Introduction
Globally, diabetic patients have quadrupled in the past 30 years and diabetes has become the ninth major cause of death. One in eleven adults worldwide suffers from diabetes and 90% of diabetes patients suffer from type 2 diabetes [1, 2]. Osteoporosis is also one of the diseases that seriously impair the health of the elderly. In the United States, osteoporosis contributed about 1.5 million fractures each year [3, 4]. Osteoporosis and diabetes mellitus are both chronic diseases and show a clear link to morbidity and mortality [5]. Complications of diabetes were also significantly associated with fractures after falling [6–9].
Metformin, an anti-diabetic drug, can significantly improve the progression of a variety of diseases, including polycystic ovary syndrome, tuberculosis, cardiovascular [10], and neurological diseases [11]. The effects of metformin on bone metabolism have also been studied [12, 13]. Existing research recognizes that Metformin played a critical role in osteoporosis by inhibiting systemic inflammation and promoting osteoclast formation [14–16]. Anti-osteoporosis drugs such as alendronate can also improve the progression of diabetes. In the clinical study, administration of 70 mg/week alendronate improves fasting plasma glucose, HbA1c, and insulin indices in postmenopausal women [17]. However, anti-osteoporotic treatment does not alter the development of diabetes in the meta-analysis [18].
Because diabetes and osteoporosis are both important metabolic diseases, it is very important to know whether the combined use of anti-osteoporosis drugs and anti-diabetic drugs will improve the progression of diabetes and the progression of diabetic osteoporosis. Therefore, the purpose of our study consisted of analyzing the effects of combined metformin and alendronate on diabetic-induced bone loss and possible mechanisms.
Materials and Methods
Animal experimental procedures
Eight-week-old C57 BL/KS db/db female mice (Lepr-KO/KO, n = 36) and non-diabetic (C57BLKS-Lepr-db/+, n = 9) were purchased from Beijing Vital River Laboratory Animal Technology (Beijing, China). A blood glucose level higher than 11.1 mmol/L was considered a hyperglycemic state. All experimental protocols and animal handling procedures were conducted according to the recommendations in the Guide for the Care and Use of Laboratory Animals, published by the National Institutes of Health (Publications No. 80-23, revised in 1996). This study was approved by the Experimental Animal Committee of our hospital.
After one week of adaptive feeding (mice were maintained in a standard animal facility with controlled temperature (22°C) and photoperiod (12 h light and 12 h dark) and free access to freshwater and food), mice were raised in the SPF animal house after treatment. and then random grouping was carried out after the treatment, divided into five groups, such as control group (Con, C57BLKS-Lepr-db/+, N = 9), diabetes mellitus group (DM, N = 9), DM with metformin group receiving daily metformin at 113.75mg/kg (DM + MET, N = 9) [19], Alendronate group receiving daily alendronate at 0.5 mg/kg (DM + ALE, N = 9) [20], metformin plus Alendronate group receiving daily alendronate at 0.5 mg/kg and metformin at 113.75 mg/kg (DM + MET + ALE, N = 9). The Control group was treated with no metformin or alendronate at all. All drugs were administered intragastrically in normal saline solution beginning at 8 weeks of age and lasting for 12 weeks.
Fasting blood glucose (FBG) and oral glucose tolerance test (GTT)
For the fasting blood glucose, glucose tolerance assays, and insulin tolerance exam as described by Amir [21]. After 12 weeks of treatment, fasted mice (16 h, paper bedding) by monitoring glucose levels after a glucose bolus (1 g/kg of body weight (BW)) or insulin (0.5 U/kg BW) by intraperitoneal (IP) injection. The exam of glucose was carried from fasted (4–16 h) or re-fed animals (15 min to 1 h). Re-feeding was conducted by injecting a bolus of glucose (1 g/kg of BW) IP as mentioned above. The first drop of blood was thrown away and then the second drop of blood was detected by the glucometer (Roche Diagnostics, Mannheim, Germany). We collected the tail blood samples at 0, 15, 30, 60, and 120 min after glucose loading and detected the blood glucose value by the glucometer (Roche Diagnostics, Mannheim, Germany).
Serum markers examination
Serum OCN, GLP-1, CTX-1, and TRAP 5b concentrations (n = 9 for each group) of all mice in the current study were quantified after fasting for 8 h using commercial enzyme-linked immunosorbent assay (ELISA) kits (CUSABIO Biotech Co., Wuhan, China) according to the manufacturer’s instructions.
Radiology examination
BMD of the whole tibiae and L4 were measured by a DXA (Faxitron® LX-60 Cabinet radiography system, US) and Micro-CT (Inveon, Siemens, Erlangen, Germany). The scanning parameters used were 70 kVp, 111 μA, and 1000 projections per 180°, resulting in a 10.5 μm isotropic voxel size and a total scan time of 13.2 min. Trabecular bone micro-architecture was assessed using the μCT Evaluation Program (Image Processing Language v. 5.0A, Scanco) [22, 23].
Biomechanical examination
The collected femurs were wrapped up using etamine soaked with normal saline, which was then stored at low temperatures. The length of the specimen was the full length of the femur. Specimens were subjected to compressive loading on a biomechanical testing machine with a loading speed of 2 mm/min [20].
Histomorphometry
The proximal tibia was dehydrated, embedded, sliced and then the calcein double-labeling sections were analyzed, which included mineral apposition rate (MAR) and bone formation rate per bone surface (BFR/BS) [24].
qPCR
Tibias were cleaned of muscle and connective tissue, flash-frozen in liquid nitrogen, and stored at −80°C. Frozen tibias were crushed under liquid nitrogen conditions with a Bessman tissue pulverizer (Spectrum Laboratories, Rancho Dominguez, CA, USA). Total RNA was extracted using Trizol reagent (Invitrogen, Carlsbad, CA, USA). The expression levels of bone metabolism, glucose metabolism, and inflammation-related genes, including ALP, OCN, BMP-2, Runx-2, Beclin-1, AMPK, OPG, COL1A1, Gfi1, OPN, GPR43, GPR41, GCG, RANKL, PC1/3, which has been upload in Supplementary Table 1. The relative change in gene expression was analyzed by the 2−ΔΔCT method. The mRNA of ALP, OCN, BMP-2, Runx-2, Beclin-1, AMPK, OPG, COL1A1, Gfi1, OPN, RANKL, is calculated with the DM group as the baseline, and the mRNA of GPR43, GPR41, GCG, PC1/3 is calculated with the control group as the baseline.
Statistical analysis
All measurements are presented as the mean ± standard deviation (SD) and a P-value of ≤0.05 was considered statistically significant. Bodyweight of the time-course study was analyzed by two-way repeated-measures analysis of variance (ANOVA). Data were analyzed for intervention and time main effects. The data were analyzed using GraphPad Prism 8.02 (La Jolla California, USA) and one-way ANOVA followed by Tukey’s multiple.
Ethical approval
All procedures performed in studies involving animal were in accordance with the ethical standards of the institutional (LA221221).
Results
The effect of metformin and alendronate on body weight and food intake
We analyzed the changes in the body weight and found that diabetic mice increased body weight significantly. After treatment with metformin and alendronate alone, the bodyweight significantly reduced compared with the diabetic group and the combined treatment also decreased significantly in the bodyweight than treatment with metformin and alendronate alone (Figure 1A, 1B). The food intake has also shown some kind of pattern with a change of body weight (Figure 1C, 1D).
