Introduction
Stroke has become a major public health problem worldwide, and the financial burden associated with stroke treatment and post-stroke care is significant, especially for older adults [1, 2]. Stroke is a common cause of death and disability in the aging population [3]. In China, stroke was the leading cause of death, disability-adjusted life years, and years of life lost in 2017 [2]. Hypertension is the most common risk factor for patients with prevalent stroke in China [4]. In addition, traditional risk factors cannot fully explain all the risks of stroke [5–7]. Therefore, it is clinically important to understand more modifiable risk factors for stroke, especially in older patients with hypertension.
Sarcopenia is a significant geriatric condition that affects aging societies and is characterized by decreased skeletal muscle mass, low muscular strength, and/or poor physical performance [8]. According to several studies, the percentage of elderly Asian individuals who have sarcopenia ranges from 6.8% to 25.7% [9–12]. Many studies have indicated that sarcopenia and heart disease in the elderly have many pathophysiological aspects in common [13–15]. Sarcopenia is independently associated with prevalent cardiovascular diseases and their associated risk factors [16–18]. Currently, muscle mass is quantified using magnetic resonance imaging, computed tomography, dual-energy X-ray absorptiometry, and bioelectrical impedance analysis [19–21]. However, these techniques have drawbacks such as radiation exposure and a lack of cost effectiveness, as well as increased needs for skilled experts and specialized equipment [22]. It was recently suggested to use the sarcopenia index (SI), a straightforward alternative screening method based on the ratio of serum creatinine (Cr) to cystatin C (CysC) levels [23]. Recent epidemiological studies have also demonstrated that in various populations, including the elderly, SI can be effectively used to measure muscle mass, strength, and functional status [23–25]. According to Hyun et al., individuals with chronic renal disease had an increased risk of all-cause mortality and cardiovascular events when their blood Cr/CysC ratio was high [26]. A low Cr/CysC ratio has also been linked to cardiovascular disease events and death in older individuals with coronary artery disease and those with obstructive coronary artery disease [27, 28]. However, little research has been done to investigate the association between SI and new-onset strokes. To date, the relationship between SI and the risk of stroke is still unknown, especially in older patients with hypertension.
Therefore, we conducted a cohort study to investigate the association between SI and the risk of stroke in elderly patients with hypertension.
Materials and Methods
Patient selection
In this retrospective cohort study, elderly hypertensive patients (age ≥ 60 years) were admitted to the People’s Hospital of Xinjiang Uygur Autonomous Region. Detailed descriptions of this study have been reported previously [29]. Those who met the following criteria were excluded: (1) patients had no data on Cr or CysC, (2) patients with kidney disease or an eGFR of less than 60 mL/min/1.73 m2, (3) loss to follow-up or follow-up duration <6 months, and (4) patients had a history of stroke, malignancy, liver cirrhosis, advanced heart failure, or chronic lung disease. After these exclusions, 5145 participants were included in the final analysis. Participant flow is illustrated in Supplementary Figure 1. Approval was obtained from the Ethical Committee of the People’s Hospital of Xinjiang Uygur Autonomous Region (No. KY2021031901). Owing to the retrospective data collection, it was not deemed to require informed consent. We followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guideline recommendations.
Data collection and measurements
Data were abstracted electronically from the patient’s medical records, including demographics, anthropometric measures, risk factors, diagnoses according to the International Classification of Diseases 10th Revision (ICD-10), prescribed medications, and laboratory data. Smoking status was dichotomized as current smokers vs. non-smokers. A similar classification was used for alcohol use (current drinkers and non-drinkers). All blood samples were collected in the morning, on fasting participants. Laboratory parameters included fasting plasma glucose (FPG), hemoglobin A1c (HbA1c), fasting lipid profile, liver and renal function tests, homocysteine (Hcy), and high-sensitivity C-reactive protein (hs-CRP). Serum Cr level was measured using an enzymatic method, and CysC levels were measured with the immunoturbidimetric assay. The SI was calculated as (serum Cr divided by serum CysC)*100 [23]. eGFR was estimated using the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation [30]. Information on disease history was obtained using ICD-10 codes. To ensure the accuracy of diagnoses, coronary heart disease (CHD) (I24 and I25), diabetes (E10-E14), atrial fibrillation (I48), and dyslipidemia (E78) were regarded as present if a participant was treated ≥ 2 times. The burden of comorbidity was measured using the Charlson Comorbidity Index (CCI) [31]. Prescription claims in the last year prior to the concomitant medications identified at baseline. The list of concomitant medications included in this study is shown in Supplementary Table 1.
Follow-up and outcome assessment
Each participant’s follow-up period (measured in person-years) was computed from the baseline date to the earliest of the following dates: incidence of stroke, death, loss to follow-up, or 31 December 2021 (the study’s end of follow-up), whichever came first. Outcomes of events since participants enrolled in the study were determined through checking medical records, interviews, contact with local disease and death registries, and electronic linkage with the national health insurance claim databases. Trained staff with no knowledge of baseline information used ICD-10 to code all diagnoses and deaths. The primary outcome was the first occurrence of total stroke (ICD-10: I60-I64), including morbidity and mortality. Secondary outcomes included first ischemic stroke (ICD-10: I63) and first hemorrhagic stroke [subarachnoid (ICD-10: I60) or intracerebral (ICD-10: I61)]. An independent clinical events committee reviewed and centrally adjudicated these outcome events.
