Introduction
Population aging is one of the significant challenges of the 21st century as the number of adults over the age of 65 continues to increase. This shift in demographics represents a substantial challenge to healthcare systems, as older adults are significantly more at risk of developing dementia and other neurodegenerative disorders [1]. Aging and cognitive decline are closely correlated. For example, median scores in the Mini-Mental State Examination (MMSE), a widely used clinical assessment for dementia, decline by approximately 10% from age 50 to 85 and up to 35% for the lowest-scoring quartile [2]. At present, we have few solutions to deal with this growing health issue with most studies focussing on specific interventions such as drugs or training programs with limited success.
Despite the clear trend for cognitive decline with aging, there is significant inter-personal variance. The reasons for this variance are not entirely understood but are suggested to be strongly dependent upon earlier lifetime experiences [3]. This is often described as ‘cognitive’ or ‘brain’ reserves, which are drawn upon in later life to slow the rate of cognitive and memory decline and to avoid or delay overt symptoms [4]. It is proposed that these experiences create multiple, redundant neural processing networks that are more resistant to disruption due to brain damage and compensatory mechanisms that help create new neural processing networks in response to such damage [5]. Previous research also suggested that these experiences can lead to increased brain size and synapse counts, which limits the magnitude of cognitive impairment until brain damage exceeds a certain critical threshold [6]. There is good evidence that the resulting reserves are influenced by genetics [7] and environmental factors, affecting the reserves in different directions. For example, educational attainment [8] has been shown to influence reserves positively, whereas alcohol consumption [9] and smoking [10] have been shown to influence reserves negatively. A considerable number of studies have investigated the impact of such prior life factors in later life, with a common finding that they typically contribute significantly to cognitive health and, thus, improve cognitive performance in aging. Although the effects of these genetic and historical life factors are considered robust, they offer little hope to current older adults because they cannot be altered in later life. What is promising and can be modified is the effect of daily activities on maintaining cognitive health and memory as adults age. Indeed, several studies have attempted to determine whether diet [11], cognitive training [11], physical exercise [12], music practice [13], meditation [14], social engagement [15, 16] or combined interventions [17] are effective at slowing cognitive decline due to aging. However, these studies have typically examined the benefits of these activities individually and the overall evidence for their benefit is inconsistent [18]. Furthermore, conclusions have been limited due to small effect sizes, low numbers of participants, healthy survivor bias, self-selection into studies, limited follow-up measurements, lack of control groups, and lack of combined interventions. As a result, we are still left with the question of ‘what can older adults do now to maintain their cognitive performance?’ Our approach is to investigate the relationship between several potential daily activities and aging memory. This alternative approach has the benefit that it allows us to investigate a broad set of factors in a data-driven manner, rather than focusing on individual factors studied in isolation and with typically low numbers of participants.
In this study, we created a new combined analysis method based on machine learning, which allowed us to investigate a large population of older adults at varying ages. Rather than conducting intervention trial studies, our investigation exploited the natural person-to-person variability in their daily activities and assessed how they affected memory at different ages in real world settings. This unique and powerful methodological approach allowed us to identify, quantify and understand how engagement in numerous key activities is related to cognitive health and to provide older adults with information that they can immediately act upon. Finally, this analysis provides a unique ability to identify the dynamics of the relationship between memory, background factors (education etc.) and daily activities, and how this relationship evolves as an individual ages.
Results
Selection of daily activities
Feature selection analyses identified 17 daily activities out of 33 that were significantly associated with changes in cognition, for at least one age group, by at least one method. These 17 variables represented cognitively challenging, physical, or social activities. Table 1 lists the selected variables with corresponding p-values for each age category. Notably, no single daily activity was significantly associated with all age groups. Only one daily activity (use computer) was identified as associated with changes in memory in four age groups. Only four daily activities (do activities with grandchildren and do word games, meet up with children, and speak on phone with other family members) were associated with changes in memory in two age groups. The rest of daily activities were significantly associated with changes in memory in only one age group.
