Introduction
Parkinson disease (PD) is a progressive neurodegenerative disorder which the majority of symptoms are caused by loss of dopaminergic neurons in the substantia nigra and pathology originate in the central nervous system (CNS) [1]. However, gastrointestinal (GI) symptoms, such as constipation and dyspepsia, are prevalent nonmotor symptoms (NMS) of PD [2]. The loss of neurons and the deposition of α-synuclein in the enteric nerve system, which represents the extra-CNS involvement of the disease pathology, are observed in people with PD (PwP) [3–5]. Furthermore, certain bowel conditions, including ulcerative colitis [6] and truncal vagotomy during gastrectomy [7, 8], affect the risk of PD.
Microorganisms, the primary inhabitants of the intestinal system, are also associated with PD. PwP tend to experience small intestinal bacterial overgrowth [9, 10], and their gut microbiota, the assemblage of microorganisms (bacteria, archaea, eukaryotes, and viruses) in the gut, is usually distinct from that of healthy controls [11]. In addition to PD, gut microbiota also plays a major role in Alzheimer disease [12] and aging [13, 14].
Some specific bacterial strains, such as Bifidobacteria and Lactobacilli, have been widely deemed to be health-promoting components of the gut microbiota [15]. Some pathobiontic bacteria, however, such as Helicobacter hepaticus, segmented filamentous bacteria, Escherichia coli, and Enterococcus faecalis may be pathologic and increase inflammation or tumorigenesis [16, 17]. The modulation of the gut microbiota can be achieved either by prebiotics (nondigestible food ingredients that, when consumed in sufficient amounts, selectively stimulate the growth and/or activity of one or a limited number of microbes in the colon), probiotics (live bacteria and yeasts that, when administered in appropriate doses, induce a beneficial effect on health), and synbiotics (the combination of both) [18]. The main purpose of microbiota modulation is to promote beneficial interactions between the gut microbiota and the host [19].
The application of prebiotics or probiotics increases the frequency of bowel movements and relieves constipation [20]. For PwP, constipation is a distressful NMS that is unresponsive to dopaminergic supplements. Laxative agents may temporarily attenuate its severity, but the response wanes rapidly [21]. The lack of proper management of constipation is a challenge, and constipation is strongly associated with a poor quality of life for PwP [22]. The etiology of constipation in PD is multifactorial, including the reduction of bowel movement frequency, less body motility, and an unhealthy gut environment [2]. A Bifidobacteria and Lactobacilli–based probiotic regimen increases bowel movement frequency and mitigates constipation among PwP [23, 24], but studies have been few and small in sample size, with consequent adverse effects on the results’ level of evidence. Furthermore, probiotics affect local inflammation, reduce pathogenic gut microorganism–related leaky gut syndrome [25, 26], and modulate metabolism [27]. These benefits may be associated with the pathogenesis of PD, leading to possible modification of disease progression.
Despite an understanding of the gut microbiota in PwP and the possible beneficial roles of probiotics, some essential points remain unclear, especially the beneficial and detrimental strains of bacteria for use in alleviating the symptoms of PD. PwP tend to have more Lactobacilli than controls do [11], and whether this difference is the cause or consequence of PD pathology is unknown. The present study reviewed the interventional clinical trials and conducted a meta-analysis of randomized controlled trials (RCTs) to investigate the effect of probiotics on PwP.
Materials and Methods
Inclusion criteria and study selection
This systematic review was performed by two reviewers (J-H.C. and C-T.H.), and any disagreements were resolved after a panel discussion involving three reviewers (J-H.C., C-T.H., and T-W. H.). Eligible studies included are (1) interventional clinical trials (RCTs or non-RCTs) (2) clearly report patient inclusion and exclusion criteria (3) comparing the effects of probiotics on PwP (4) available data on GI symptoms including bowel movement and stool consistency. Studies were excluded if the intervention (1) did not include PwP (2) did not use probiotics as intervention (3) did not clearly report the effect on GI symptoms (4) was not published in English. This study is registered with PROSPERO (CRD42021262036).
Literature search strategy
We searched for interventional clinical trials published before May 2022 in the PubMed, Embase, and Cochrane databases following the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) guidelines [28]. The search keywords were as follows: {(Parkinson disease [Title/Abstract] OR Parkinson’s disease [Title/Abstract]) AND (probiotics [Title/Abstract] OR Bifidobacterium [Title/Abstract] OR Lactobacillus [Title/Abstract] OR Enterococcus [Title/Abstract] OR Limosilactobacillus [Title/Abstract]). Only studies published in English were included.
Data extraction
Baseline and outcome data were independently retrieved by two reviewers (J-H.C. and C-T.H.). Furthermore, data on study designs, study population characteristics, inclusion and exclusion criteria, main outcomes and adverse events were extracted. Decisions recorded individually by the reviewers were compared, and disagreements were resolved by a third reviewer (T-W.H.).
Outcomes
All outcomes of the included studies were summarized for the literature review. The primary outcome of the meta-analysis was the effect of probiotics on constipation, including bowel movement and stool consistency.
Appraisal of methodological quality
Two reviewers (C-T.H. and J-H.C.) independently assessed the methodological quality of each study using the revised Risk of Bias (version 2.0) method for the RCTs [29] and Risk of Bias Assessment Tool for Systematic Reviews-I (ROBIS-I) for the non-RCT, as recommended by the Cochrane Collaboration [30]. The included RCTs were scored to determine whether they had a high, medium, or low overall risk of bias. The risk of bias was calculated through the assessment of five domains: bias resulting from the randomization process, bias resulting from deviation from intended interventions, bias resulting from missing outcome data, bias in the measurement of outcomes, and bias in the selection of reported results. Serious, moderate, or low overall risk among non-RCTs was assessed for seven domains: bias resulting from confounding, bias in selection of participants into the study, bias in classification of interventions, bias resulting from deviations from intended interventions, bias resulting from missing data, bias in measurement of outcomes, and bias in selection of the reported result.
