Introduction
The importance of the bidirectional communication between the gut and central nervous system (the brain-gut-microbiome axis) is continually growing with our greater understanding of the process. Microbiota is involved in the pathophysiology of various cellular mechanisms, such as immune system, homeostasis, and disease, and is implicated in both health and disease [1, 2]. Particularly, healthy individuals present distinct human gut microbiota compositions compared to those with several neurological diseases, including Parkinson’s disease, Alzheimer’s disease (AD), autism spectrum disorder, multiple sclerosis, and amyotrophic lateral sclerosis [2, 3]. Changes in the diversity and richness of gut microbiota are correlated with disease progression, particularly with diminished cognitive function [4]. Furthermore, there is an interplay between gastrointestinal pathology and neuropsychiatric conditions [5]. In other words, if alteration of microbial species may induce immune signaling, it results in the change in host homeostasis and CNS disease progression. Therefore, the clinical features and development of various neurological disorders may be influenced by gut microbiota. Thus, fecal microbiota transplantation (FMT), in which fecal solution from a healthy donor is administered into a patient’s intestinal tract, may provide therapeutic benefit to patients.
Previous animal studies have demonstrated that FMT can improve emotional behaviors, learning, memory, and recognition [6]. Cognitive decline may occur in transplanted adult mice after FMT for spatial learning and memory through changes in the hippocampal synaptic plasticity-associated and neurotransmission-related proteins [7, 8]. These microbially derived changes may intrinsically regulate the host homeostasis, including the blood-brain barrier and intestinal integrity after FMT [6, 9]. However, no association between FMT and cognitive improvement has been established in humans. This study aims to establish that FMT leads to significant improvements in cognitive performance and discuss the relationship between cognition and the gut microbiome.
Results
Clinical overview and cognitive function differences before and after FMT
Among 10 patients, 5 representative cases are briefly described below for easy understanding of this study.
Case 1: An 85-year-old woman was admitted to the intensive care unit for intracranial aneurysmal coiling treatment (Table 1). Subsequently, she suffered from diarrhea and was diagnosed with CDI. Antibiotic treatment, including vancomycin and metronidazole, failed. She had been experiencing a gradual decline in memory and cognition and was taking donepezil (10 mg) for AD. The patient scored 8 and 10 on her recent Mini-Mental State Examination (MMSE) and Clinical Dementia Rating Scale Sum of Boxes (CDR-SB) scores, respectively, while the Glasgow Coma Scale (GCS) score was 14. She no longer appeared to enjoy socializing and required considerable assistance with basic tasks, such as food preparation, bathing, and taking medication. She underwent FMT after 1 month, and her MMSE and CDR-SB scores improved to 13 and 8, respectively, and the GCS score was 14 (Table 2, Figure 1, and Supplementary Table 1). Following FMT, her severe gastrointestinal symptoms improved, and a stool test for CDI was negative.
Table 1. Clinical characteristics of the study participants.
| Characteristics | Value | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FMT group (N = 10) | Control group (N = 10) | P value | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sex, female, n (%) | 8 (80) | 8 (80) | 0.71 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age, years, median (IQR) | 76 (63–90) | 77 (62–91) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| mRS, median (IQR) | 3 (1–4) | 3 (1–4) | 1.00 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Geriatric Depression Scale, median (IQR) | 17 (13–22) | 16 (10–23) | 0.84 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Nutrition supplement status | 0.22 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Oral feeding, n (%) | 9 (90) | 8 (80) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Enteral feeding, n (%) | 1 (10) | 2 (20) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Peripheral feeding, n (%) | 0 (0) | 0 (0) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Presenting symptoms of CDI | 0.79 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diarrhea, n (%) | 9 (90) | 9 (90) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abdominal pain, n (%) | 4 (40) | 5 (50) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Fever, n (%) | 4 (40) | 4 (40) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Antibiotics for CDI | 0.80 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Vancomycin, n (%) | 2 (20) | 3 (30) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Metronidazole, n (%) | 2 (20) | 2 (20) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Vancomycin + metronidazole, n (%) | 6 (60) | 5 (50) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FMT dose, g | 60 | – | – | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Complications after FMT | – | – | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| None, n (%) | 8 (80) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Nausea, n (%) | 1 (10) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abdominal pain, n (%) | 1 (10) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Fever, n (%) | 1 (10) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Medication for dementia | 0.50 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Donepezil, n (%) | 7 (77.8) | 6 (60) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Memantine, n (%) | 1 (11.1) | 2 (20) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Donepezil + Memantine, n (%) | 1 (11.1) | 1 (10) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Duration of dementia, years, median (IQR) | 3.5 (1.8–5.3) | 3.8 (1.7–5.4) | 0.73 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: mRS: modified Rankin Scale; FMT: fecal microbiota transplantation; CDI: Clostridioides difficile infection; IOR: interquartile range. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 2. Cognitive function difference between the FMT and control groups.
| Cognitive function test | FMT group | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Before FMT, median (IQR) | After FMT, median (IQR) | Before-After, mean (SD) | P valuea | P valueb | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MMSE | 10.0 (7.0–14.0) | 16.0 (13.0–18.0) | 4.7 (1.6) | 0.010 | 0.005 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CDR-SB score | 10.0 (5.0–12.0) | 5.5 (4.0–8.0) | 3.1 (2.6) | 0.048 | 0.005 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cognitive function test | Control group | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Before antibiotics treatment, median (IQR) | After antibiotics treatment, median (IQR) | Before-After. mean (SD) | P valuea | P valueb | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MMSE | 14.0 (12.8–16.0) | 10.0 (9.8–15.3) | 2.1 (2.8) | 0.206 | 0.072 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CDR-SB score | 10.0 (8.4–11.3) | 8.0 (7.9–12.5) | 0.3 (1.3) | 0.492 | 0.470 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: FMT: fecal microbiota transplantation; MMSE: Mini-mental state examination; CDR-SB score: Clinical Dementia Rating Scale Sum of Boxes Score; IQR: interquartile range; CDI: Clostridioides difficile infection; SD: standard deviation. aKruskal-Wallis H. bWilcoxon signed rank. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
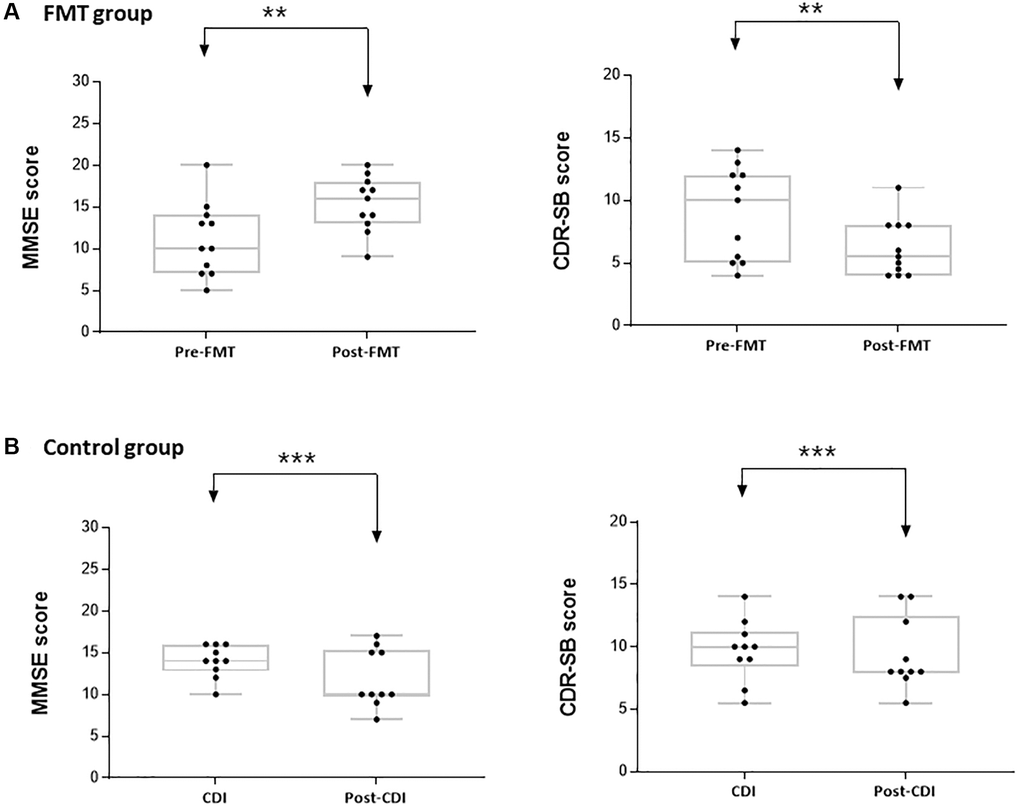
Figure 1. Mini-mental state examination (MMSE) and clinical dementia rating scale sum of boxes (CDR-SB) scores in the study groups. (A) Difference in the MMSE and CDR-SB scores before and after FMT. (B) Difference in the MMSE and CDR-SB scores before and after CDI. **P < 0.05, ***P > 0.05.
