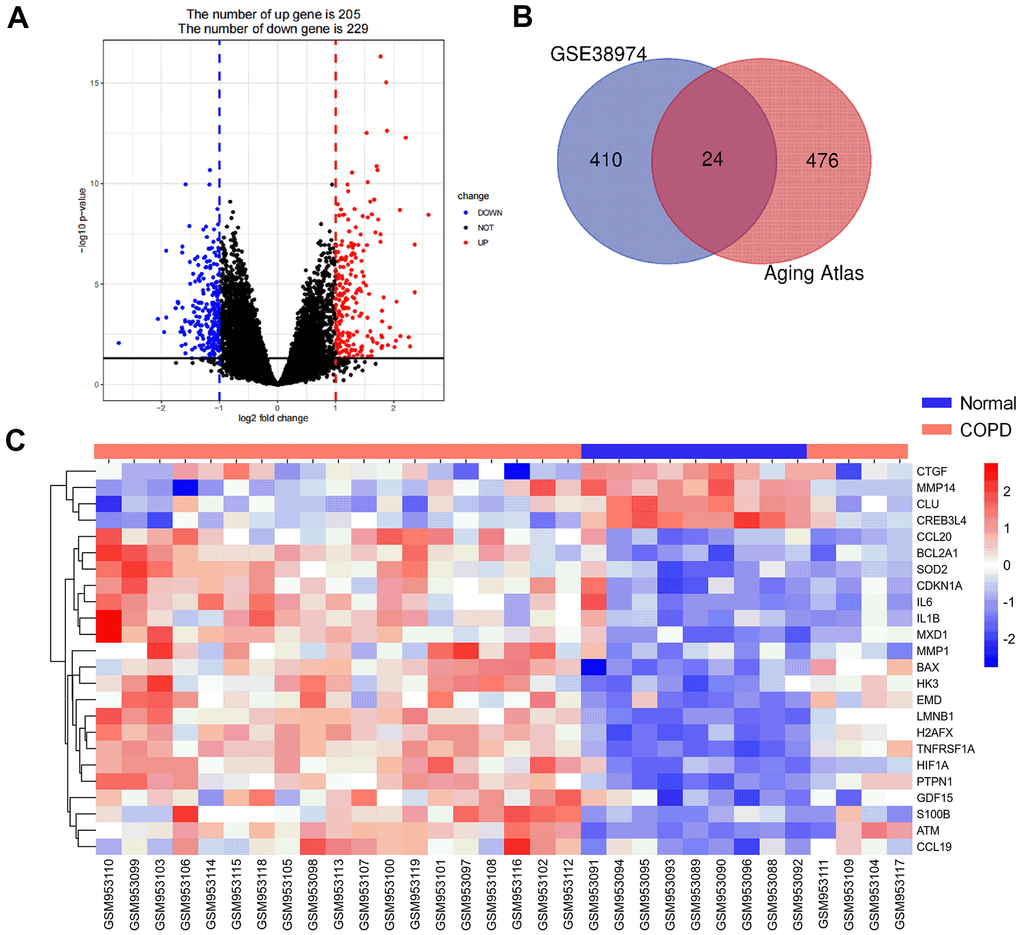
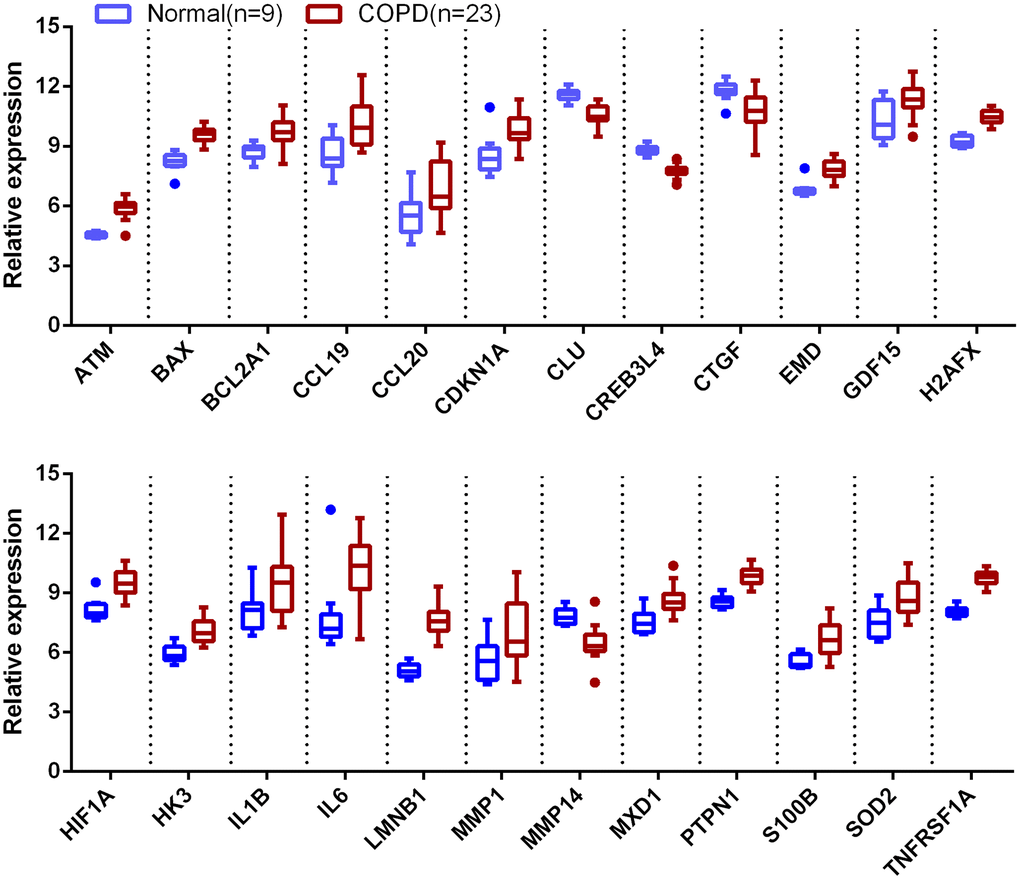
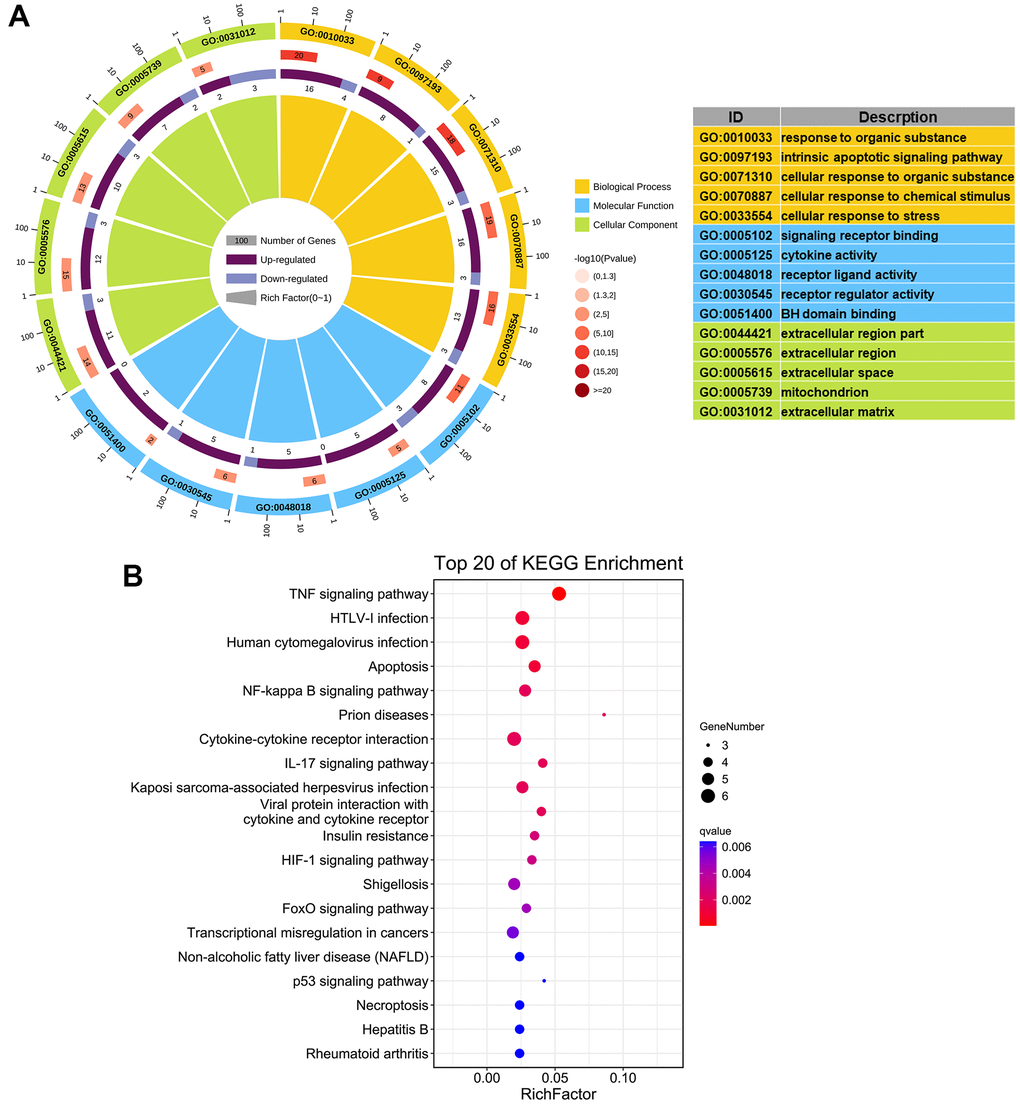
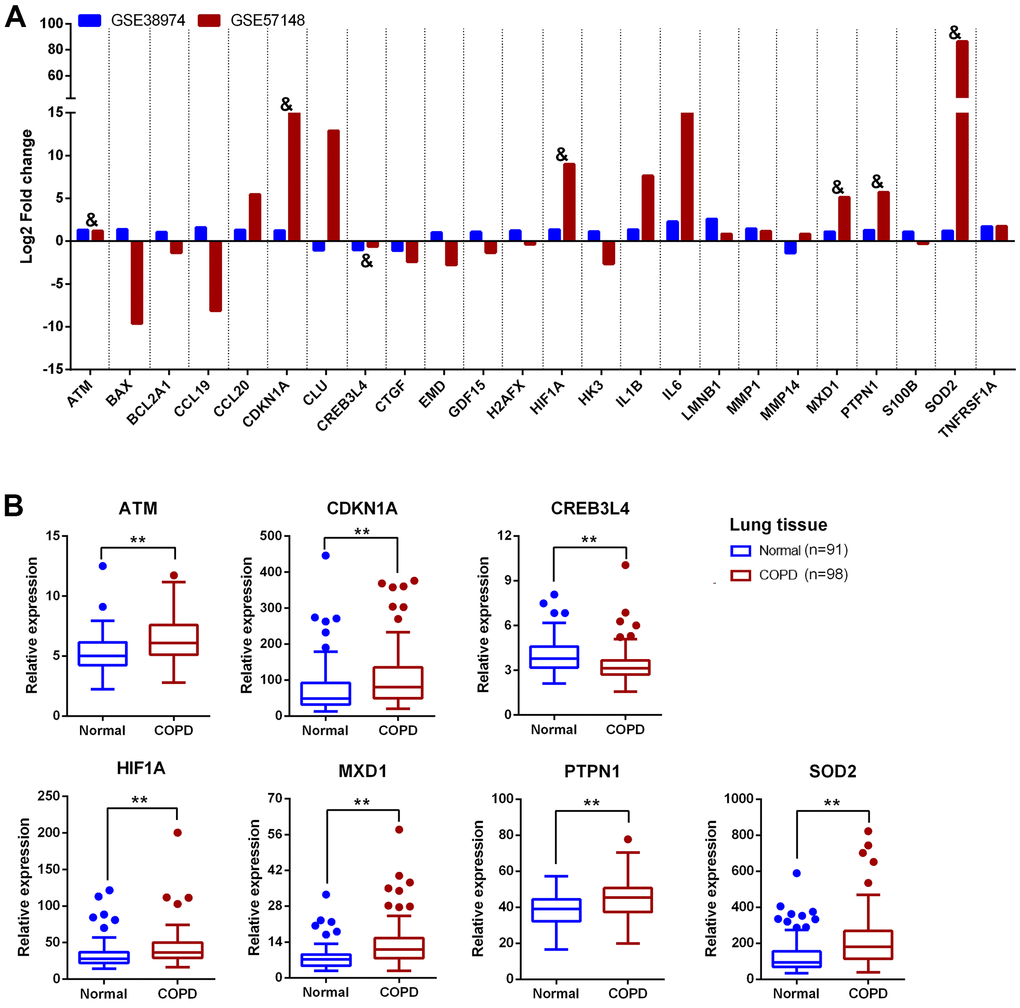
Introduction
Chronic obstructive pulmonary disease (COPD) is a complicated and heterogeneous respiratory condition with a high morbidity and mortality over three million people died from this disease worldwide per year, which causes a huge burden to medical and financial systems globally [1, 2]. Although previous studies reported that the main risks for COPD are long-term exposure to cigarette smoke or air pollution and genetic factors, the aging process is also important in the pathogenesis of COPD [3–6]. A recent study indicated that the prevalence of COPD increases with age (5.1% in 35–54 year-olds, 13.3% in 55–64 year-olds, and 21.7% in those older than 65) [7] and patients older than 65 year-old had a five-fold increase at risk of COPD process compared to patients younger than 40 [8]. Investigation of the relationship between aging associated genes and progression of COPD may provide new biomarkers for diagnosis and personalized therapy of this disease.
Aging is the important physiological and pathophysiological processes in human life, which involves inflammation, oxidative stress, mitochondrial dysfunction, epigenetic alterations, cell senescence and death, and regulates COPD development. Dysregulation of aging-related genes, such as SIRT1 and SIRT6, were recently demonstrated to be associated with COPD [9–14]. Previous studies showed that the reduction of SIRT1 and SIRT6 expression could exacerbate the response to oxidative stress, premature senescence and chronic inflammation, which further accelerate the aging process in the lung [11–13]. The activity of SIRT1 may be partially enhanced by treatment with the anti-inflammatory molecule Resveratrol to reverse the progression of CSE-induced COPD [14]. Li et al. found that Klotho, an anti-aging protein, could inhibit the expression of inflammatory mediators such as MMP-9, TNF-α, and IL-6 via the NF-κB pathway in COPD [15]. Another study demonstrated that Klotho down-regulation in COPD was associated with accelerated lung aging in COPD development and increased oxidative stress, inflammation, and apoptosis of airway epithelial cells [16]. However, the roles of aging-related genes during COPD development remain largely unknown.