Statistical analysis
Data for the meta-analysis were entered and analyzed using Review Manager 5.4 (The Cochrane Collaboration, Oxford, England). A meta-analysis was performed following the PRISMA guidelines. The standard deviation was calculated using the provided confidence interval (CI) limits, standard errors, or interquartile ranges, where appropriate. The effect sizes of continuous outcomes were reported as the standardized mean difference (SMD). The precision of effect sizes was reported using a 95% CI. A pooled estimate of the weighted mean difference (WMD) was computed using the DerSimonian and Laird random-effects method. A statistically significant result was indicated by a p value of <0.05 or a 95% CI that did not include 1 in the relative risk ratio and 0 in the WMD estimation. Statistical heterogeneity and inconsistencies in treatment effects across the studies were evaluated using the Cochrane Q test and I2 statistic, respectively. Statistical significance was set at a p value of <0.10 for the Cochrane Q test. Statistical heterogeneity across the studies was assessed using the I2 statistic, which quantifies the proportion of total outcome variability across studies.
Data availability statement (DAS)
The authors confirm that the data supporting the findings of this study are available within the article and its Supplementary Materials. These data were derived from the following resources available in the public domain.
Results
Search results and study characteristics
Figure 1 displays the study selection flowchart. Our initial search yielded 151 studies, 19 of which were eliminated because of duplication. The remaining 132 studies were subjected to title and abstract screening, and 118 were excluded. The final 14 studies were entered in the full text review. Two studies were excluded because they were conference posters, and another four were excluded because they reported on ongoing clinical trials. The remaining eight clinical trials, including two open-label, single-arm studies [24, 31] and six eligible RCTs [23, 32–36], were included in the review. These studies were published between 2011 and 2021 and had sample sizes ranging from 25 to 120 PwP. All eight studies recruited PwP who had idiopathic PD diagnoses, and five of the studies specifically enrolled patients with functional constipation. The composition, amount, and treatment duration of probiotics varied, but Lactobacilli and Bifidobacterium were administered in most of the studies. Adverse events were reported in 6 studies, including bloating, abdominal distention, dizziness and lethargy. All of the adverse events were reversible and there was no serious adverse event noted (Table 1).
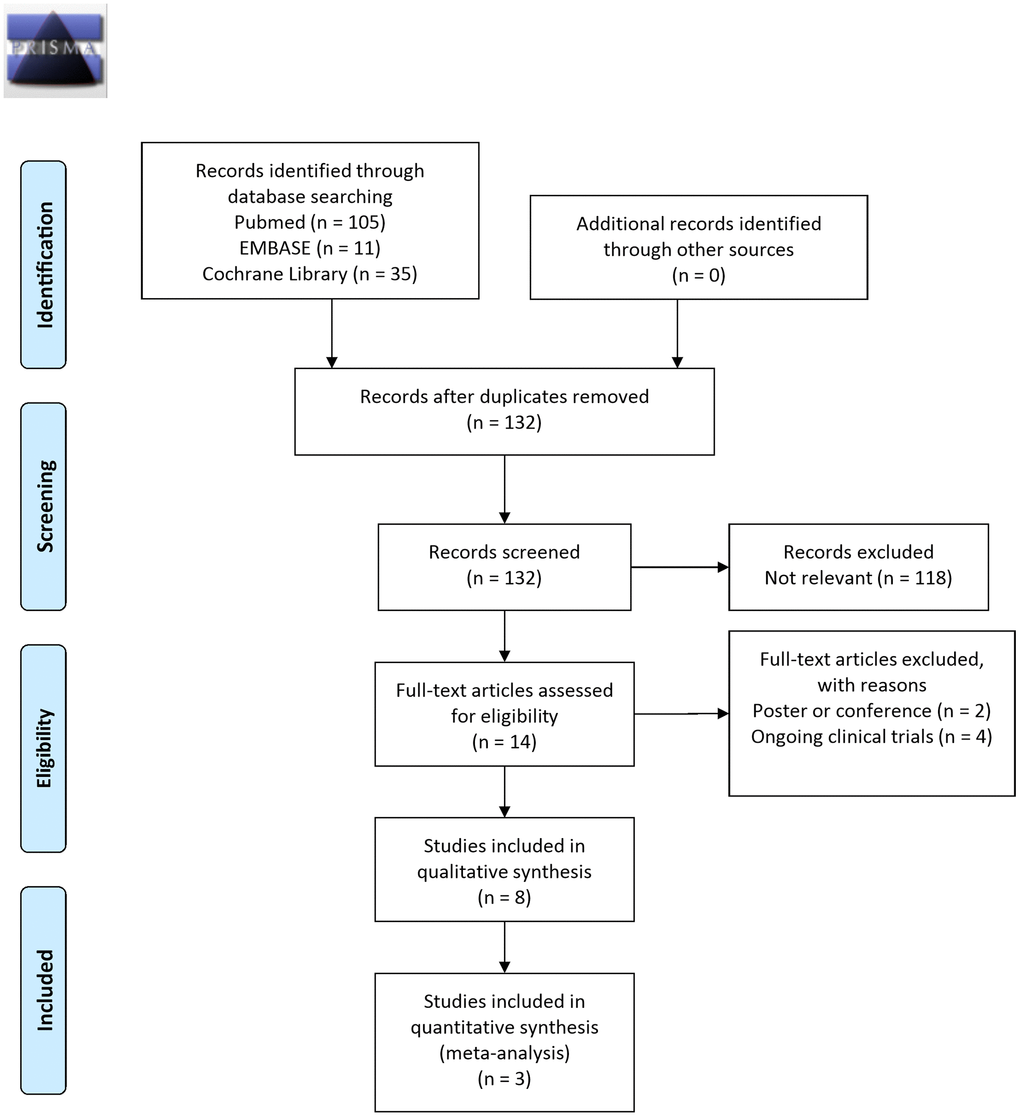
Figure 1. Study selection flowchart (RCT, randomized controlled trial).