Case 2: A 73-year-old woman with a history of AD, acute kidney injury, and atrial fibrillation was admitted for diarrhea and fever (Table 1). She was diagnosed with CDI, and intravenous vancomycin treatment failed. She also presented with AD, including depression, memory loss, and flattened affect. She took memantine (20 mg) and had MMSE and CDR-SB scores of 13 and 7, respectively, and GCS score of 13. After undergoing FMT, her MMSE and CDR-SB scores improved to 17 and 6, respectively, and the GCS score was 14 (Table 2, Figure 1, and Supplementary Table 1). Her severe gastrointestinal discomfort improved with negative CDI stool results 1 week after FMT.
Case 3: A 90-year-old woman with a history of AD, diabetes mellitus, hypertension, and chronic kidney disease was admitted for diarrhea and fever (Table 1). On hospital day 3, she was diagnosed with CDI. She was treated unsuccessfully with vancomycin with metronidazole. She was taking donepezil (10 mg) for AD. Her most recent MMSE and CDR-SB scores were 15 and 5.5, respectively, and GCS score was 14. After FMT, her MMSE score improved to 18 and CDR-SB score to 5 (Table 2, Figure 1). She no more experienced diarrhea and fever, with a negative CDI stool test after FMT. Three months after the first FMT, she suffered from diarrhea, fever (38.0°C), continuous abdominal pain, and progressively worsening conditions. She was re-diagnosed with a recurrent severe CDI with a positive CDI stool test, and was unsuccessfully treated with antibiotics. She underwent a second FMT with the same modalities as the first FMT. Her MMSE and CDR-SB scores before the second FMT procedure were 20 and 5.5, respectively, which improved immediately after the first FMT. One week after the second FMT, her severe gastrointestinal discomfort improved with negative CDI stool results. Nevertheless, her scores were stable (MMSE 20, CDR-SB 4). However, no difference was observed in GCS scores before and after FMT (Supplementary Table 1).
Case 4: A 63-year-old man with a history of diabetes mellitus, hypertension, stroke, and vascular dementia was admitted for pneumonia with dyspnea (Table 1). On hospital day 62, he was diagnosed with CDI and treated with oral metronidazole. Because of additional unsuccessful treatment with vancomycin, he underwent FMT. Before FMT, he presented with depression and memory loss, and his MMSE and CDR-SB scores were 14 and 5, respectively, and GCS score was 14. After FMT, his MMSE and CDR-SB scores improved to 19 and 4, respectively (Table 2, Figure 1). However, GCS was not different compared to that before FMT (Supplementary Table 1). He reported a marked improvement from previous symptoms.
Case 5: An 84-year-old woman with a history of ischemic cardiomyopathy and AD was admitted for type 2 acute respiratory failure (Table 1). On hospital day 7, CDI was diagnosed and treated with vancomycin and metronidazole. Because of additional unsuccessful treatment with oral antibiotics, she underwent FMT. Before FMT, she presented with depression and memory loss, and her MMSE and CDR-SB scores were 5 and 11, respectively. After FMT, her MMSE and CDR-SB scores improved to 12 and 8, respectively (Table 2, Figure 1). Following FMT, her stool test for CDI was negative.
Baseline characteristics
Baseline characteristics of the study population are provided in Table 1. Subjects with dementia and severe CDI suffered from CDI symptoms, such as abdomen pain, diarrhea, vomiting, and fever. They were divided into two groups: those with FMT (FMT group) and without FMT (control group). In the FMT group, there were 8 women and 2 men; the median age at the time of FMT was 76 years (range, 63–90 years). All subjects were treated with 1–2 treatment courses of oral antibiotics before FMT, 8 patients (80.0%) were administered oral vancomycin, and 8 were administered metronidazole. The main manifestations of CDI symptoms were abdominal pain and diarrhea in FMT patients. Nine patients (90.0%) had prescriptions for dementia based on cognitive test results or brain imaging. The control group was not different compare to FMT group. They also included 8 women and 2 men; the median age at the time of CDI diagnosis was 77 years (range, 62–91 years). All control subjects were also treated with 1–2 treatment courses of oral antibiotics and had prescriptions for dementia.
Clinical responses with or without FMT
A total of 10 FMT were performed; 9 (90.0%) patients were cured by a single FMT, and 1 (10.0%) was cured with two FMTs. There were no serious adverse events reported after FMT. Transient nausea was reported in 1 patient on the day of FMT and disappeared after 3 hours. Transient abdominal pain was reported in 1 patient after the procedure. In addition, 1 patient vomited during the procedure. No recurrence of CDI and FMT-related adverse events were reported by the caregivers of subjects after FMT by phone call due to COVID-19.
GCS scores were not significantly different between pre- and post-FMT. Most patients’ scores were 13 or 14 without any acute need for change in the management or neuroimaging. All patients performed significantly better on the cognitive function post-FMT than pre-FMT. Median MMSE scores were higher post-FMT (16.0, Median; 13.0–18.0, interquartile range [IQR]) than pre-FMT (10.0, Median; 7.0–14.0, IQR, Table 2 and Supplementary Table 1, Figure 1). The difference in the MMSE score pre- and post-FMT was 4.7 (mean, P = 0.010). As the result of Wilcoxon signed rank test pre- and post-FMT, the difference in the MMSE scores was significant (P = 0.005) for an average of 10 patients. Median CDR-SB score was lower post-FMT (5.5, Median; 4.0–8.0, IQR) than pre-FMT (10.0, Median 5.0–12.0, IQR, Table 2, Figure 1). The difference in the CDR-SB scores between pre- and post-FMT was 3.1 (mean, P = 0.048). Wilcoxon signed rank test pre- and post-FMT showed that the difference in the CDR-SB scores was significant (P = 0.005) in the 10 patients. In the control group, 90.0% of the patients were cured by the antibiotic treatment. Further, all control subjects did not show significantly altered cognitive function at admission and after CDI treatment (Figure 1). Median MMSE scores were rather low after CDI treatment (10.0, Median; 9.8–15.3, interquartile range [IQR]) than at admission (14.0, Median; 12.8–16.0, IQR, Table 2 and Supplementary Table 2). Median CDR-SB score also did not significantly change after CDI treatment (8.0, Median) compared to that at admission (10.00, Median, P = 0.470, Table 2). Thus, FMT caused a significant improvement in cognitive functions.
Alternation of patients’ fecal composition
Twenty fecal samples were collected from 10 patients with CDI to analyze gut microbiota before and after FMT. We also collected samples from the control group at admission due to CDI and after CDI treatment. For patients treated with FMT twice, pre-FMT fecal samples were collected before the first FMT, and post-FMT samples were collected after the last FMT procedure.