MicroRNAs (miRNAs) are small non-coding RNA 18–22 nucleotides in length that block protein translation through miRNA-mRNA interactions, or increase mRNA degradation [17]. Abnormal miRNA expression is important in cancer and other human diseases due to their importance in pathophysiological processes and several miRNA-based therapeutics have applied to clinical testing, such as miR-34 mimic reached phase I clinical trials for cancer treatment and miR-122 antagonist reached phase II trials for hepatitis treatment [18, 19]. The roles of miRNA in COPD have been examined over the past few years [20, 21]. Several studies highlighted that miR-34a was up-regulated in patients with severe COPD and involved in the pathogenesis of COPD by affecting the HIF-1α-dependent lung structure maintenance program [22], regulating the apoptosis of human pulmonary microvascular endothelial cells by directly targeting Notch1 [23], and orchestrating the oxidative stress response by regulating the expression of SIRT1 and SIRT6 [24]. Another study indicated that elevated miR-125a-5p facilitated the senescence of lung epithelial cells to participate in the pathogenesis of smoking-induced COPD [25].
The development and application of high-throughput sequencing technology and multiple public databases have facilitated for screening new disease-related biomarkers using enrichment analyses of gene expression profiles from the gene expression omnibus (GEO) database [26]. In this study, we used gene expression profiles of COPD, aging related databases, bioinformatic analyses and validation tests to screen aging-related genes as biomarkers of COPD development, and to investigate the effects of miRNAs on these candidate aging-related genes. A workflow of this study is shown in Figure 1.