Table 1. Characteristics of the included clinical trials.
| Author [Year]/study design | Inclusion criteria | No. of patients (male, %) | Age/disease duration (years) | Probiotics: strains; CFU; treatment duration | Outcome | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cassani [2011]/ Open-label, single-arm | PD with poor drug response constipation | 40 (36, 90) | 71.9/9.75 | Lactobacillus; 6.5·109; 5 weeks | BM, abdominal symptoms, stool consistency | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Barichella [2016]/ RCT | PD with functional constipation | E: 80 (41, 51.2) C: 40 (24, 60.0) | E: 71.8/10.9 C: 69.5/9.6 | Streptococcus, Enterococcus, Lactobacillus, Bifidobacterium; 250·109; 4 weeks | BM, CBM, stool consistency | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Georgescu [2016]/ RCT | PD with mid-to- moderate GI symptoms | probiotics: 20 (10, 50) trimebutine: 20 (7, 35) | probiotics: 69.8/7.05 trimebutine: 75.65/7.5 | Lactobacillus and Bifidobacterium; 120 mg/day; 3 months | GI symptoms | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Borzabadi [2018]/ RCT | PD | E: 25 (17, 68) C: 25 (16, 74) | E: 66.9/5.0 C: 66.7/5.4 | Lactobacillus and Bifidobacterium; 8·109; 12 weeks | Gene expression related to metabolism, biomarkers of inflammation and oxidative stress | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tamtaji [2018]/ RCT | PD | E:30 C:30 | E: 68.2 C: 67.7 | Lactobacillus and Bifidobacterium; 8·109; 12 weeks | Metabolism profile | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ibrahim [2020]/ RCT | PD with functional constipation | E: 27 (16, 59.3) C: 28 (17, 60.7) | E: 69.0/6 C: 70.5/6.5 | Lactobacillus Bifidobacterium; 30·109, 8 weeks | BM, Garrigues Questionnaire, Gut transit time | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tan [2021]/ RCT | PD with functional constipation | E: 34 (20, 58.8) C: 38 (28, 73.7) | E: 70.9/9.7C: 68.6/10.1 | Enterococcus, Lactobacillus, Bifidobacterium, Limosilactobacillus;10·109; 4 weeks | BM, stool consistency, constipation severity score, quality of life | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lu [2021]/ open-label, single-arm | PD with OFF> 3 hours/day | 25 (17, 68) | 61.81/10.12 | Lactobacillus plantarum; 3·109; 12 weeks | UPDRS-III, PDQ-39, NMSS, BDI-II, PAC-SYM and PGI-C | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BDI-II, Beck Depression Inventory-II; BM, bowel movement; CBM, complete bowel movement; C, control; CFU, colony-forming units; E: experimental; GI, gastrointestinal; PAC-SYM, Patient Assessment of Constipation Symptoms; PDQ-39, 39-item Parkinson’s Disease Questionnaire; PGI-C, Patient Global Impression of Change; RCT, randomized controlled trials; UPDRS-III, Unified Parkinson’s Disease Rating Scale part III. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Risk of bias assessment
The results of risk of bias assessment are shown in Figure 2A, 2B. Overall, risk of bias was medium across the included RCT studies. All RCT studies had risk of some concerns regarding performance bias. One trial had another medium risk of allocation bias, measurement bias, and reporting bias. For non-RCT studies, both had a moderate overall risk of bias, especially on bias due to confounding, bias in selection of participants into the study, bias in measurement of outcomes, and bias in selection of the reported result.

Figure 2. Risk of bias assessment (A) randomized controlled trials (B) nonrandomized controlled trials. green light with cross: low risk; yellow light with question mark: some concerns.
Review of include studies
Five studies investigated the effect of probiotics on GI symptoms, especially constipation, of PwP. Cassani et al. conducted an open-label, single-arm study to evaluate the benefits of probiotics on bowel movements, stool consistency, and clinical abdominal symptoms among PwP with constipation with poor drug response [24]. They found that 6-week probiotic treatment normalized the stool consistency and alleviated abdominal symptoms. Georgescu et al., however, conducted an RCT to compare probiotics with trimebutine, an intestinal spasmolytic agent, in PwP with mild-to-moderate GI symptoms [34]. Despite being inferior to trimebutine, probiotics significantly reduced the severity of abdominal pain and bloating when compared with data collected before treatment. However, because of the lack of a placebo control, this information can only be utilized as a single-arm, baseline-controlled trial to demonstrate the effect of probiotics. The remaining three RCTs, published between 2016 and 2021, evaluated the therapeutic benefit of probiotics on the GI symptoms of PwP. Two of the three studies were from Malaysia and added Streptococcus and Enterococcus to the Lactobacillus and Bifidobacterium–based regimen. The meta-analysis of these three studies demonstrated that the prescription of probiotics significantly increased weekly bowel movement (MD: 1.06, 95% CI: 0.61–1.51, p < 0.001, I2 = 40%; Figure 3). Stool consistency was evaluated in two studies, which presented a significant normalization of stool consistency (SMD: 0.61, 95% CI = 0.31–0.91, I2 = 0%; Figure 4). Ibrahim et al. also demonstrated probiotics’ significant reduction of gut transit time, which was measured using red carmine capsules to dye the stool.
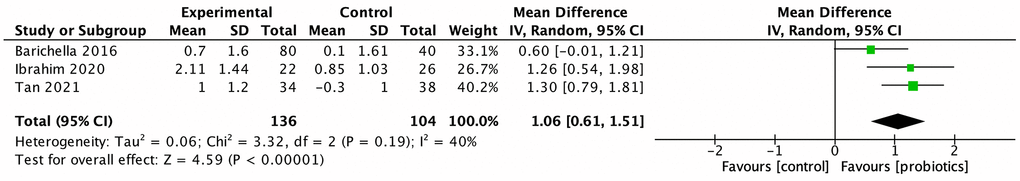
Figure 3. Effect of probiotics on frequency of bowel movements of people with Parkinson disease.
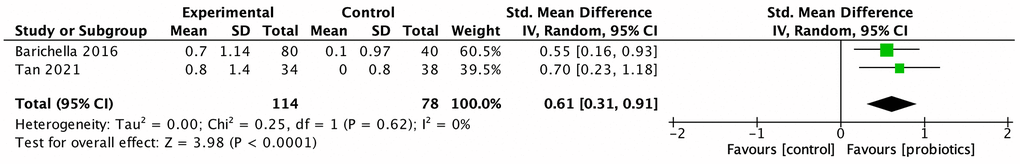
Figure 4. Effect of probiotics on the stool consistency of people with Parkinson disease.