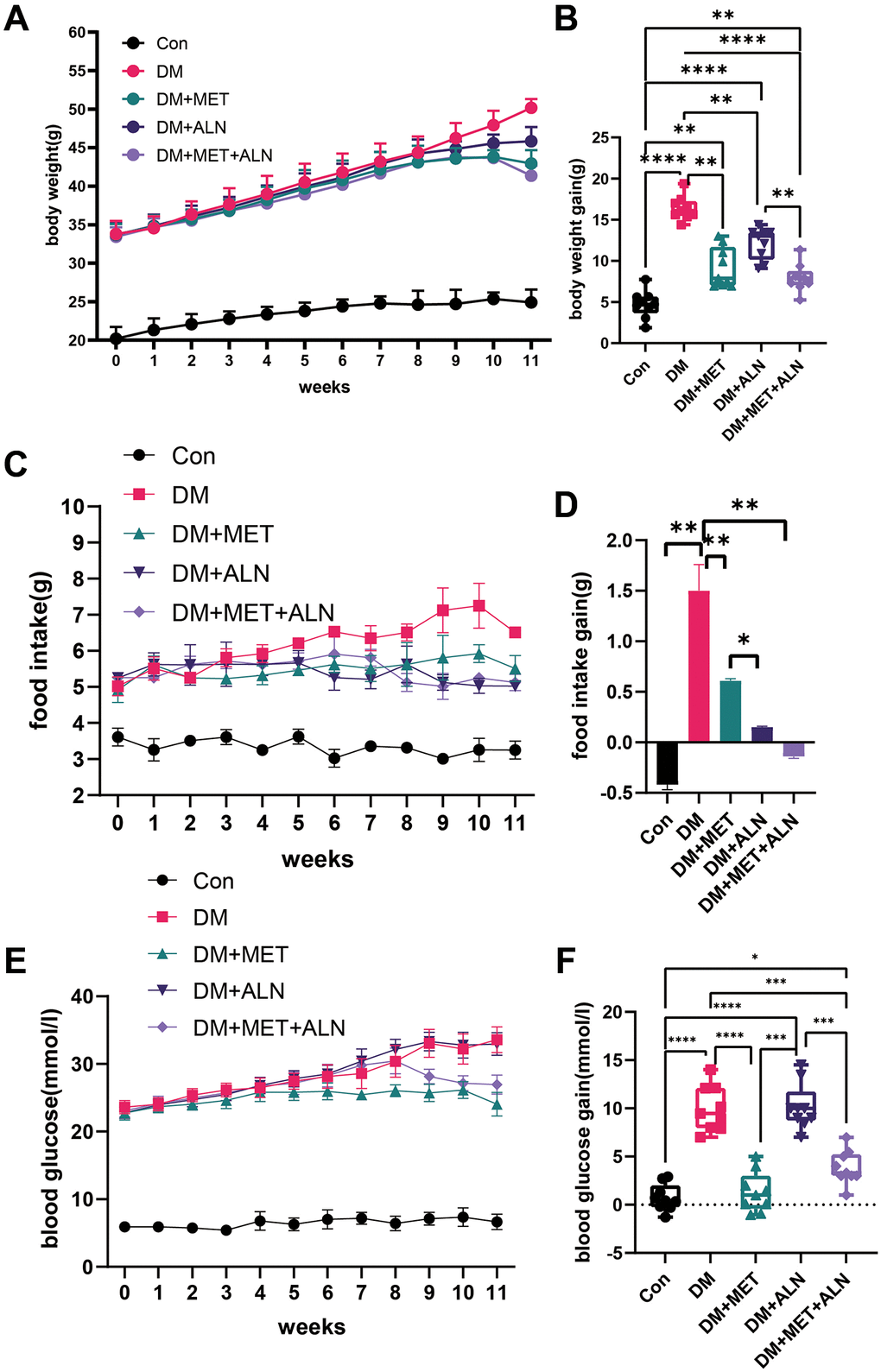
Figure 1. Effect of body weight, food intake and blood glucose in metformin and alendronate in diabetes mice. (A, B) Change of body weight in four group. (C, D) Change of food intake. (E, F) Change of blood glucose.
The effect of metformin and alendronate on glucose metabolism
Because metformin and alendronate also have significant effects on glucose metabolism, we assessed the effects of using alone or in combination alendronate and metformin treatment on glucose metabolism. We found that the use of metformin alone and the combined use of metformin and alendronate significantly improved blood glucose levels, but the use of alendronate alone did not improve blood glucose significantly (Figure 1E, 1F). The experiment of serum glucose test after 4 hour fasting and 16-hour fasting, GTT test and insulin levels and insulin sensitivity can only be improved in both the use of metformin alone and the combined use of metformin and alendronate (Figure 2A–2C) but the use of alendronate has a weaker effect on glucose metabolism (Figure 2D, 2E).
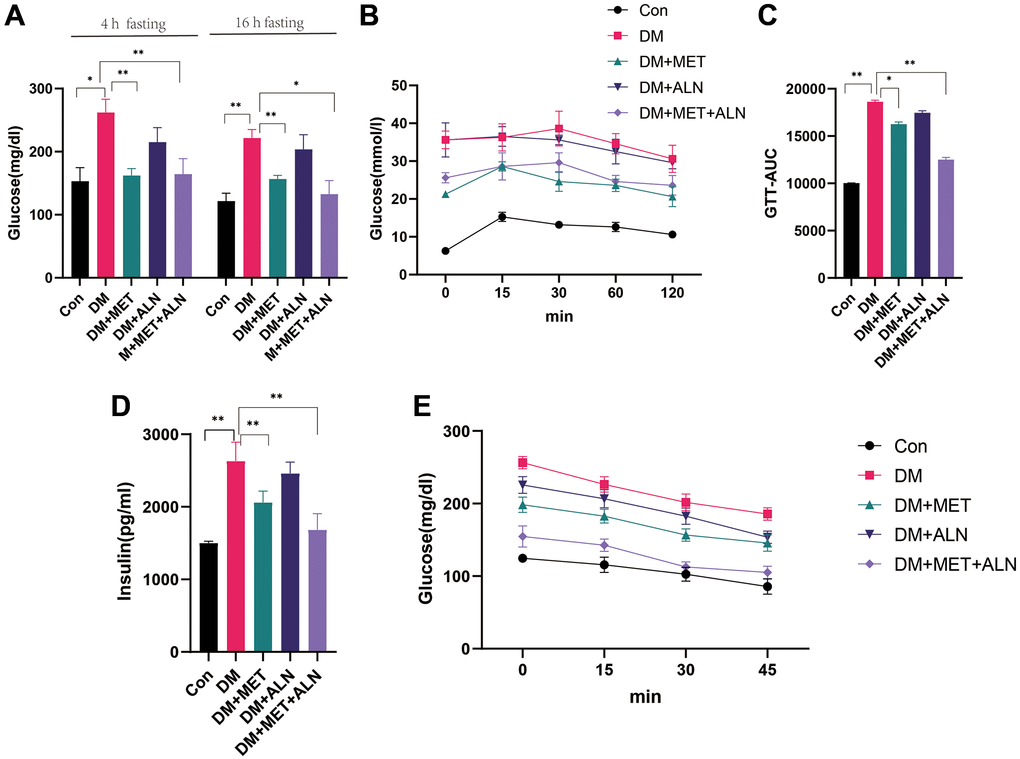
Figure 2. Effect of glucose metabolism in metformin and alendronate in diabetes mice. (A) serum glucose after 4 hour and 16 hours fasting. (B, C). GTT test. (D, E) Serum insulin and Insulin sensitivity.