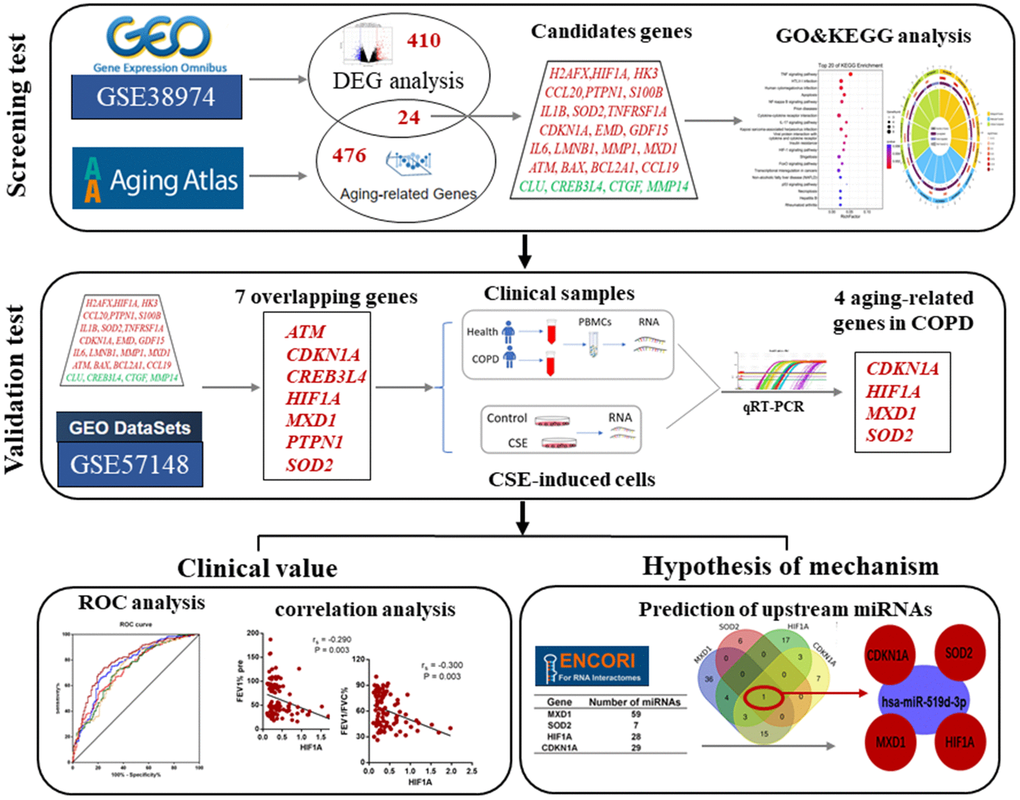
Figure 1. The work-flow of this study.
Results
Identification of miRNA upstream of CDKN1A, HIF1A, MXD1 and SOD2
Upstream miRNAs for CDKN1A, HIF1A, MXD1, and SOD2 were predicted using the StarBase3.0 software, and miRNA candidates that were identified by at least four different target-predicting programs were considered hits. The predicted miRNAs are shown in Supplementary Table 3. There were 29, 28, 59 and 7 miRNAs predicted to target CDKN1A, HIF1A, MXD1, and SOD2, respectively. hsa-miR-519d-3p was the only miRNA predicted to target all four genes (Figure 10A) with 3, 1, 2, and 3 potential interaction sites on the CDKN1A, HIF1A, MXD1, and SOD2 mRNA sequences, respectively. The seeding matched sequences are shown in Figure 10B. Subsequently, the expression of hsa-miR-519d-3p in PBMCs from COPD and normal groups was quantified by qRT-PCR, with hsa-miR-519d-3p expression significantly decreased in COPD patients, compared with the normal controls (Figure 10C). Furthermore, overexpression of hsa-miR-519d-3p in Beas-2B cell using a mimic (Figure 10D) significantly decreased the expression of CDKN1A, HIF1A, MXD1, and SOD2 (Figure 10E). Luciferase reporter assays were used to investigate the interaction of hsa-miR-519d-3p with the four genes. As shown in Figure 11A, the luciferase activities of psiCHECK2 vectors harboring wild-type 3’UTR sequences for these four genes were all reduced after co-transfection with the hsa-miR-519d-3p mimic, and the CDKN1A considerably more reduced than the other three genes. The luciferase activity of psiCHECK2 vectors harboring mutational 3’UTR sequences of CDKN1A was also measured, while the luciferase activity not significantly different in the presence of hsa-miR-519d-3p after mutating all predicted interaction sites (Figure 11B). This suggests that hsa-miR-519d-3p can bind to 3’-UTR position of the CDKN1A transcript to inhibit expression. Finally, overexpression of has-miR-519d-3p decreased the expression of p21 (the protein encoded by CDKN1A) in Beas-2B cells (Figure 11C).
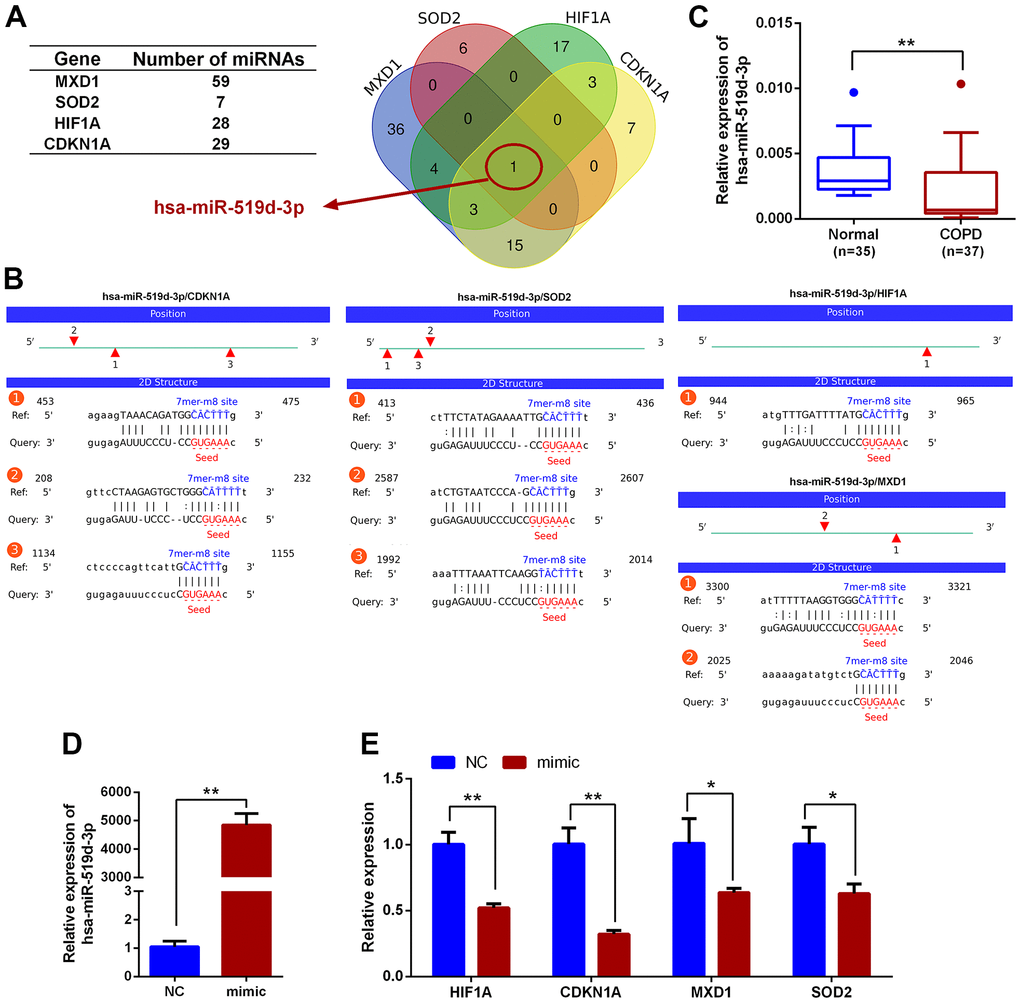
Figure 10. Prediction of miRNAs that could regulate HIF1A, CDKN1A, MXD1 and SOD2. (A) Venn diagram of miRNAs predicted to target CDKN1A, HIF1A, MXD1 and SOD2. (B) The predicted interactions of hsa-miR-519d-3p with CDKN1A, HIF1A, MXD1 and SOD2. (C) qRT-PCR expression analysis of hsa-miR-519d-3p in PBMCs from COPD and healthy volunteers; data are presented as 2(-ΔCт) relative to U6. (D) Expression analysis of hsa-miR-519d-3p in Beas-2B cell following transfection with hsa-miR-519d-3p mimic. (E) Expression of CDKN1A, HIF1A, MXD1 and SOD2 in Beas-2B cell following transfecting with hsa-miR-519d-3p mimic; data are presented as 2(-ΔΔCт) relative to GAPDH. *P <0.05; **P <0.01.