Statistical analysis
Details of the missing covariates are shown in Supplementary Table 2. We performed multiple imputation to recover missing covariates. Baseline characteristics were compared between groups categorized by SI levels. The age-adjusted incidence rates were determined by calculating age-specific incidence rates within 1-year age categories. Time to first stroke event was examined using Kaplan-Meier survival curves and compared using log-rank test. Multicollinearity was tested using the variance inflation factor (Supplementary Table 3). Testing for proportional hazards used Schoenfeld residuals (Supplementary Figure 2). Cox regression analysis was used to assess the association between SI and stroke and its subtypes, and hazard ratios (HRs) with 95% confidence intervals (CIs) were estimated. Values for trend tests were assigned using the within-quartile medians. Additionally, we used restricted cubic splines to investigate the non-linear associations between SI and the outcomes. Furthermore, subgroup analyses were done and differences were examined using tests for interaction. Sensitivity analyses assessed robustness of results. First, we specified a 1-year exposure lag to circumvent the potential bias of reverse causation. Second, we did competing-risk analysis that treats non-stroke-related deaths as a competing risk. Third, sensitivity analyses were performed that excluded all individuals with CCI ≥ 2. Fourth, participants with atrial fibrillation were excluded. Lastly, potential unmeasured confounding was examined by calculating E-values. The additional value of adding SI to the conventional model was evaluated by C-statistics, the net reclassification index (NRI), and the integrated discrimination index (IDI). More details are found in the Supplementary Materials and Methods.
All analyses were performed using R 4.1.1 software, with a two-sided significance p-value < 0.05.
Results
Study population and characteristics
As illustrated in the flowchart (Supplementary Figure 1), a total of 5145 eligible participants were included in the present study. Among the included participants, the average age was 66.54 ± 4.79 years, and 2481 (48.22%) participants were female. The distribution of SI is shown in Supplementary Figure 3. The mean SI index was 90.41 ± 24.04, and we categorized the population into four groups based on the quartiles of the SI (Table 1).
Table 1. Population characteristics by quartiles of SI.
| Variables | Quartiles of SI | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Quartile 1 | Quartile 2 | Quartile 3 | Quartile 4 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| (≤74.03) | (74.04–87.27) | (87.31–103.69) | (≥103.70) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Participants, N | 1286 | 1286 | 1286 | 1287 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age, years | 66.74 (4.87) | 66.43 (4.82) | 66.39 (4.54) | 66.60 (4.92) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Male, N (%) | 590 (45.88%) | 620 (48.21%) | 717 (55.75%) | 737 (57.26%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hypertension duration, years | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ≤5 | 800 (62.21%) | 1024 (79.63%) | 942 (73.25%) | 1009 (78.46%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| >5 to ≤10 | 146 (11.35%) | 77 (5.99%) | 180 (14.00%) | 179 (13.92%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| >10 | 340 (26.44%) | 185 (14.39%) | 164 (12.75%) | 98 (7.62%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Heart rate, bpm | 79.68 (9.14) | 80.23 (8.99) | 79.68 (9.20) | 80.12 (9.11) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SBP, mmHg | 141.43 (17.77) | 140.76 (16.63) | 140.23 (17.06) | 139.03 (17.38) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| DBP, mmHg | 84.61 (12.66) | 86.24 (12.66) | 87.31 (12.62) | 89.36 (12.54) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BMI, kg/m2 | 24.34 (2.37) | 24.44 (2.40) | 24.30 (2.33) | 24.59 (2.36) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Current smoker, N (%) | 509 (39.58%) | 443 (34.45%) | 391 (30.40%) | 229 (17.81%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Current drinker, N (%) | 482 (37.48%) | 381 (29.63%) | 322 (25.04%) | 180 (14.00%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Comorbidities, N (%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Coronary heart disease | 225 (17.50%) | 197 (15.32%) | 209 (16.25%) | 220 (17.09%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diabetes | 326 (25.35%) | 384 (29.86%) | 363 (28.23%) | 361 (28.05%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dyslipidemia | 799 (62.13%) | 807 (62.75%) | 771 (59.95%) | 783 (60.84%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atrial fibrillation | 39 (3.03%) | 43 (3.34%) | 42 (3.27%) | 31 (2.41%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Charlson comorbidity index | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 0 | 415 (32.27%) | 577 (44.87%) | 629 (48.91%) | 673 (52.29%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1 | 414 (32.19%) | 368 (28.62%) | 335 (26.05%) | 344 (26.73%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ≥2 | 457 (35.54%) | 341 (26.52%) | 322 (25.04%) | 270 (20.98%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Laboratory parameters | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ALT, U/L | 21.87 (14.00–31.62) | 23.00 (14.36–34.02) | 23.98 (15.00–34.30) | 25.20 (16.60–34.50) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AST, U/L | 21.00 (16.00–28.00) | 21.45 (16.89–27.64) | 21.26 (16.39–27.28) | 21.01 (16.00–27.66) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GGT, U/L | 24.22 (15.14–36.60) | 25.30 (16.00–38.58) | 28.03 (17.94–40.14) | 29.00 (19.00–42.22) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Creatinine, mg/dL | 0.66 (0.13) | 0.77 (0.17) | 0.82 (0.19) | 0.89 (0.20) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| UA, μmol/L | 303.68 (79.50) | 325.87 (81.20) | 334.40 (85.55) | 349.45 (85.95) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BUN, mmol/L | 5.29 (1.37) | 5.17 (1.41) | 5.30 (1.42) | 5.26 (1.38) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cystatin C, mg/L | 1.07 (0.22) | 0.96 (0.21) | 0.87 (0.20) | 0.75 (0.19) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| eGFR, ml/min/1.73 m2 | 94.00 (17.75) | 93.28 (18.18) | 93.56 (17.64) | 95.70 (18.54) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TC, mmol/L | 4.47 (3.79–5.10) | 4.52 (3.77–5.20) | 4.52 (3.85–5.15) | 4.47 (3.85–5.03) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TG, mmol/L | 1.50 (1.06–2.15) | 1.49 (1.06–2.25) | 1.64 (1.11–2.32) | 1.67 (1.15–2.41) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HDL-C, mmol/L | 1.08 (0.92–1.26) | 1.04 (0.89–1.23) | 1.06 (0.89–1.24) | 1.02 (0.88–1.21) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LDL-C, mmol/L | 2.73 (2.15–3.27) | 2.80 (2.20–3.32) | 2.79 (2.24–3.33) | 2.79 (2.26–3.26) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HbA1c, % | 6.08 (0.91) | 6.12 (0.90) | 6.07 (0.94) | 6.10 (0.89) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FPG, mmol/L | 5.13 (1.13) | 5.09 (1.11) | 4.98 (0.98) | 5.06 (1.13) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hcy, μmol/L | 14.26 (5.52) | 14.62 (5.90) | 15.33 (5.74) | 15.36 (6.31) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hs-CRP, mg/L | 1.96 (1.10–3.34) | 2.00 (1.13–3.46) | 2.10 (1.18–3.46) | 1.98 (1.11–3.18) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Medications, N (%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ACEI/ARB | 969 (75.35%) | 919 (71.46%) | 916 (71.23%) | 883 (68.61%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Beta-blocker | 495 (38.49%) | 490 (38.10%) | 459 (35.69%) | 458 (35.59%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Calcium channel blockers | 1039 (80.79%) | 1046 (81.34%) | 1015 (78.93%) | 1017 (79.02%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diuretic | 292 (22.71%) | 284 (22.08%) | 320 (24.88%) | 299 (23.23%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Insulin | 131 (10.19%) | 152 (11.82%) | 125 (9.72%) | 121 (9.40%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Oral antidiabetic drugs | 196 (15.24%) | 273 (21.23%) | 230 (17.88%) | 235 (18.26%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Statins | 597 (46.42%) | 553 (43.00%) | 633 (49.22%) | 598 (46.46%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Aspirins | 901 (70.06%) | 907 (70.53%) | 883 (68.66%) | 906 (70.40%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Variables were presented as mean (SD), median (IQR), or N (%). Abbreviations: SI: sarcopenia index; SBP: systolic blood pressure; DBP: diastolic blood pressure; BMI: body mass index; ALT: alanine aminotransferase; AST: aspartate aminotransferase; GGT: gamma-glutamyl transferase; TC: total cholesterol; TG: triglyceride; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; HbA1c: hemoglobin A1c; FPG: fasting plasma glucose; Hcy: homocysteine; UA: uric acid; Cr: blood creatinine; BUN: blood urea nitrogen; hs-CRP: high-sensitivity C-reactive protein; eGFR: estimated glomerular filtration rate; ACEI: angiotensin-converting enzyme inhibitor; IQR: interquartile range; SD: standard deviation. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Association of SI with total stroke and its subtypes
Over a median follow-up of 38 months (IQR: 19–64 months), we identified 607 (11.80%) individuals with total stroke, of whom 507 (9.85%) had ischemic stroke and 93 (1.81%) had hemorrhagic stroke. The age-adjusted incidence of total strokes decreased substantially with the magnitude of SI (quartiles), reaching a maximum incidence of 43.34 per 1000 person-years in quartile 4 (Figure 1). Similarly, the risks of ischemic and hemorrhagic stroke decreased as SI quartiles increased. The Kaplan-Meier curve showed that participants in the quartile 1 group had a higher risk of total stroke, ischemic stroke, and hemorrhagic stroke than those in other groups (log-rank test, P < 0.001, Figure 2A; P < 0.001, Figure 2B; P = 0.007, Figure 2C). Overall, lower SI was significantly associated with higher hazards of stroke and its subtypes among elderly patients with hypertension (Figure 3). In the fully-adjusted model that measured the SI as a continuous variable, each 10-unit increment in the SI was associated with a 12% lower risk of total stroke (HR 0.88, 95% CI 0.85–0.92; Table 2). The cumulative hazard of total stroke also decreased with increasing SI, and this trend persisted even after adjusting for potential confounding factors in Model 3 (P for trend < 0.001). The HRs were 0.85 (95% CI, 0.69–1.05), 0.57 (95% CI, 0.45–0.72), and 0.46 (95% CI, 0.35–0.59) for the quartile 2, quartile 3, and quartile 4 groups versus the quartile 1 group of SI (Table 2). The results were similar when the association between SI and ischemic stroke and hemorrhagic stroke was examined (Table 2). In the restricted cubic spline analysis, we observed a significant dose-response relationship between SI and the risk of total stroke (P for non-linear association = 0.003) (Figure 3A). Similar results were observed in ischemic strokes (P for non-linear association = 0.002) (Figure 3B), but not in hemorrhagic strokes (P for non-linear association = 0.525) (Figure 3C).
