Introduction
The World Health Organization (WHO) published the World Report on Aging and Health in 2015 and the Integrated Care for Older People (ICOPE) in 2017, which has transformed health services from the traditional disease-focused approach into a function-centered one in the scheme of healthy aging. Healthy aging is defined as the process of developing and maintaining functional ability to ensure wellbeing in later life; IC and FA are proposed to describe and estimate the state of healthy aging [1, 2]. IC, defined as a composite measure for all physical and mental capacities of an individual, is conceptualized as a dynamic construct to serve as the potential functional reserve in the aging process [3]. Interacting with environmental factors, such as facilitators or barriers, IC may be a proxy to estimate the FA of an individual and the status of healthy aging [1]. Notably, as conceptualized, IC declines occur earlier before clinical manifestations of FA declines, so it is critical to capture IC declines in the life course for healthy longevity [4].
In the WHO Integrated Care for Older People, IC consists of five elements, i.e., locomotion, sensory, vitality, psychological, and cognition, which represent the physiological competence of individuals to support their FA [1]. Although there are strong theoretical and consensual bases to support this multidomain conceptualization of IC [5–8], most empirical studies have focused on the associations between IC and disability [4, 9], falls [10], quality of life [11], and mortality [12–14]. Unique diet, lifestyle, culture and policy may interact with IC, and contribute to FA, particularly in Asia [8]. In this context, functioning was used as a target to build an IC model instead of exploring the role of FA on health outcomes. Nevertheless, questions remained as there might be a gap between IC and FA against mortality due to their different conceptualization regarding a person’s competency and ability. To the best of our knowledge, no study has examined the impacts of these two distinct constructs on mortality in parallel to distinguish their potential impacts on clinical outcomes. A systemic review exploring possible biomarkers related to aging showed that lipids, glucose, inflammatory biomarkers, dehydroepiandrosterone sulfate (DHEA-S), growth hormone and insulin-like growth Factor 1 (IGF-1) were candidate biomarkers [15, 16]. Other studies indicated that elevated systemic inflammatory biomarkers, such as high-sensitivity C-reactive protein (hsCRP) and homocysteine, were associated with slow gait speed, weak muscle strength, and low IC [17, 18]. However, conflicting results have been reported regarding the associations between specific biomarkers and IC or FA. Although the underlying biological mechanisms of healthy aging remain unclear, identifying the biological features of healthy aging helps to capture the heterogenicity of aging over time [19]. Moreover, assessing the biological features of IC enables researchers and clinicians to understand potential pathophysiological mechanism, and to design personalized intervention programs to promote healthy aging.
To address the abovementioned knowledge gaps, the study aimed 1) to examine the association between declines in IC or FA and 10-year mortality risk and 2) to further explore the biological features of IC based on its longitudinal changes.
Results
Demographic characteristics of participants stratified by IC and FA status
The demographic characteristics of the participants (mean age 63.9 ± 9.3 years, 47.5% men) are shown in Supplementary Table 1. Compared to the high IC group (n = 1190), those with low IC (n = 649) were older (70.0 ± 8.8 vs. 60.6 ± 7.7 years old), mainly women (63.0% vs. 46.8%), had fewer years of education (2.5 ± 3.4 vs. 8.2 ± 4.5 years), were less likely to consume alcohol (23.0% vs. 38.5%), and had a higher disease burden (CCI 1.6 ± 1.4 vs. 0.7 ± 1.1). Compared to the high FA group (n = 1769), those with low FA (n = 70) were older (76.0 ± 9.9 vs. 63.5 ± 8.9 years old), had fewer years of education (2.9 ± 4.1 vs. 6.4 ± 4.9 years), were less likely to consume alcohol (15.7% vs. 33.7%) and had a higher disease burden (CCI 2.3 ± 1.6 vs. 1.0 ± 1.2). Comparisons of subcategories of IC were shown in Supplementary Table 2. The distributions of IC and FA were left-skewed (Supplementary Figure 1). Overall, IC declined progressively from 86.0 ± 5.1 to 80.5 ± 5.1 points, and FA declined from 99.9 ± 1.3 to 99.3 ± 4.1 points in a mean follow-up period of 6.5 ± 0.8 years.
Survival analysis for IC and FA status
There were 238 deaths in a mean follow-up period of 8.5 ± 1.5 years. Kaplan–Meier analysis showed that low FA (Figure 1A, log-rank test, p < 0.001) and low IC (Figure 1B, log-rank test, p < 0.001) were significantly associated with mortality. A one-point (percent) increase in IC score decreased the odds of mortality by 5% (HR 0.95, 95% CI 0.93–0.97, p < 0.001), and those with low IC had a greater risk of mortality (HR 1.94, 95% CI 1.39–2.70, p < 0.001). The associations between FA and mortality were attenuated after adjusting for relevant confounders (Table 1).
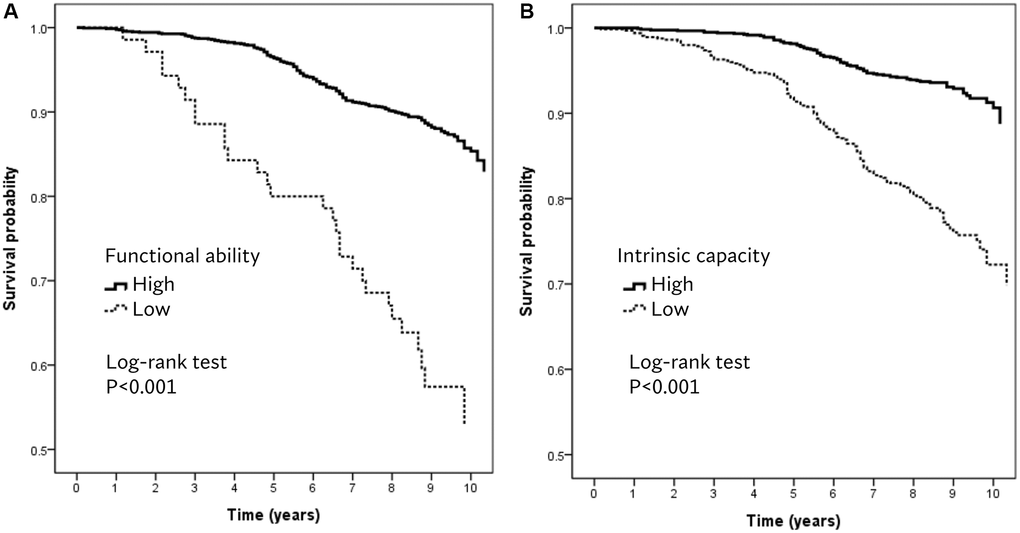
Figure 1. Kaplan–Meier survival plots for (A) functional ability and (B) intrinsic capacity.