Another two RCTs published by Iranian groups systematically investigated the influence of probiotics. These two RCTs recruited PwP and evaluated the changes in their metabolic profiles and gene expressions after 12 weeks of probiotics treatment. Tamtaji et al. argued that probiotic treatment resulted in a significant decrease in the Movement Disorder Society-Unified Parkinson Disease Rating Score (MDS-UPDRS) and a reduction of systemic inflammation (assessed by high-sensitivity C-reactive protein) and oxidative stress (assessed by malondialdehyde and enhanced glutathione) compared with placebo treatment [32]. Additionally, probiotic treatment also caused a statistically significant reduction in insulin levels and insulin resistance and a statistically significant rise in insulin sensitivity. Borzabadi et al. found that probiotic treatment downregulated the gene expression of proinflammatory cytokines (interleukin-1 and tumor necrosis factor-α) in peripheral blood mononuclear cells compared with controls [35]. Furthermore, probiotics upregulated the gene expression of the anti-inflammatory cytokine, transforming growth factor-β, and the metabolism regulator, peroxisome proliferator-activated receptor gamma. These two RCTs indicated that probiotics’ manipulation of gut microbiota may work not only on GI symptoms but also systemic pathology.
Last, Lu et al. published the latest, open-label, single-arm study that investigated the influence of probiotics on the motor fluctuation of PwP [31]. The prescribed probiotic was a single strain, Lactobacillus plantarum, and after 12 weeks of supplementation, UPDRS motor scores improved significantly in both the “OFF” and “ON” states, and the daily “OFF” period of time was reduced. However, no significant improvement occurred in the NMS of PwP.
Of the eight total studies, three studies mentioned adverse effects of probiotics. In general, these were tolerable and resolved after probiotic discontinuation. Tan et al. reported one case of lethargy after treatment with probiotics [36], and Ibrahim et al. reported two patients with abdominal symptoms and two with dizziness [33].
Meta-analysis results for probiotics treatment
The meta-analysis of three studies demonstrated that the prescription of probiotics significantly increased weekly bowel movement (MD: 1.06, 95% CI: 0.61–1.51, p < 0.001, I2 = 40%; Figure 3). Stool consistency was evaluated in two studies, which presented a significant normalization of stool consistency (SMD: 0.61, 95% CI = 0.31–0.91, I2 = 0%; Figure 4).
Discussion
The present study conducted a literature review of interventional clinical trials to determine the effect of probiotics on PwP and performed a meta-analysis of RCTs to investigate the benefit of probiotics on GI symptoms, especially constipation, among PwP. In general, despite the heterogenicity of the bacterial strains and variations in the amount of probiotics and durations of the treatment regimes, probiotics were shown to significantly increase the frequency of bowel movements and normalize stool consistency among PwP. The side effects are generally tolerable. The present study provides evidence for the prescription of probiotics for PwP, especially for their GI symptoms.
The gut microbiota contains more genomes than any other body location [37]. Altered gut microbiota is associated with many health conditions and has a bidirectional causal relationship [38–40]. An association between the gut and PD has been suspected [22]. Constipation heralds the onset of PD motor symptoms decades ahead of time [41, 42], and the PD pathology, namely α-synuclein aggregation, may originate from the myenteric plexus in the intestine [43, 44]. PwP are also known to embrace gut dysbiosis [45, 46]. Although the intestines’ sterile condition attenuates PD pathology in clinical in vivo models [47], possible methods of manipulating the gut microbiota may only be possible by using prebiotics, probiotics, or symbiotics. According to the present review and meta-analysis, however, how probiotics affect PD-specific disease mechanisms, such as the degeneration of dopaminergic neurons and α-synuclein aggregation, has not been a research focus. Rather, the primary outcomes of most of the clinical trials were mainly GI symptoms, and only two studies from Iranian groups assessed the influence of oxidative stress and inflammation on patient metabolic profiles and gene expression. Metabolic syndrome, oxidative stress, and inflammation are contributors to neurodegeneration in PD [48, 49]. Neuroinflammation is one of the major pathogeneses of PD, and the activation of microglia was noted in post-mortem substantia nigra of PD brains [50]. Systemic inflammation also contributed to an increase in the risk of PD [51]. PwP exhibited elevated blood cytokine levels in serum and plasma extracellular vesicles compared with healthy controls [52, 53]. (Gut dysbiosis is a substantial risk factor of elevated systemic inflammation through the destruction of the intestinal epithelial membrane and the entrance of pathogens and toxins from the intestinal lumen into the systemic circulation [54]. On the hand, diabetes is a well-known risk factor for PD [55], and poor glucose control is a predictor of rapid disease progression [56]. Insulin resistance triggers the significant PD pathogeneses, namely mitochondrial dysfunction and α-synuclein accumulation, and neuronal insulin resistance is remarkable in PwP [57, 58]. Tamtaji et al. demonstrated that probiotic treatment resulted in a reduction in inflammation (assessed by a high-sensitivity C-reactive protein test) and oxidative stress (assessed by glutathione levels) and an increase in insulin sensitivity [32]. With regard to the disease course of PD, the short-term studies used in this meta-analysis cannot support a claim of a disease modification effect based on their short-term benefits. Further long-term studies are necessary to determine the effects of probiotic-related anti-inflammation, antioxidation, and antimetabolic syndrome on decelerating the neurodegenerative process of PD.