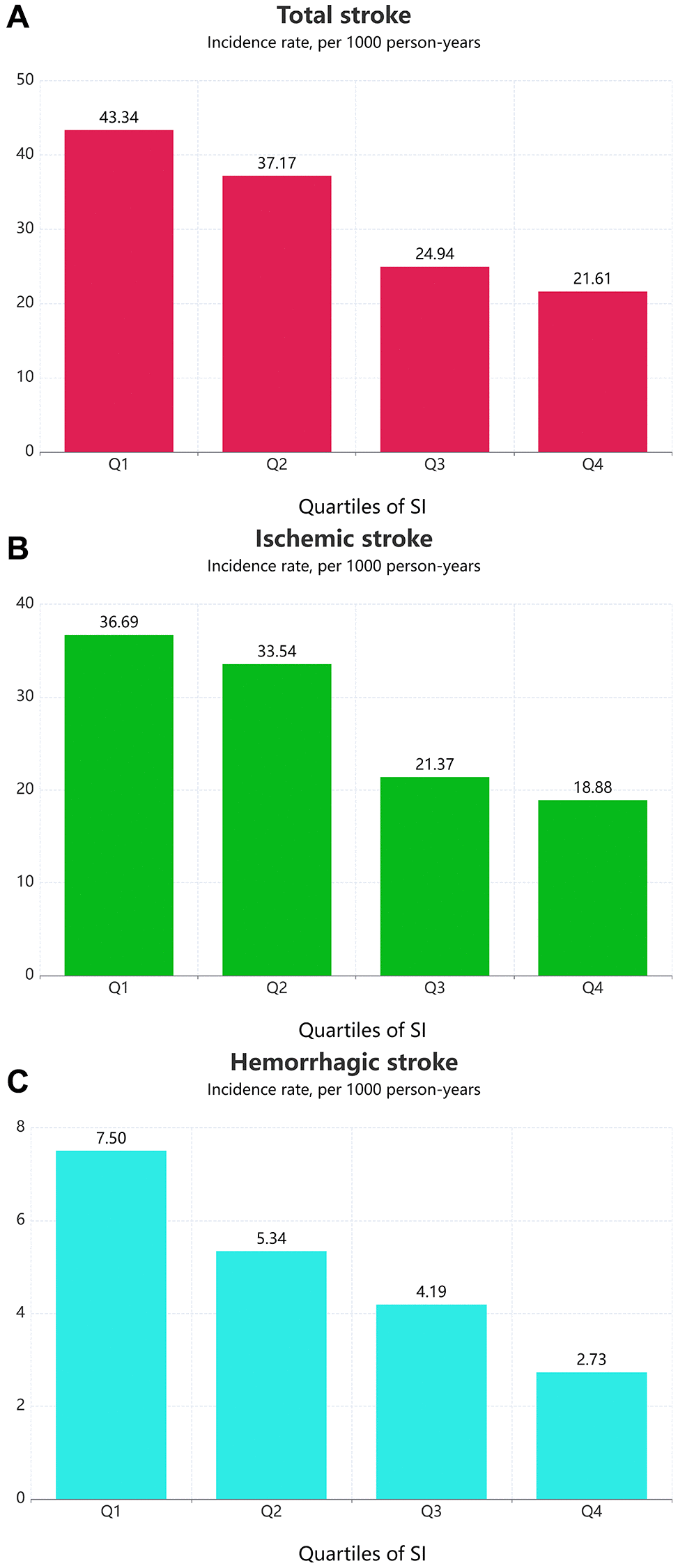
Figure 1. Age-adjusted incidence of outcomes according to the SI quartiles. (A) Total stroke; (B) Ischemic stroke; (C) Hemorrhagic stroke.
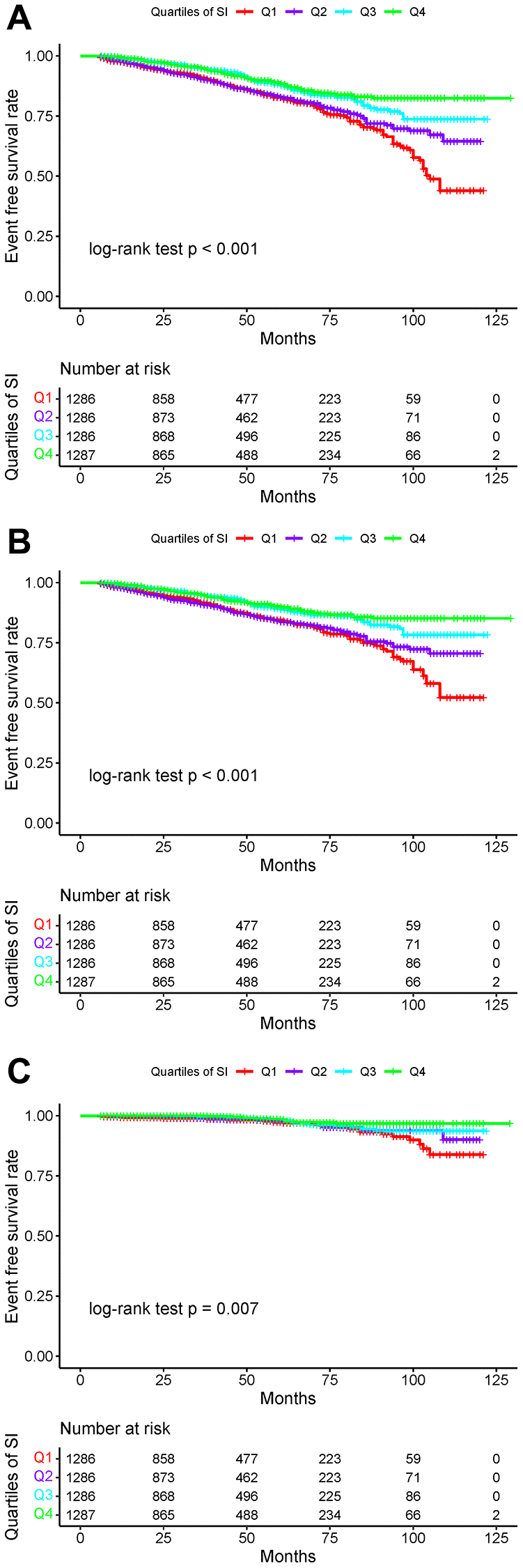
Figure 2. Kaplan-Meier survival curves for total strokes and individual outcomes based on SI quartiles. (A) Total stroke; (B) Ischemic stroke; (C) Hemorrhagic stroke.