Table 1. Relationship between Intrinsic capacity, functional ability and mortality.
| Model Ia | p | Model IIb | p | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HR (95%CI) | HR (95%CI) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Intrinsic capacityc | 0.94 (0.92–0.96) | <0.001 | 0.95 (0.93–0.97) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Functional abilityc | 0.97 (0.94–1.00) | 0.025 | 0.97 (0.94–1.00) | 0.089 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Low Intrinsic capacityd | 2.19 (1.58–3.04) | <0.001 | 1.94 (1.39–2.70) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Low Functional abilityd | 1.53 (1.00–2.33) | 0.049 | 1.29 (0.84–1.97) | 0.248 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Components of intrinsic capacityc | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Locomotion | 0.97 (0.96–0.98) | <0.001 | 0.97 (0.96–0.98) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cognition | 0.99 (0.98–1.00) | 0.010 | 0.99 (0.98–1.00) | 0.018 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Psychology | 0.99 (0.97–1.00) | 0.022 | 0.99 (0.98–1.00) | 0.139 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Vitality | 0.97 (0.95–0.98) | <0.001 | 0.98 (0.96–0.99) | 0.006 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sensory | 0.98 (0.95–1.00) | 0.047 | 0.98 (0.96–1.01) | 0.182 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| aCox proportional hazards model adjusted for age, gender, and education years. bCox proportional hazards model adjusted for age, gender, education years, smoke, drink and charlson comorbidity index. cnumerical variables. dcategorical variables. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Biomarkers associated with low IC
The results of adjusted logistic regression analysis for the associations between biomarkers and low IC are shown in Figure 2. Participants with higher levels of fasting glucose had higher odds of having low IC (OR 1.40, 95% CI 1.09–1.81, p = 0.010), but those with higher levels of cholesterol (OR 0.60, 95% CI 0.47–0.79, p < 0.001) and LDL-C (OR 0.72, 95% CI 0.55–0.94, p = 0.015) were less likely to have low IC. Those in the lowest tertile of DHEA-S had higher odds for low IC (OR 3.33, 95% CI 1.32–8.41, p = 0.011). Of the inflammatory biomarkers, higher levels of hsCRP (OR 1.45, 95% CI 1.05–2.00, p = 0.023) and neutrophil-to-lymphocyte ratio (NLR, highest tertile OR 1.77, 95% CI 1.29–2.43, p < 0.001) were significantly associated with low IC (Figure 2).
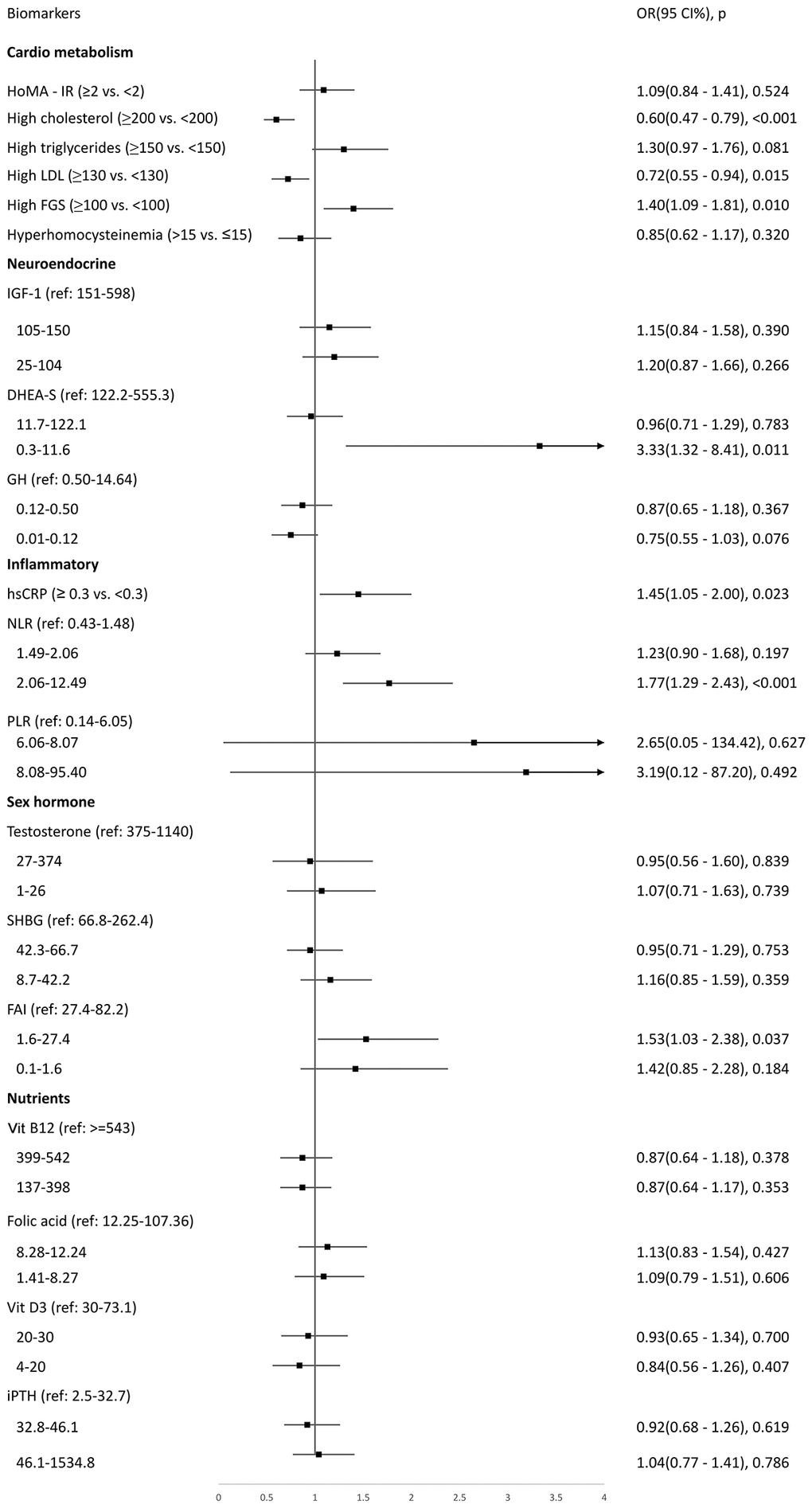
Figure 2. Logistic regression to explore biomarkers associated with low intrinsic capacity at baseline.