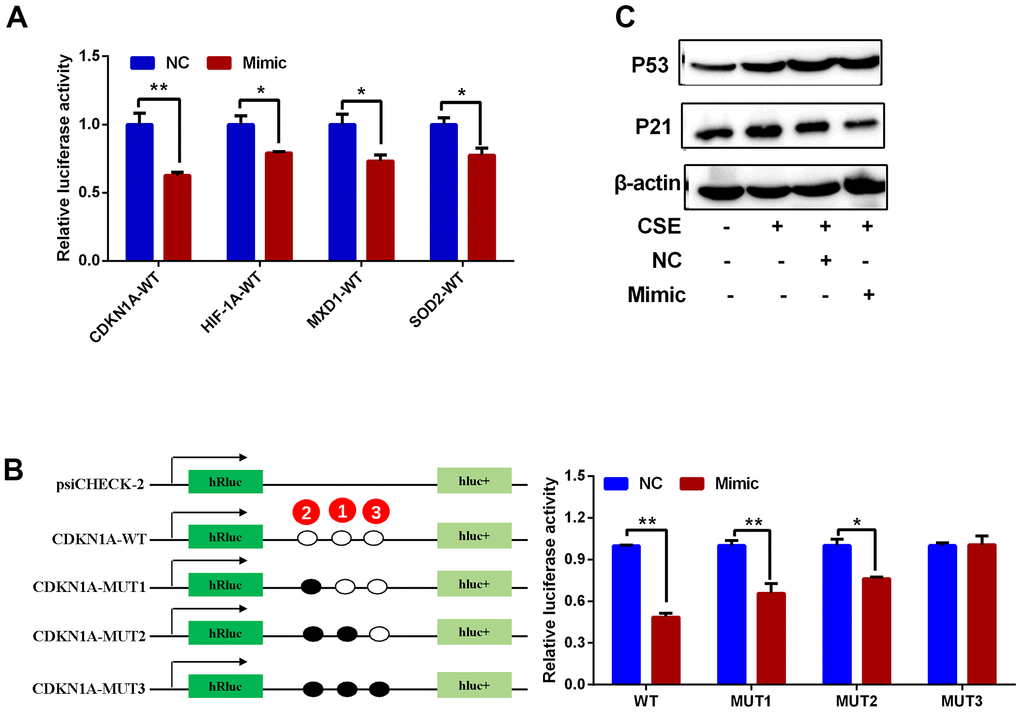
Figure 11. Regulatory interactions between hsa-miR-519d-3p and CDKN1A, HIF1A, MXD1 and SOD2. (A) Luciferase reporter assay for detection of the interaction of hsa-miR-519d-3p with 4 candidate genes. (B) Results of luciferase reporter assay demonstrate that hsa-miR-519d-3p directly binds to CDKN1A. (C) Western blot analysis of the effect of hsa-miR-519d-3p on CSE-induced aging-related proteins. *P <0.05; **P <0.01.
Discussion
The incidence of COPD is increasing worldwide. Importantly, most elderly patients suffering from COPD show age-related changes of the lung [27–29] accompanied with other chronic diseases such as cardiovascular disease, hypertension, metabolic disorder, cognitive impairment, and gastrointestinal conditions. All of these co-morbidities are linked to aging-associated pathological mechanisms [9, 10, 30, 31]. Although, the underlying mechanisms of COPD are a topic of active research, current therapeutics for COPD are mostly borrowed pharmacological therapy from the treatment of asthma [21], effective therapies are still absent for this irreversible disease, especially for older patients. It is therefore crucial to identify novel therapeutic targets specifically for COPD.
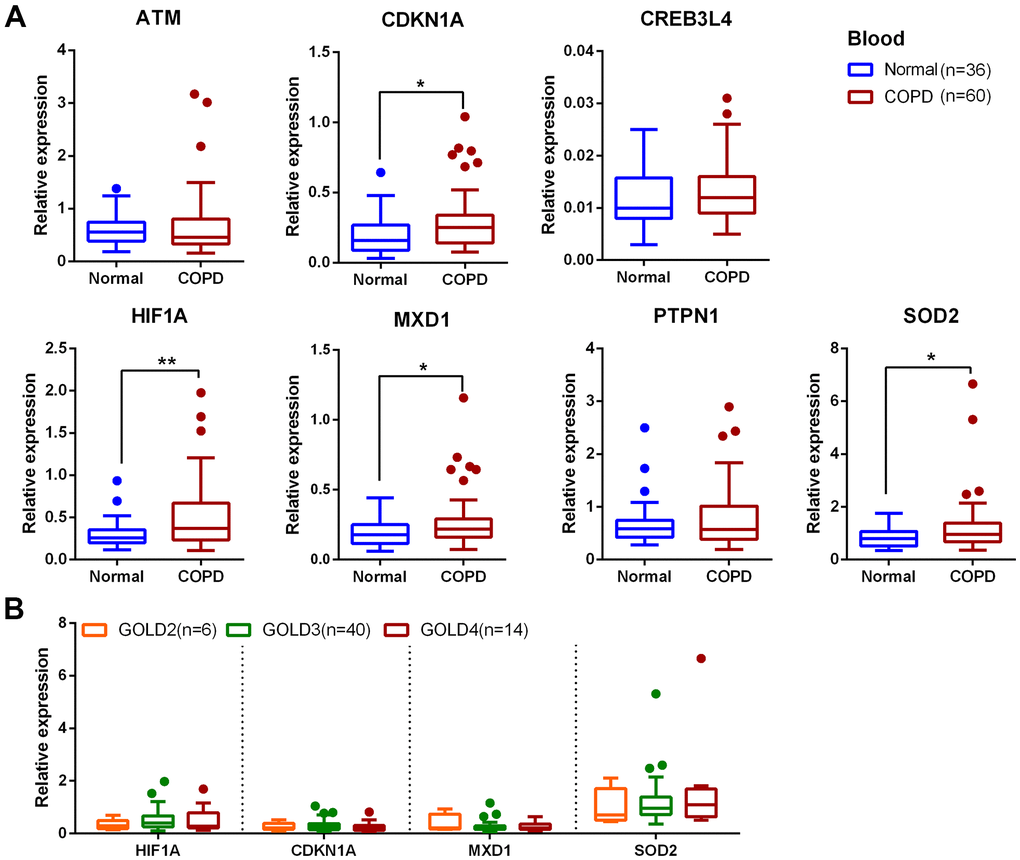
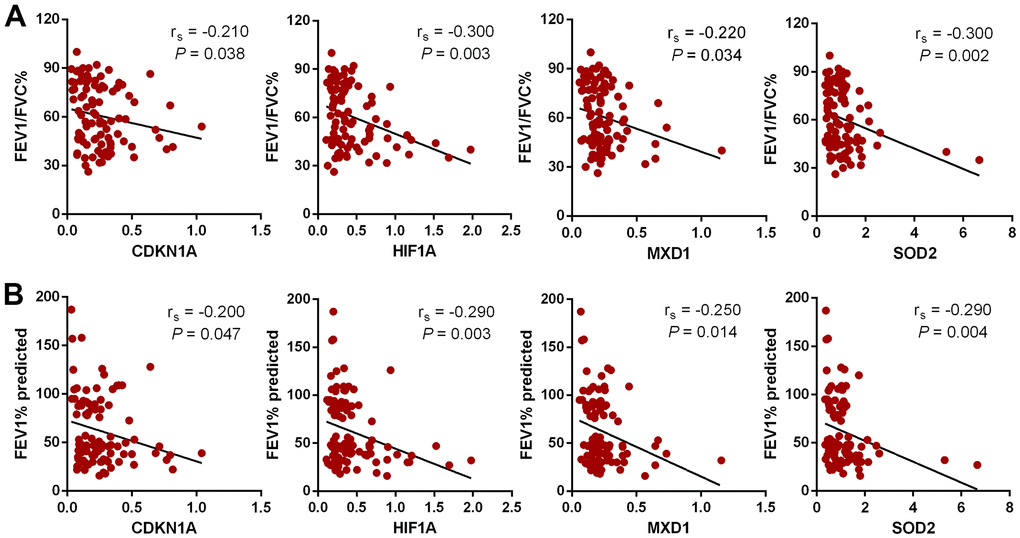
In this study 24 aging- and COPD-correlated genes were identified using a variety of bioinformatic approaches. These 24 genes principally involved in TNF, NF-κB, IL-17, HIF-1 signaling pathways, which were associated with COPD pathophysiology, such as aging, inflammation, and oxidative stress [32–34]. The genes identified in this study (CDKN1A, HIF1A, MXD1, and SOD2) were significantly upregulated in lung tissues in two different clinical COPD transcriptomics datasets, and had the potential to develop as therapeutics targets of COPD. Although the lung is the principal target organ in COPD, the blood biomarkers are increasingly applied to in diagnosis and prognosis of conditions due to the collection and dynamic monitoring easily. Hence, we validated the above four genes in blood samples from a new third cohort, and have found that the trend of the relative expression of these genes are consistent in blood and lung tissue. Furthermore, the expression of these genes was negatively correlated with pulmonary function in our validated cohorts.