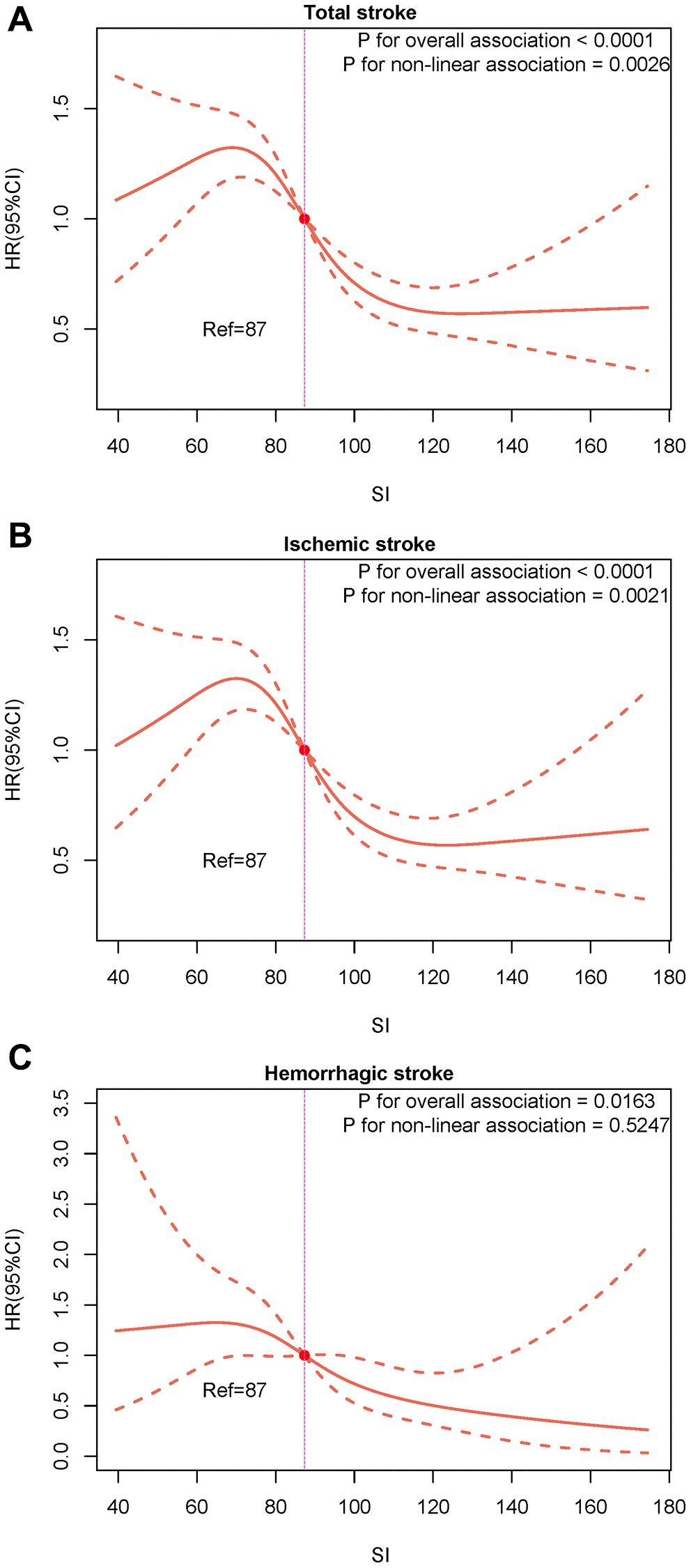
Figure 3. Restricted cubic splines for the associations of SI with the risk of total stroke and its subtypes. (A) Total stroke; (B) Ischemic stroke; (C) Hemorrhagic stroke.