The effect of metformin and alendronate on bone microstructure
We found that combined utilization of alendronate and metformin can improve the BV/TV, Tb.N, Tb.Sp of the spine and BV/TV, Tb.Th and Tb.N of the tibia than diabetes group. besides, the use of alendronate alone can be increased significantly in BV/TV of tibia and decreased significantly in Tb.Sp of spine. The use of metformin alone can improve Tb.N and Tb.Sp of spine than diabetes mice (Figure 3A–3H).
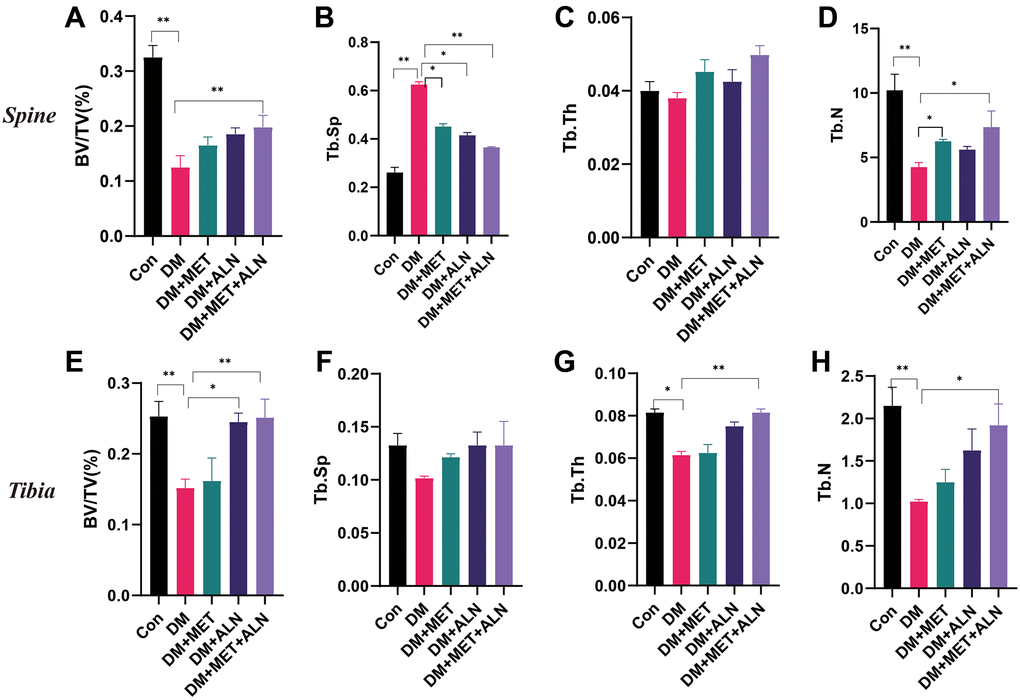
Figure 3. The effect of metformin and alendronate on bone microstructure. (A) BV/TV in spine. (B) Tb.Sp in spine. (C) Tb.Th in spine. (D) Tb.N in spine. (E ) BV/TV in tibia. (F) Tb.Sp in tibia. (G) Tb.Th in tibia. (H) Tb.N in tibia.
DXA analysis found that diabetes can decrease significantly in bone mass density (BMD) of the spine and tibia. The use of alendronate alone and the combined use of alendronate and metformin can significantly improve bone loss, but for metformin, it does not significantly improve BMD of the spine and tibia (Figure 4, Figure 5A, 5B).
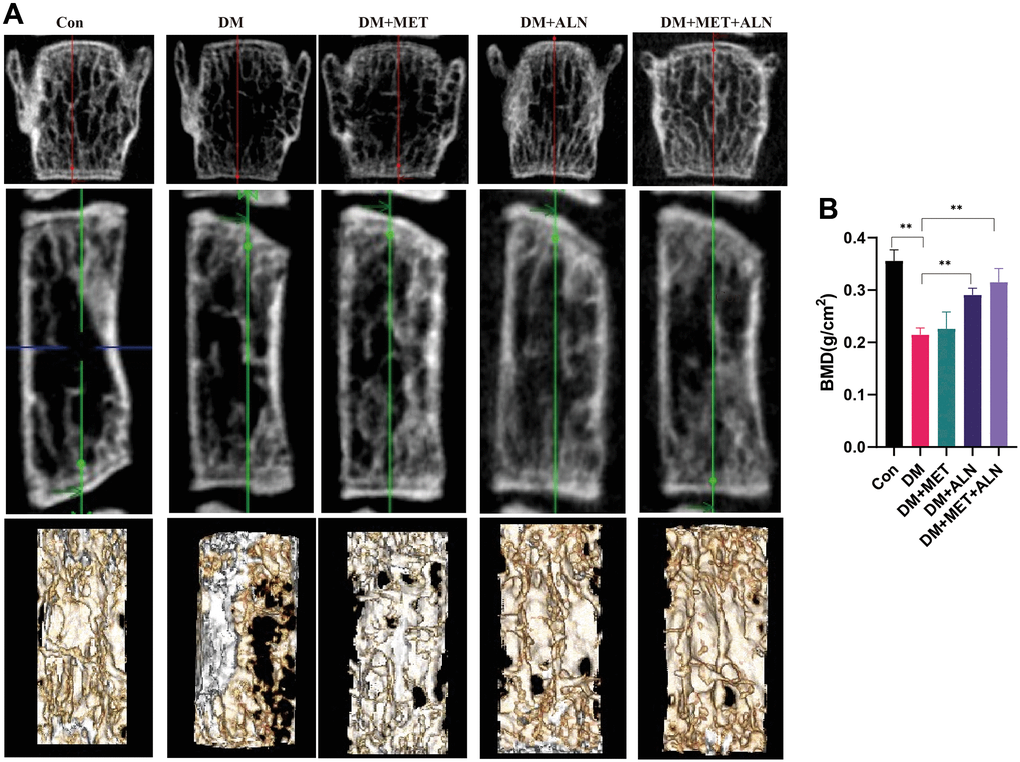
Figure 4. The effect of metformin and alendronate on bone microstructure in lumbar vertebra. (A) 3D image of Micro-CT. (B) BMD of spine.