Several studies have already demonstrated that dysregulation of CDKN1A, HIF1A and SOD2 are associated with the aging phenotype in COPD patients [35–41]. For example, CDKN1A (which encodes the p21 protein) is an important cyclin-dependent kinase inhibitor, which can potentiate the inflammatory response and cellular senescence, and is reported to be significantly upregulated in COPD [35, 36]. Decreasing the expression of CDKN1A can attenuate multiple pro-inflammatory stimuli-mediated lung oxidative and inflammatory responses and plays a critical role in cigarette smoke induced senescence of lung cells during COPD pathogenesis [36]. Zhang et al. identified 40 potential autophagy-related genes which were differentially expressed in COPD, including CDKN1A and HIF1A, and suggested that overexpression of CDKN1A and HIF1A might be implicated in the pathogenesis of COPD by regulating autophagy [37]. In addition, HIF1A expression was increased in the airway epithelial cells of COPD patients and serves as an important transcriptional regulator to promote the cellular response to inflammatory and oxidative stress [38, 39]. SOD2, an antioxidant-related gene, is found significantly increased in alveolar macrophages after exposure to fine atmospheric particulate matter [40]. Another study indicates that the expression of SOD2 protein is increased in bronchial epithelial cells from COPD donors following infection with rhinovirus [41]. MXD1 is a transcription factor that belongs to the MYC/MXD/MAX family, and is implicated in the pathophysiology of avian influenza virus infections [42], intracerebral hemorrhage [43], and various cancers such as osteosarcoma, lung adenocarcinoma and B cell lymphoma [44–46]. MXD1 can antagonize the transcriptional activation of c-Myc, serving as a transcription repressor [47, 48]. It can also be regulated by miRNAs to form a potential tumor-suppressing positive feedback loop [49]. However, MXD1 has not been studied in COPD. Future functional research is required to investigate the contribution of MXD1 to COPD development.
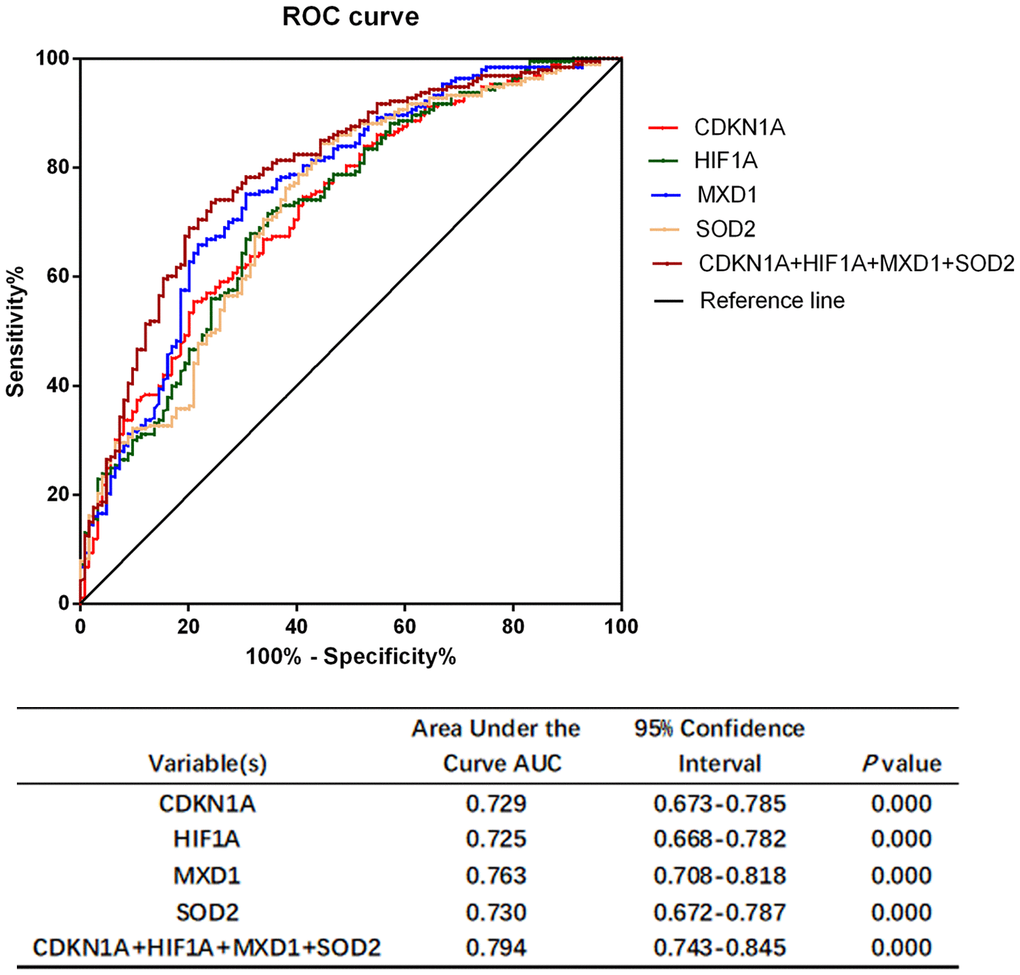
The severity of COPD is largely related to symptoms, exacerbations and comorbidities, while underdiagnosis of COPD is a common problem worldwide. The current diagnosis of COPD is depending on spirometry and patients’ demographic characteristics that could contributed to overdiagnosis or misdiagnosis aperiodically [50]. Diagnostic tests based on biomarker combinations are often evaluated by the area under the ROC curve analysis to discriminate individuals who have disease and individuals who are disease-free to assisting the clinical diagnosis [51]. In this study, the AUC values of CDKN1A, HIF1A, MXD1, and SOD2 were all greater than 0.7, and the combination of the four genes showed a better discrimination (AUC = 0.794). This suggested that CDKN1A, HIF1A, MXD1, and SOD2 had excellent discriminate ability between COPD patients and health subjects. However, whether these candidate genes can be applied to clinical practice for prevention of and intervention into COPD development is currently unclear, further studies are required to explore the molecular mechanisms of COPD in vivo and in vitro.
Previous studies have identified miRNA-based therapeutics, applied to clinical testing, and dysregulated miRNAs in COPD, such as miR-195 and miR-181c, which have potential promise in alleviating COPD in vivo, and may serve as therapeutic targets for COPD in the near future [18, 20, 21]. Therefore, we identified miRNAs potentially upstream of CDKN1A, HIF1A, MXD1, and SOD2 expression changes. hsa-miR-519d-3p was predicted to target CDKN1A, HIF1A, MXD1, and SOD2, and its expression was significantly downregulated in COPD. Previous studies have reported that hsa-miR-519d-3p is involved in tumorigenesis in gastric cancer, pancreatic cancer, and lung cancer [52–54], and correlates with cell proliferation and migration in trophoblastic cell lines [55]. The biological function of hsa-miR-519d-3p in COPD is unknown. In this study, hsa-miR-519d-3p was shown to directly bind to CDKN1A, expression of which was associated with the aging phenotype in COPD patients. Further in-depth study of hsa-miR-519d-3p may provide a new biomarker for COPD.
In conclusion, this study identified hsa-miR-519d-3p and four aging- and COPD-related genes (HIF1A, CDKN1A, MXD1, and SOD2) that may influence the development of COPD. Further study of these new targets may lead to new insight into the pathogenesis, diagnosis, and treatment of COPD.
Materials and Methods
Biological function and signaling pathway enrichment analysis
A webserver (https://www.omicshare.com/) was used to functionally annotate the genes that were associated with aging and COPD. GO enrichment analysis was used to identify enriched molecular functions, cellular components, and biological processes. KEGG enrichment analysis was used to identify signaling pathways enriched in the list of aging- and COPD-related genes.
Enrolment of subjects and collection of samples
A total of 60 COPD patients and 36 aging-matched healthy controls were enrolled from the first affiliated hospital of Wenzhou medical university from December 2017 to December 2019. All participants were asked to read a document approved by the Human Medical Ethics Committee of the First Affiliated Hospital of Wenzhou Medical University (approval no.: 2016131) and written informed consent was provided by all participants. All participants were aged 40–80 years old and COPD patients suffered from respiratory symptoms, and fitted the GOLD diagnostic criteria. Patients with other complications such as cancer, cardiac conditions, and other respiratory diseases (e.g., bronchiectasis, bronchial asthma pulmonary fibrosis and/or active tuberculosis) were excluded. Peripheral blood samples were collected, and peripheral blood mononuclear cells (PBMCs) were isolated from 10 mL blood samples using human lymphocyte separation medium (Solarbio, China). All PBMCs samples were immediately stored at -80° C.