Table 1. List of daily activities, which were found to be significantly associated with changes in memory, at least in one age category, at least by one method.
| Daily Activity | Age Group | ||||
| 65–69 | 70–74 | 75–79 | 80–84 | 85–89 | |
| DO ACTIVITIES WITH GRANDCHILDREN | .30 | .05 | .33 | <.01 | .11 |
| VOLUNTEER YOUTH | .54 | .13 | .39 | .03 | .63 |
| ATTEND SPORTS/SOCIAL/CLUB | .02 | .32 | .48 | .59 | .21 |
| READ | .03 | .14 | .26 | .17 | .13 |
| DO WORD GAMES | .25 | .05 | .05 | .14 | .90 |
| PLAY CARDS AND GAMES | .28 | .21 | .68 | .16 | .01 |
| USE COMPUTER | <.01 | <.01 | .04 | .01 | .27 |
| BAKE OR COOK | .13 | .15 | .73 | .04 | .45 |
| SEW OR KNIT | .02 | .52 | .75 | .15 | .26 |
| WALK FOR 20 MINS | .61 | .33 | .52 | .04 | .39 |
| MEET UP WITH CHILDREN | .17 | <.01 | .51 | .05 | .39 |
| SPEAK ON PHONE WITH CHILDREN | .55 | .19 | .03 | .10 | .85 |
| WRITE OR EMAIL CHILDREN | .16 | .30 | .11 | .44 | <.01 |
| SPEAK ON PHONE WITH OTHER FAMILY MEMBERS | .38 | .02 | .01 | .43 | .15 |
| WRITE OR EMAIL OTHER FAMILY MEMBERS | .05 | .06 | .11 | .19 | .51 |
| SPEAK ON PHONE WITH FRIENDS | .26 | .06 | .02 | .74 | .19 |
| WRITE OR EMAIL FRIENDS | .05 | .38 | .14 | .06 | .19 |
Importance of individual daily activities
The relative importance of daily activities at predicting memory changes was quantified through sensitivity analyses for each age group. Figure 1 shows the relative importance of individual daily activities for each age group. No clear pattern of age-related changes in the relative importance of individual daily activities for predicting cognition changes was identified.
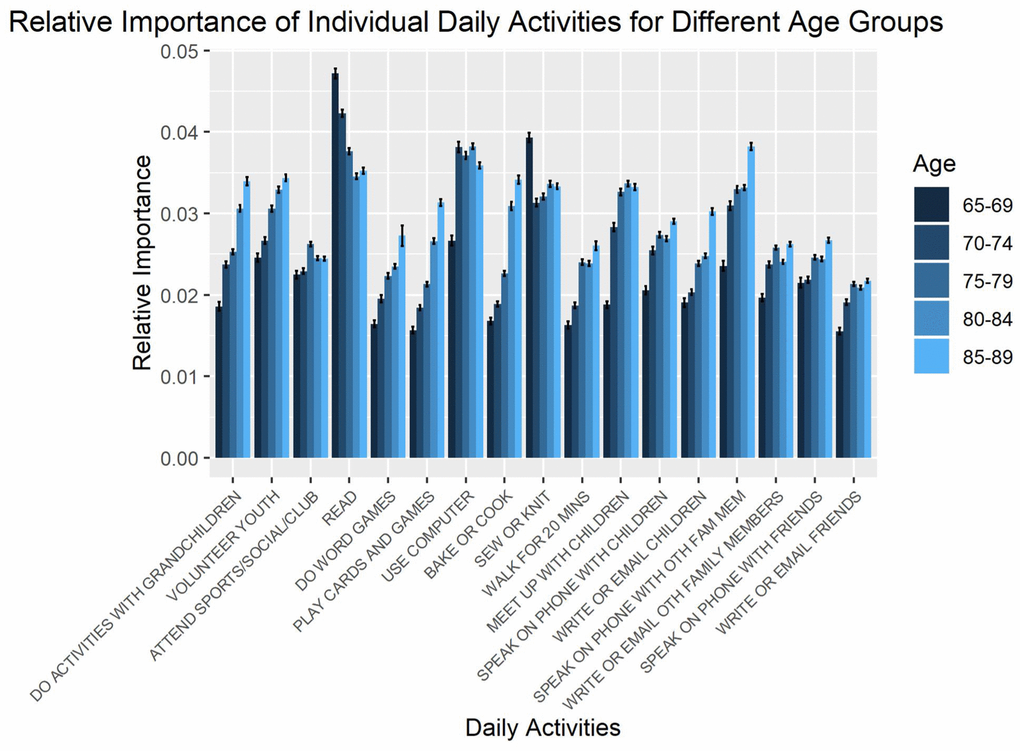
Figure 1. Relative importance of daily activities for predicting changes in memory, as estimated from sensitivity analysis. Error bars represent standard errors across repetitions in the sensitivity analysis.
The combined effect of daily activities on memory
The relative importance of daily activities from Figure 1 were combined for each age group. Figure 2 shows the relative importance of these combined daily activities compared with education and baseline cognition for each age group, demonstrating their power in predicting memory changes. The relative importance of the combined daily activities was computed as an arithmetic sum of the individual relative importance. Although finding regular patterns in the predictive power of individual daily activities was elusive, clear trends could be observed by considering their combined effects.