Table 2. Association of SI with incident stroke.
| Outcome | HR (95% CI) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Model 1 | Model 2 | Model 3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total stroke | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Per 10-unit increment | 0.89 (0.86, 0.92) | 0.89 (0.86, 0.92) | 0.88 (0.85, 0.92) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Quartiles | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Q1 | Reference | Reference | Reference | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Q2 | 0.86 (0.70, 1.05) | 0.85 (0.69, 1.04) | 0.85 (0.69, 1.05) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Q3 | 0.57 (0.45, 0.71) | 0.58 (0.46, 0.73) | 0.57 (0.45, 0.72) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Q4 | 0.49 (0.39, 0.63) | 0.48 (0.37, 0.61) | 0.46 (0.35, 0.59) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| P for trend | <0.001 | <0.001 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ischemic stroke | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Per 10-unit increment | 0.89 (0.86, 0.93) | 0.89 (0.85, 0.93) | 0.88 (0.85, 0.92) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Quartiles | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Q1 | Reference | Reference | Reference | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Q2 | 0.91 (0.73, 1.13) | 0.90 (0.72, 1.12) | 0.90 (0.73, 1.13) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Q3 | 0.58 (0.45, 0.74) | 0.58 (0.46, 0.75) | 0.58 (0.45, 0.74) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Q4 | 0.51 (0.40, 0.66) | 0.49 (0.37, 0.63) | 0.46 (0.35, 0.61) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| P for trend | <0.001 | <0.001 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hemorrhagic stroke | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Per 10-unit increment | 0.86 (0.79, 0.95) | 0.86 (0.78, 0.95) | 0.85 (0.77, 0.94) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Quartiles | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Q1 | Reference | Reference | Reference | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Q2 | 0.71 (0.42, 1.18) | 0.71 (0.42, 1.20) | 0.71 (0.42, 1.20) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Q3 | 0.55 (0.32, 0.95) | 0.54 (0.31, 0.94) | 0.52 (0.29, 0.93) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Q4 | 0.36 (0.19, 0.67) | 0.35 (0.18, 0.68) | 0.33 (0.17, 0.64) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| P for trend | 0.001 | 0.001 | 0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Model 1, adjusted for age and sex. Model 2, adjusted for variables in model 1 plus SBP, DBP, BMI, hypertension duration, heart rate, smoking status, drinking status, and comorbidities. Model 3, adjusted for variables in model 2 plus ALT, AST, GGT, UA, BUN, eGFR, TC, TG, HDL-C, LDL-C, HbA1c, FPG, Hcy, use of statins, use of aspirins, use of insulins, use of oral antidiabetic drugs, and antihypertensive drugs. Abbreviations: CI: confidence interval; HR: hazard ratio. Other abbreviations as presented in Table 1. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
To investigate the robustness of our findings, we conducted multiple sensitivity analyses. In the sensitivity analyses, the associations of SI with the risk of stroke and its subtypes were not materially changed after excluding participants who developed strokes within the first year of follow-up (Supplementary Table 4), participants with CCI ≥ 2 (Supplementary Table 5), participants with atrial fibrillation (Supplementary Table 6), or participants with a competing risk model (Supplementary Table 7). Sensitivity analyses using E-values also revealed that strong unmeasured confounding is required for the observed association to be null (Supplementary Table 8 and Supplementary Figure 4).
Stratified analyses
We further performed exploratory subgroup analyses to assess the association between SI (per 10-unit increment) and the risk of stroke and its subtypes among elderly patients with hypertension (Figure 4). In the stratified analyses, age, sex, BMI, smoking status, drinking status, eGFR, hypertension duration, dyslipidemia, CCI, and coronary heart disease did not significantly modify the association between the SI and the risk of new-onset total strokes (all P-interactions > 0.05) (Figure 4A). Although the P value for the interaction of diabetes status in elderly hypertensive patients was less than 0.05, due to the similar directionality of the association, the result may not be clinically significant (Figure 4A). None of the variables significantly modified the association between SI and the risk of new-onset ischemic stroke and hemorrhagic stroke (all P for interactions > 0.05) (Figure 4B and 4C).

Figure 4. Adjusted hazard ratios of stroke associated with per 10 unit increase in SI. (A) Total stroke; (B) Ischemic stroke; (C) Hemorrhagic stroke.
Incremental predictive value of SI
We assessed whether SI would boost the traditional model’s prediction power even further (Supplementary Table 9). With the addition of SI, the conventional model’s C statistics greatly improved (Δ C-statistics = 0.02). With an IDI of 0.03 (95% CI, 0.02–0.04) and an NRI of 0.17 (95% CI, 0.10–0.23), the discriminating power and risk reclassification also seemed to be much superior. Both ischemic and hemorrhagic strokes had similar results.
Discussion
This study examined the relationship between SI and incident stroke in elderly hypertensive patients. After controlling for multiple confounders, the results showed that the risk of stroke was substantially associated with SI. Furthermore, in the restricted cubic spline analysis, we observed a significant dose-response relationship between SI and the risk of total strokes and ischemic strokes. These findings were consistent in both the sensitivity and subgroup analyses. Overall, the current investigation showed that low SI was associated with a higher risk of incident stroke, independent of other traditional risk variables. To our knowledge, this is the first large cohort study to demonstrate a link between SI and the risk of incident stroke among elderly hypertensive patients.
