X, Liu X, Li W, Holmes JA, Kruger AJ, Yang C, Li Y, Xu M, Ye H, Li S, Liao X, Sheng Q, Chen D, et al. Microrna-130a downregulates HCV replication through an atg5-dependent autophagy pathway. Cells. 2019; 8:338. https://doi.org/10.3390/cells8040338 [PubMed]