Biomarkers associated with longitudinal preservation or deterioration of IC
Adjusted logistic regressions showed that predictors for rapid deterioration of IC were lower levels of dehydroepiandrosterone sulfate (DHEA-S, middle tertile OR 1.84, 95% CI 1.15–2.95, p = 0.012; lowest tertile OR 2.25, 95% CI 1.34–3.77, p = 0.002) and vitamin D deficiency (OR 1.82, 95% CI 1.02–3.27, p = 0.044) (Figure 3). Inverse probability weighting regressions were used to reduce potential selection bias from excluded participants, and findings were similar (OR 1.41, 95% CI 1.09–1.83, p = 0.008 for lowest tertile of DHEA-S; OR 1.06, 95% CI 1.01–1.12, p = 0.030 for vitamin D deficiency). Supplementary Figure 2 shows adjusted logistic regression to explore potential biomarkers for maintained or improved IC. All biomarkers showed insignificant associations.
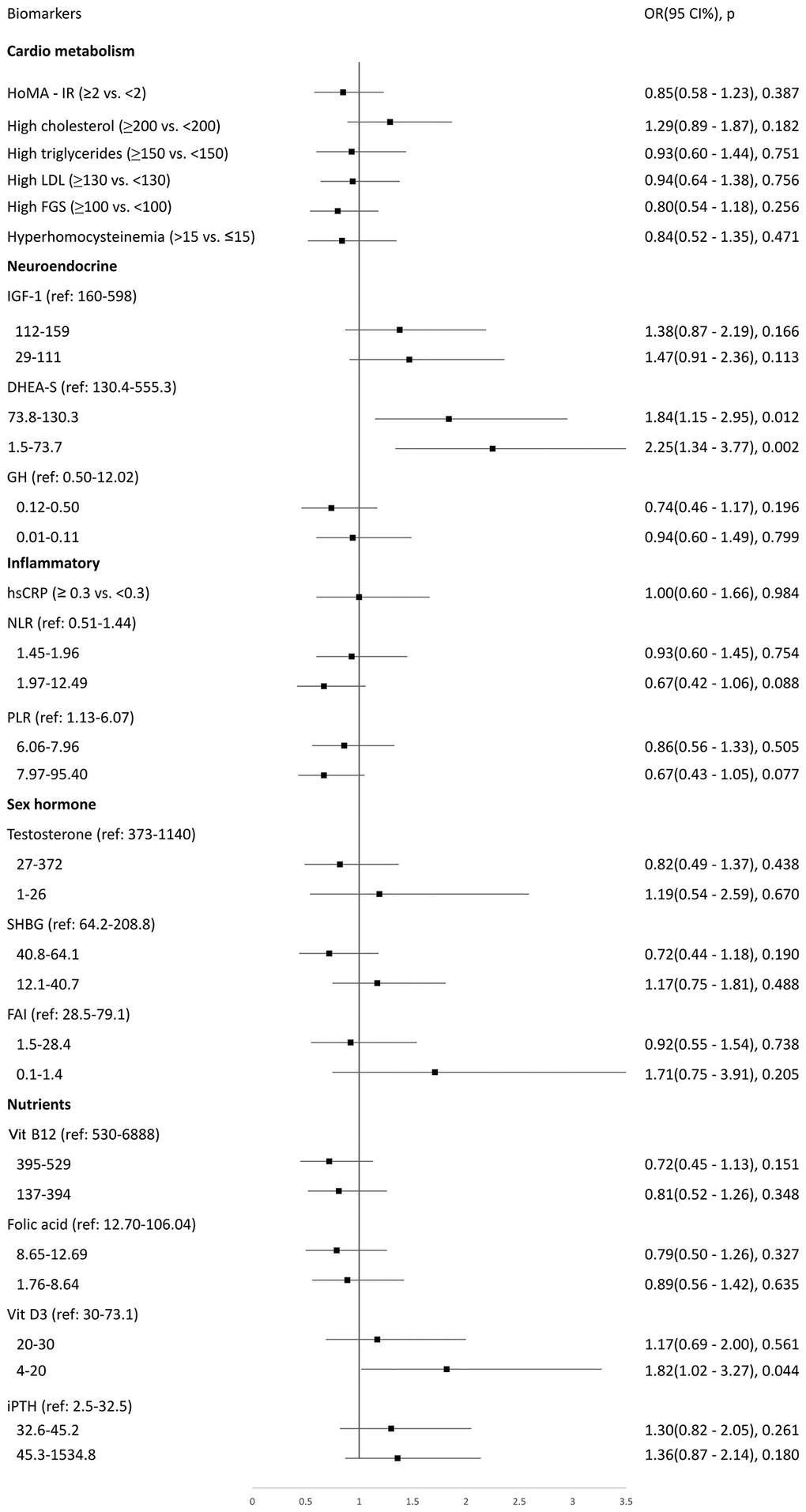
Figure 3. Logistic regression to explore biomarkers associated with rapidly deteriorated intrinsic capacity at a 7-year follow-up period.
Discussion
The WHO conceptualized the scheme of healthy aging and addressed the importance of IC and FA in the life course that shifted health care services from disease-centric models to function-centric ones, as well as the focus on positive attributes of health. This study used a longitudinal cohort to compare the 10-year mortality risk between IC and FA. Unlike IC, the mortality risk of FA attenuated after adjustment for smoking, drinking, and disease burden, which supports the idea that environmental factors modify health outcomes in older age. However, the constant mortality risk of IC clearly reflects its adverse impacts on health outcomes in the aging process. In addition, this study found that hyperglycemia, proinflammatory status of hsCRP and NLR, and low serum levels of DHEA-S were associated with low IC. In particular, lower serum levels of DHEA-S and vitamin D deficiency were associated with rapid deterioration of IC. These findings suggest the potential roles of inflammation and endocrine and musculoskeletal systems in healthy aging through their influences on IC.
Based on the conceptual framework of healthy aging, the impacts of environmental or modifiable factors, including interventions on diseases and healthy behaviors, were emphasized to constitute FA instead of focusing on age-related declines in IC. The results of the current study completely support the conceptual framework of healthy aging. In contrast to frailty characterized by increased vulnerability to stressors from disrupted homeostasis of multiple physical systems, IC aimed to capture the nature of physiological reserves and residual capacities in aging, which is particularly suitable for longitudinal assessments [3]. Although frailty and IC are two interrelated but distinct constructs, both of them focus on function-centric health services with comprehensive assessment and are provided in a multidisciplinary fashion. Our previous study indicated that multidomain interventions combined with integrated primary care significantly improve physical function, cognitive performance, and quality of life among older adults with multimorbidity, which supported the core concepts of healthy aging [20, 21]. Nevertheless, the importance of FA should be addressed as well because of the significant modifying effects of environmental factors in older age.