Post-FMT, the bacterial richness of gut microbiota was increased in the patients (Figure 2A, 2B). At the phylum level, Proteobacteria was enriched (44.25%, pre-FMT vs. 20.68%, post-FMT); moreover, Bacteroidetes were enriched in patients post FMT (10.44%, pre-FMT vs. 34.78%, post-FMT) (P = 0.893). At the genus level, relative abundances of Hafnia (9.08%), Enterobacteriaceae (6.34%), Sutterella (1.99%), and Klebsiella (2.48%) were higher in the pre-FMT samples. Bacteroides (8.46%, pre-FMT vs. 27.15%, post-FMT), Alistipes (0.00%, pre-FMT vs. 4.18%, post-FMT), Blautia (0.00%, pre-FMT vs. 2.03%, post-FMT), and Bifidobacterium (1.20%, pre-FMT vs. 5.78%, post-FMT) were the more enriched taxonomies after FMT (P = 0.161). Compared to the control group, especially after CDI treatment, in the FMT group, Bacteroidetes were more enriched at the phylum level (22.73% vs. 34.78%, P = 0.427, Figure 2C). Firmicutes were more enriched in the control group than in the post-FMT group (52.96% vs. 38.36%, P = 0.241). Bacteroides (17.44%, post-CDI), Alistipes (1.96%, post-CDI), and Bifidobacterium (3.02%, post-CDI) were the less enriched taxonomies at the genus level in the control group compared with post-FMT samples (P = 0.473, Figure 2D).
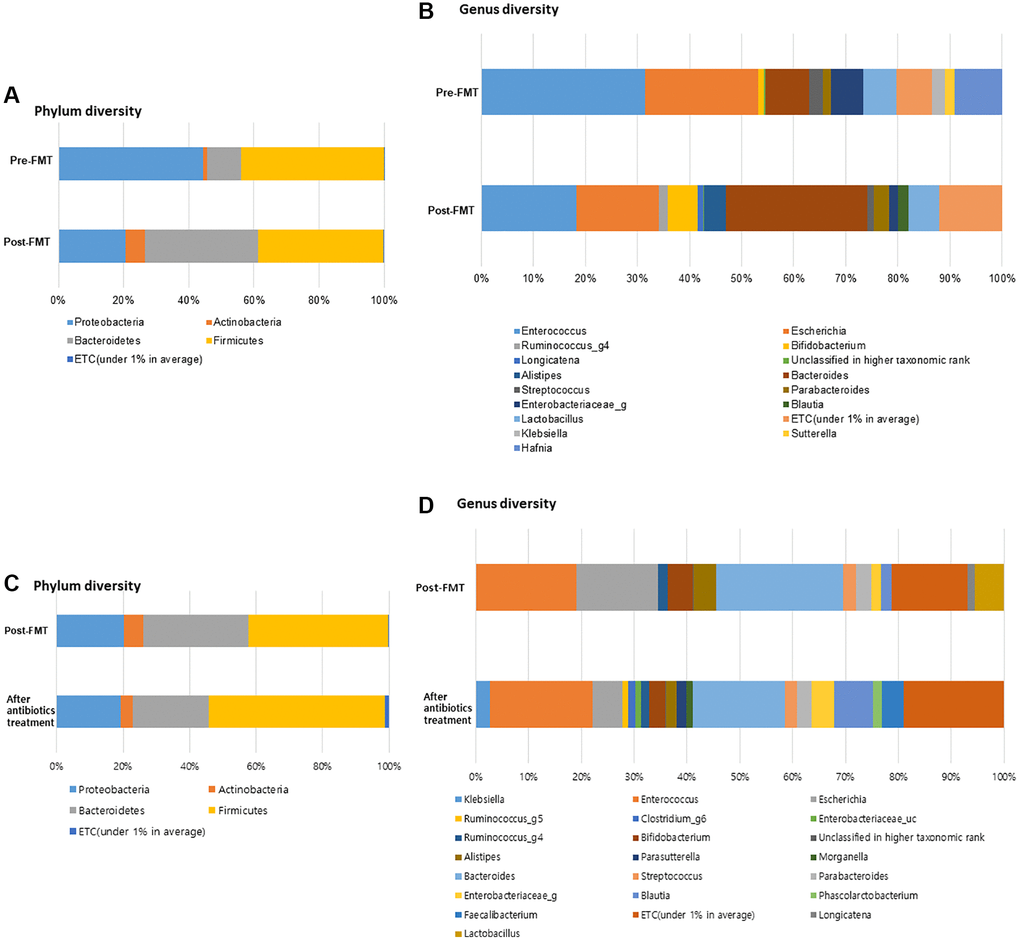
Figure 2. FMT alter the gut microbiome composition in dementia patients with severe Clostridioides difficile infection (CDI). (A) Phylum diversity pre- and post-FMT. (B) Genus diversity pre- and post-FMT. (C) Phylum diversity post-FMT and after antibiotics treatment. (D) Genus diversity post-FMT and after antibiotics treatment. *P > 0.05, **P < 0.05.
The alpha diversities were significantly different before and after FMT (Chao, P = 0.11; Shannon, P = 0.026, Figure 3). In the control group, the alpha diversities were not significantly different at admission and after CDI treatment (Chao, P = 0.650; Shannon, P = 0.496, Figure 4). In other words, the alpha diversity indices were increased after FMT. This indicates that FMT changes the species composition of recipient feces. The beta diversity showed a significant difference between pre- and post- FMT groups according to the permutational multivariate analysis of variance (PERMANOVA, P = 0.046, Figure 3) [10]. There were no significant differences in beta diversity at admission and after CDI treatment according to PERMANOVA (P = 0.659, Figure 4).
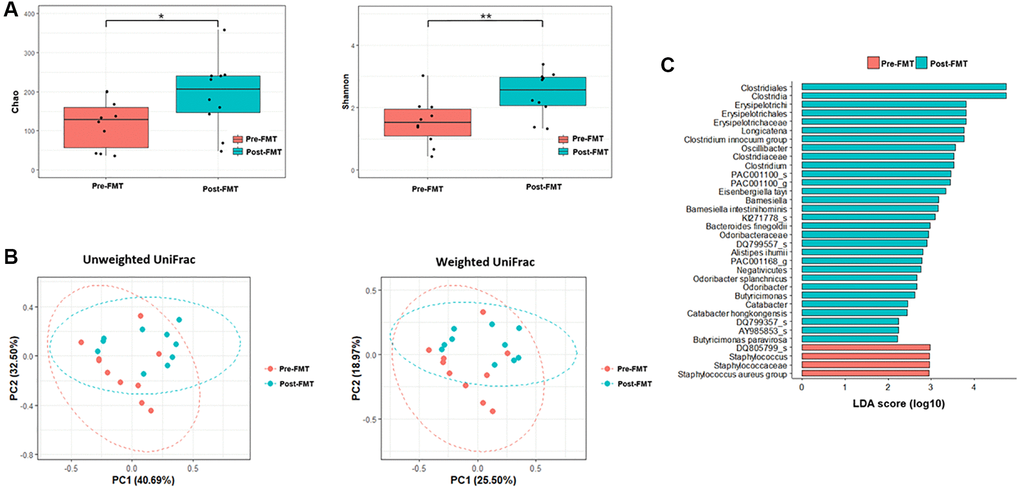
Figure 3. Microbiome analysis before and after fecal microbiota transplantation (FMT). (A) Alpha diversity was calculated using the number of overserved Chao and Shannon. (B) Principal coordinate analysis (PCoA) profile of microbial diversity across all samples using unweighted and weighted UniFrac. (C) Linear discriminant analysis effect size analysis (P < 0.05). *P > 0.05, **P < 0.05.
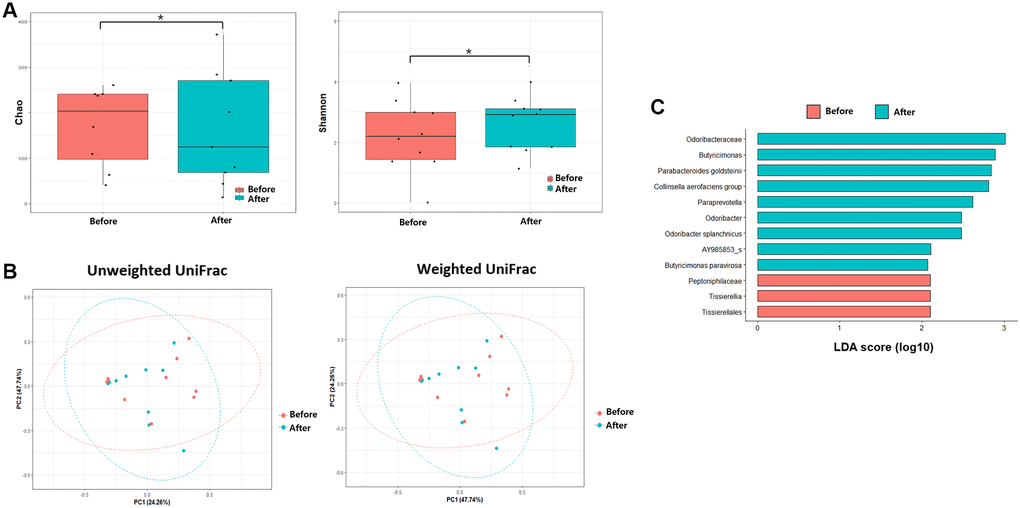
Figure 4. Microbiome analysis before antibiotics treatment and after antibiotics treatment. (A) Alpha diversity was calculated using the number of overserved Chao and Shannon. (B) Principal coordinate analysis (PCoA) profile of microbial diversity across all samples using unweighted and weighted UniFrac. (C) Linear discriminant analysis effect size analysis (P < 0.05). *P > 0.05.