The gut microbiota plays an essential role in digestion and is strongly associated with health conditions of the GI system [59]. Probiotic supplementation, which introduces beneficial strains to the gut, may help restore the balance of the gut microbiota [60]. That Lactobacilli and Bifidobacterium in fermented foods relieve GI symptoms and promote bowel movements has been recognized for decades [61]. Studies have determined the abundance of Lactobacilli among PwP is greater than among controls [11, 46]. This suggests that subsequent investigations are needed to assess the ability of exogenous supplements of Lactobacilli to attenuate constipation and other GI symptoms among PwP. The present review and meta-analysis determined the consistent benefit of probiotic supplements containing Lactobacilli on the frequency of bowel movements and normalization of stool consistency. These results may instigate further investigations into whether the association between the greater abundance of Lactobacilli among PwP is a causal disease-pathognomonic relationship or constipation-related compensatory change. However, no sufficient evidence exists to clearly delineate this matter at this time.
The present study provided meta-analysis-based evidence for the benefit of probiotic therapy on constipation among PwP. The latest evidence-based medicine recommendations, based on the RCT published by Barichella et al., for managing constipation among PwP indicate that probiotics are “efficacious” and “clinically useful” [21, 23]. The present meta-analysis could further consolidate this concept after further RCTs are conducted and studied. Additionally, the present study also revealed a possible systemic effect, including the enhancement of probiotics on PD, which may lead to a possible disease modification effect or facilitate the body’s drug response through the manipulation of inflammation, oxidative stress, insulin sensitivity and metabolism.
However, the present study also involved certain limitations. First, only eight studies conducted in five countries, of which three were in Asia and two were in Europe, were included in this study. A person’s environment and diet greatly affect the gut microbiota [62]; therefore, the lack of studies from Africa, Oceania, and the Americas potentially limits the study’s global representation. Besides, the present review is limited to studies published in English, which means some studies may be omitted. Second, the present study could not determine whether the increase of Lactobacilli in PwP is detrimental or not. The regimens of probiotics in all studies contained Lactobacilli, despite the awareness that PwP already have more Lactobacilli than controls do [11]. Because the longest treatment period was only 12 weeks, probiotics’ long-term effects remain uncertain. Third, none of the studies investigated the change in stool short-chain fatty acids (SCFAs), which are potent antioxidants and one of the indicators of healthy gut microbiota [63]. Evidence of increased SCFAs in PwP’s stool after probiotic treatment would further enhance the argument for the beneficial effect of probiotic treatment on PwP. Fourth, the results of the meta-analysis were similar to each of enrolled study, which did not provide new information about the efficacy of probiotic on PwP. However, with the effort of meta-analysis, the findings were more consolidated and able to be listed in the future treatment suggestions of PwP. Last, the endogenous gut microbiota overwhelms supplementary probiotics, and the strict and competitive environment of the intestines reduces the possibility of long-lasting inhabitation of supplementary probiotics [64]. Future studies are suggested to compare the gut microbiota before and after treatment to demonstrate the direct effect of probiotics on PwP’s gut microbiota.
Conclusions
This review and meta-analysis determined that probiotic treatments, mainly Lactobacilli and Bifidobacterium–based regimens, effectively alleviated constipation. Adverse effects are generally tolerable. However, considering the gut microbiota is highly associated with a person’s environment and diet, studies from other continents are required to establish the benefit of probiotics on constipation. Moreover, probiotic treatment is likely to affect the systemic inflammation and metabolism of PwP, but further studies are warranted to investigate the possibility of the disease modification effect on PD.
Author Contributions
1. Research project: A. Conception: JH Chen, TW Huang, CT Hong B. Organization: CT Hong C. Execution: JH Chen, TW Huang, CT Hong.
2. Statistical Analysis: A. Design: TW Huang, CT Hong. B. Execution: JH Chen, TW Huang, CT Hong C. Review and Critique: TW Huang, CT Hong.
3. Manuscript Preparation: A. Writing of the first draft: JH Chen, TW Huang, CT Hong B. Review and Critique: JH Chen, TW Huang, CT Hong.
Acknowledgments
This manuscript was edited by Wallace Academic Editing.
Conflicts of Interest
The authors report no conflicts of interest.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
References
- 1. Gaig C, Tolosa E. When does Parkinson's disease begin? Mov Disord. 2009; 24 Suppl 2:S656–64. https://doi.org/10.1002/mds.22672 [PubMed]
- 2. Fasano A, Visanji NP, Liu LW, Lang AE, Pfeiffer RF. Gastrointestinal dysfunction in Parkinson’s disease. Lancet Neurol. 2015; 14:625–39. https://doi.org/10.1016/S1474-4422(15)00007-1 [PubMed]
- 3. Beach TG, Adler CH, Sue LI, Vedders L, Lue L, White Iii CL, Akiyama H, Caviness JN, Shill HA, Sabbagh MN, Walker DG, and Arizona Parkinson’s Disease Consortium. Multi-organ distribution of phosphorylated alpha-synuclein histopathology in subjects with Lewy body disorders. Acta Neuropathol. 2010; 119:689–702. https://doi.org/10.1007/s00401-010-0664-3 [PubMed]
- 4. Gelpi E, Navarro-Otano J, Tolosa E, Gaig C, Compta Y, Rey MJ, Martí MJ, Hernández I, Valldeoriola F, Reñé R, Ribalta T. Multiple organ involvement by alpha-synuclein pathology in Lewy body disorders. Mov Disord. 2014; 29:1010–8. https://doi.org/10.1002/mds.25776 [PubMed]