By examining associations between IC and biomarkers, this study disclosed the multidimensional nature of biological mechanisms in the process of healthy aging. In this study, low IC status was associated with glucose metabolism, chronic inflammation, and neuroendocrine diseases. Previous studies have suggested associations between hsCRP, NLR, poor locomotion and vitality [17, 22]. This study further extended these associations from two specific domains (locomotion and vitality) to the whole composite IC, which strengthens the roles of inflammation in the aging process. On the other hand, cholesterol and LDL-C are well-known cardiovascular risk factors, but the associations are attenuated in late life [23]. A systemic review concluded that lower serum cholesterol levels were associated with greater mortality risk in older adults [24], which is in line with our study that lower cholesterol and low-density lipoprotein cholesterol (LDL-C) increased the risk of poor IC. These data justified the debates on statin treatment in older adults that routine statin use was not encouraged for those aged 75 years old and older. In addition, deprescription of statin therapy in those who have developed frailty is usually recommended [25]. Hyperglycemia is also a factor contributing to incident frailty and disability [26], which explained its association with low IC in our study.
DHEA-S is being widely used in the general public to prevent age-related disease. However, oral supplementation with DHEA-S failed to demonstrate a positive impact on the improvement of cognitive performance in healthy older people [27], whereas it preserved bone health [28]. Vitamin D is crucial to musculoskeletal health [29], but a systemic review of 81 randomized controlled trials reported disappointing results on falls, fracture preventions, or meaningful effects on bone mineral density [30]. Previous studies have suggested potential associations between DHEA-S and inflammatory [31] as well as the benefits for DHEA-S supplement administration on libido improvement and bone turnover biomarkers improvement [32]. In this study, low DHEA-S and vitamin D deficiency were both associated with rapid deterioration of IC but were not associated with maintained or improved IC in a follow-up period of up to 7 years. These findings highlight the importance of musculoskeletal health in preventing IC decline but also reflect the fact that neither monodimensional factor could counter the effects of age-related IC decline stemming from deficits in multiple physiological systems. Further interventional studies to explore the effects of oral supplementation with DHEA-S and vitamin D on IC are needed to further guide the optimal use of these supplements.
Despite all efforts made in this study, there are some limitations to note. First, environmental factors such as access to health facilities or means of transportation, etc., could not be adjusted for FA due to the limitation of data availability. Nevertheless, FA measured by the Functional Autonomy Measurement System (SMAF) considered available resources and environmental factors to compensate for functional impairment. Second, information regarding the cause of death could not be available in this study, which precludes cause-specific analysis. Further biological marker analysis could provide information on IC decline and potential mechanisms. Third, those excluded from the second part of this study might introduce selection bias. However, we have conducted an inverse probability weighting analysis to adjust for such potential selection bias and yields similar results. Fourth, as the participants from ILAS were community-dwelling adults, they are relative healthy. That is why the cutoff value of functional ability was quite high. Last, subdomains of vision and hearing were obtained from self-report questionnaires, which may probably underestimate the prevalence of vision and hearing impairment.
In conclusion, IC and FA predicted 10-year mortality, whereas the association for FA diminished after adjusting for smoking, drinking, and disease burden. Chronic inflammatory markers of hsCRP and NLR, hyperglycemia, and low DHEA-S were associated with low IC status. DHEA-S and vitamin D deficiency aggravated IC deterioration, which implies the importance of musculoskeletal health in healthy aging.
Materials and Methods
Participants and study design
Data from the first and third waves of the I-Lan Longitudinal Aging Study (ILAS) were collected for the baseline cross-sectional (wave 1) and longitudinal cohort analyses (wave 1 and 3). ILAS was a prospective cohort study focused on interactions between sarcopenia, frailty, and cognitive functioning throughout the aging process. The details of the study design, participant recruitment, and data collection of ILAS have been reported previously [33]. Briefly, ILAS recruited community-dwelling adults aged ≥50 years without severe disability, dementia or communication difficulties, limited life expectancy due to major illness or being institutionalized. The first part of this study was a survival analysis of a 10-year follow-up, and the second part was a 7-year longitudinal study to capture biological features based on the status of IC changes (maintained, improved and rapidly deteriorated). In the first part of the study, data from 1,839 participants from the ILAS wave 1 survey in 2011 were used to evaluate the 10-year mortality risk of IC and FA (Figure 4A). In the second part of the study, 1019 participants who recruited in ILAS wave 1 and completed the ILAS wave 3 survey since 2018 (216 participants died before wave 3 survey and 604 participants declined to participate in wave 3 survey) were enrolled (Figure 4B). Compared to those enrolled in the second part of this study, those excluded were older (67.5 ± 10.0 vs. 61.1 ± 7.5 years, p < 0.001), less educated (4.5 ± 4.5 vs. 7.6 ± 4.8 years, p < 0.001), higher disease burden of Charlson comorbidity index (1.4 ± 1.4 vs. 0.7 ± 1.1, p < 0.001), but similar sex proportions.