Preparation of cigarette smoke extract (CSE) and cell cultures
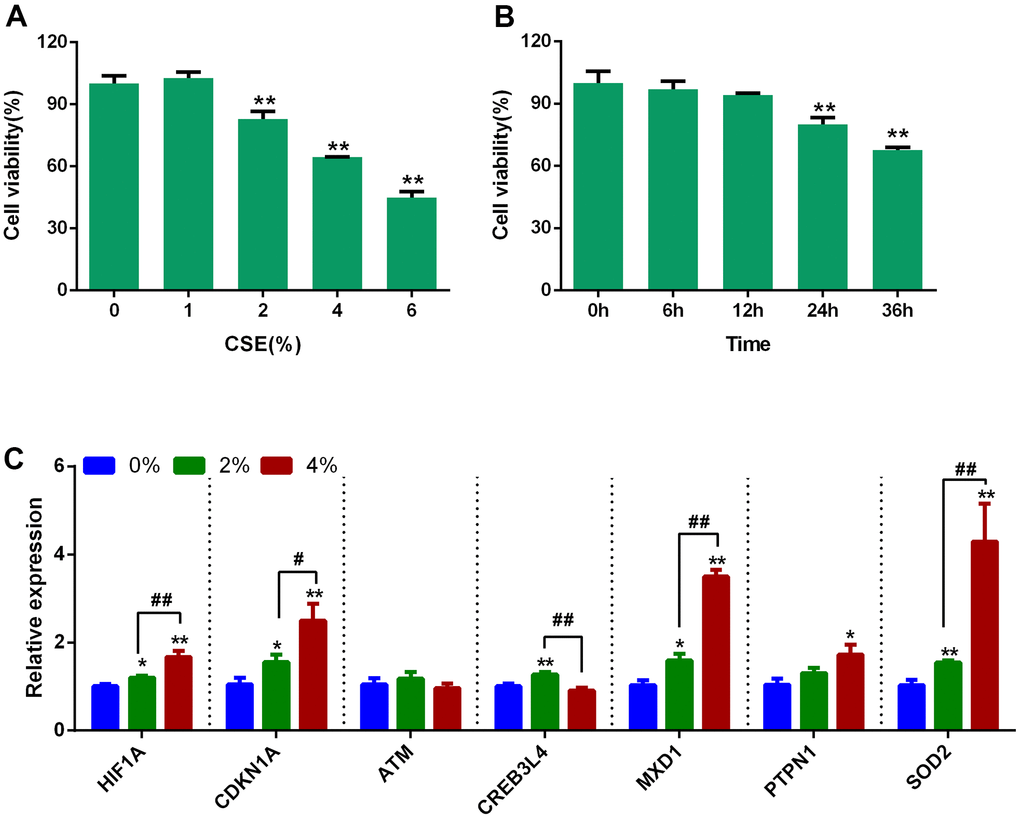
Two unfiltered cigarettes (0.8 mg of nicotine and 9 mg of tar per cigarette) were used to bubble smoke into 10 mL Dulbecco’s modified Eagle’s medium (DMEM; Gibco, USA) at a speed of 2 minutes per cigarette. The medium was sterilized using a 0.22 μm filter. This solution was considered as a 100% CSE solution to apply in subsequent experiments.
Human bronchial epithelial cells (Beas-2B) were cultured in DMEM supplemented with 10% fetal bovine serum (FBS; Gibco, USA) and under a humidified atmosphere of 5% CO2 at 37° C. The cells were grown to approximately 90% confluence before experiments using CSE or transfection with 40 nM hsa-miR-519d-3p mimic or negative control (NC) oligoribonucleotides (Sangon Biotech, China) using Lipofectamine™ 3000 (Invitrogen, USA) according to the manufacturer’s instructions.
Cell viability assay
Beas-2B cells were seeded into 96-well plates at a density of 5 × 103 cells/well and incubated overnight, then treated with CSE at 0, 1, 2, 4 and 6%. Cell viability was measured at different times (0, 6, 12, 24, and 36 hours) using cell counting kit-8 (CCK-8) reagent (YEASEN, China) according to the manufacturer’s instructions. After an incubation with CCK-8 reagent for 2 hours, the optical density was measured at 450 nm using a microplate reader (Bio-Rad Laboratories, USA). Cell viability was calculated by the following formula: (absorbance of treatment group/absorbance of control group) × 100%.
Quantitative real-time PCR (qRT-PCR)
Total RNA was extracted from PBMCs or cultured cells using M5 Hiper Universal Plus RNA Mini Kit (Mei5 Biotechnology, China). cDNA was synthesized using the cDNA synthesis kit or Mir-X miRNA First-Strand Synthesis Kit (both TaKaRa, Japan). Primers for qRT-PCR were designed via a public resource named PrimerBank (https://pga.mgh.harvard.edu/primerbank/) and synthesized by Sangon Biotech, which are listed in Table 3. qRT-PCR amplification was performed using SYBR Green PCR Premix Ex Taq™ II reagents (TaKaRa, Japan) with the Quant Studio 6 FlexI real-time PCR system (Applied Biosystems, USA), following the protocols from the commercial kits. Expression levels of tested genes were determined with the 2-ΔCt or 2-ΔΔCt method based on the endogenous control (GAPDH or U6).
Table 3. Primer sequences for validating gene candidates.
| Gene name | Primer sequence (5’-3’) | |
| Forward primer | Reverse primer | |
| HIF1A | ATCCATGTGACCATGAGGAAATG | TCGGCTAGTTAGGGTACACTTC |
| CDKN1A | TGTCCGTCAGAACCCATGC | AAAGTCGAAGTTCCATCGCTC |
| ATM | GGCTATTCAGTGTGCGAGACA | TGGCTCCTTTCGGATGATGGA |
| CREB3L4 | CAGACGCTAATTGCTCAAACTTC | CCACTTGGGTCTCCAGATTTTCT |
| MXD1 | CGTGGAGAGCACGGACTATC | CCAAGACACGCCTTGTGACT |
| PTPN1 | TCCCTTTGACCATAGTCGGAT | GTGACCGCATGTGTTAGGCA |
| SOD2 | TTTCAATAAGGAACGGGGACAC | GTGCTCCCACACATCAATCC |
| GAPDH | CAATGACCCCTTCATTGACC | TTGATTTTGGAGGGATCTCG |
| hsa-miR-519d-3p | CAAAGTGCCTCCCTTTAGAGTG | |
Prediction of upstream miRNAs
StarBase3.0 (http://starbase.sysu.edu.cn/), which includes seven miRNA target prediction programs (PITA, RNA22, miRmap, DIANA-microT, miRanda, PicTar and TargetScan) was used to predict the miRNAs that may regulate the genes we identified. Hits were chosen which appeared in at least 4 of the 7 prediction programs. The interactions between miRNA and mRNA were visualized using Miranda software.
Luciferase activity assay
The psiCHECK2 luciferase reporter vectors (Promega, USA) harboring wild-type (WT) or mutated (MUT) 3’UTR sequences were co-transfected with hsa-miR-519d-3p mimic or the corresponding negative control (NC) into human embryonic kidney (HEK) 293T cells. After a 48-hour incubation, cells were lysed and luciferase activity was measured using a dual-luciferase reporter assay system (Promega, USA), according to the manufacturer’s protocol.
Western blot
Total proteins were extracted from Beas-2B cells by using radio-immunoprecipitation assay (RIPA) buffer with protease inhibitor (Roche Applied Science, USA), then the protein concentration was measured using a BCA kit (Thermo, USA). Protein samples were separated on 12% SDS-PAGE gels at 80 V for 120 minutes and transferred onto a nitrocellulose membrane (Millipore Co, USA) at 300 mA for 1 hour using a wet transfer method. The membranes were incubated with primary antibody against p21 (10355-1-AP; Proteintech, USA), p53 (#2524; Cell Signaling Technology, USA) and β-actin (#3700; Cell Signaling Technology, USA) at 4° C overnight. Horseradish peroxidase (HRP)-conjugated secondary antibodies (A0208 and A0216; Beyotime, China) were incubated at room temperature for 1 hour, and visualized using an enhanced chemiluminescence kit and Image Lab (Bio-Rad, USA).
Statistical analysis
GraphPad Prism 6.0 (GraphPad Software Inc., San Diego, CA, USA) and SPSS 21.0 were used for statistical analysis. Student's t-test or Mann-Whitney U-test was applied for analyzing the data between two groups based on the normality of data. Spearman’s rank correlation coefficient was used to investigate the correlation between gene expression and pulmonary function of COPD patients. The area under the curve (AUC) of the receiver operating characteristics curve (ROC) was evaluated to assess effectiveness in discriminating patients with COPD from healthy participants. P <0.05 was considered statistically significant.
Availability of data and material
The data used to support the findings of this study are available from the corresponding author upon request.
Author Contributions
SZ contributed to project design, data collection, data analysis and original draft preparation; NL and GZ contributed to data collection and data analysis; LY and CC contributed to project design, sample collection, and provided clinical information; ZH contributed to project design and draft editing; YW contributed to project design, sample collection, data collection, data analysis, and draft editing. All authors have read and approved the final version of the manuscript.
Acknowledgments
The authors are grateful to the investigators who submitted the data we used to the public databases.
Conflicts of Interest
All authors declare that they have no conflicts of interest.
Funding
This study was supported by the National key R&D Program (No. 2016YFC1304000) and Shenzhen Collaborative Innovation Science and Technology Program (No. JCYJ20190808122413582).