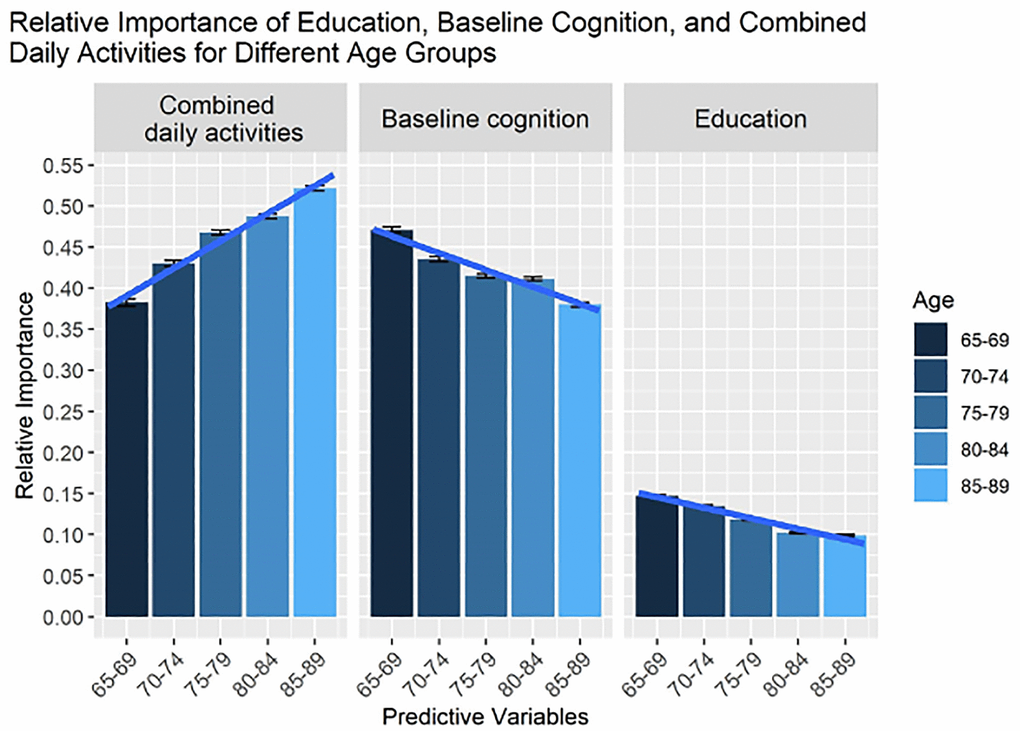
Figure 2. The relative importance of all life activities, education, and baseline memory for predicting changes in memory, as estimated from sensitivity analysis. In contrast to Figure 1, we show the combined importance of life activities (total sum). Note that on average age-related changes are characterized as an increasing trend for the combined effect of life activities on changes in cognitive health, whereas effects of education and baseline cognition are decreasing.
The importance of daily activities increases with age
As can be seen from Figure 2, the relative importance of combined daily activities on memory increased with age, which was confirmed by ANOVA (p < 0.001). Furthermore, pairwise comparisons confirmed significant differences in the relative importance of combined daily activities between age groups with p < 0.0002 for all pairs. In contrast to the combined effects of daily activities, the importance of education and baseline cognition on memory decreased with age (see Figure 2), which was confirmed by ANOVA (p < 0.001). Pairwise comparisons also showed significant differences between age groups (p < 0.001) except the 75–79 and 80–84 age groups for baseline cognition and 80–84 and 85–89 age groups for education were not significantly different.
Discussion
Current health and medical literatures provide inconclusive information on what factors can impact aging memory, leading to confusion amongst both medical and general communities. Some of this confusion could be explained by methodological limitations such as a low number of study participants, or the complexity of measuring the impact of daily activities across a lifespan. To avoid these limitations, this study used the extensive database of the Health Retirement Study [19] and created machine-learning analyses to investigate the relationship between daily activities (identified from literature) and aging memory, controlling for robust factors such as level of education and baseline memory.
Our analyses replicated the results of previous studies that showed baseline cognition and memory health [20] and level of education [8] are both critical factors that influence the rate of memory decline. Such studies found a moderate association between the rate of memory change and baseline memory, which suggests that changes in memory reflect person-specific factors and are not an inevitable result of the aging process. A novel aspect of our results is the confirmation of these findings using real-life context data and much larger sample size.