Currently, China has the world’s largest elderly population [32]. According to the results of the seventh national census in 2020, the population aged 60 and above accounted for 18.7% of China’s total population, amounting to 264 million people [33]. Population aging has become a significant trend in China’s social development, and the aging of China’s population has further intensified. In our aging society, sarcopenia, or reduced muscle mass, is a growing health concern [34]. Sarcopenia, which is a widespread and gradual reduction in skeletal muscle mass and function that impairs mobility and work capability in elderly people, is regarded as a geriatric syndrome [8]. Skeletal muscle degeneration, according to epidemiological research, starts after the age of 40 and gets worse with time. Skeletal muscle deteriorates in both number and quality at a rate of 8% each year [35]. The results of a recent meta-analysis showed that the prevalence of sarcopenia among the Chinese elderly aged 60 years and older was about 11.2% to 33.7% [36]. Stroke is the leading cause of disability and death among the elderly in China [37]. Some studies have linked skeletal muscle weakness to eating disorders, lack of exercise, insulin resistance, inflammation, and atherosclerosis and found that these are independent risk factors for cardiovascular disease in older adults [17, 38–41]. Sarcopenia imposes a significant burden on society by significantly increasing hospitalization and mortality rates in elderly patients [42, 43]. Screening for sarcopenia in elderly hypertensive patients at an early stage is therefore critical in clinical practice.
In this context, the quantification of skeletal muscle mass is particularly important. To assess skeletal muscle mass, several options have been proposed: magnetic resonance imaging, computed tomography, dual-energy X-ray absorptiometry, and bioelectrical impedance analysis [44]. However, these measurements are not universally applicable in clinical practice due to their high cost, possible radiation exposure, and the requirements for specialized technicians and sophisticated equipment [45]. Therefore, there is a need for more applicable and reliable alternative serum biomarkers for the assessment of sarcopenia in elderly patients with hypertension. Recently, the SI has been proposed as a simple alternative screening tool [23]. The results of numerous studies have suggested that the Cr/CysC ratio may serve as a marker for predicting muscle atrophy and dysfunction [44–48]. The Cr/CysC ratio has been positively correlated with muscle mass and strength in various populations [46, 49, 50]. In particular, the accuracy of the Cr/CysC ratio as a measure of muscle mass has been validated by computed tomography in the assessment of muscle mass in different populations, including the elderly and cancer patients [45, 51, 52]. Furthermore, evidence that the Cr/CysC ratio is superior to bioelectrical impedance analysis in the detection of muscle weakness further supports its accuracy [52].
In this study, we confirmed that SI was independently associated with an increased risk of stroke in elderly patients with hypertension. And we also observed a significant dose-response relationship between SI and the risk of total and ischemic strokes. Our findings are consistent with those of several previous studies. The difference is that previous studies have focused on patients with chronic renal insufficiency, older patients undergoing transcatheter aortic valve replacement, and patients with obstructive coronary artery disease [22, 53–55]. Further, they usually focus on looking at the risk of SI with cardiovascular death, all-cause death, and major adverse cardiovascular events. The underlying mechanism leading to this association may be multifactorial and has not yet been identified. Several potential explanations may account for this result. Skeletal muscle is a major site of glucose uptake, deposition, and actin secretion [56]. Therefore, the reduced glucose uptake caused by low skeletal muscle mass loss may contribute to enhanced insulin resistance [57, 58]. Insulin resistance can produce chronic hyperglycemia, which in turn triggers oxidative stress, causing an inflammatory response and cell damage [59]. Insulin resistance can also alter systemic lipid metabolism, leading to the development of dyslipidemia. This, combined with endothelial dysfunction, insulin resistance, and dyslipidemia, can all lead to atherosclerosis and eventually progress to ischemic stroke [60]. Increased muscle strength is associated with lower blood pressure and improved hemodynamics, suggesting a protective role for muscle in the development of atherosclerosis [61]. Another possible mechanism of poor prognosis is a decrease in skeletal muscle and endocrine function as secretory organs [62]. Myogenic factors are cytokines or other peptides produced, expressed, and released by skeletal muscle fibers that may help regulate beneficial cardiovascular effects [63]. Myocytes perform endocrine functions by secreting cardiovascular-beneficial myokines [64]. In patients with low muscle mass, decreased muscle cell numbers and decreased endocrine function may lead to adverse clinical outcomes [65]. These factors, together with pre-existing chronic comorbidities, explain the high risk of stroke. There are also studies showing that in older patients with skeletal myasthenia, long-term systemic chronic inflammation appears to be associated with the overall course of cardiovascular disease [66, 67]. Aging-related secretory phenotyping, one of the key factors in the chronic inflammation-induced instability of atherosclerotic plaques, is part of the pathogenesis of atherosclerosis, and is an independent risk factor for cardiovascular disease and cardiovascular mortality [66, 68, 69]. Research has also shown that inflammation activates the body’s catabolic pathway and promotes the hydrolysis of muscle proteins, leading to an imbalance between protein synthesis and catabolism, which further exacerbates the development of sarcopenia [70].