Inter-group comparisons of taxonomic profiles revealed that the gut microbiome altered in the abundance of several taxa. LEfSe was performed to determine the significant taxonomic difference between pre- and post-FMT (Figure 3). After FMT, some bacteria were uplifted or reduced. At the phylum level, Bacteroidetes was the more enriched taxonomy after FMT (LEfSe >2, P < 0.05). At the genus level, Longicatena, Barmesiella, and Odoribacter were also enriched taxonomy after FMT (LEfSe >2, P < 0.05). In the control group, Bacteroidota and Actinomycetota were the more enriched taxonomies at the phylum level after CDI treatment (LEfSe >2, P < 0.05, Figure 4). At the genus level, Butyricimonas and Odoribacter were enriched after CDI treatment (LEfSe >2, P < 0.05, Figure 4).
Functional biomarker analysis using the Kruskal-Wallis H test was performed between the pre- and post-FMT groups. Alanine, aspartate, and glutamate metabolism pathways were found to be significantly modulated between before and after FMT (Figure 5, P = 0.034).
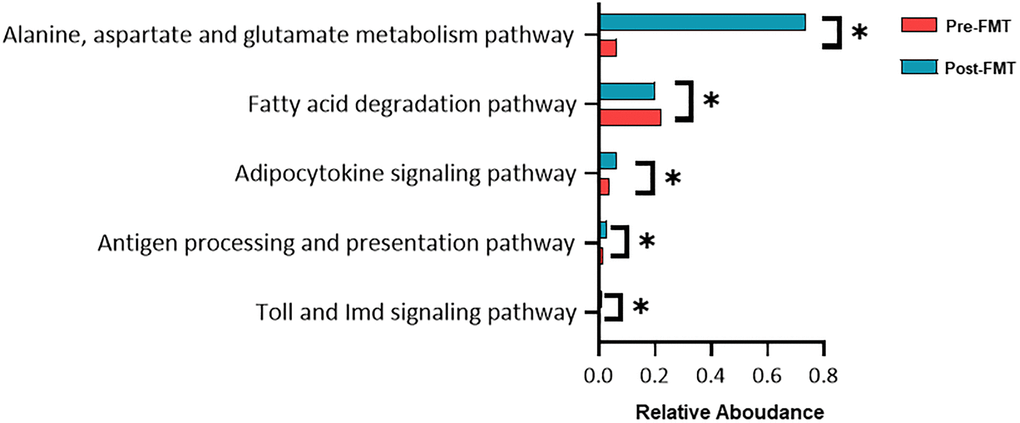
Figure 5. Functional composition difference pre- and post-fecal microbiota transplantation (FMT). Relative abundances of the most abundant microbial pathways among the different groups. Data shown are the means ± SD. *P < 0.05.
Discussion
To our best knowledge, this is an interesting study to describe cognitive improvements in dementia patients with recurrent severe CDI after FMT. In our study, the MMSE, CDR-GS, and CDR-SB scores improved in all patients after FMT. Alterations in gut microbiome composition consistently influenced the gut microbiome, which may be closely related to cognitive function. Therefore, this study provides clinical evidence in support of the brain-gut-microbiome association. Improving cognitive function by modulating the gut microbiota is a novel area for the bidirectional communication pathway, and this study is a meaningful presentation of a link between the brain and gut [11].
Dementia is a neurodegenerative disorder, specifically accompanied by progressive cognitive decline. Effective and definitive therapeutic management for cognitive decline has not yet been established. Therefore, various mice model was used to investigate dementia treatments [12]. Previous studies have demonstrated the relationship between cognitive function and gut microbiome in animal models [6]. Since most animal models are based on gene modification and validation, the limitation generated to demonstrate disease’ onset and similarity of humans. However, we constructed dementia patients with FMT to correlate if pathogenesis affected cognition.
The study results suggest that FMT positively affects cognitive function. Gut microbial diversity was increased after FMT. Indeed, the gut microbiota of dementia patients had low diversity compared to healthy people, which is consistent with the findings of previous animal model studies [5, 13]. In addition, a dramatic increase in Proteobacteria and Bacteroidetes was observed before and after FMT, respectively. Primarily, these genera are associated with positive effects in cognitive function [14–17]. In addition, Longicatena, Barmesiella, and Odoribacter are associated with cognitive function at the genus level. These microbiomes improve the cognitive function in AD individuals. On the contrary, Butyricimonas and Odoribacter are correlated with cognitive dysfunction [18], suggesting that the microbiome composition changes after FMT as compared to CDI treatment without FMT. These results suggest that altered fecal microbiome composition affects cognitive function. The intervention of microbiome composition, such as FMT, could affect cognitive function via many metabolic pathways. Therefore, the gut microbiome might influence the pathophysiological effects of cognitive function through various mechanisms.
Our study hypothesized that gut microbial composition affects the gut metabolite pathway and alters the gut microbiome affecting the brain, such as bile acids, amino acids, and short-chain fatty acids. Alternation of gut metabolite also might correlate to changes in the gut microbiota. Among various gut metabolite, changes in amino acid metabolism have been observed in AD brain [19, 20]. Recent metabolomics studies showed changes in the levels of various amino acids in the brain and plasma of AD patients. It is essential to consider that changes in amino acid metabolism are a driving force for disease progression. Therefore, the balance of amino acid metabolites is associated with dementia progression [21–26]. Further investigation of the role for metabolic dysregulation of amino acids in AD pathogenesis is needed [27–31].
This study showed that alanine, aspartate, and glutamate (amino acid metabolite) levels differed after FMT. These amino acids have been shown to decrease in the brain and plasma of patients with AD, particularly in the temporal lobe cortex [22–34]. Reduced glutamate levels are associated with impaired cognitive function, while decreased hippocampal glutamate levels are associated with episodic memory performance in dementia, including mild cognitive impairment. During gut metabolism, microbiome-mediated molecular mimicry may be responsible for decreased levels of amino acids essential for the normal brain function [35]. Because molecular mimicry occurs between brain and gut microbiota in cross-reaction immune cells including T or B cells, gut microbiota is considered a well-assumed strategy for increasing gut microbiota metabolites.
Importantly, these results imply that changing the amino acid composition affects glucose metabolism and eventually, the brain. AD is associated with dysfunctional energy metabolism and neurons contain the fatty acid beta-oxidation enzymes. Amino acid catabolism, including lactate and glucose metabolism, plays an important role in maintaining cellular adenosine triphosphate (ATP) levels in AD neurons [36, 37]. Therefore, FMT could change the amino acid metabolite composition and affect the brain. This study showed that FMT affects amino acid metabolism, especially affecting cognition [19, 20].
Another possible clue is that amyloid plaques in the brain might be metabolized by the gut microbiota [34]. Amyloid plaques, including amyloid-beta (Aβ), are pathophysiologically associated with neuroinflammation. Previous studies have reported microbe- associated and dementia-associated Aβ signals, required for the microglia activation, through the same Toll-like receptor (TLR)2/TLR1 complex [37]. Aβ deposition affects microglial activation; therefore, it has emerged as a key factor in AD pathogenesis. Aβ accumulation and microglia activation are present in the brain of AD patients. Aβ proportion was also found to be increased in a mouse model with cognitive impairment, as observed in humans [38, 39]. FMT improved the cognitive deficits and reduced Aβ deposition in mice [6]. Injection of healthy mouse feces into the gut of cognitively impaired mice resulted in an improvement in the cognitive functions. Further, a reduction in Aβ accumulation was also observed. FMT can downregulate pro-inflammatory cytokines and inhibit protein transcription factors elevated in AD [2, 40]. This gut microbiota modulate recipient homeostasis via the blood-brain barrier and intestinal integrity after FMT, resulting in improved cognitive function.