To further explore whether other factors influence aging memory, this study was extended to investigate the impact of several daily activities. Several previous studies have reported a positive relationship between some daily activities such as physical activity [12] or music [13] on the cognitive aging process. Our findings confirm that the contribution of individual daily activities for predicting changes in memory is significant but relatively small. Specifically, we estimate that the relative, quantitative importance of individual daily activities does not exceed a 10% change in the baseline memory. This result is consistent with those from previous studies, in which the effect of physical interventions on cognitive functioning was similarly estimated to be around 10% and independent of the length of those interventions [21, 22]. These small effects may also explain the conflicting or inconsistent findings in the literature and the limitation of focusing on the benefits of a single activity.
A potential limit could be the interplay between ‘self-reported frequency of daily activities’ and more objective values such as ‘hour per month’. This relationship could be complex. However, this issue was addressed in previous studies and their results showed that self-reported values, rather than more objective indicators, represent the issue in questions more accurately [23, 24]. Given that older adults do not typically engage in a singular daily activity but in an assortment of them, the combined effects of various daily activities were investigated. The findings of this study clearly showed that the effect of combined, rather than single, daily activities on memory significantly increases with age. Indeed, by the age of 90, the importance of combined daily activities on memory had increased by almost 40%. Interestingly, this age-dependent trend was not apparent when considered separately for individual daily activities. A clear, monotonic pattern of age-related changes in the relative importance of individual daily activities was not observed. More importantly, not a single daily activity was found to be significant for all five age groups. It was only after combining individual daily activities that age-related changes in the importance of daily activities for predicting changes in memory were observed. Although additional studies are required to model the key factors linking daily activities to cognitive benefits in older adults, our findings indicate that, as individuals age, their cognitive health is less influenced by historical factors and is increasingly influenced by more immediate factors such as daily tasks and leisure activities. This suggests that the decline in cognition associated with aging can be reduced by having an individual remain active both physically and mentally.
Future research and study limitations
While the results of this study are encouraging, the findings are based on a statistical model that uses historical observations relating to daily activities, education and cognition baseline as predicting variables. In addition, the study had two other significant limitations: i) The data set did not include other potentially influential factors such as diet or socioeconomic factors. ii) Causal relationships were not considered. For example, part of the memory baseline may be explained by education, which may ultimately affect the quantitative assessment of relative importance. Similarly, engagement in one daily activity may be directly affected by the engagement in another activity. This relationship between daily activities needs to be understood further to develop causal relationship models in the future. Despite these limitations, and because the considered set of daily activities is unlikely to be highly interconnected, we feel that the results of this present study are valid, but additional studies need to be conducted to confirm our findings substantially.
Policy recommendations
Care for patients with dementia is challenging, labor-intensive, and chronic, which generates high costs for health systems. Currently, in the United States, the combined direct and indirect care cost for dementia is estimated to be about $277 billion per year [2], almost certainly increasing in the coming years. Since the 1980s, significant efforts have been made to develop drug treatments for dementia, but these efforts have largely been unsuccessful and current drug-based treatments have little or no efficacy in rehabilitating or reducing the symptoms of dementia. Moreover, the development of drug-based therapies is costly and slow. It means that by the time an effective drug is discovered, validated, and brought to market, a crucial window may have closed in which we could have slowed the onset of dementia and sustained normal cognitive health. Our research suggests that it is possible to improve the well-being of the older adults. This may be achieved by non-pharmacological interventions that promote active aging and engagement in a rich array of daily activities. This would likely have significant benefits in improving the cognitive health of older adults and would reduce demands on health services and family caregivers stemming from dementia and other health conditions.
Methods
Data source
Data obtained through interviews and surveys were taken from the 2008 and 2014 releases of the Health and Retirement Study (HRS), a longitudinal cohort study on health, retirement, and aging [19]. In particular, the study contained information from individuals with a broad range of ages on cognition and daily activities.
Sample selection
Three thousand five hundred seventy participants were selected based on: i) aged between 65 and 89 years (2014 release); ii) education record (2014 release); iii) memory assessment (2008 and 2014 release); iv) at least one daily activity (2014 release; see next section); and v) having gone through typical testing procedures (i.e., no interruptions, or problems with hearing). The participants were then reduced to 3,210 based by removing those with more than 15% missing data across all variables. Any remaining missing data were replaced with the median across participants, separately for each variable. The selected 3,210 participants were then grouped into five age ranges: 65–69 years (757); 70–74 years (840); 75–79 years (800); 80–84 years (518); and 85–89 years (295). It is worth noting that the upper age limit for the third age category corresponded to the USA’s life expectancy, which is about 79 years of age.