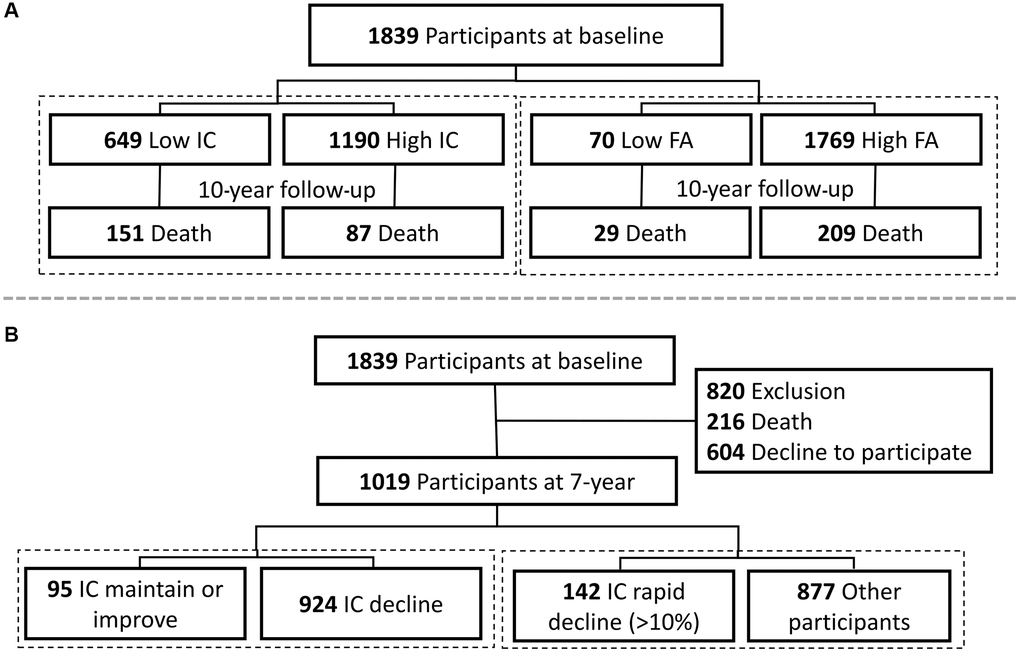
Figure 4. Study flowcharts of (A) survival analysis and (B) longitudinal study for biological features of intrinsic capacity. Abbreviations: IC: denotes intrinsic capacity; FA: denotes functional ability.
This study was designed and conducted in accordance with the principles of the 1964 Declaration of Helsinki and later amendments. The institutional review board of National Yang Ming University (YM103008) and Taipei Veterans General Hospital (2018-05-003B) approved the study protocol, and written informed consent was obtained from each individual before inclusion. The observational design and reporting format followed the STROBE guidelines [34].
Intrinsic capacity (IC)
The five IC elements, i.e., cognition, locomotion, vitality, psychology, and sensory, were selected based on the principle proposed by WHO ICOPE [5].
Cognition was evaluated by the Chinese version of the Mini-Mental State Examination (MMSE), with scores ranging from 0 to 30, and a higher score on the MMSE indicated better cognition [35]. Locomotion was assessed by a timed 6-meter gait speed at the usual pace in meters/second based on a consensus recommended by the Asian Working Group for Sarcopenia (AWGS) [36]. Vitality was assessed by the Mini Nutritional Assessment (MNA), with scores ranging from 0 to 30, and a higher score indicated better nutrition [37]. Psychology was measured with the Center for Epidemiologic Studies—Depression scale (CESD) with scores ranging from 0 to 60, and a higher score denoted a greater level of depressive symptoms [38]. Psychological scores were obtained by multiplying the original CESD scores by −1 in this study. The reason why we would like to adopt this approach is mainly because IC was constructed based on positive capacity for physical and mental reserves. However, we used CESD to measure mental reserve, but a higher CESD score indicates the higher level of depression. We thus rescale our CESD by multiplying the original CESD by −1. Sensory impairment was assessed through a self-reported score comprising visual and hearing impairments based on a previous study [12]. A sensory score was built from two questions of vision and hearing: score from 0 (independent), −1 (need supervision), −2 (need help), and −3 (dependent), yielding a total score ranging from −6 to 0. We further used the percent of maximum possible method - calculated by [100 × (observed − minimum)/(maximum − minimum)]- to rescale all individual variables with a ranging from 0 (minimum possible) to 100 (maximum possible) [39] to make the composite values of IC comparable longitudinally [40]. Psychological scores were obtained by 100 minus the rescaled CESD scores. For each study participant, we calculated their IC scores as the mean of the sum of subscores obtained in each of five elements.
Functional ability (FA)
Functional ability (FA) was measured by SMAF through the percent of maximum possible method to rescale the original score with the range from 0 to 100. SMAF is a 29-item four-level measurement scale based on WHO’s classification of impairments, disabilities, and handicap with consideration of available resources and environmental factors to compensate for functional impairment [41]. The SMAF assessed functional ability in 5 areas: activities of daily living (ADL) [7 items], mobility [6 items], communication [3 items], mental functions [5 items] and instrumental activities of daily living (IADL) [8 items]. For each item, the disability was scored on a 4-level scale: 0 (independent), −0.5 (with difficulty), −1 (needs supervision), −2 (needs help), −3 (dependent). Resources available to compensate for the disability were also evaluated, and a handicap score was deducted. The stability of the resources was also assessed. A disability score (on −87) can be calculated, together with subscores for each dimension. Given the involvement of environmental factors, the SMAF evaluated a person’s FA rather than their IC alone [42].
Acquisition of mortality
All participants in ILAS received a phone call by research nurses every 3 months to check their health conditions and survival status. These survival data were calculated from the index interview day until the last phone contact before 31 March 2022.
Other variables
Potential confounding variables were identified from previous literature, which included age, sex, education years, smoking and alcohol consumption in the past six months (yes versus no). The burden of disease was assessed by Charlson’s comorbidity index (CCI) [43]. Venous blood samples were collected from all ILAS participants after a 10-hour overnight fast. Biomarkers related to cardiometabolic health, hormones and biochemistry were tested for all participants, including fasting glucose, total cholesterol, triglycerides, LDL, insulin level, and homeostasis model assessment-insulin resistance (HOMA-IR)-insulin resistance. Inflammatory biomarkers (NLR, platelet-to-lymphocyte ratio, homocysteine, and hsCRP), age-related hormones (growth hormone, IGF-1, DHEA-S, testosterone, sex hormone binding globulin (SHBG), intact parathyroid hormone, and micronutrients (vitamin B12, folic acid and vitamin D, measured by 25-OH vitamin D) were also tested for all participants. Details of the machine, limit of detection, and intra- and inter-assay coefficients of variation for serum biomarkers are shown in the appendix (Supplementary Table 3). The free androgen index (FAI) was calculated from testosterone and SHBG [44].
Statistical analysis
Numerical variables were expressed as the mean ± standard deviation, and categorical variables were expressed as numbers with percentages. Comparisons of baseline characteristics between different IC or FA groups were performed by Student’s t test for continuous variables and chi-square tests or Fisher’s exact test for categorical variables. In the first part of this study, IC (≥82.7 vs. <82.7) and FA (≥98.9 vs. <98.9) at baseline were assigned to high IC/FA or low IC/FA groups according to the abovementioned cutoff values. Receiver operating curve (ROC) analysis and Youden’s index maxima were used to determine the optimal cutoff values that achieved optimal discrimination (Supplementary Table 4). Subcategories of IC as mobility (≥39.8 vs. <39.8), cognition (≥86.7 vs. <86.7), psychological (≥96.7 vs. <96.7), vitality (≥88.3 vs. <88.3), and sensory (≥83.3 vs. <83.3) were categorized into high and low for comparisons of baseline characteristics. Multivariable logistic regression was used to assess the associations between identified biomarkers and low IC. Cox proportional hazard regression was used to evaluate the association between IC, FA, and mortality. Schoenfeld residuals were used to test proportionality assumptions in Cox proportional hazard models. Kaplan–Meier survival plots and log-rank tests were applied to investigate the association between IC, FA, and mortality. In the second part of this study, multivariable logistic regressions were used to identify biomarkers associated with maintained or improved IC or rapidly deteriorated IC. The rapidly deteriorated IC group was defined as those with a decline in IC score >10% (equal to the 1 standard deviation (SD) lower than the group mean of decline in IC score) between wave 1 and 3 survey.