- 5. Ohlsson B, Englund E. Atrophic Myenteric and Submucosal Neurons Are Observed in Parkinson’s Disease. Parkinsons Dis. 2019; 2019:7935820. https://doi.org/10.1155/2019/7935820 [PubMed]
- 6. Brudek T. Inflammatory Bowel Diseases and Parkinson’s Disease. J Parkinsons Dis. 2019; 9:S331–44. https://doi.org/10.3233/JPD-191729 [PubMed]
- 7. Liu B, Fang F, Pedersen NL, Tillander A, Ludvigsson JF, Ekbom A, Svenningsson P, Chen H, Wirdefeldt K. Vagotomy and Parkinson disease: A Swedish register-based matched-cohort study. Neurology. 2017; 88:1996–2002. https://doi.org/10.1212/WNL.0000000000003961 [PubMed]
- 8. Svensson E, Horváth-Puhó E, Thomsen RW, Djurhuus JC, Pedersen L, Borghammer P, Sørensen HT. Vagotomy and subsequent risk of Parkinson’s disease. Ann Neurol. 2015; 78:522–9. https://doi.org/10.1002/ana.24448 [PubMed]
- 9. Fasano A, Bove F, Gabrielli M, Petracca M, Zocco MA, Ragazzoni E, Barbaro F, Piano C, Fortuna S, Tortora A, Di Giacopo R, Campanale M, Gigante G, et al. The role of small intestinal bacterial overgrowth in Parkinson's disease. Mov Disord. 2013; 28:1241–9. https://doi.org/10.1002/mds.25522 [PubMed]
- 10. Gabrielli M, Bonazzi P, Scarpellini E, Bendia E, Lauritano EC, Fasano A, Ceravolo MG, Capecci M, Rita Bentivoglio A, Provinciali L, Tonali PA, Gasbarrini A. Prevalence of small intestinal bacterial overgrowth in Parkinson's disease. Mov Disord. 2011; 26:889–92. https://doi.org/10.1002/mds.23566 [PubMed]
- 11. Romano S, Savva GM, Bedarf JR, Charles IG, Hildebrand F, Narbad A. Meta-analysis of the Parkinson’s disease gut microbiome suggests alterations linked to intestinal inflammation. NPJ Parkinsons Dis. 2021; 7:27. https://doi.org/10.1038/s41531-021-00156-z [PubMed]
- 12. Jiang C, Li G, Huang P, Liu Z, Zhao B. The Gut Microbiota and Alzheimer’s Disease. J Alzheimers Dis. 2017; 58:1–15. https://doi.org/10.3233/JAD-161141 [PubMed]
- 13. Bosco N, Noti M. The aging gut microbiome and its impact on host immunity. Genes Immun. 2021; 22:289–303. https://doi.org/10.1038/s41435-021-00126-8 [PubMed]
- 14. DeJong EN, Surette MG, Bowdish DM. The Gut Microbiota and Unhealthy Aging: Disentangling Cause from Consequence. Cell Host Microbe. 2020; 28:180–9. https://doi.org/10.1016/j.chom.2020.07.013 [PubMed]
- 15. Dimidi E, Christodoulides S, Scott SM, Whelan K. Mechanisms of Action of Probiotics and the Gastrointestinal Microbiota on Gut Motility and Constipation. Adv Nutr. 2017; 8:484–94. https://doi.org/10.3945/an.116.014407 [PubMed]
- 16. Akin H, Tözün N. Diet, microbiota, and colorectal cancer. J Clin Gastroenterol. 2014. https://doi.org/10.1097/MCG.0000000000000252 [PubMed]
- 17. Kamada N, Chen GY, Inohara N, Núñez G. Control of pathogens and pathobionts by the gut microbiota. Nat Immunol. 2013; 14:685–90. https://doi.org/10.1038/ni.2608 [PubMed]
- 18. Pandey KR, Naik SR, Vakil BV. Probiotics, prebiotics and synbiotics- a review. J Food Sci Technol. 2015; 52:7577–87. https://doi.org/10.1007/s13197-015-1921-1 [PubMed]
- 19. Ale EC, Binetti AG. Role of Probiotics, Prebiotics, and Synbiotics in the Elderly: Insights Into Their Applications. Front Microbiol. 2021; 12:631254. https://doi.org/10.3389/fmicb.2021.631254 [PubMed]
- 20. Ford AC, Quigley EM, Lacy BE, Lembo AJ, Saito YA, Schiller LR, Soffer EE, Spiegel BM, Moayyedi P. Efficacy of prebiotics, probiotics, and synbiotics in irritable bowel syndrome and chronic idiopathic constipation: systematic review and meta-analysis. Am J Gastroenterol. 2014; 109:1547–61. https://doi.org/10.1038/ajg.2014.202 [PubMed]
- 21. Seppi K, Ray Chaudhuri K, Coelho M, Fox SH, Katzenschlager R, Perez Lloret S, Weintraub D, Sampaio C, and the collaborators of the Parkinson’s Disease Update on Non-Motor Symptoms Study Group on behalf of the Movement Disorders Society Evidence-Based Medicine Committee. Update on treatments for nonmotor symptoms of Parkinson’s disease-an evidence-based medicine review. Mov Disord. 2019; 34:180–98. https://doi.org/10.1002/mds.27602 [PubMed]
- 22. Travagli RA, Browning KN, Camilleri M. Parkinson disease and the gut: new insights into pathogenesis and clinical relevance. Nat Rev Gastroenterol Hepatol. 2020; 17:673–85. https://doi.org/10.1038/s41575-020-0339-z [PubMed]
- 23. Barichella M, Pacchetti C, Bolliri C, Cassani E, Iorio L, Pusani C, Pinelli G, Privitera G, Cesari I, Faierman SA, Caccialanza R, Pezzoli G, Cereda E. Probiotics and prebiotic fiber for constipation associated with Parkinson disease: An RCT. Neurology. 2016; 87:1274–80. https://doi.org/10.1212/WNL.0000000000003127 [PubMed]
- 24. Cassani E, Privitera G, Pezzoli G, Pusani C, Madio C, Iorio L, Barichella M. Use of probiotics for the treatment of constipation in Parkinson’s disease patients. Minerva Gastroenterol Dietol. 2011; 57:117–21. [PubMed]
- 25. Tsai YL, Lin TL, Chang CJ, Wu TR, Lai WF, Lu CC, Lai HC. Probiotics, prebiotics and amelioration of diseases. J Biomed Sci. 2019; 26:3. https://doi.org/10.1186/s12929-018-0493-6 [PubMed]
- 26. Ahmadi S, Wang S, Nagpal R, Wang B, Jain S, Razazan A, Mishra SP, Zhu X, Wang Z, Kavanagh K, Yadav H. A human-origin probiotic cocktail ameliorates aging-related leaky gut and inflammation via modulating the microbiota/taurine/tight junction axis. JCI Insight. 2020; 5:e132055. https://doi.org/10.1172/jci.insight.132055 [PubMed]
- 27. Falcinelli S, Rodiles A, Hatef A, Picchietti S, Cossignani L, Merrifield DL, Unniappan S, Carnevali O. Influence of Probiotics Administration on Gut Microbiota Core: A Review on the Effects on Appetite Control, Glucose, and Lipid Metabolism. J Clin Gastroenterol. 2018. https://doi.org/10.1097/MCG.0000000000001064 [PubMed]
- 28. Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JP, Clarke M, Devereaux PJ, Kleijnen J, Moher D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol. 2009; 62:e1–34. https://doi.org/10.1016/j.jclinepi.2009.06.006 [PubMed]
- 29. Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA (editors). Cochrane Handbook for Systematic Reviews of Interventions version 6.2. Cochrane. 2021. https://training.cochrane.org/handbook.