Measures
Baseline memory and memory changes
The goal of our research was to explore the influence of background factors and current daily activities on memory and to examine how the importance of background and current daily activities change with age. Memory was assessed in the HRS by ‘immediate’ and ‘delayed’ word recall tests that are commonly used in dementia diagnosis [25]. Memory changes that often precede cognitive decline are widely used to measure cognitive health in older adults [26, 27]. During the recall test, participants were asked to memorize a list of ten words, which was read aloud by the interviewer [25]. Participants were then asked to recall as many words as possible (immediate recall). After five minutes of answering other questions, participants were then asked again to recall as many words as possible (delayed recall). The baseline memory measure was the sum of words correctly remembered during both the immediate and delayed recalls (i.e., ranging between 0 and 20). The changes in memory were subsequently defined as the difference between the HRS 2008 and 2014 baseline memory measures.
Daily activities
33 daily activities (see Table 2) were selected from the HRS 2008 database that covered diverse aspects of daily life, including cognitively challenging, mild physical, housework and leisure activities. Daily activities associated with personal hygiene, dressing, eating, maintaining continence were excluded. Participants had been asked to indicate their level of engagement in each activity ranging from ‘never’ or ‘at least once a month’ to ‘several times a month’ up to ‘daily’. Although continuous by design, the daily activities were mapped into six or seven ordered categories.
Table 2. List of all daily activities included in our initial data set.
| Number | Daily Activity |
| 1 | CARE ADULT |
| 2 | DO ACTIVITIES WITH GRANDCHILDREN |
| 3 | VOLUNTEER YOUTH |
| 4 | CHARITY WORK |
| 5 | EDUCATION |
| 6 | ATTEND SPORTS/SOCIAL/CLUB |
| 7 | ATTEND NON RELIGIOUS ORGS |
| 8 | PRAY PRIVATELY |
| 9 | READ |
| 10 | WATCH TELEVISION |
| 11 | DO WORD GAMES |
| 12 | PLAY CARDS AND GAMES |
| 13 | DO WRITING |
| 14 | USE COMPUTER |
| 15 | MAINTENANCE/GARDENING |
| 16 | BAKE OR COOK |
| 17 | SEW OR KNIT |
| 18 | DO HOBBY |
| 19 | PLAY SPORT/EXERCISE |
| 20 | WALK FOR 20 MINS |
| 21 | PARTICIPATE COMMUNITY ARTS GRP |
| 22 | MEET UP WITH CHILDREN |
| 23 | SPEAK ON PHONE WITH CHILDREN |
| 24 | WRITE OR EMAIL CHILDREN |
| 25 | COMMUNICATE BY SOCIAL MEDIA |
| 26 | MEET UP WITH OTHER FAMILY MEMBERS |
| 27 | SPEAK ON PHONE WITH OTHER FAMILY MEMBERS |
| 28 | WRITE OR EMAIL OTH FAMILY MEMBERS |
| 29 | COMMUNICATE BY SOCIAL MEDIA WITH FAMILY MEMBERS |
| 30 | MEET UP WITH FRIENDS |
| 31 | SPEAK ON PHONE WITH FRIENDS |
| 32 | WRITE OR EMAIL FRIENDS |
| 33 | COMMUNICATE BY SOCIAL MEDIA WITH FRIENDS |
Historical factors
The level of education in years from the HRS 2008 database was also included in the analysis as it is considered a well-established measure of cognitive and memory reserve [8].
Data analyses
Taking into account education, machine learning was used to predict age-related changes in memory from participation in daily activities. Given the growing body of evidence indicating non-linear relationships between daily activities and physical health outcomes, no assumptions were made regarding the relationship between daily activities and potential cognitive outcomes. To that end, a two-step approach was followed to identify and quantify the relationship between daily activities and memory function.
Step 1. Feature selection to identify relevant predictive variables
In step one, a combination of non-linear techniques (Distance Correlation [28] and Random Forest [29]) was used to identify the most relevant variables with the goal of dimensionality reduction and support vector regression (SVR) for constructing predictive models. It should be noted that the use of SVR was not to focus on predictive accuracy but rather to investigate the importance of each predicting variable in explaining the target variable and exploring how a variable’s importance changes as a function of age.
Still, we estimated the predictive accuracy by randomly splitting dataset into training and testing subsets, with 80% used for training, and 20% used for testing for each age category, and further computing the coefficient of determination for the testing set. We repeated this estimation for 100 times, with mean coefficient of determination being 0.51. Non-linear techniques were considered most appropriate due to their versatile ability to capture functional relationships between variables of interest and that linearity between variables could not be assumed. Besides, non-linear techniques are better at accurately capturing strong associations between non-linearly dependent variables, while retaining the ability to capture strong associations between linearly dependent variables [30].
Distance Correlation is a measure of dependence between two random variables, not necessarily linearly related, which is zero only when variables are independent [28]. The distance correlation between memory changes and the daily activities was computed separately for each age group. The significance of correlations was tested with a bootstrap procedure, based on ~300 samples, as implemented in the R statistical toolbox ‘Energy’ [31].