In our study, the attention and calculation domains and daily living abilities of patients significantly improved after FMT. A previous study demonstrated that microbial dysbiosis increased hippocampal neurodegeneration and disrupted neurogenesis [41]. Thus, alterations in microbiome composition have been associated with cognitive activity, such as learning, environmental enrichment, and exercise. Further evaluation is necessary to correlate unique regions of cognitive domains and FMT. This report also presents a patient (Case 3) who experienced a step-wise improvement in the cognitive function following repeated FMT in cognitive domains of orientation, attention, and short-term memory, in addition to daily living ability [14]. Whether FMT has a continuous and cumulative effect on cognitive function is challenging to confirm.
To determine whether the cognitive function is affected by patients' general condition, laboratory findings were also checked 1 week before and after FMT. The laboratory findings suggested that patients’ medical condition did not significantly affect cognitive function before and after FMT (Table 3). Therefore, this study provided clinical evidence indicating improved cognitive function after FMT and comparatively improved cognitive function within a short time after FMT administration.
Table 3. Laboratory findings in the FMT group.
| Value (before FMT/after FMT) | Case | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1 | 2 | 3-1 | 3-2 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BUN, mg/dL (normal range: 6.0–20.0) | 13.3/3.7 | 7.7/6.7 | 37.2/26.8 | 23.4/16.6 | 2.1/1.9 | 23.4/12.8 | 11.8/12.8 | 18.6/22.7 | 23.6/12.0 | 10.7/13.1 | 7.3/6.5 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Creatinine, mg/dl (normal range: 0.5–1.20) | 0.36/0.30 | 0.57/0.39 | 1.88/1.97 | 2.27/2.09 | 0.45/0.32 | 1.07/0.79 | 0.75/0.79 | 0.57/0.58 | 2.13/2.07 | 0.77/0.85 | 0.26/0.28 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CRP, mg/dl (normal range: 0.0–0.5) | 6.34/4.51 | 1.39/0.13 | 1.20/3.60 | 6.17/2.29 | 1.56/2.28 | 0.63/1.43 | 0.75/0.54 | 3.25/5.94 | 3.12/0.70 | 9.29/5.87 | 1.06/1.78 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ESR, mm/hr (normal range: 1–22) | 22.0/7.0 | 37.2/26.8 | 37.2/26.8 | 67/NA | 21.0/50.0 | 63.0/101.0 | 20.0/121.0 | 16.0/7.0 | 50.0/41.0 | 69.0/43.0 | 14.0/8.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Procalcitonin, ng/ml (normal range: 0.0–0.50) | NA/0.07 | 0.37/0.09 | 0.73/0.14 | 0.10/0.14 | 0.71/0.47 | 0.79/0.14 | 0.06/0.06 | 0.68/4.79 | 0.14/0.19 | 0.18/0.15 | 0.08/0.07 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ammonia, μg/dl (normal range: 12–66) | 35.0/NA | 67.0/NA | 42.0/NA | NA/NA | NA/19.0 | NA/NA | NA/NA | NA/NA | NA/NA | 39.0/NA | NA/NA | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Albumin, g/dL (normal range: 3.5–5.2) | 1.9/1.8 | 3.5/2.8 | 2.7/2.7 | 2.7/3.4 | 2.1/2.3 | 3.0/3.1 | 3.0/2.6 | 1.9/1.9 | 2.7/2.5 | 2.9/2.7 | 3.0/3.1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lactic acid, mmol/L (normal range: 0.5–2.20) | 1.40/NA | NA/1.52 | 2.22/1.21 | 0.76/1.38 | 1.13/1.02 | 1.44/1.23 | 0.84/1.23 | 2.37/1.29 | 1.19/0.73 | 1.38/0.99 | 0.92/NA | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: FMT: fecal microbiota transplantation; NA: not applicable. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
This study has limitations. First, we adjusted the follow-up timing for cognitive function analysis to reduce the bias that appears to improve cognitive function. Therefore, we checked the patients’ laboratory results before and after FMT. Second, this study could not perform various cognitive function tests due to hospital policy regarding CDI patients. Patients with CDI have limited permission to go outside the hospital room except for important evaluations. Therefore, we did limited cognitive tests. Although MMSE was limited, it was clinically useful [40]. In addition, MMSE could evaluate the mild-to-moderate stages of dementia [41]. Enrolled patients in our study had mild-to-moderate stages of dementia, and hence, MMSE was implemented. In addition, to reinforce MMSE’s accuracy, we additionally used CDR because it is associated with good interrater reliability and criterion validity [42]. However, a more diverse set of cognitive function tests must be evaluated in future studies to confirm our findings. Third, it is necessary to analyze gut microbiome differences, taxa, and additional mechanisms in patients with cognitive decline and cognitive improvement for larger patient groups and multiple centers. In addition, the FMT results are not consistent across studies, likely due to the different methodologies and instrumentation used. Since we did not check if enteric neuron receptors of FMT promoted metabolite composition and affected gastrointestinal motility, further studies are needed on transmitting information of metabolites across the epithelium to the brain barrier and changing concentrations of metabolites in the intestinal lamina propria. Fourth, although FMT is a relatively safe procedure and our patients did not present with fatal side effects, adverse effects such as fever, abdominal pain, and abdominal distension can occur after FMT (Table 1). Therefore, patient selection before FMT and monitoring after FMT is necessary. Fifth, although we analyzed laboratory tests before and after FMT, various general conditions (diarrhea, fever, nutrition status, ambulation, mood, etc.) and medical conditions can affect cognitive function. Most patients were on oral feeding (90%) in this study. However, there are individual differences in the amount eaten, which could not reflect the required daily energy that was enough for individual patients. Therefore, there seems to be a need for a further study to consider the association between patient’s nutrition support status and gut microbiome before and after FMT. In addition, because of the COVID-19 pandemic and associated hospital policies, we could perform limited test for patients. Therefore, we need to further evaluate the patient’s mood and functional activity changes. Finally, we did not evaluate dementia using other diagnostic tools such as the brain magnetic resonance imaging (MRI). Because we assessed the patients’ cognition before and after 1 month of FMT, the duration was short to perform brain MRI during follow-up. In addition, it was difficult to show the change in GCS scores before and after FMT because it demonstrates a state of consciousness and is mainly used in trauma patients [42]. Therefore, we plan to evaluate the cognition change after FMT during long-term follow-up in future studies.
FMT might affect the dysbiosis of the patient’s microbiome. We suggest that FMT alters the diversity and specificity of gut microbiota, which affects the brain-gut-microbiome axis. The positive effects of FMT may be associated with changes in the gut microbiota and an increase in profitable brain metabolites, similar to those found in mice studies [43]. This study suggests that FMT is a viable treatment option for patients with cognitive decline, thereby providing a new opportunity for practical improvements in patients with dementia. It also further highlights the critical importance of the association between the gut microbiome and cognitive function.
Materials and Methods
Patients
Patients were screened and enrolled between November 2019 and July 2021 via the Dementia and Microbiome centers of Inha University Hospital, Incheon, Republic of Korea. We initially screened patients admitted to our institution because of dementia diagnosis by a neurologist, especially Alzheimer’s dementia (AD). Simultaneously, we screened for severe CDI patients who had previously failed to improve with several antibiotics and had a relapse of symptoms with a positive stool test [44]. Patients were enrolled if they met all the following inclusion criteria: 1) over 19 years of age, 2) with dementia, 3) with CDI, and 4) absolutely required FMT. Patients who were unsuitable for FMT due to severe systemic disease (immunocompromised, acute inflammation, or infectious diseases) and those whose cognitive function could not be tested were excluded initially. Sixty-eight patients with written informed consent were enrolled; however, 27 were lost to follow-up, and 31 were not appreciate fecal analysis due to patients’ fecal samples (Supplementary Table 3). Consequently, 10 patients were enrolled for the analysis. Additionally, we matched 10 dementia patients with CDI having similar characteristics as that of the FMT group. They were treated with antibiotics and enrolled as the control group using 90% power and 5% Type I error rate.