- 30. Sterne JA, Hernán MA, Reeves BC, Savović J, Berkman ND, Viswanathan M, Henry D, Altman DG, Ansari MT, Boutron I, Carpenter JR, Chan AW, Churchill R, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ. 2016; 355:i4919. https://doi.org/10.1136/bmj.i4919 [PubMed]
- 31. Lu CS, Chang HC, Weng YH, Chen CC, Kuo YS, Tsai YC. The Add-On Effect of Lactobacillus plantarum PS128 in Patients With Parkinson's Disease: A Pilot Study. Front Nutr. 2021; 8:650053. https://doi.org/10.3389/fnut.2021.650053 [PubMed]
- 32. Tamtaji OR, Taghizadeh M, Daneshvar Kakhaki R, Kouchaki E, Bahmani F, Borzabadi S, Oryan S, Mafi A, Asemi Z. Clinical and metabolic response to probiotic administration in people with Parkinson’s disease: A randomized, double-blind, placebo-controlled trial. Clin Nutr. 2019; 38:1031–5. https://doi.org/10.1016/j.clnu.2018.05.018 [PubMed]
- 33. Ibrahim A, Ali RA, Manaf MR, Ahmad N, Tajurruddin FW, Qin WZ, Desa SH, Ibrahim NM. Multi-strain probiotics (Hexbio) containing MCP BCMC strains improved constipation and gut motility in Parkinson’s disease: A randomised controlled trial. PLoS One. 2020; 15:e0244680. https://doi.org/10.1371/journal.pone.0244680 [PubMed]
- 34. Georgescu D, Ancusa OE, Georgescu LA, Ionita I, Reisz D. Nonmotor gastrointestinal disorders in older patients with Parkinson’s disease: is there hope? Clin Interv Aging. 2016; 11:1601–8. https://doi.org/10.2147/CIA.S106284 [PubMed]
- 35. Borzabadi S, Oryan S, Eidi A, Aghadavod E, Daneshvar Kakhaki R, Tamtaji OR, Taghizadeh M, Asemi Z. The Effects of Probiotic Supplementation on Gene Expression Related to Inflammation, Insulin and Lipid in Patients with Parkinson’s Disease: A Randomized, Double-blind, PlaceboControlled Trial. Arch Iran Med. 2018; 21:289–95. [PubMed]
- 36. Tan AH, Lim SY, Chong KK, A Manap MA, Hor JW, Lim JL, Low SC, Chong CW, Mahadeva S, Lang AE. Probiotics for Constipation in Parkinson Disease: A Randomized Placebo-Controlled Study. Neurology. 2021; 96:e772–82. https://doi.org/10.1212/WNL.0000000000010998 [PubMed]
- 37. O'Toole PW, Jeffery IB. Gut microbiota and aging. Science. 2015; 350:1214–5. https://doi.org/10.1126/science.aac8469 [PubMed]
- 38. Zheng D, Liwinski T, Elinav E. Interaction between microbiota and immunity in health and disease. Cell Res. 2020; 30:492–506. https://doi.org/10.1038/s41422-020-0332-7 [PubMed]
- 39. Al Khodor S, Shatat IF. Gut microbiome and kidney disease: a bidirectional relationship. Pediatr Nephrol. 2017; 32:921–31. https://doi.org/10.1007/s00467-016-3392-7 [PubMed]
- 40. Dutta D, Lim SH. Bidirectional interaction between intestinal microbiome and cancer: opportunities for therapeutic interventions. Biomark Res. 2020; 8:31. https://doi.org/10.1186/s40364-020-00211-6 [PubMed]
- 41. Goldman JG, Postuma R. Premotor and nonmotor features of Parkinson's disease. Curr Opin Neurol. 2014; 27:434–41. https://doi.org/10.1097/WCO.0000000000000112 [PubMed]
- 42. Lin CH, Lin JW, Liu YC, Chang CH, Wu RM. Risk of Parkinson’s disease following severe constipation: a nationwide population-based cohort study. Parkinsonism Relat Disord. 2014; 20:1371–5. https://doi.org/10.1016/j.parkreldis.2014.09.026 [PubMed]
- 43. Barrenschee M, Zorenkov D, Böttner M, Lange C, Cossais F, Scharf AB, Deuschl G, Schneider SA, Ellrichmann M, Fritscher-Ravens A, Wedel T. Distinct pattern of enteric phospho-alpha-synuclein aggregates and gene expression profiles in patients with Parkinson’s disease. Acta Neuropathol Commun. 2017; 5:1. https://doi.org/10.1186/s40478-016-0408-2 [PubMed]
- 44. Phillips RJ, Walter GC, Wilder SL, Baronowsky EA, Powley TL. Alpha-synuclein-immunopositive myenteric neurons and vagal preganglionic terminals: autonomic pathway implicated in Parkinson’s disease? Neuroscience. 2008; 153:733–50. https://doi.org/10.1016/j.neuroscience.2008.02.074 [PubMed]
- 45. Wallen ZD, Appah M, Dean MN, Sesler CL, Factor SA, Molho E, Zabetian CP, Standaert DG, Payami H. Characterizing dysbiosis of gut microbiome in PD: evidence for overabundance of opportunistic pathogens. NPJ Parkinsons Dis. 2020; 6:11. https://doi.org/10.1038/s41531-020-0112-6 [PubMed]
- 46. Nishiwaki H, Ito M, Ishida T, Hamaguchi T, Maeda T, Kashihara K, Tsuboi Y, Ueyama J, Shimamura T, Mori H, Kurokawa K, Katsuno M, Hirayama M, Ohno K. Meta-Analysis of Gut Dysbiosis in Parkinson’s Disease. Mov Disord. 2020; 35:1626–35. https://doi.org/10.1002/mds.28119 [PubMed]