Random forest is an ensemble learning algorithm that is an extension of decision trees. An algorithm progressively learns to predict the value of a target variable based on several input variables. Random forests are a way of averaging multiple decision trees, trained on subsamples of the same training data, to reduce the variance of the trained model [29]. One advantage of using random forests is the ability to identify, as a batch, groups of predicting variables that are related to the target variable. Random forest regression was used to predict changes in memory based on daily activities, education, and memory baseline for each age group. Each feature’s statistical significance was estimated for each age group using a permutation test based on ~300 samples, as implemented in the R package ‘pRF’ [32]. For further analysis, a subset of daily activities, which were found to be associated with memory changes at the 95% confidence interval (at least in one age category and by one method), was identified.
Step 2. Sensitivity analysis to quantify the relative importance of daily activities
The next step was to predict changes in memory using a sub-set of daily activities, which were identified as significant in the previous analysis, as well as education level and baseline cognition from the 2008 HRS release. Each age category was analyzed separately to quantify the relative importance of each predicting variable in its ability to explain the variability of the target variable. This procedure was performed using an SVR (based on a Gaussian kernel with a 10-range grid search for hyper-parameter optimization [33]) in combination with sensitivity analysis. Sensitivity analysis is based on the idea that changes in a relevant feature should yield substantial changes in the target variable. Features (variables) with low sensitivity are considered less important, whereas those with high sensitivity are considered more valuable. A random selection of several training sub-samples of the data with the corresponding responses of the target variable, taking into account the physiological well-being of an individual, was implemented in the R statistical toolbox ‘rminer’ [34]. The measure of importance was defined as a relative measure of dispersion in the target variable caused by changes in a given feature. Therefore, the importance of different features represents a unified measure that can be subsequently summed up.
The sensitivity analysis was repeated 200 times for each age group. As a result, each predicting variable was associated with a distribution of its important values, describing its ability to cause changes in the predicting variable. Analysis of variance (ANOVA - a generalization of the two-sample t-test to more than two groups with the null hypothesis that the group means are equal) was applied to test for significance of the differences in a variable’s importance between age groups. To further compare the variable’s importance between age groups, a series of two-sample Tukey Honest Significant Differences tests [35] was performed on a pairwise basis.
Consent for publication
Corresponding Author has the right to grant on behalf of all authors and does grant on behalf of all authors, an exclusive licence (or nonexclusive for government employees) on a worldwide basis to the BMJ Publishing Group Ltd to permit this article (if accepted) to be published in BMJ editions and any other BMJPGL products and sublicences such use and exploit all subsidiary rights, as set out in our licence.
Author Contributions
Olga Krakovska, Gregory J. Christie, Faranak Farzan, Andrew Sixsmith, Martin Ester, Sylvain Moreno. Olga Krakovska had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Study concept and design: Martin Ester, Sylvain Moreno, Olga Krakovska. Acquisition, analysis, or interpretation of data: All authors. Drafting of the manuscript: All authors. Critical revision of the manuscript for important intellectual content: All authors. Statistical analysis: Olga Krakovska. Obtained funding: Martin Ester, Sylvain Moreno, Greg Christie. Administrative, technical, or material support: Martin Ester, Sylvain Moreno, Faranak Farzan, Andrew Sixsmith. Study supervision: Martin Ester, Sylvain Moreno, Andrew Sixsmith. All authors provided input into the design, edited and revised the manuscript. All authors read and approved the final manuscript. Dissemination of the results to study participants is impossible.
Acknowledgments
The Health and Retirement Study (HRS) is sponsored by the National Institute on Aging (grant number NIA U01AG009740) and is conducted by the University of Michigan.
Conflicts of Interest
All authors have completed the Unified Competing Interest form (available on request from the corresponding author) and declare: no support from any organisation for the submitted work [or describe if any]; no financial relationships with any organisations that might have an interest in the submitted work in the previous three years, no other relationships or activities that could appear to have influenced the submitted work.
Funding
This study was supported by grants from the Simon Fraser University Community Trust Endowment Fund and an AGE-WELL Catalyst grant to M.E. and S.M. The study’s funders had no role in the study design, data collection, analysis, interpretation and report writing. The corresponding author had full access to all the data and had final responsibility for the decision to submit for publication.
References
- 1. Alzheimer's Association. Alzheimer's Disease Facts and Figures. 2018. https://www.alz.org/alzheimers-dementia/facts-figures.