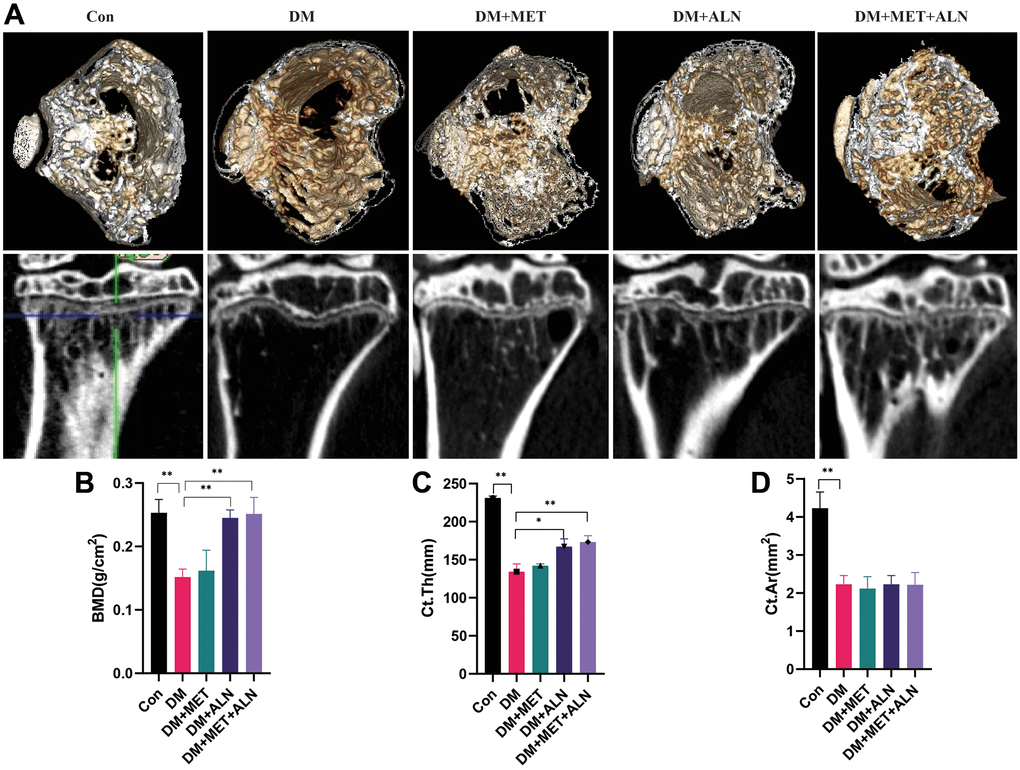
Figure 5. The effect of metformin and alendronate on bone microstructure in tibia. (A) 3D image of Micro-CT in tibia. (B) BMD of tibia. (C) Ct.Th in tibia. (D) Ct.Ar in tibia.
For the results of tibial cortical bone, it was found that the use of alendronate alone and the combined use of alendronate and metformin can improve the thickness of cortical bone, but there is no significant effect on the area of cortical bone (Ct.Ar) (Figure 5C, 5D).
The effect of metformin and alendronate on pathology and biomechanics
We found that the combination of alendronate and metformin can increase the result of MAR and BFR/BS than diabetic mice and shown that combined medications can promote bone formation. We also analyzed the biomechanics of the tibia and found that combination drugs significantly improved maximum force. Stiffness, and energy absorption but the use of metformin alone also had no significant effect on the maximum force (Figure 3, Figure 6A–6F).
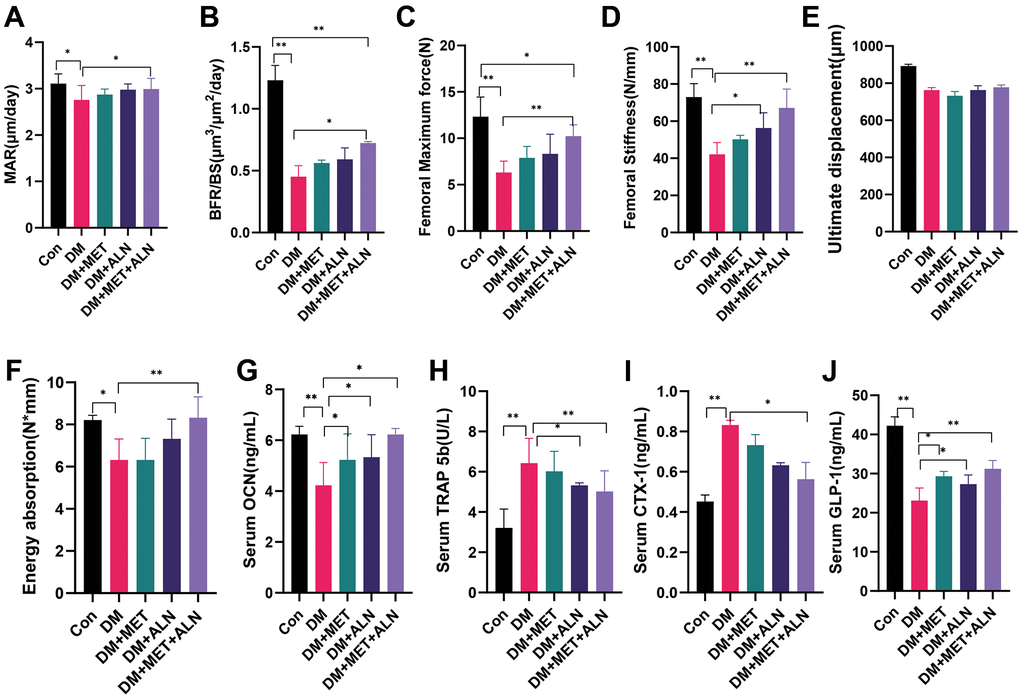
Figure 6. The effect of metformin and alendronate on serum biomarker, pathology and biomechanics. (A) mineral apposition rate (MAR). (B) bone formation rate per bone surface (BFR/BS). (C) maximum load. (D) stiffness. (E) ultimate displacement. (F) energy absorption. (G) serum OCN. (H) Serum TRAP 5b. (I) Serum CTX-1. (J) Serum GLP-1.
The effect of metformin and alendronate on serum biomarkers and mRNA level in the tibia
We further analyzed the results of serological markers and found that diabetic mice significantly reduced OCN and GLP-1 levels, but significantly increased TRAP 5b and CTX-1 levels. Combined medication can significantly reverse the process. In addition, the use of alendronate alone can significantly reduce TRAP 5b levels and increase GLP-1 and the use of metformin alone can significantly increase GLP-1 and OCN levels (Figure 6G–6J).
We firstly analyzed the expression of bone metabolism-related genes in the tibia. Diabetic mice decrease significantly in ALP, OCN, Runx2, Col-1, BMP-2, OPG, AMPK, Gfi1, and OPN and decreased significantly in Beclin-1 than the control group (Figure 7A). Besides, the use of Metformin alone can only increase significantly in ALP and the use of alendronate alone can increase significantly in OCN, AMPK, Gfi1 mRNA level than the control group. Secondly, we further analyzed the level of mRNA related to glucose metabolism in the tibia and found that the combined use of metformin and alendronate can significantly increase the levels of GPR43 and GPR41, GCG, and PC1/3 than diabetic mice. The use of metformin alone can also increase significantly in GPR43 and GPR41 and PC1/3 than in diabetic mice (Figure 7B).