Fecal microbiota transplantation
Stool donors with no gastrointestinal problems or other health problems underwent blood and stool tests and responded to a specific questionnaire for FMT donor selection [44]. Informed consent was obtained before screening procedures. Donated stool (50–70 g) was filtered to make a stool suspension and stored at −80°C. Thereafter, the stool suspension was strained and poured into a sterile container. Stool suspensions (60 g/300 mL) were thawed at 37°C for 6 h before FMT. Recipients’ antibiotic prescriptions for CDI were stopped 48 h before the FMT procedure. Recipients underwent bowel preparation according to doctor’s instructions before FMT. Stool suspensions (60 g) were injected into the patient through a colonoscopy by a skilled gastroenterologist, following evidence-based FMT guidelines [44]. After FMT, recipients were monitored for any side effects of FMT at the microbiome center.
Cognitive function and laboratory evaluations
Cognitive function and mental status evaluations were performed individually on all dementia patients for approximately 20–30 min by the same neurologist. The MMSE (range, 0–30), CDR (range, 0–3), and GCS (range, 3–15) were administered 1 month before and 1 month after FMT in the FMT group. Control group patients were also assessed at admission due to CDI and 1 month after CDI. The sensitivity and specificity of the MMSE are 27% to 89% and 32% to 90%, respectively [45]. Therefore, MMSE had limited diagnostic accuracy, especially for distinguishing mild cognitive impairment from AD dementia [45]. Although MMSE has limitations, we used it because it has long been used to detect and monitor dementia progression. The CDR has been widely used and has been validated as a reliable tool to grade dementia severity [46, 47]. The CDR scale yields Sum of Boxes (CDR-SB) scores (range, 0–18) with the global score regularly used in clinical and research settings to stage dementia severity [17, 45]. Higher scores on the MMSE and lower scores on CDR-SB suggest better cognitive performance. The GCS is the sum of the scores as well as the individual elements (3 being the worst and 15 being the highest) [41]. For example, a score of 12 might be expressed as GCS12 = E4V5M3. We performed laboratory tests before and after FMT or at admission due to CDI and after CDI treatment at the same time as possible when performing cognitive function tests (maximum 3 days before and after cognitive test) to further confirm whether the patients' cognition is affected by the general condition.
Microbiome analysis
We collected patients’ fecal samples before FMT and compared them with the microbiota compositions 3 weeks later. Fecal samples of control group patients were also collected at admission and 3 weeks after the CDI treatment. All samples were stored at −80°C until shipping to the Macrogen Biotech Lab (Seoul, Korea) for DNA extraction and sequencing. Metagenomic DNA was extracted from fecal samples, and amplification of the V3-V4 region of the bacterial 16S rRNA gene was conducted using barcoded universal primers. Sequencing was carried out using a MiSeq sequencer on the Illumina platform (Macrogen Inc., Seoul, Korea) according to the manufacturer’s specifications. Microbiome profiling was conducted with the 16S-based Microbial taxonomic profiling (MTP) platform of EzBio-Cloud Apps (ChunLab Inc., Seoul, Korea). After taxonomic profiling of each sample, the comparative MTP analyzer of EzBioCloud Apps was used for comparative analysis of the samples. In the MTP platform of ChunLab Inc., Seoul, Korea, preprocessing of the sequencing reads was conducted using the following five steps: 1) filtering of low-quality reads, 2) merging of the paired-end reads, 3) removal of barcode and primer sequences, 4) taxonomic assignment of the reads, and 5) removal of chimeric sequences. Taxonomic assignment of the reads was conducted with ChunLab’s 16S rRNA database (DB ver. PKSSU4.0) [48]. OUT picking was conducted with UCLUST and CD-HIT with a 97% similarity cutoff [49]. Then, Good’s coverage, rarefaction, and alpha-diversity indices, including Chao and Shannon found in MTP, were calculated. Beta-diversity was analyzed using the PERMANOVA test [10]. Overall differences in the microbiome structure were evaluated through Principal Coordinate Analysis (PCoA) to unweighted and weighted UniFrac distance [50, 51]. Linear discriminant analysis (LDA) effect size (LEfSe) was performed to determine the taxonomic difference between pre- and post-FMT [52]. Functional biomarker analysis using phylogenetic investigation of communities by reconstruction of unobserved states (PICRUSt) software package was performed between the pre and post-FMT groups using the EzBio-Cloud Apps [53].
Outcome
The primary end point was a correlation between FMT and cognitive function in dementia patients with CDI compared to the control group. In addition, we analyzed the patients’ gut microbiome before and after FMT to investigate microbiome metabolites that affect cognition.
Statistical analysis
Data are presented as the mean, range, median, or number (percentage) as appropriate. Statistical significance between the two groups was determined using the Kruskal-Wallis test, a non-parametric method for testing whether samples originate from the same distribution. In addition, between two groups comparison were conducted using the Wilcoxon signed rank test, a non-parametric statistical test. Statistical analysis was performed using GraphPad Prism (Version 9.0, GraphPad Software, San Diego, CA, USA), R statistics package (R Foundation, Vienna, Austria; https://www.r-project.org), and SPSS 26.0 for Windows (SPSS Inc., Chicago, IL, USA). P values < 0.05 were considered significant.
Supplementary Materials
Author Contributions
SHP, JHL, JBS, JHI, BRC, SJL, KSK and YWS contributed to data collection and conception of the work. SHP, JHL, JBS, JSK, KSK and SHC did the initial data interpretation. SHP wrote the manuscript with contributions from JHL, JBS, TJK and SBK. The sponsor collected and analyzed the data in conjunction with the authors, who contributed to manuscript draft revisions, provided critical comment, and approved submission for publication.
Acknowledgments
We thank the study participants, their families and caregivers.
Conflicts of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as potential conflicts of interest.
Ethical Statement
This prospective observational study was conducted according to the tenets of the Declaration of Helsinki and was approved by the Institutional Review Board of Inha University Hospital (2018-08-015, 2020-11-004). All subjects provided informed written consent.
Funding
This paper was supported by the National Research Foundation of Korea (NRF) under Grant (2021R1F1A1049320), by Bumsuk Academic Research Fund in 2021, by Memorial Foundation for Dr. Suh Succjo by named Hyangseal, Korean Neurological Association (KNA-21-HS-08), by the Ministry of Science and ICT of Republic of Korea (NRF-2020M3E5D2A01084721), and Inha University.