Biomarkers were dichotomized based on definitions used in previous literature: HOMA-IR (≥2 vs. <2) [45], high cholesterol (≥200 or taking lipid lower drugs vs. <200 mg/dL) [46], high triglycerides (≥150 or taking lipid lower drugs vs. <150 mg/dL) [46], high LDL-C (≥130 or taking lipid lower drugs vs. <130 mg/dL) [46], high fasting blood glucose (≥100 or taking oral anti-diabetic drugs vs. <100 mg/dL) [46], hyperhomocysteinemia (>15 vs. ≤15 μmol/L), and hsCRP (≥0.3 vs. <0.3 mg/dL) [47]. All other biomarkers, including the FAI, were tertilized [44]. Vitamin D insufficiency and deficiency were defined by 25-OH vitamin D <20 and <10 ng/mL, respectively [29].
A p value from two-sided tests <0.05 and 95% CIs not spanning the null hypothesis values were considered to be statistically significant. All analyses were performed using the SAS statistical package version 9.4 (SAS Institute, Inc., Cary, NC, USA).
Author Contributions
Conceptualization, W-J.L., L-N.P., M-H.L., C-H. L., and L-K.C.; Methodology, W-J.L.; Validation, L-N.P., M-H.L., C-H. L., and L-K.C.; Formal analysis, W-J.L.; Investigation, W-J.L.; Resources, L-K. C.; Data curation, W-J.L.; Writing – original draft preparation, W-J.L.; Writing –review and editing, W-J.L., L-N.P., M-H.L., C-H. L.,F-Y.H., and L-K.C.; Visualization, W-J.L.; Supervision, L-K.C.; Project administration, W-J.L.
Acknowledgments
We express our gratitude for the support from the Center for Geriatrics and Gerontology of Taipei Veterans General Hospital and Department of Family Medicine of Taipei Veterans General Hospital Yuanshan Branch.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
Ethical Statement and Consent
This study was designed and conducted in accordance with the principles of the 1964 Declaration of Helsinki and later amendments. The institutional review board of National Yang Ming University (YM103008) and Taipei Veterans General Hospital (2018-05-003B) approved the study protocol, and written informed consent was obtained from each individual before inclusion. The observational design and reporting format followed the STROBE guidelines [34].
Funding
The whole study was funded by the National Science and Technology Council (NSTC-108-2634-F-010-001, NSTC 111-2314-B-A49-048-MY2, NSTC 111-2321-B-A49-006, and NSTC 111-2321-B-A49-008), National Health Research Institute (NHRI-11A1-CG-CO-01-2225-1), and Taipei Veterans General Hospital (110VACS-001).
References
- 1. World Health Organization. World report on ageing and health: World Health Organization. 2015. https://apps.who.int/iris/handle/10665/186463.
- 2. Beard JR, Officer A, de Carvalho IA, Sadana R, Pot AM, Michel JP, Lloyd-Sherlock P, Epping-Jordan JE, Peeters GME, Mahanani WR, Thiyagarajan JA, Chatterji S. The World report on ageing and health: a policy framework for healthy ageing. Lancet. 2016; 387:2145–54. https://doi.org/10.1016/S0140-6736(15)00516-4 [PubMed]
- 3. Belloni G, Cesari M. Frailty and Intrinsic Capacity: Two Distinct but Related Constructs. Front Med (Lausanne). 2019; 6:133. https://doi.org/10.3389/fmed.2019.00133 [PubMed]
- 4. Stolz E, Mayerl H, Freidl W, Roller-Wirnsberger R, Gill TM. Intrinsic Capacity Predicts Negative Health Outcomes in Older Adults. J Gerontol A Biol Sci Med Sci. 2022; 77:101–5. https://doi.org/10.1093/gerona/glab279 [PubMed]
- 5. Cesari M, Araujo de Carvalho I, Amuthavalli Thiyagarajan J, Cooper C, Martin FC, Reginster JY, Vellas B, Beard JR. Evidence for the Domains Supporting the Construct of Intrinsic Capacity. J Gerontol A Biol Sci Med Sci. 2018; 73:1653–60. https://doi.org/10.1093/gerona/gly011 [PubMed]
- 6. George PP, Lun P, Ong SP, Lim WS. A Rapid Review of the Measurement of Intrinsic Capacity in Older Adults. J Nutr Health Aging. 2021; 25:774–82. https://doi.org/10.1007/s12603-021-1622-6 [PubMed]
- 7. Gonzalez-Bautista E, Andrieu S, Gutiérrez-Robledo LM, García-Chanes RE, de Souto Barreto P. In the quest of a Standard Index of Intrinsic Capacity. A Critical Literature Review. J Nutr Health Aging. 2020; 24:959–65. https://doi.org/10.1007/s12603-020-1394-4 [PubMed]
- 8. Fang EF, Xie C, Schenkel JA, Wu C, Long Q, Cui H, Aman Y, Frank J, Liao J, Zou H, Wang NY, Wu J, Liu X, et al. A research agenda for ageing in China in the 21st century (2nd edition): Focusing on basic and translational research, long-term care, policy and social networks. Ageing Res Rev. 2020; 64:101174. https://doi.org/10.1016/j.arr.2020.101174 [PubMed]
- 9. Yu R, Leung G, Leung J, Cheng C, Kong S, Tam LY, Woo J. Prevalence and Distribution of Intrinsic Capacity and Its Associations with Health Outcomes in Older People: The Jockey Club Community eHealth Care Project in Hong Kong. J Frailty Aging. 2022; 11:302–8. https://doi.org/10.14283/jfa.2022.19 [PubMed]
- 10. Liu S, Yu X, Wang X, Li J, Jiang S, Kang L, Liu X. Intrinsic Capacity predicts adverse outcomes using Integrated Care for Older People screening tool in a senior community in Beijing. Arch Gerontol Geriatr. 2021; 94:104358. https://doi.org/10.1016/j.archger.2021.104358 [PubMed]