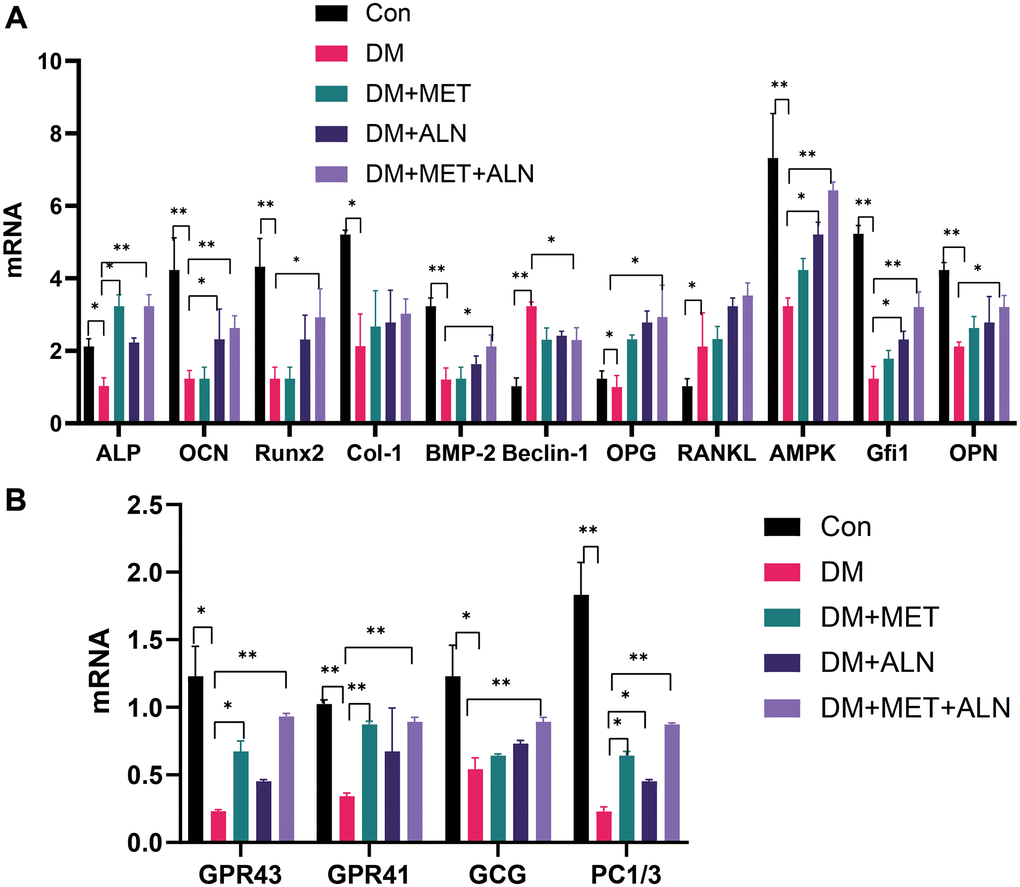
Figure 7. The relative mRNA level of tibia. (A) bone metabolism relative mRNA level. (B) glucose metabolism relative mRNA level.
Discussion
In our research, the study found that the combined use of alendronate and metformin can substantially improve bone loss and glucose metabolism disorders than the diabetes group. However, the hypoglycemic effect or anti-osteoporosis effect of combining the use of alendronate and metformin is not obvious compared with the use of metformin alone or alendronate alone. It means that they appear to act no obvious synergistically between alendronate and metformin.
Osteoporosis is often accompanied by diabetes [25] and bone remodeling is compromised in both type 1 and 2 diabetes [26, 27]. Both body weight and food intake are significantly affected by diabetes and medication. Diabetes will enhance the weight of the patient and anti-diabetic treatment can improve the weight change of the patient [28–30]. This is also compatible with our research results. At the same time, alendronate can also substantially improve weight gain [31, 32]. The improvement in body weight and food intake may better reflect the results of the improvement in bone metabolism and glucose metabolism [33–36].
The treatment of diabetes with anti-osteoporosis drugs has drawn attention [37, 38]. In our studies, we found that the use of alendronate alone can decrease significantly in 4- and 16-hour fasting glucose and increased GLP-1 level but has not significantly decreased glucose level and insulin sensitivity in diabetic mice. It is In accordance with the present results, previous studies (e.g. Konstantinos et al.) have demonstrated that alendronate was associated with a significant 50% reduction in the risk of incident T2DM [39]. Yang et al. also found that antiresorptive therapy was not a risk factor for diabetes [40]. Alendronate also play important role in improving fasting plasma glucose and insulin sensitivity and decreases insulin resistance in prediabetic osteopenic postmenopausal women [17]. However, in our animal experiments, we found that the use of alendronate alone has a weaker effect on glucose metabolism than the use of metformin which can improve blood glucose and insulin sensitivity.
At the same time, we have also observed that the use of metformin alone can improve the bone microstructure of the spine than the diabetes group. Consistent with the literature, this research found that participants who reported that metformin use is involved in a lower risk of osteoporosis in adult women independent of type 2 diabetes mellitus and obesity [11, 41]. This also accords with our earlier observations, which showed that the metformin decreased bone turnover marker and influence bone formation in clinical studies [42, 43]. However, we have also noticed that the effect of metformin on bone metabolism is significantly weaker than the use of alendronate, so the use of metformin alone in the treatment of diabetic osteoporosis may require further verification. In addition, contrary to expectations, this study did not find a significant difference between the anti-osteoporosis effect of using the alendronate alone or the anti-diabetes effect of use in the alendronate and combination medication. These results further support the idea that the synergy between metformin and alendronate may be poor. However, the levels observed in this investigation are far below those observed by Lyudmila et al. It is suggested that metformin combined with alendronate significantly reduced the degree of cartilage degeneration and show the synergy in osteoarthritis. In addition, metformin can reverse some of the complications caused by alendronate, such as stomach damage [44]. A probable explanation for these results may be the lack of adequate observation time. Therefore, in our future research, we will further increase the observation time to determine whether the combined use of metformin and alendronate has a synergistic effect.
Metformin increased ALP and OCN secretion, enhanced BMP-2 expression, improved bone mineral density (BMD) [45]. In our study, it was found that metformin can improve the secretion of OCN, but not significant improvements in osteoclast-related biological markers (for example, CTX-1 and TRAP 5b), which may indicate that metformin may play important role in the process of osteoblast [46]. Metformin can also improve the progression of diabetes through the AMPK regulation and affect the expression of BMP-2 levels, and it can also improve bone metabolism [47, 48]. AMPK-Gfi1-OPN axis also play important role in bone and glucose metabolism [49]. Mai et al. found that metformin stimulates OPN and reduces RANKL expression in osteoblasts and ovariectomized rats [50–52]. OPN mediated AMPK regulation of osteogenesis and inhibited adipogenesis [49]. Besides, metformin also can inhibit the gene level of Runx2 which is associated with osteoblast differentiation markers such as OCN [53].
The effect and related mechanism of bisphosphonate on osteoporosis treatment has been widely studied [54–57]. Alendronate has a certain effect on the development of diabetes [58, 59]. Ikeda et al. found that alendronate that produces a reduction in urinary NTx and inhibition of decrease in BMD may have a clinical significance to reduce the risk of bone fracture in postmenopausal type 2 diabetic women [60]. In this study, we found that alendronate can increase the secretion of GPL-1 and proconvertase 1/3 (PC1/3) activity which may improve glucose metabolism [61, 62].
In our study, it was further verified that combination applications had significant improvements in diabetic osteoporosis, but the use of metformin or bisphosphonates alone did not improve osteoporosis or diabetes significantly, which also indicated that single medication in diabetic osteoporosis treatment may not be effective and further study in the clinical practical application of combination drugs.