- 47. Sampson TR, Debelius JW, Thron T, Janssen S, Shastri GG, Ilhan ZE, Challis C, Schretter CE, Rocha S, Gradinaru V, Chesselet MF, Keshavarzian A, Shannon KM, et al. Gut Microbiota Regulate Motor Deficits and Neuroinflammation in a Model of Parkinson’s Disease. Cell. 2016; 167:1469–80.e12. https://doi.org/10.1016/j.cell.2016.11.018 [PubMed]
- 48. Schapira AH, Jenner P. Etiology and pathogenesis of Parkinson's disease. Mov Disord. 2011; 26:1049–55. https://doi.org/10.1002/mds.23732 [PubMed]
- 49. Zhang P, Tian B. Metabolic syndrome: an important risk factor for Parkinson's disease. Oxid Med Cell Longev. 2014; 2014:729194. https://doi.org/10.1155/2014/729194 [PubMed]
- 50. Wang Q, Liu Y, Zhou J. Neuroinflammation in Parkinson’s disease and its potential as therapeutic target. Transl Neurodegener. 2015; 4:19. https://doi.org/10.1186/s40035-015-0042-0 [PubMed]
- 51. Ferrari CC, Tarelli R. Parkinson’s disease and systemic inflammation. Parkinsons Dis. 2011; 2011:436813. https://doi.org/10.4061/2011/436813 [PubMed]
- 52. Chan L, Chung CC, Chen JH, Yu RC, Hong CT. Cytokine Profile in Plasma Extracellular Vesicles of Parkinson’s Disease and the Association with Cognitive Function. Cells. 2021; 10:604. https://doi.org/10.3390/cells10030604 [PubMed]
- 53. Lin CH, Chen CC, Chiang HL, Liou JM, Chang CM, Lu TP, Chuang EY, Tai YC, Cheng C, Lin HY, Wu MS. Altered gut microbiota and inflammatory cytokine responses in patients with Parkinson’s disease. J Neuroinflammation. 2019; 16:129. https://doi.org/10.1186/s12974-019-1528-y [PubMed]
- 54. Dutta SK, Verma S, Jain V, Surapaneni BK, Vinayek R, Phillips L, Nair PP. Parkinson’s Disease: The Emerging Role of Gut Dysbiosis, Antibiotics, Probiotics, and Fecal Microbiota Transplantation. J Neurogastroenterol Motil. 2019; 25:363–76. https://doi.org/10.5056/jnm19044 [PubMed]
- 55. Chohan H, Senkevich K, Patel RK, Bestwick JP, Jacobs BM, Bandres Ciga S, Gan-Or Z, Noyce AJ. Type 2 Diabetes as a Determinant of Parkinson’s Disease Risk and Progression. Mov Disord. 2021; 36:1420–9. https://doi.org/10.1002/mds.28551 [PubMed]
- 56. Ou R, Wei Q, Hou Y, Zhang L, Liu K, Lin J, Jiang Z, Song W, Cao B, Shang H. Effect of diabetes control status on the progression of Parkinson’s disease: A prospective study. Ann Clin Transl Neurol. 2021; 8:887–97. https://doi.org/10.1002/acn3.51343 [PubMed]
- 57. Chou SY, Chan L, Chung CC, Chiu JY, Hsieh YC, Hong CT. Altered Insulin Receptor Substrate 1 Phosphorylation in Blood Neuron-Derived Extracellular Vesicles From Patients With Parkinson’s Disease. Front Cell Dev Biol. 2020; 8:564641. https://doi.org/10.3389/fcell.2020.564641 [PubMed]
- 58. Hong CT, Chen KY, Wang W, Chiu JY, Wu D, Chao TY, Hu CJ, Chau KD, Bamodu OA. Insulin Resistance Promotes Parkinson’s Disease through Aberrant Expression of α-Synuclein, Mitochondrial Dysfunction, and Deregulation of the Polo-Like Kinase 2 Signaling. Cells. 2020; 9:740. https://doi.org/10.3390/cells9030740 [PubMed]
- 59. Nagao-Kitamoto H, Kitamoto S, Kuffa P, Kamada N. Pathogenic role of the gut microbiota in gastrointestinal diseases. Intest Res. 2016; 14:127–38. https://doi.org/10.5217/ir.2016.14.2.127 [PubMed]
- 60. Hemarajata P, Versalovic J. Effects of probiotics on gut microbiota: mechanisms of intestinal immunomodulation and neuromodulation. Therap Adv Gastroenterol. 2013; 6:39–51. https://doi.org/10.1177/1756283X12459294 [PubMed]
- 61. Kok CR, Hutkins R. Yogurt and other fermented foods as sources of health-promoting bacteria. Nutr Rev. 2018 (Suppl 1); 76:4–15. https://doi.org/10.1093/nutrit/nuy056 [PubMed]
- 62. Dong TS, Gupta A. Influence of Early Life, Diet, and the Environment on the Microbiome. Clin Gastroenterol Hepatol. 2019; 17:231–42. https://doi.org/10.1016/j.cgh.2018.08.067 [PubMed]
- 63. Dalile B, Van Oudenhove L, Vervliet B, Verbeke K. The role of short-chain fatty acids in microbiota-gut-brain communication. Nat Rev Gastroenterol Hepatol. 2019; 16:461–78. https://doi.org/10.1038/s41575-019-0157-3 [PubMed]
- 64. Wieërs G, Belkhir L, Enaud R, Leclercq S, Philippart de Foy JM, Dequenne I, de Timary P, Cani PD. How Probiotics Affect the Microbiota. Front Cell Infect Microbiol. 2020; 9:454. https://doi.org/10.3389/fcimb.2019.00454 [PubMed]