- 2. Crum RM, Anthony JC, Bassett SS, Folstein MF. Population-based norms for the Mini-Mental State Examination by age and educational level. JAMA. 1993; 269:2386–91. https://doi.org/10.1001/jama.1993.03500180078038 [PubMed]
- 3. Stern Y. Cognitive reserve. Neuropsychologia. 2009; 47:2015–28. https://doi.org/10.1016/j.neuropsychologia.2009.03.004 [PubMed]
- 4. Wrenn KA, Maurer TJ. Beliefs about older workers' learning and development behavior in relation to beliefs about malleability of skills, age-related decline, and control. J Appl Soc Psychol. 2004; 34:223–42. https://doi.org/10.1111/j.1559-1816.2004.tb02546.x
- 5. Stern Y. What is cognitive reserve? Theory and research application of the reserve concept. J Int Neuropsychol Soc. 2002; 8:448–60. https://doi.org/10.1017/S1355617702813248 [PubMed]
- 6. Satz P. Brain reserve capacity on symptom onset after brain injury: a formulation and review of evidence for threshold theory. Neuropsychology. 1993; 7:273–95. https://doi.org/10.1037/0894-4105.7.3.273
- 7. McClearn GE, Johansson B, Berg S, Pedersen NL, Ahern F, Petrill SA, Plomin R. Substantial genetic influence on cognitive abilities in twins 80 or more years old. Science. 1997; 276:1560–63. https://doi.org/10.1126/science.276.5318.1560 [PubMed]
- 8. Stern Y, Albert S, Tang MX, Tsai WY. Rate of memory decline in AD is related to education and occupation: cognitive reserve? Neurology. 1999; 53:1942–47. https://doi.org/10.1212/wnl.53.9.1942 [PubMed]
- 9. Ganguli M, Vander Bilt J, Saxton JA, Shen C, Dodge HH. Alcohol consumption and cognitive function in late life: a longitudinal community study. Neurology. 2005; 65:1210–17. https://doi.org/10.1212/01.wnl.0000180520.35181.24 [PubMed]
- 10. Anstey KJ, von Sanden C, Salim A, O'Kearney R. Smoking as a risk factor for dementia and cognitive decline: a meta-analysis of prospective studies. Am J Epidemiol. 2007; 166:367–78. https://doi.org/10.1093/aje/kwm116 [PubMed]
- 11. Valls-Pedret C, Sala-Vila A, Serra-Mir M, Corella D, de la Torre R, Martínez-González MÁ, Martínez-Lapiscina EH, Fitó M, Pérez-Heras A, Salas-Salvadó J, Estruch R, Ros E. Mediterranean Diet and Age-Related Cognitive Decline: A Randomized Clinical Trial. JAMA Intern Med. 2015; 175:1094–103. https://doi.org/10.1001/jamainternmed.2015.1668 [PubMed]
- 12. Laurin D, Verreault R, Lindsay J, MacPherson K, Rockwood K. Physical activity and risk of cognitive impairment and dementia in elderly persons. Arch Neurol. 2001; 58:498–504. https://doi.org/10.1001/archneur.58.3.498 [PubMed]
- 13. Hanna-Pladdy B, MacKay A. The relation between instrumental musical activity and cognitive aging. Neuropsychology. 2011; 25:378–86. https://doi.org/10.1037/a0021895 [PubMed]
- 14. Pagnoni G, Cekic M. Age effects on gray matter volume and attentional performance in Zen meditation. Neurobiol Aging. 2007; 28:1623–27. https://doi.org/10.1016/j.neurobiolaging.2007.06.008 [PubMed]
- 15. Bassuk SS, Glass TA, Berkman LF. Social disengagement and incident cognitive decline in community-dwelling elderly persons. Ann Intern Med. 1999; 131:165–73. https://doi.org/10.7326/0003-4819-131-3-199908030-00002 [PubMed]
- 16. Zunzunegui MV, Alvarado BE, Del Ser T, Otero A. Social networks, social integration, and social engagement determine cognitive decline in community-dwelling Spanish older adults. J Gerontol B Psychol Sci Soc Sci. 2003; 58:S93–100. https://doi.org/10.1093/geronb/58.2.s93 [PubMed]
- 17. Ngandu T, Lehtisalo J, Solomon A, Levälahti E, Ahtiluoto S, Antikainen R, Bäckman L, Hänninen T, Jula A, Laatikainen T, Lindström J, Mangialasche F, Paajanen T, et al. A 2 year multidomain intervention of diet, exercise, cognitive training, and vascular risk monitoring versus control to prevent cognitive decline in at-risk elderly people (FINGER): a randomised controlled trial. Lancet. 2015; 385:2255–63. https://doi.org/10.1016/S0140-6736(15)60461-5 [PubMed]
- 18. Christie GJ, Hamilton T, Manor BD, Farb NAS, Farzan F, Sixsmith A, Temprado JJ, Moreno S. Do Lifestyle Activities Protect Against Cognitive Decline in Aging? A Review. Front Aging Neurosci. 2017; 9:381. https://doi.org/10.3389/fnagi.2017.00381 [PubMed]
- 19. Health and Retirement Study. HRS 2014 Core (Final V2.0), and HRS 2008 Core (Final V3.0)) public use dataset. Produced and distributed by the University of Michigan with funding from the National Institute on Aging (grant number NIA U01AG009740). Ann Arbor, MI. 2014.