Conclusions
In conclusion, the present study demonstrated that combining metformin and alendronate prevent the emergence of diabetes and diabetic-relative bone loss by up-regulating AMPK gene expression and secreting GLP-1, stimulating bone formation and suppressing bone resorption than diabetes group. But they appear to act no obvious synergistically between alendronate and metformin in a shorter observation period. Future studies on the current topic are therefore recommended to focus on the effect of combining the use of alendronate and metformin in diabetes and diabetic-relative bone loss.
Supplementary Materials
Author Contributions
Conception, design and performed experiments mentioned: QYZ, ZQG, FSL, YJX. Acquisition, analysis, and interpretation of the data: GH, HC, XJ, KT, SFL. Drafting and writing: QYZ, YJX. Final approval of the article: QYZ, ZQG, GH, HC, XJ, ZYG.
Acknowledgments
We would like to thank all participants and our hospital.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
Funding
This work was partly supported by the Clinical Chinese Medicine Plateau Discipline Construction Project of Shanghai Pudong New Area Health Committee [grant number PDZY-2018-0604].
References
- 1. Zheng Y, Ley SH, Hu FB. Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat Rev Endocrinol. 2018; 14:88–98. https://doi.org/10.1038/nrendo.2017.151 [PubMed]
- 2. American Diabetes Association. 2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes-2018. Diabetes Care. 2018 (Suppl 1); 41:S13–27. https://doi.org/10.2337/dc18-S002 [PubMed]
- 3. Johnston CB, Dagar M. Osteoporosis in Older Adults. Med Clin North Am. 2020; 104:873–84. https://doi.org/10.1016/j.mcna.2020.06.004 [PubMed]
- 4. Black DM, Rosen CJ. Clinical Practice. Postmenopausal Osteoporosis. N Engl J Med. 2016; 374:254–62. https://doi.org/10.1056/NEJMcp1513724 [PubMed]
- 5. Kurra S, Fink DA, Siris ES. Osteoporosis-associated fracture and diabetes. Endocrinol Metab Clin North Am. 2014; 43:233–43. https://doi.org/10.1016/j.ecl.2013.09.004 [PubMed]
- 6. Walsh JS, Vilaca T. Obesity, Type 2 Diabetes and Bone in Adults. Calcif Tissue Int. 2017; 100:528–35. https://doi.org/10.1007/s00223-016-0229-0 [PubMed]
- 7. Paschou SA, Dede AD, Anagnostis PG, Vryonidou A, Morganstein D, Goulis DG. Type 2 Diabetes and Osteoporosis: A Guide to Optimal Management. J Clin Endocrinol Metab. 2017; 102:3621–34. https://doi.org/10.1210/jc.2017-00042 [PubMed]
- 8. Napoli N, Chandran M, Pierroz DD, Abrahamsen B, Schwartz AV, Ferrari SL, and IOF Bone and Diabetes Working Group. Mechanisms of diabetes mellitus-induced bone fragility. Nat Rev Endocrinol. 2017; 13:208–19. https://doi.org/10.1038/nrendo.2016.153 [PubMed]
- 9. Eller-Vainicher C, Cairoli E, Grassi G, Grassi F, Catalano A, Merlotti D, Falchetti A, Gaudio A, Chiodini I, Gennari L. Pathophysiology and Management of Type 2 Diabetes Mellitus Bone Fragility. J Diabetes Res. 2020; 2020:7608964. https://doi.org/10.1155/2020/7608964 [PubMed]
- 10. Dziubak A, Wójcicka G, Wojtak A, Bełtowski J. Metabolic Effects of Metformin in the Failing Heart. Int J Mol Sci. 2018; 19:2869. https://doi.org/10.3390/ijms19102869 [PubMed]
- 11. Bahrambeigi S, Yousefi B, Rahimi M, Shafiei-Irannejad V. Metformin; an old antidiabetic drug with new potentials in bone disorders. Biomed Pharmacother. 2019; 109:1593–601. https://doi.org/10.1016/j.biopha.2018.11.032 [PubMed]
- 12. Zhao J, Li Y, Zhang H, Shi D, Li Q, Meng Y, Zuo L. Preventative effects of metformin on glucocorticoid-induced osteoporosis in rats. J Bone Miner Metab. 2019; 37:805–14. https://doi.org/10.1007/s00774-019-00989-y [PubMed]
- 13. Gürler EB, Çilingir-Kaya ÖT, Peker Eyüboglu I, Ercan F, Akkiprik M, Reiter RJ, Yegen BÇ. Melatonin supports alendronate in preserving bone matrix and prevents gastric inflammation in ovariectomized rats. Cell Biochem Funct. 2019; 37:102–12. https://doi.org/10.1002/cbf.3379 [PubMed]
- 14. Fan KJ, Wu J, Wang QS, Xu BX, Zhao FT, Wang TY. Metformin inhibits inflammation and bone destruction in collagen-induced arthritis in rats. Ann Transl Med. 2020; 8:1565. https://doi.org/10.21037/atm-20-3042 [PubMed]
- 15. Jiating L, Buyun J, Yinchang Z. Role of Metformin on Osteoblast Differentiation in Type 2 Diabetes. Biomed Res Int. 2019; 2019:9203934. https://doi.org/10.1155/2019/9203934 [PubMed]
- 16. Malta FS, Garcia RP, Azarias JS, Ribeiro GKD, Miranda TS, Shibli JA, Bastos MF. Impact of hyperglycemia and treatment with metformin on ligature-induced bone loss, bone repair and expression of bone metabolism transcription factors. PLoS One. 2020; 15:e0237660. https://doi.org/10.1371/journal.pone.0237660 [PubMed]
- 17. Karimi Fard M, Aminorroaya A, Kachuei A, Salamat MR, Hadi Alijanvand M, Aminorroaya Yamini S, Karimifar M, Feizi A, Amini M. Alendronate improves fasting plasma glucose and insulin sensitivity, and decreases insulin resistance in prediabetic osteopenic postmenopausal women: A randomized triple-blind clinical trial. J Diabetes Investig. 2019; 10:731–7. https://doi.org/10.1111/jdi.12944 [PubMed]
- 18. Anagnostis P, Paschou SA, Gkekas NN, Artzouchaltzi AM, Christou K, Stogiannou D, Vryonidou A, Potoupnis M, Goulis DG. Efficacy of anti-osteoporotic medications in patients with type 1 and 2 diabetes mellitus: a systematic review. Endocrine. 2018; 60:373–83. https://doi.org/10.1007/s12020-018-1548-x [PubMed]
- 19. Zhang W, Xu JH, Yu T, Chen QK. Effects of berberine and metformin on intestinal inflammation and gut microbiome composition in db/db mice. Biomed Pharmacother. 2019; 118:109131. https://doi.org/10.1016/j.biopha.2019.109131 [PubMed]