- 11. Salinas-Rodríguez A, González-Bautista E, Rivera-Almaraz A, Manrique-Espinoza B. Longitudinal trajectories of intrinsic capacity and their association with quality of life and disability. Maturitas. 2022; 161:49–54. https://doi.org/10.1016/j.maturitas.2022.02.005 [PubMed]
- 12. Sánchez-Sánchez JL, Rolland Y, Cesari M, de Souto Barreto P. Associations Between Intrinsic Capacity and Adverse Events Among Nursing Home Residents: The INCUR Study. J Am Med Dir Assoc. 2022; 23:872–76.e4. https://doi.org/10.1016/j.jamda.2021.08.035 [PubMed]
- 13. Meng LC, Huang ST, Peng LN, Chen LK, Hsiao FY. Biological Features of the Outcome-Based Intrinsic Capacity Composite Scores From a Population-Based Cohort Study: Pas de Deux of Biological and Functional Aging. Front Med (Lausanne). 2022; 9:851882. https://doi.org/10.3389/fmed.2022.851882 [PubMed]
- 14. Yu R, Lai ETC, Leung G, Ho SC, Woo J. Intrinsic capacity and 10-year mortality: Findings from a cohort of older people. Exp Gerontol. 2022; 167:111926. https://doi.org/10.1016/j.exger.2022.111926 [PubMed]
- 15. Engelfriet PM, Jansen EH, Picavet HS, Dollé ME. Biochemical markers of aging for longitudinal studies in humans. Epidemiol Rev. 2013; 35:132–51. https://doi.org/10.1093/epirev/mxs011 [PubMed]
- 16. Belsky DW, Caspi A, Houts R, Cohen HJ, Corcoran DL, Danese A, Harrington H, Israel S, Levine ME, Schaefer JD, Sugden K, Williams B, Yashin AI, et al. Quantification of biological aging in young adults. Proc Natl Acad Sci U S A. 2015; 112:E4104–10. https://doi.org/10.1073/pnas.1506264112 [PubMed]
- 17. Lee WJ, Peng LN, Loh CH, Chen LK. Sex-different associations between serum homocysteine, high-sensitivity C-reactive protein and sarcopenia: Results from I-Lan Longitudinal Aging Study. Exp Gerontol. 2020; 132:110832. https://doi.org/10.1016/j.exger.2020.110832 [PubMed]
- 18. Giudici KV, de Souto Barreto P, Guerville F, Beard J, Araujo de Carvalho I, Andrieu S, Rolland Y, Vellas B, and MAPT/DSA group. Associations of C-reactive protein and homocysteine concentrations with the impairment of intrinsic capacity domains over a 5-year follow-up among community-dwelling older adults at risk of cognitive decline (MAPT Study). Exp Gerontol. 2019; 127:110716. https://doi.org/10.1016/j.exger.2019.110716 [PubMed]
- 19. Beard JR, Jotheeswaran AT, Cesari M, Araujo de Carvalho I. The structure and predictive value of intrinsic capacity in a longitudinal study of ageing. BMJ Open. 2019; 9:e026119. https://doi.org/10.1136/bmjopen-2018-026119 [PubMed]
- 20. Lee WJ, Peng LN, Lin CH, Chen RC, Lin SZ, Loh CH, Kao SL, Hung TS, Chang CY, Huang CF, Tang TC, Huang ST, Wen YW, et al, and Taiwan Integrated Geriatric Care Study Group. Effects of incorporating multidomain interventions into integrated primary care on quality of life: a randomised controlled trial. Lancet Healthy Longev. 2021; 2:e712–23. https://doi.org/10.1016/S2666-7568(21)00248-8 [PubMed]
- 21. Lee WJ, Peng LN, Lin MH, Lin CH, Chen LK. Clinical Efficacy of Multidomain Interventions among Multimorbid Older People Stratified by the Status of Physio-Cognitive Declines: A Secondary Analysis from the Randomized Controlled Trial for Healthy Aging. J Nutr Health Aging. 2022; 26:909–17. https://doi.org/10.1007/s12603-022-1843-3 [PubMed]
- 22. Öztürk ZA, Kul S, Türkbeyler İH, Sayıner ZA, Abiyev A. Is increased neutrophil lymphocyte ratio remarking the inflammation in sarcopenia? Exp Gerontol. 2018; 110:223–9. https://doi.org/10.1016/j.exger.2018.06.013 [PubMed]
- 23. Schatz IJ, Masaki K, Yano K, Chen R, Rodriguez BL, Curb JD. Cholesterol and all-cause mortality in elderly people from the Honolulu Heart Program: a cohort study. Lancet. 2001; 358:351–5. https://doi.org/10.1016/S0140-6736(01)05553-2 [PubMed]
- 24. Ravnskov U, Diamond DM, Hama R, Hamazaki T, Hammarskjöld B, Hynes N, Kendrick M, Langsjoen PH, Malhotra A, Mascitelli L, McCully KS, Ogushi Y, Okuyama H, et al. Lack of an association or an inverse association between low-density-lipoprotein cholesterol and mortality in the elderly: a systematic review. BMJ Open. 2016; 6:e010401. https://doi.org/10.1136/bmjopen-2015-010401 [PubMed]
- 25. Grundy SM, Stone NJ, Bailey AL, Beam C, Birtcher KK, Blumenthal RS, Braun LT, de Ferranti S, Faiella-Tommasino J, Forman DE, Goldberg R, Heidenreich PA, Hlatky MA, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2019; 139:e1082–143. https://doi.org/10.1161/CIR.0000000000000625 [PubMed]
- 26. Kalyani RR, Tian J, Xue QL, Walston J, Cappola AR, Fried LP, Brancati FL, Blaum CS. Hyperglycemia and incidence of frailty and lower extremity mobility limitations in older women. J Am Geriatr Soc. 2012; 60:1701–7. https://doi.org/10.1111/j.1532-5415.2012.04099.x [PubMed]
- 27. Grimley Evans J, Malouf R, Huppert F, van Niekerk JK. Dehydroepiandrosterone (DHEA) supplementation for cognitive function in healthy elderly people. Cochrane Database Syst Rev. 2006; 2006:CD006221. https://doi.org/10.1002/14651858.CD006221 [PubMed]
- 28. von Mühlen D, Laughlin GA, Kritz-Silverstein D, Bergstrom J, Bettencourt R. Effect of dehydroepiandrosterone supplementation on bone mineral density, bone markers, and body composition in older adults: the DAWN trial. Osteoporos Int. 2008; 19:699–707. https://doi.org/10.1007/s00198-007-0520-z [PubMed]