References
- 1. GBD 2016 Neurology Collaborators. Global, regional, and national burden of neurological disorders, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2019; 18:459–80. https://doi.org/10.1016/S1474-4422(18)30499-X [PubMed]
- 2. Vendrik KEW, Ooijevaar RE, de Jong PRC, Laman JD, van Oosten BW, van Hilten JJ, Ducarmon QR, Keller JJ, Kuijper EJ, Contarino MF. Fecal Microbiota Transplantation in Neurological Disorders. Front Cell Infect Microbiol. 2020; 10:98. https://doi.org/10.3389/fcimb.2020.00098 [PubMed]
- 3. Martin CR, Osadchiy V, Kalani A, Mayer EA. The Brain-Gut-Microbiome Axis. Cell Mol Gastroenterol Hepatol. 2018; 6:133–48. https://doi.org/10.1016/j.jcmgh.2018.04.003 [PubMed]
- 4. Hazan S. Rapid improvement in Alzheimer's disease symptoms following fecal microbiota transplantation: a case report. J Int Med Res. 2020; 48:300060520925930. https://doi.org/10.1177/0300060520925930 [PubMed]
- 5. Ma Q, Xing C, Long W, Wang HY, Liu Q, Wang RF. Impact of microbiota on central nervous system and neurological diseases: the gut-brain axis. J Neuroinflammation. 2019; 16:53. https://doi.org/10.1186/s12974-019-1434-3 [PubMed]
- 6. Sun J, Xu J, Ling Y, Wang F, Gong T, Yang C, Ye S, Ye K, Wei D, Song Z, Chen D, Liu J. Fecal microbiota transplantation alleviated Alzheimer's disease-like pathogenesis in APP/PS1 transgenic mice. Transl Psychiatry. 2019; 9:189. https://doi.org/10.1038/s41398-019-0525-3 [PubMed]
- 7. D'Amato A, Di Cesare Mannelli L, Lucarini E, Man AL, Le Gall G, Branca JJV, Ghelardini C, Amedei A, Bertelli E, Regoli M, Pacini A, Luciani G, Gallina P, et al. Faecal microbiota transplant from aged donor mice affects spatial learning and memory via modulating hippocampal synaptic plasticity- and neurotransmission-related proteins in young recipients. Microbiome. 2020; 8:140. https://doi.org/10.1186/s40168-020-00914-w [PubMed]
- 8. Li Y, Ning L, Yin Y, Wang R, Zhang Z, Hao L, Wang B, Zhao X, Yang X, Yin L, Wu S, Guo D, Zhang C. Age-related shifts in gut microbiota contribute to cognitive decline in aged rats. Aging (Albany NY). 2020; 12:7801–17. https://doi.org/10.18632/aging.103093 [PubMed]
- 9. Le Chatelier E, Nielsen T, Qin J, Prifti E, Hildebrand F, Falony G, Almeida M, Arumugam M, Batto JM, Kennedy S, Leonard P, Li J, Burgdorf K, et al, and MetaHIT consortium. Richness of human gut microbiome correlates with metabolic markers. Nature. 2013; 500:541–6. https://doi.org/10.1038/nature12506 [PubMed]
- 10. Anderson MJ. Permutational Multivariate Analysis of Variance (PERMANOVA). In Balakrishnan N, Colton T, Everitt B, Piegorsch W, Ruggeri F, Teugels JL, eds. Wiley StatsRef: Statistics Reference Online. 2017. https://doi.org/10.1002/9781118445112.stat07841
- 11. Saito T, Matsuba Y, Mihira N, Takano J, Nilsson P, Itohara S, Iwata N, Saido TC. Single App knock-in mouse models of Alzheimer's disease. Nat Neurosci. 2014; 17:661–3. https://doi.org/10.1038/nn.3697 [PubMed]
- 12. Vogt NM, Kerby RL, Dill-McFarland KA, Harding SJ, Merluzzi AP, Johnson SC, Carlsson CM, Asthana S, Zetterberg H, Blennow K, Bendlin BB, Rey FE. Gut microbiome alterations in Alzheimer's disease. Sci Rep. 2017; 7:13537. https://doi.org/10.1038/s41598-017-13601-y [PubMed]
- 13. Bäuerl C, Collado MC, Diaz Cuevas A, Viña J, Pérez Martínez G. Shifts in gut microbiota composition in an APP/PSS1 transgenic mouse model of Alzheimer's disease during lifespan. Lett Appl Microbiol. 2018; 66:464–71. https://doi.org/10.1111/lam.12882 [PubMed]
- 14. Park SH, Lee JH, Shin J, Kim JS, Cha B, Lee S, Kwon KS, Shin YW, Choi SH. Cognitive function improvement after fecal microbiota transplantation in Alzheimer's dementia patient: a case report. Curr Med Res Opin. 2021; 37:1739–44. https://doi.org/10.1080/03007995.2021.1957807 [PubMed]
- 15. Gu X, Zhou J, Zhou Y, Wang H, Si N, Ren W, Zhao W, Fan X, Gao W, Wei X, Yang J, Bian B, Zhao H. Huanglian Jiedu decoction remodels the periphery microenvironment to inhibit Alzheimer's disease progression based on the "brain-gut" axis through multiple integrated omics. Alzheimers Res Ther. 2021; 13:44. https://doi.org/10.1186/s13195-021-00779-7 [PubMed]
- 16. Guerrero-Berroa E, Luo X, Schmeidler J, Rapp MA, Dahlman K, Grossman HT, Haroutunian V, Beeri MS. The MMSE orientation for time domain is a strong predictor of subsequent cognitive decline in the elderly. Int J Geriatr Psychiatry. 2009; 24:1429–37. https://doi.org/10.1002/gps.2282 [PubMed]
- 17. Angelucci F, Cechova K, Amlerova J, Hort J. Antibiotics, gut microbiota, and Alzheimer's disease. J Neuroinflammation. 2019; 16:108. https://doi.org/10.1186/s12974-019-1494-4 [PubMed]
- 18. Ren T, Gao Y, Qiu Y, Jiang S, Zhang Q, Zhang J, Wang L, Zhang Y, Wang L, Nie K. Gut Microbiota Altered in Mild Cognitive Impairment Compared With Normal Cognition in Sporadic Parkinson's Disease. Front Neurol. 2020; 11:137. https://doi.org/10.3389/fneur.2020.00137 [PubMed]
- 19. Griffin JW, Bradshaw PC. Amino Acid Catabolism in Alzheimer's Disease Brain: Friend or Foe? Oxid Med Cell Longev. 2017; 2017:5472792. https://doi.org/10.1155/2017/5472792 [PubMed]
- 20. Liu P, Fleete MS, Jing Y, Collie ND, Curtis MA, Waldvogel HJ, Faull RL, Abraham WC, Zhang H. Altered arginine metabolism in Alzheimer's disease brains. Neurobiol Aging. 2014; 35:1992–2003. https://doi.org/10.1016/j.neurobiolaging.2014.03.013 [PubMed]
- 21. Coloma E, Prieto-González S, López-Giraldo A, López-Soto A. Hyperammonemic encephalopathy due to a urinary diversion: an uncommon cause of reversible dementia. J Am Geriatr Soc. 2011; 59:930–2. https://doi.org/10.1111/j.1532-5415.2011.03377.x [PubMed]
- 22. Gropman AL, Summar M, Leonard JV. Neurological implications of urea cycle disorders. J Inherit Metab Dis. 2007; 30:865–79. https://doi.org/10.1007/s10545-007-0709-5 [PubMed]
- 23. Wiesinger H. Arginine metabolism and the synthesis of nitric oxide in the nervous system. Prog Neurobiol. 2001; 64:365–91. https://doi.org/10.1016/s0301-0082(00)00056-3 [PubMed]
- 24. Esposito Z, Belli L, Toniolo S, Sancesario G, Bianconi C, Martorana A. Amyloid β, glutamate, excitotoxicity in Alzheimer’s disease: are we on the right track? CNS Neurosci Ther. 2013; 19:549–55. https://doi.org/10.1111/cns.12095 [PubMed]
- 25. Jęśko H, Wilkaniec A, Cieślik M, Hilgier W, Gąssowska M, Lukiw WJ, Adamczyk A. Altered Arginine Metabolism in Cells Transfected with Human Wild-Type Beta Amyloid Precursor Protein (βAPP). Curr Alzheimer Res. 2016; 13:1030–9. https://doi.org/10.2174/1567205013666160314150348 [PubMed]
- 26. Ibáñez C, Simó C, Martín-Álvarez PJ, Kivipelto M, Winblad B, Cedazo-Mínguez A, Cifuentes A. Toward a predictive model of Alzheimer's disease progression using capillary electrophoresis-mass spectrometry metabolomics. Anal Chem. 2012; 84:8532–40. https://doi.org/10.1021/ac301243k [PubMed]