- 20. Mao Z, Zhu Y, Hao W, Chu C, Su H. MicroRNA-155 inhibition up-regulates LEPR to inhibit osteoclast activation and bone resorption via activation of AMPK in alendronate-treated osteoporotic mice. IUBMB Life. 2019; 71:1916–28. https://doi.org/10.1002/iub.2131 [PubMed]
- 21. Zarrinpar A, Chaix A, Xu ZZ, Chang MW, Marotz CA, Saghatelian A, Knight R, Panda S. Antibiotic-induced microbiome depletion alters metabolic homeostasis by affecting gut signaling and colonic metabolism. Nat Commun. 2018; 9:2872. https://doi.org/10.1038/s41467-018-05336-9 [PubMed]
- 22. Zhang Y, Li XL, Sha NN, Shu B, Zhao YJ, Wang XL, Xiao HH, Shi Q, Wong MS, Wang YJ. Differential response of bone and kidney to ACEI in db/db mice: A potential effect of captopril on accelerating bone loss. Bone. 2017; 97:222–32. https://doi.org/10.1016/j.bone.2017.01.029 [PubMed]
- 23. Zhou X, Guan Z, Jin X, Zhao J, Chen G, Ding J, Ren Y, Zhai X, Zhou Q, Guan Z. Reversal of alopecia areata, osteoporosis follow treatment with activation of Tgr5 in mice. Biosci Rep. 2021; 41:BSR20210609. https://doi.org/10.1042/BSR20210609 [PubMed]
- 24. Li J, Zeng Z, Zhao Y, Jing D, Tang C, Ding Y, Feng X. Effects of low-intensity pulsed electromagnetic fields on bone microarchitecture, mechanical strength and bone turnover in type 2 diabetic db/db mice. Sci Rep. 2017; 7:10834. https://doi.org/10.1038/s41598-017-11090-7 [PubMed]
- 25. Lecka-Czernik B. Diabetes, bone and glucose-lowering agents: basic biology. Diabetologia. 2017; 60:1163–9. https://doi.org/10.1007/s00125-017-4269-4 [PubMed]
- 26. Khosla S, Hofbauer LC. Osteoporosis treatment: recent developments and ongoing challenges. Lancet Diabetes Endocrinol. 2017; 5:898–907. https://doi.org/10.1016/S2213-8587(17)30188-2 [PubMed]
- 27. Ferrari SL, Abrahamsen B, Napoli N, Akesson K, Chandran M, Eastell R, El-Hajj Fuleihan G, Josse R, Kendler DL, Kraenzlin M, Suzuki A, Pierroz DD, Schwartz AV, Leslie WD, and Bone and Diabetes Working Group of IOF. Diagnosis and management of bone fragility in diabetes: an emerging challenge. Osteoporos Int. 2018; 29:2585–96. https://doi.org/10.1007/s00198-018-4650-2 [PubMed]
- 28. Mavian AA, Miller S, Henry RR. Managing type 2 diabetes: balancing HbA1c and body weight. Postgrad Med. 2010; 122:106–17. https://doi.org/10.3810/pgm.2010.05.2148 [PubMed]
- 29. Lau DC, Teoh H. Impact of Current and Emerging Glucose-Lowering Drugs on Body Weight in Type 2 Diabetes. Can J Diabetes. 2015 (Suppl 5); 39:S148–54. https://doi.org/10.1016/j.jcjd.2015.09.090 [PubMed]
- 30. Fromenty B, Vadrot N, Massart J, Turlin B, Barri-Ova N, Lettéron P, Fautrel A, Robin MA. Chronic ethanol consumption lessens the gain of body weight, liver triglycerides, and diabetes in obese ob/ob mice. J Pharmacol Exp Ther. 2009; 331:23–34. https://doi.org/10.1124/jpet.109.155168 [PubMed]
- 31. Reid IR, Horne AM, Mihov B, Stewart A, Bastin S, Gamble GD. Zoledronate Slows Weight Loss and Maintains Fat Mass in Osteopenic Older Women: Secondary Analysis of a Randomized Controlled Trial. Calcif Tissue Int. 2020; 106:386–91. https://doi.org/10.1007/s00223-019-00653-7 [PubMed]
- 32. Kaji H, Hisa I, Inoue Y, Naito J, Sugimoto T, Kasuga M. Analysis of factors affecting increase in bone mineral density at lumbar spine by bisphosphonate treatment in postmenopausal osteoporosis. J Bone Miner Metab. 2009; 27:76–82. https://doi.org/10.1007/s00774-008-0005-y [PubMed]
- 33. Izzi-Engbeaya C, Ma Y, Buckley NW, Ratnasabapathy R, Richardson E, Counsell JR, Fernandes-Freitas I, Norton M, Farooq G, Mirza Z, Cai M, Cheetham S, Seckl J, et al. Effects of corticosterone within the hypothalamic arcuate nucleus on food intake and body weight in male rats. Mol Metab. 2020; 36:100972. https://doi.org/10.1016/j.molmet.2020.02.015 [PubMed]
- 34. Gibbons C, Blundell J, Tetens Hoff S, Dahl K, Bauer R, Baekdal T. Effects of oral semaglutide on energy intake, food preference, appetite, control of eating and body weight in subjects with type 2 diabetes. Diabetes Obes Metab. 2021; 23:581–8. https://doi.org/10.1111/dom.14255 [PubMed]
- 35. Beccuti G, Monagheddu C, Evangelista A, Ciccone G, Broglio F, Soldati L, Bo S. Timing of food intake: Sounding the alarm about metabolic impairments? A systematic review. Pharmacol Res. 2017; 125:132–41. https://doi.org/10.1016/j.phrs.2017.09.005 [PubMed]
- 36. Argyrakopoulou G, Simati S, Dimitriadis G, Kokkinos A. How Important Is Eating Rate in the Physiological Response to Food Intake, Control of Body Weight, and Glycemia? Nutrients. 2020; 12:1734. https://doi.org/10.3390/nu12061734 [PubMed]
- 37. Mohsin S, Baniyas MM, AlDarmaki RS, Tekes K, Kalász H, Adeghate EA. An update on therapies for the treatment of diabetes-induced osteoporosis. Expert Opin Biol Ther. 2019; 19:937–48. https://doi.org/10.1080/14712598.2019.1618266 [PubMed]
- 38. Panagiotakou A, Yavropoulou M, Nasiri-Ansari N, Makras P, Basdra EK, Papavassiliou AG, Kassi EN. Extra-skeletal effects of bisphosphonates. Metabolism. 2020; 110:154264. https://doi.org/10.1016/j.metabol.2020.154264 [PubMed]
- 39. Toulis KA, Nirantharakumar K, Ryan R, Marshall T, Hemming K. Bisphosphonates and glucose homeostasis: a population-based, retrospective cohort study. J Clin Endocrinol Metab. 2015; 100:1933–40. https://doi.org/10.1210/jc.2014-3481 [PubMed]
- 40. Yang S, Leslie WD, Morin SN, Majumdar SR, Lix LM. Antiresorptive therapy and newly diagnosed diabetes in women: a historical cohort study. Diabetes Obes Metab. 2016; 18:875–81. https://doi.org/10.1111/dom.12678 [PubMed]
- 41. Blümel JE, Arteaga E, Aedo S, Arriola-Montenegro J, López M, Martino M, Miranda C, Miranda O, Mostajo D, Ñañez M, Ojeda E, Pilnik S, Rojas J, et al. Metformin use is associated with a lower risk of osteoporosis in adult women independent of type 2 diabetes mellitus and obesity. REDLINC IX study. Gynecol Endocrinol. 2020; 36:421–5. https://doi.org/10.1080/09513590.2020.1718092 [PubMed]