References
- 1. GBD 2015 Chronic Respiratory Disease Collaborators. Global, regional, and national deaths, prevalence, disability-adjusted life years, and years lived with disability for chronic obstructive pulmonary disease and asthma, 1990-2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet Respir Med. 2017; 5:691–706. https://doi.org/10.1016/S2213-2600(17)30293-X [PubMed]
- 2. Anees Ur Rehman, Ahmad Hassali MA, Muhammad SA, Shah S, Abbas S, Hyder Ali IAB, Salman A. The economic burden of chronic obstructive pulmonary disease (COPD) in the USA, Europe, and Asia: results from a systematic review of the literature. Expert Rev Pharmacoecon Outcomes Res. 2020; 20:661–72. https://doi.org/10.1080/14737167.2020.1678385 [PubMed]
- 3. Silverman EK. Genetics of COPD. Annu Rev Physiol. 2020; 82:413–31. https://doi.org/10.1146/annurev-physiol-021317-121224 [PubMed]
- 4. Salvi S. Tobacco smoking and environmental risk factors for chronic obstructive pulmonary disease. Clin Chest Med. 2014; 35:17–27. https://doi.org/10.1016/j.ccm.2013.09.011 [PubMed]
- 5. Ito K, Barnes PJ. COPD as a disease of accelerated lung aging. Chest. 2009; 135:173–80. https://doi.org/10.1378/chest.08-1419 [PubMed]
- 6. Mercado N, Ito K, Barnes PJ. Accelerated ageing of the lung in COPD: new concepts. Thorax. 2015; 70:482–9. https://doi.org/10.1136/thoraxjnl-2014-206084 [PubMed]
- 7. Gershon AS, McGihon RE, Luo J, Blazer AJ, Kendzerska T, To T, Aaron SD. Trends in Chronic Obstructive Pulmonary Disease Prevalence, Incidence, and Health Services Use in Younger Adults in Ontario, Canada, 2006-2016. Am J Respir Crit Care Med. 2021; 203:1196–9. https://doi.org/10.1164/rccm.202006-2495LE [PubMed]
- 8. Raherison C, Girodet PO. Epidemiology of COPD. Eur Respir Rev. 2009; 18:213–21. https://doi.org/10.1183/09059180.00003609 [PubMed]
- 9. Schuliga M, Read J, Knight DA. Ageing mechanisms that contribute to tissue remodeling in lung disease. Ageing Res Rev. 2021; 70:101405. https://doi.org/10.1016/j.arr.2021.101405 [PubMed]
- 10. Easter M, Bollenbecker S, Barnes JW, Krick S. Targeting Aging Pathways in Chronic Obstructive Pulmonary Disease. Int J Mol Sci. 2020; 21:6924. https://doi.org/10.3390/ijms21186924 [PubMed]
- 11. Chun P. Role of sirtuins in chronic obstructive pulmonary disease. Arch Pharm Res. 2015; 38:1–10. https://doi.org/10.1007/s12272-014-0494-2 [PubMed]
- 12. Conti V, Corbi G, Manzo V, Pelaia G, Filippelli A, Vatrella A. Sirtuin 1 and aging theory for chronic obstructive pulmonary disease. Anal Cell Pathol (Amst). 2015; 2015:897327. https://doi.org/10.1155/2015/897327 [PubMed]
- 13. Takasaka N, Araya J, Hara H, Ito S, Kobayashi K, Kurita Y, Wakui H, Yoshii Y, Yumino Y, Fujii S, Minagawa S, Tsurushige C, Kojima J, et al. Autophagy induction by SIRT6 through attenuation of insulin-like growth factor signaling is involved in the regulation of human bronchial epithelial cell senescence. J Immunol. 2014; 192:958–68. https://doi.org/10.4049/jimmunol.1302341 [PubMed]
- 14. Wang XL, Li T, Li JH, Miao SY, Xiao XZ. The Effects of Resveratrol on Inflammation and Oxidative Stress in a Rat Model of Chronic Obstructive Pulmonary Disease. Molecules. 2017; 22:1529. https://doi.org/10.3390/molecules22091529 [PubMed]
- 15. Li L, Wang Y, Gao W, Yuan C, Zhang S, Zhou H, Huang M, Yao X. Klotho Reduction in Alveolar Macrophages Contributes to Cigarette Smoke Extract-induced Inflammation in Chronic Obstructive Pulmonary Disease. J Biol Chem. 2015; 290:27890–900. https://doi.org/10.1074/jbc.M115.655431 [PubMed]
- 16. Gao W, Yuan C, Zhang J, Li L, Yu L, Wiegman CH, Barnes PJ, Adcock IM, Huang M, Yao X. Klotho expression is reduced in COPD airway epithelial cells: effects on inflammation and oxidant injury. Clin Sci (Lond). 2015; 129:1011–23. https://doi.org/10.1042/CS20150273 [PubMed]
- 17. Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004; 116:281–97. https://doi.org/10.1016/s0092-8674(04)00045-5 [PubMed]
- 18. Rupaimoole R, Slack FJ. MicroRNA therapeutics: towards a new era for the management of cancer and other diseases. Nat Rev Drug Discov. 2017; 16:203–22. https://doi.org/10.1038/nrd.2016.246 [PubMed]
- 19. Rupaimoole R, Calin GA, Lopez-Berestein G, Sood AK. miRNA Deregulation in Cancer Cells and the Tumor Microenvironment. Cancer Discov. 2016; 6:235–46. https://doi.org/10.1158/2159-8290.CD-15-0893 [PubMed]
- 20. Huang X, Zhu Z, Guo X, Kong X. The roles of microRNAs in the pathogenesis of chronic obstructive pulmonary disease. Int Immunopharmacol. 2019; 67:335–47. https://doi.org/10.1016/j.intimp.2018.12.013 [PubMed]
- 21. Mei D, Tan WSD, Tay Y, Mukhopadhyay A, Wong WSF. Therapeutic RNA Strategies for Chronic Obstructive Pulmonary Disease. Trends Pharmacol Sci. 2020; 41:475–86. https://doi.org/10.1016/j.tips.2020.04.007 [PubMed]
- 22. Mizuno S, Bogaard HJ, Gomez-Arroyo J, Alhussaini A, Kraskauskas D, Cool CD, Voelkel NF. MicroRNA-199a-5p is associated with hypoxia-inducible factor-1α expression in lungs from patients with COPD. Chest. 2012; 142:663–72. https://doi.org/10.1378/chest.11-2746 [PubMed]
- 23. Long YJ, Liu XP, Chen SS, Zong DD, Chen Y, Chen P. miR-34a is involved in CSE-induced apoptosis of human pulmonary microvascular endothelial cells by targeting Notch-1 receptor protein. Respir Res. 2018; 19:21. https://doi.org/10.1186/s12931-018-0722-2 [PubMed]
- 24. Baker JR, Vuppusetty C, Colley T, Papaioannou AI, Fenwick P, Donnelly L, Ito K, Barnes PJ. Oxidative stress dependent microRNA-34a activation via PI3Kα reduces the expression of sirtuin-1 and sirtuin-6 in epithelial cells. Sci Rep. 2016; 6:35871. https://doi.org/10.1038/srep35871 [PubMed]
- 25. Wu H, Ma H, Wang L, Zhang H, Lu L, Xiao T, Cheng C, Wang P, Yang Y, Wu M, Wang S, Zhang J, Liu Q. Regulation of lung epithelial cell senescence in smoking-induced COPD/emphysema by microR-125a-5p via Sp1 mediation of SIRT1/HIF-1a. Int J Biol Sci. 2022; 18:661–74. https://doi.org/10.7150/ijbs.65861 [PubMed]
- 26. Edgar R, Domrachev M, Lash AE. Gene Expression Omnibus: NCBI gene expression and hybridization array data repository. Nucleic Acids Res. 2002; 30:207–10. https://doi.org/10.1093/nar/30.1.207 [PubMed]
- 27. Kennedy BK, Berger SL, Brunet A, Campisi J, Cuervo AM, Epel ES, Franceschi C, Lithgow GJ, Morimoto RI, Pessin JE, Rando TA, Richardson A, Schadt EE, et al. Geroscience: linking aging to chronic disease. Cell. 2014; 159:709–13. https://doi.org/10.1016/j.cell.2014.10.039 [PubMed]
- 28. Meiners S, Eickelberg O, Königshoff M. Hallmarks of the ageing lung. Eur Respir J. 2015; 45:807–27. https://doi.org/10.1183/09031936.00186914 [PubMed]
- 29. Ito K, Mercado N. STOP accelerating lung aging for the treatment of COPD. Exp Gerontol. 2014; 59:21–7. https://doi.org/10.1016/j.exger.2014.03.014 [PubMed]