- 27. Kaddurah-Daouk R, Zhu H, Sharma S, Bogdanov M, Rozen SG, Matson W, Oki NO, Motsinger-Reif AA, Churchill E, Lei Z, Appleby D, Kling MA, Trojanowski JQ, et al, and Pharmacometabolomics Research Network. Alterations in metabolic pathways and networks in Alzheimer's disease. Transl Psychiatry. 2013; 3:e244. https://doi.org/10.1038/tp.2013.18 [PubMed]
- 28. Graham SF, Chevallier OP, Elliott CT, Hölscher C, Johnston J, McGuinness B, Kehoe PG, Passmore AP, Green BD. Untargeted metabolomic analysis of human plasma indicates differentially affected polyamine and L-arginine metabolism in mild cognitive impairment subjects converting to Alzheimer's disease. PLoS One. 2015; 10:e0119452. https://doi.org/10.1371/journal.pone.0119452 [PubMed]
- 29. Trushina E, Dutta T, Persson XM, Mielke MM, Petersen RC. Identification of altered metabolic pathways in plasma and CSF in mild cognitive impairment and Alzheimer's disease using metabolomics. PLoS One. 2013; 8:e63644. https://doi.org/10.1371/journal.pone.0063644 [PubMed]
- 30. González-Domínguez R, García-Barrera T, Gómez-Ariza JL. Metabolite profiling for the identification of altered metabolic pathways in Alzheimer's disease. J Pharm Biomed Anal. 2015; 107:75–81. https://doi.org/10.1016/j.jpba.2014.10.010 [PubMed]
- 31. Gueli MC, Taibi G. Alzheimer's disease: amino acid levels and brain metabolic status. Neurol Sci. 2013; 34:1575–9. https://doi.org/10.1007/s10072-013-1289-9 [PubMed]
- 32. Mattson MP, Cheng B, Davis D, Bryant K, Lieberburg I, Rydel RE. beta-Amyloid peptides destabilize calcium homeostasis and render human cortical neurons vulnerable to excitotoxicity. J Neurosci. 1992; 12:376–89. https://doi.org/10.1523/JNEUROSCI.12-02-00376.1992 [PubMed]
- 33. Wu Z, Guo Z, Gearing M, Chen G. Tonic inhibition in dentate gyrus impairs long-term potentiation and memory in an Alzheimer's [corrected] disease model. Nat Commun. 2014; 5:4159. https://doi.org/10.1038/ncomms5159 [PubMed]
- 34. Panov A, Orynbayeva Z, Vavilin V, Lyakhovich V. Fatty acids in energy metabolism of the central nervous system. Biomed Res Int. 2014; 2014:472459. https://doi.org/10.1155/2014/472459 [PubMed]
- 35. Mächler P, Wyss MT, Elsayed M, Stobart J, Gutierrez R, von Faber-Castell A, Kaelin V, Zuend M, San Martín A, Romero-Gómez I, Baeza-Lehnert F, Lengacher S, Schneider BL, et al. In Vivo Evidence for a Lactate Gradient from Astrocytes to Neurons. Cell Metab. 2016; 23:94–102. https://doi.org/10.1016/j.cmet.2015.10.010 [PubMed]
- 36. Rapsinski GJ, Newman TN, Oppong GO, van Putten JPM, Tükel Ç. CD14 protein acts as an adaptor molecule for the immune recognition of Salmonella curli fibers. J Biol Chem. 2013; 288:14178–88. https://doi.org/10.1074/jbc.M112.447060 [PubMed]
- 37. Jones L, Holmans PA, Hamshere ML, Harold D, Moskvina V, Ivanov D, Pocklington A, Abraham R, Hollingworth P, Sims R, Gerrish A, Pahwa JS, Jones N, et al. Genetic evidence implicates the immune system and cholesterol metabolism in the aetiology of Alzheimer's disease. PLoS One. 2010; 5:e13950. https://doi.org/10.1371/journal.pone.0013950 [PubMed]
- 38. Fan Z, Brooks DJ, Okello A, Edison P. An early and late peak in microglial activation in Alzheimer's disease trajectory. Brain. 2017; 140:792–803. https://doi.org/10.1093/brain/aww349 [PubMed]
- 39. Sarlus H, Heneka MT. Microglia in Alzheimer's disease. J Clin Invest. 2017; 127:3240–9. https://doi.org/10.1172/JCI90606 [PubMed]
- 40. Celorrio M, Abellanas MA, Rhodes J, Goodwin V, Moritz J, Vadivelu S, Wang L, Rodgers R, Xiao S, Anabayan I, Payne C, Perry AM, Baldridge MT, et al. Gut microbial dysbiosis after traumatic brain injury modulates the immune response and impairs neurogenesis. Acta Neuropathol Commun. 2021; 9:40. https://doi.org/10.1186/s40478-021-01137-2 [PubMed]
- 41. Chapman KR, Bing-Canar H, Alosco ML, Steinberg EG, Martin B, Chaisson C, Kowall N, Tripodis Y, Stern RA. Mini Mental State Examination and Logical Memory scores for entry into Alzheimer's disease trials. Alzheimers Res Ther. 2016; 8:9. https://doi.org/10.1186/s13195-016-0176-z [PubMed]
- 42. Jain S, Iverson LM. Glasgow Coma Scale. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2022. [PubMed]
- 43. Silva YP, Bernardi A, Frozza RL. The Role of Short-Chain Fatty Acids From Gut Microbiota in Gut-Brain Communication. Front Endocrinol (Lausanne). 2020; 11:25. https://doi.org/10.3389/fendo.2020.00025 [PubMed]
- 44. van Nood E, Vrieze A, Nieuwdorp M, Fuentes S, Zoetendal EG, de Vos WM, Visser CE, Kuijper EJ, Bartelsman JF, Tijssen JG, Speelman P, Dijkgraaf MG, Keller JJ. Duodenal infusion of donor feces for recurrent Clostridium difficile. N Engl J Med. 2013; 368:407–15. https://doi.org/10.1056/NEJMoa1205037 [PubMed]
- 45. Arevalo-Rodriguez I, Smailagic N, Roqué I Figuls M, Ciapponi A, Sanchez-Perez E, Giannakou A, Pedraza OL, Bonfill Cosp X, Cullum S. Mini-Mental State Examination (MMSE) for the detection of Alzheimer's disease and other dementias in people with mild cognitive impairment (MCI). Cochrane Database Syst Rev. 2015; 2015:CD010783. https://doi.org/10.1002/14651858.CD010783.pub2 [PubMed]
- 46. Wada-Isoe K, Kikuchi T, Umeda-Kameyama Y, Mori T, Akishita M, Nakamura Y, and ABC Dementia Scale Research Group. Global Clinical Dementia Rating Score of 0.5 May Not Be an Accurate Criterion to Identify Individuals with Mild Cognitive Impairment. J Alzheimers Dis Rep. 2019; 3:233–9. https://doi.org/10.3233/ADR-190126 [PubMed]
- 47. Morris JC. The Clinical Dementia Rating (CDR): current version and scoring rules. Neurology. 1993; 43:2412–4. https://doi.org/10.1212/wnl.43.11.2412-a [PubMed]
- 48. Yoon SH, Ha SM, Kwon S, Lim J, Kim Y, Seo H, Chun J. Introducing EzBioCloud: a taxonomically united database of 16S rRNA gene sequences and whole-genome assemblies. Int J Syst Evol Microbiol. 2017; 67:1613–7. https://doi.org/10.1099/ijsem.0.001755 [PubMed]
- 49. Edgar RC. Search and clustering orders of magnitude faster than BLAST. Bioinformatics. 2010; 26:2460–1. https://doi.org/10.1093/bioinformatics/btq461 [PubMed]
- 50. Zakrzewski M, Proietti C, Ellis JJ, Hasan S, Brion MJ, Berger B, Krause L. Calypso: a user-friendly web-server for mining and visualizing microbiome-environment interactions. Bioinformatics. 2017; 33:782–3. https://doi.org/10.1093/bioinformatics/btw725 [PubMed]
- 51. Lozupone CA, Hamady M, Kelley ST, Knight R. Quantitative and qualitative beta diversity measures lead to different insights into factors that structure microbial communities. Appl Environ Microbiol. 2007; 73:1576–85. https://doi.org/10.1128/AEM.01996-06 [PubMed]
- 52. Segata N, Izard J, Waldron L, Gevers D, Miropolsky L, Garrett WS, Huttenhower C. Metagenomic biomarker discovery and explanation. Genome Biol. 2011; 12:R60. https://doi.org/10.1186/gb-2011-12-6-r60 [PubMed]
- 53. Langille MG, Zaneveld J, Caporaso JG, McDonald D, Knights D, Reyes JA, Clemente JC, Burkepile DE, Vega Thurber RL, Knight R, Beiko RG, Huttenhower C. Predictive functional profiling of microbial communities using 16S rRNA marker gene sequences. Nat Biotechnol. 2013; 31:814–21. https://doi.org/10.1038/nbt.2676 [PubMed]