- 42. Lingaiah S, Morin-Papunen L, Risteli J, Tapanainen JS. Metformin decreases bone turnover markers in polycystic ovary syndrome: a post hoc study. Fertil Steril. 2019; 112:362–70. https://doi.org/10.1016/j.fertnstert.2019.04.013 [PubMed]
- 43. van Lierop AH, Hamdy NA, van der Meer RW, Jonker JT, Lamb HJ, Rijzewijk LJ, Diamant M, Romijn JA, Smit JW, Papapoulos SE. Distinct effects of pioglitazone and metformin on circulating sclerostin and biochemical markers of bone turnover in men with type 2 diabetes mellitus. Eur J Endocrinol. 2012; 166:711–6. https://doi.org/10.1530/EJE-11-1061 [PubMed]
- 44. Nolêto IRS, Iles B, Alencar MS, Lopes ALF, Oliveira AP, Pacheco G, Sousa FBM, Araújo AR, Alves EHP, Vasconcelos DFP, Leal LKA, Araújo AJ, Filho JDB, Medeiros JVR. Alendronate-induced gastric damage in normoglycemic and hyperglycemic rats is reversed by metformin. Eur J Pharmacol. 2019; 856:172410. https://doi.org/10.1016/j.ejphar.2019.172410 [PubMed]
- 45. Zheng L, Shen X, Ye J, Xie Y, Yan S. Metformin alleviates hyperglycemia-induced apoptosis and differentiation suppression in osteoblasts through inhibiting the TLR4 signaling pathway. Life Sci. 2019; 216:29–38. https://doi.org/10.1016/j.lfs.2018.11.008 [PubMed]
- 46. Park SH, Kang MA, Moon YJ, Jang KY, Kim JR. Metformin coordinates osteoblast/osteoclast differentiation associated with ischemic osteonecrosis. Aging (Albany NY). 2020; 12:4727–41. https://doi.org/10.18632/aging.102796 [PubMed]
- 47. McCarthy AD, Cortizo AM, Sedlinsky C. Metformin revisited: Does this regulator of AMP-activated protein kinase secondarily affect bone metabolism and prevent diabetic osteopathy. World J Diabetes. 2016; 7:122–33. https://doi.org/10.4239/wjd.v7.i6.122 [PubMed]
- 48. Kanazawa I, Yamaguchi T, Yano S, Yamauchi M, Sugimoto T. Metformin enhances the differentiation and mineralization of osteoblastic MC3T3-E1 cells via AMP kinase activation as well as eNOS and BMP-2 expression. Biochem Biophys Res Commun. 2008; 375:414–9. https://doi.org/10.1016/j.bbrc.2008.08.034 [PubMed]
- 49. Wang YG, Qu XH, Yang Y, Han XG, Wang L, Qiao H, Fan QM, Tang TT, Dai KR. AMPK promotes osteogenesis and inhibits adipogenesis through AMPK-Gfi1-OPN axis. Cell Signal. 2016; 28:1270–82. https://doi.org/10.1016/j.cellsig.2016.06.004 [PubMed]
- 50. Shin HS, Sun HJ, Whang YM, Park YJ, Park DJ, Cho SW. Metformin Reduces Thyroid Cancer Tumor Growth in the Metastatic Niche of Bone by Inhibiting Osteoblastic RANKL Productions. Thyroid. 2021; 31:760–71. https://doi.org/10.1089/thy.2019.0851 [PubMed]
- 51. Mai QG, Zhang ZM, Xu S, Lu M, Zhou RP, Zhao L, Jia CH, Wen ZH, Jin DD, Bai XC. Metformin stimulates osteoprotegerin and reduces RANKL expression in osteoblasts and ovariectomized rats. J Cell Biochem. 2011; 112:2902–9. https://doi.org/10.1002/jcb.23206 [PubMed]
- 52. Araújo AA, Pereira ASB, Medeiros CAC, Brito GAC, Leitão RFC, Araújo LS, Guedes PMM, Hiyari S, Pirih FQ, Araújo Júnior RF. Effects of metformin on inflammation, oxidative stress, and bone loss in a rat model of periodontitis. PLoS One. 2017; 12:e0183506. https://doi.org/10.1371/journal.pone.0183506 [PubMed]
- 53. Kasai T, Bandow K, Suzuki H, Chiba N, Kakimoto K, Ohnishi T, Kawamoto S, Nagaoka E, Matsuguchi T. Osteoblast differentiation is functionally associated with decreased AMP kinase activity. J Cell Physiol. 2009; 221:740–9. https://doi.org/10.1002/jcp.21917 [PubMed]
- 54. Sharpe M, Noble S, Spencer CM. Alendronate: an update of its use in osteoporosis. Drugs. 2001; 61:999–1039. https://doi.org/10.2165/00003495-200161070-00010 [PubMed]
- 55. Porras AG, Holland SD, Gertz BJ. Pharmacokinetics of alendronate. Clin Pharmacokinet. 1999; 36:315–28. https://doi.org/10.2165/00003088-199936050-00002 [PubMed]
- 56. Pérez-López FR. Postmenopausal osteoporosis and alendronate. Maturitas. 2004; 48:179–92. https://doi.org/10.1016/j.maturitas.2003.12.006 [PubMed]
- 57. Leder BZ, Mitlak B, Hu MY, Hattersley G, Bockman RS. Effect of Abaloparatide vs Alendronate on Fracture Risk Reduction in Postmenopausal Women With Osteoporosis. J Clin Endocrinol Metab. 2020; 105:938–43. https://doi.org/10.1210/clinem/dgz162 [PubMed]
- 58. Chuguransky SR, Cortizo AM, McCarthy AD. Alendronate Can Improve Bone Alterations in Experimental Diabetes by Preventing Antiosteogenic, Antichondrogenic, and Proadipocytic Effects of AGEs on Bone Marrow Progenitor Cells. Biomed Res Int. 2016; 2016:5891925. https://doi.org/10.1155/2016/5891925 [PubMed]
- 59. Berti-Couto SA, Vasconcelos AC, Iglesias JE, Figueiredo MA, Salum FG, Cherubini K. Diabetes mellitus and corticotherapy as risk factors for alendronate-related osteonecrosis of the jaws: a study in Wistar rats. Head Neck. 2014; 36:84–93. https://doi.org/10.1002/hed.23260 [PubMed]
- 60. Ikeda T, Manabe H, Iwata K. Clinical significance of alendronate in postmenopausal type 2 diabetes mellitus. Diabetes Metab. 2004; 30:355–8. https://doi.org/10.1016/s1262-3636(07)70128-3 [PubMed]
- 61. Wideman RD, Gray SL, Covey SD, Webb GC, Kieffer TJ. Transplantation of PC1/3-Expressing alpha-cells improves glucose handling and cold tolerance in leptin-resistant mice. Mol Ther. 2009; 17:191–8. https://doi.org/10.1038/mt.2008.219 [PubMed]
- 62. Sancho V, Daniele G, Lucchesi D, Lupi R, Ciccarone A, Penno G, Bianchi C, Dardano A, Miccoli R, Del Prato S. Metabolic regulation of GLP-1 and PC1/3 in pancreatic α-cell line. PLoS One. 2017; 12:e0187836. https://doi.org/10.1371/journal.pone.0187836 [PubMed]