- 30. Negewo NA, Gibson PG, McDonald VM. COPD and its comorbidities: Impact, measurement and mechanisms. Respirology. 2015; 20:1160–71. https://doi.org/10.1111/resp.12642 [PubMed]
- 31. Eapen MS, Sohal SS. Update on the Pathogenesis of COPD. N Engl J Med. 2019; 381:2483–4. https://doi.org/10.1056/NEJMc1914437 [PubMed]
- 32. Barnes PJ. Oxidative stress-based therapeutics in COPD. Redox Biol. 2020; 33:101544. https://doi.org/10.1016/j.redox.2020.101544 [PubMed]
- 33. Barnes PJ. Inflammatory mechanisms in patients with chronic obstructive pulmonary disease. J Allergy Clin Immunol. 2016; 138:16–27. https://doi.org/10.1016/j.jaci.2016.05.011 [PubMed]
- 34. Alharbi KS, Fuloria NK, Fuloria S, Rahman SB, Al-Malki WH, Javed Shaikh MA, Thangavelu L, Singh SK, Rama Raju Allam VS, Jha NK, Chellappan DK, Dua K, Gupta G. Nuclear factor-kappa B and its role in inflammatory lung disease. Chem Biol Interact. 2021; 345:109568. https://doi.org/10.1016/j.cbi.2021.109568 [PubMed]
- 35. Yang D, Yan Y, Hu F, Wang T. CYP1B1, VEGFA, BCL2, and CDKN1A Affect the Development of Chronic Obstructive Pulmonary Disease. Int J Chron Obstruct Pulmon Dis. 2020; 15:167–75. https://doi.org/10.2147/COPD.S220675 [PubMed]
- 36. Yao H, Yang SR, Edirisinghe I, Rajendrasozhan S, Caito S, Adenuga D, O’Reilly MA, Rahman I. Disruption of p21 attenuates lung inflammation induced by cigarette smoke, LPS, and fMLP in mice. Am J Respir Cell Mol Biol. 2008; 39:7–18. https://doi.org/10.1165/rcmb.2007-0342OC [PubMed]
- 37. Sun S, Shen Y, Wang J, Li J, Cao J, Zhang J. Identification and Validation of Autophagy-Related Genes in Chronic Obstructive Pulmonary Disease. Int J Chron Obstruct Pulmon Dis. 2021; 16:67–78. https://doi.org/10.2147/COPD.S288428 [PubMed]
- 38. Shukla SD, Walters EH, Simpson JL, Keely S, Wark PAB, O’Toole RF, Hansbro PM. Hypoxia-inducible factor and bacterial infections in chronic obstructive pulmonary disease. Respirology. 2020; 25:53–63. https://doi.org/10.1111/resp.13722 [PubMed]
- 39. Kusko RL, Brothers JF 2nd, Tedrow J, Pandit K, Huleihel L, Perdomo C, Liu G, Juan-Guardela B, Kass D, Zhang S, Lenburg M, Martinez F, Quackenbush J, et al. Integrated Genomics Reveals Convergent Transcriptomic Networks Underlying Chronic Obstructive Pulmonary Disease and Idiopathic Pulmonary Fibrosis. Am J Respir Crit Care Med. 2016; 194:948–60. https://doi.org/10.1164/rccm.201510-2026OC [PubMed]
- 40. Martin PJ, Héliot A, Trémolet G, Landkocz Y, Dewaele D, Cazier F, Ledoux F, Courcot D. Cellular response and extracellular vesicles characterization of human macrophages exposed to fine atmospheric particulate matter. Environ Pollut. 2019; 254:112933. https://doi.org/10.1016/j.envpol.2019.07.101 [PubMed]
- 41. Menzel M, Ramu S, Calvén J, Olejnicka B, Sverrild A, Porsbjerg C, Tufvesson E, Bjermer L, Akbarshahi H, Uller L. Oxidative Stress Attenuates TLR3 Responsiveness and Impairs Anti-viral Mechanisms in Bronchial Epithelial Cells From COPD and Asthma Patients. Front Immunol. 2019; 10:2765. https://doi.org/10.3389/fimmu.2019.02765 [PubMed]
- 42. Yim HCH, Leon TYY, Li JCB. MXD1 regulates the H9N2 and H1N1 influenza A virus-induced chemokine expression and their replications in human macrophage. J Leukoc Biol. 2020; 108:1631–40. https://doi.org/10.1002/JLB.4MA0620-703RR [PubMed]
- 43. Durocher M, Ander BP, Jickling G, Hamade F, Hull H, Knepp B, Liu DZ, Zhan X, Tran A, Cheng X, Ng K, Yee A, Sharp FR, Stamova B. Inflammatory, regulatory, and autophagy co-expression modules and hub genes underlie the peripheral immune response to human intracerebral hemorrhage. J Neuroinflammation. 2019; 16:56. https://doi.org/10.1186/s12974-019-1433-4 [PubMed]
- 44. Zheng D, Wu W, Dong N, Jiang X, Xu J, Zhan X, Zhang Z, Hu Z. Mxd1 mediates hypoxia-induced cisplatin resistance in osteosarcoma cells by repression of the PTEN tumor suppressor gene. Mol Carcinog. 2017; 56:2234–44. https://doi.org/10.1002/mc.22676 [PubMed]
- 45. Xu Q, Chen Y. An Aging-Related Gene Signature-Based Model for Risk Stratification and Prognosis Prediction in Lung Adenocarcinoma. Front Cell Dev Biol. 2021; 9:685379. https://doi.org/10.3389/fcell.2021.685379 [PubMed]
- 46. Li S, He X, Gan Y, Zhang J, Gao F, Lin L, Qiu X, Yu T, Zhang X, Chen P, Tong J, Qian W, Xu Y. Targeting miR-21 with NL101 blocks c-Myc/Mxd1 loop and inhibits the growth of B cell lymphoma. Theranostics. 2021; 11:3439–51. https://doi.org/10.7150/thno.53561 [PubMed]
- 47. Grandori C, Cowley SM, James LP, Eisenman RN. The Myc/Max/Mad network and the transcriptional control of cell behavior. Annu Rev Cell Dev Biol. 2000; 16:653–99. https://doi.org/10.1146/annurev.cellbio.16.1.653 [PubMed]
- 48. Lüscher B. MAD1 and its life as a MYC antagonist: an update. Eur J Cell Biol. 2012; 91:506–14. https://doi.org/10.1016/j.ejcb.2011.07.005 [PubMed]
- 49. Wu Q, Yang Z, An Y, Hu H, Yin J, Zhang P, Nie Y, Wu K, Shi Y, Fan D. MiR-19a/b modulate the metastasis of gastric cancer cells by targeting the tumour suppressor MXD1. Cell Death Dis. 2014; 5:e1144. https://doi.org/10.1038/cddis.2014.110 [PubMed]
- 50. Global Strategy for Diagnosis, Management and Prevention of COPD. The Global Initiative for Chronic Obstructive Lung Diseases (GOLD). 2020 report. https://goldcopd.org/gold-reports/.
- 51. Meisner A, Parikh CR, Kerr KF. Biomarker combinations for diagnosis and prognosis in multicenter studies: Principles and methods. Stat Methods Med Res. 2019; 28:969–85. https://doi.org/10.1177/0962280217740392 [PubMed]
- 52. Sun L, Li J, Yan W, Yao Z, Wang R, Zhou X, Wu H, Zhang G, Shi T, Chen W. H19 promotes aerobic glycolysis, proliferation, and immune escape of gastric cancer cells through the microRNA-519d-3p/lactate dehydrogenase A axis. Cancer Sci. 2021; 112:2245–59. https://doi.org/10.1111/cas.14896 [PubMed]
- 53. Liang J, Liu Y, Zhang L, Tan J, Li E, Li F. Overexpression of microRNA-519d-3p suppressed the growth of pancreatic cancer cells by inhibiting ribosomal protein S15A-mediated Wnt/β-catenin signaling. Chem Biol Interact. 2019; 304:1–9. https://doi.org/10.1016/j.cbi.2019.02.026 [PubMed]
- 54. Zhang G, Hu Y, Yuan W, Qiu H, Yu H, Du J. miR-519d-3p Overexpression Inhibits P38 and PI3K/AKT Pathway via Targeting VEGFA to Attenuate the Malignant Biological Behavior of Non-Small Cell Lung Cancer. Onco Targets Ther. 2020; 13:10257–66. https://doi.org/10.2147/OTT.S252795 [PubMed]
- 55. Chaiwangyen W, Murrieta-Coxca JM, Favaro RR, Photini SM, Gutiérrez-Samudio RN, Schleussner E, Markert UR, Morales-Prieto DM. MiR-519d-3p in Trophoblastic Cells: Effects, Targets and Transfer to Allogeneic Immune Cells via Extracellular Vesicles. Int J Mol Sci. 2020; 21:3458. https://doi.org/10.3390/ijms21103458 [PubMed]