- 20. Wilson RS, Beckett LA, Barnes LL, Schneider JA, Bach J, Evans DA, Bennett DA. Individual differences in rates of change in cognitive abilities of older persons. Psychol Aging. 2002; 17:179–93. https://doi.org/10.1037/0882-7974.17.2.179 [PubMed]
- 21. Oken BS, Zajdel D, Kishiyama S, Flegal K, Dehen C, Haas M, Kraemer DF, Lawrence J, Leyva J. Randomized, controlled, six-month trial of yoga in healthy seniors: effects on cognition and quality of life. Altern Ther Health Med. 2006; 12:40–47. [PubMed]
- 22. Legault C, Jennings JM, Katula JA, Dagenbach D, Gaussoin SA, Sink KM, Rapp SR, Rejeski WJ, Shumaker SA, Espeland MA, and SHARP-P Study Group. Designing clinical trials for assessing the effects of cognitive training and physical activity interventions on cognitive outcomes: the Seniors Health and Activity Research Program Pilot (SHARP-P) study, a randomized controlled trial. BMC Geriatr. 2011; 11:27. https://doi.org/10.1186/1471-2318-11-27 [PubMed]
- 23. Chirikos TN, Nestel G. Economic determinants and consequences of self-reported work disability. J Health Econ. 1984; 3:117–36. https://doi.org/10.1016/0167-6296(84)90002-x [PubMed]
- 24. Anderson KH, Burkhauser RV. The Retirement-Health Nexus: A New Measure of an Old Puzzle. J Hum Resour. 1985; 20:315–30. https://doi.org/10.2307/145884
- 25. Ofstedal MG, Fisher GG, Herzog AR. Documentation of Cognitive Functioning Measures in the Health and Retirement Study. 2005. Ann Arbor, MI: University of Michigan. https://doi.org/10.7826/ISR-UM.06.585031.001.05.0010.2005
- 26. Albert M, Soldan A, Gottesman R, McKhann G, Sacktor N, Farrington L, Grega M, Turner R, Lu Y, Li S, Wang MC, Selnes O. Cognitive changes preceding clinical symptom onset of mild cognitive impairment and relationship to ApoE genotype. Curr Alzheimer Res. 2014; 11:773–84. https://doi.org/10.2174/156720501108140910121920 [PubMed]
- 27. Hedden T, Gabrieli JD. Insights into the ageing mind: a view from cognitive neuroscience. Nat Rev Neurosci. 2004; 5:87–96. https://doi.org/10.1038/nrn1323 [PubMed]
- 28. Székely GJ, Rizzo ML, Bakirov NK. Measuring and testing dependence by correlation of distances. Ann Stat. 2007; 35:2769–94. https://doi.org/10.1214/009053607000000505
- 29. Genuer R, Poggi JM, Tuleau-Malot C. Variable selection using random forests. Pattern Recognit Lett. 2010; 31:2225–36. https://doi.org/10.1016/j.patrec.2010.03.014
- 30. Krakovska O, Christie G, Sixsmith A, Ester M, Moreno S. Performance comparison of linear and non-linear feature selection methods for the analysis of large survey datasets. PLoS One. 2019; 14:e0213584. https://doi.org/10.1371/journal.pone.0213584 [PubMed]
- 31. Rizzo ML, Szekely GJ. energy: E-Statistics: Multivariate Inference via the Energy of Data. 2016. https://CRAN.R-project.org/package=energy.
- 32. Chakravarthy A. pRF: Permutation Significance for Random Forests. 2015. https://cran.r-project.org/web/packages/pRF.
- 33. Platt J. Probabilistic outputs for support vector machines and comparisons to regularized likelihood methods. Advances in large margin classifiers. 1999; 10:61–74.
- 34. Cortez P. rminer: Data Mining Classification and Regression Methods. 2016. https://cran.r-project.org/web/packages/rminer.
- 35. Tukey JW. Comparing individual means in the analysis of variance. Biometrics. 1949; 5:99–114. https://doi.org/10.2307/3001913 [PubMed]