- 29. Chen CH, Liu LK, Chen MJ, Lee WJ, Lin MH, Peng LN, Chen LK. Associations between vitamin D deficiency, musculoskeletal health, and cardiometabolic risk among community-living people in Taiwan: Age and sex-specific relationship. Medicine (Baltimore). 2018; 97:e13886. https://doi.org/10.1097/MD.0000000000013886 [PubMed]
- 30. Bolland MJ, Grey A, Avenell A. Effects of vitamin D supplementation on musculoskeletal health: a systematic review, meta-analysis, and trial sequential analysis. Lancet Diabetes Endocrinol. 2018; 6:847–58. https://doi.org/10.1016/S2213-8587(18)30265-1 [PubMed]
- 31. Young DG, Skibinski G, Mason JI, James K. The influence of age and gender on serum dehydroepiandrosterone sulphate (DHEA-S), IL-6, IL-6 soluble receptor (IL-6 sR) and transforming growth factor beta 1 (TGF-beta1) levels in normal healthy blood donors. Clin Exp Immunol. 1999; 117:476–81. https://doi.org/10.1046/j.1365-2249.1999.01003.x [PubMed]
- 32. Baulieu EE, Thomas G, Legrain S, Lahlou N, Roger M, Debuire B, Faucounau V, Girard L, Hervy MP, Latour F, Leaud MC, Mokrane A, Pitti-Ferrandi H, et al. Dehydroepiandrosterone (DHEA), DHEA sulfate, and aging: contribution of the DHEAge Study to a sociobiomedical issue. Proc Natl Acad Sci U S A. 2000; 97:4279–84. https://doi.org/10.1073/pnas.97.8.4279 [PubMed]
- 33. Liu LK, Lee WJ, Chen LY, Hwang AC, Lin MH, Peng LN, Chen LK. Sarcopenia, and its association with cardiometabolic and functional characteristics in Taiwan: results from I-Lan Longitudinal Aging Study. Geriatr Gerontol Int. 2014 (Suppl 1); 14:36–45. https://doi.org/10.1111/ggi.12208 [PubMed]
- 34. von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP, and STROBE Initiative. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. Prev Med. 2007; 45:247–51. https://doi.org/10.1016/j.ypmed.2007.08.012 [PubMed]
- 35. Guo NW, Liu HC, Wong PF, Liao KK, Yan SH, Lin KP, Chang CY, Hsu TC. Chinese Version and Norms of the Mini-Mental State Examination. J Phys Med Rehabil. 1988; 52–9.
- 36. Chen LK, Woo J, Assantachai P, Auyeung TW, Chou MY, Iijima K, Jang HC, Kang L, Kim M, Kim S, Kojima T, Kuzuya M, Lee JSW, et al. Asian Working Group for Sarcopenia: 2019 Consensus Update on Sarcopenia Diagnosis and Treatment. J Am Med Dir Assoc. 2020; 21:300–7.e2. https://doi.org/10.1016/j.jamda.2019.12.012 [PubMed]
- 37. Vellas B, Villars H, Abellan G, Soto ME, Rolland Y, Guigoz Y, Morley JE, Chumlea W, Salva A, Rubenstein LZ, Garry P. Overview of the MNA--Its history and challenges. J Nutr Health Aging. 2006; 10:456–63. [PubMed]
- 38. Lewinsohn PM, Seeley JR, Roberts RE, Allen NB. Center for Epidemiologic Studies Depression Scale (CES-D) as a screening instrument for depression among community-residing older adults. Psychol Aging. 1997; 12:277–87. https://doi.org/10.1037/0882-7974.12.2.277 [PubMed]
- 39. Cohen P, Cohen J, Aiken LS, West SG. The Problem of Units and the Circumstance for POMP. Multivariate Behav Res. 1999; 34:315–46. https://doi.org/10.1207/S15327906MBR3403_2
- 40. Moeller J. A word on standardization in longitudinal studies: don't. Front Psychol. 2015; 6:1389. https://doi.org/10.3389/fpsyg.2015.01389 [PubMed]
- 41. Hebert R, Carrier R, Bilodeau A. The Functional Autonomy Measurement System (SMAF): description and validation of an instrument for the measurement of handicaps. Age Ageing. 1988; 17:293–302. https://doi.org/10.1093/ageing/17.5.293 [PubMed]
- 42. Moreno-Agostino D, Prina M, Chua KC, Jotheeswaran AT, Sadana R, Officer A, Kamenov K, Cieza A. Measuring functional ability in healthy ageing: a nationwide cross-sectional survey in the Philippine older population. BMJ Open. 2021; 11:e050827. https://doi.org/10.1136/bmjopen-2021-050827 [PubMed]
- 43. Charlson M, Szatrowski TP, Peterson J, Gold J. Validation of a combined comorbidity index. J Clin Epidemiol. 1994; 47:1245–51. https://doi.org/10.1016/0895-4356(94)90129-5 [PubMed]
- 44. Brannian JD, Long P, Kreger DO. Is the free androgen index a useful clinical marker in male patients? S D J Med. 1998; 51:449–51. [PubMed]
- 45. Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985; 28:412–9. https://doi.org/10.1007/BF00280883 [PubMed]
- 46. Bozkurt B, Aguilar D, Deswal A, Dunbar SB, Francis GS, Horwich T, Jessup M, Kosiborod M, Pritchett AM, Ramasubbu K, Rosendorff C, Yancy C, and American Heart Association Heart Failure and Transplantation Committee of the Council on Clinical Cardiology, and Council on Cardiovascular Surgery and Anesthesia, and Council on Cardiovascular and Stroke Nursing, and Council on Hypertension, and Council on Quality and Outcomes Research. Contributory Risk and Management of Comorbidities of Hypertension, Obesity, Diabetes Mellitus, Hyperlipidemia, and Metabolic Syndrome in Chronic Heart Failure: A Scientific Statement From the American Heart Association. Circulation. 2016; 134:e535–78. https://doi.org/10.1161/CIR.0000000000000450 [PubMed]
- 47. Casas JP, Shah T, Hingorani AD, Danesh J, Pepys MB. C-reactive protein and coronary heart disease: a critical review. J Intern Med. 2008; 264:295–314. https://doi.org/10.1111/j.1365-2796.2008.02015.x [PubMed]