The present study has multiple strengths. Initially, this study is the first to use data from a cohort study to identify SI as a predictor of stroke in elderly patients with hypertension. Our findings should be considered in clinical practice and prospective clinical trials to prevent or treat pre-existing sarcopenia in older patients with hypertension. Second, we adjusted for as many confounders as possible in our study to improve the reliability of the results. Finally, this study used subgroup analysis and restricted cubic spline curve analysis, which helped enrich the interpretation of the relationship between SI and the risk of stroke. This study still has the following limitations: First, the observational, retrospective design limits the determination of causality. Second, we did not use methods such as dual energy X-ray absorptiometry or magnetic resonance imaging to assess actual residual muscle mass in these patients or analyze the association between SI and skeletal muscle mass, or sarcopenia. However, our goal was to focus more on the prognostic value of SI and obtain results that might be helpful in actual clinical practice. Third, we used measurements from only one point in time, so trends and changes in SI could not be determined. Fourth, this study was conducted only in hypertensive patients aged 60 years and older, and therefore our results may not be fully generalizable to younger people or other populations. Finally, although we examined measured covariates for potential confounding, residual confounding resulting from unmeasured factors such as frailty, physical activity, and dietary factors cannot be excluded.
In summary, this study found that elevated SI was negatively associated with the risk of stroke in elderly patients with hypertension. Uncovering the causality behind the relationship requires further prospective study.
Author Contributions
Conception and design of the work: CXT, HJL, and LNF. Provision of study materials or patients: LNF. Acquisition of data: YWB, DYJ, and WW. Analysis of data: CXT, WMR, WJY, and HJ. CXT drafted the manuscript. LNF, LQ, and HJL substantially revised the manuscript. All authors read and approved the final manuscript.
Acknowledgments
Contributions from all participants are appreciated.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
Ethical Statement and Consent
The study procedures were carried out by the Declaration of Helsinki. Approval was obtained from the Ethical Committee of People’s Hospital of Xinjiang Uygur Autonomous Region (No. KY2021031901). The need for written informed consent was waived by the (People’s Hospital of Xinjiang Uygur Autonomous Region) ethics committee due to retrospective nature of the study.
Funding
This work was supported by the Key Research and Development Project of Xinjiang Uygur Autonomous Region (Nos. 2022B03009 and 2022B03009-1).
References
- 1. Ma Q, Li R, Wang L, Yin P, Wang Y, Yan C, Ren Y, Qian Z, Vaughn MG, McMillin SE, Hay SI, Naghavi M, Cai M, et al. Temporal trend and attributable risk factors of stroke burden in China, 1990-2019: an analysis for the Global Burden of Disease Study 2019. Lancet Public Health. 2021; 6:e897–906. https://doi.org/10.1016/S2468-2667(21)00228-0 [PubMed]
- 2. Zhou M, Wang H, Zeng X, Yin P, Zhu J, Chen W, Li X, Wang L, Wang L, Liu Y, Liu J, Zhang M, Qi J, et al. Mortality, morbidity, and risk factors in China and its provinces, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2019; 394:1145–58. https://doi.org/10.1016/S0140-6736(19)30427-1 [PubMed]
- 3. Lindley RI. Stroke Prevention in the Very Elderly. Stroke. 2018; 49:796–802. https://doi.org/10.1161/STROKEAHA.117.017952 [PubMed]
- 4. Tu WJ, Hua Y, Yan F, Bian H, Yang Y, Lou M, Kang D, He L, Chu L, Zeng J, Wu J, Chen H, Han J, et al. Prevalence of stroke in China, 2013-2019: A population-based study. Lancet Reg Health West Pac. 2022; 28:100550. https://doi.org/10.1016/j.lanwpc.2022.100550 [PubMed]
- 5. Nie J, Xie L, Zhao BX, Li Y, Qiu B, Zhu F, Li GF, He M, Wang Y, Wang B, Liu S, Zhang H, Guo H, et al. Serum Trimethylamine N-Oxide Concentration Is Positively Associated With First Stroke in Hypertensive Patients. Stroke. 2018; 49:2021–8. https://doi.org/10.1161/STROKEAHA.118.021997 [PubMed]
- 6. Xie L, Zhao BX, Luo J, Li Y, Zhu F, Li GF, He M, Wang B, Zhang H, Cai Y, Huo Y, Wang X, Hou FF, et al. A U-shaped association between serum betaine and incident risk of first ischemic stroke in hypertensive patients. Clin Nutr. 2020; 39:2517–24. https://doi.org/10.1016/j.clnu.2019.11.011 [PubMed]
- 7. Wang Z, Ma H, Song Y, Lin T, Liu L, Zhou Z, Wei Y, Huang X, Chen P, Liu C, Li Y, Wang B, Li J, et al. Plasma selenium and the risk of first stroke in adults with hypertension: a secondary analysis of the China Stroke Primary Prevention Trial. Am J Clin Nutr. 2022; 115:222–31. https://doi.org/10.1093/ajcn/nqab320 [PubMed]
- 8. Batsis JA, Villareal DT. Sarcopenic obesity in older adults: aetiology, epidemiology and treatment strategies. Nat Rev Endocrinol. 2018; 14:513–37. https://doi.org/10.1038/s41574-018-0062-9 [PubMed]
- 9. Tanimoto Y, Watanabe M, Sun W, Sugiura Y, Tsuda Y, Kimura M, Hayashida I, Kusabiraki T, Kono K. Association between sarcopenia and higher-level functional capacity in daily living in community-dwelling elderly subjects in Japan. Arch Gerontol Geriatr. 2012; 55:e9–13. https://doi.org/10.1016/j.archger.2012.06.015 [PubMed]
- 10. Kim YS, Lee Y, Chung YS, Lee DJ, Joo NS, Hong D, Song Ge, Kim HJ, Choi YJ, Kim KM. Prevalence of sarcopenia and sarcopenic obesity in the Korean population based on the Fourth Korean National Health and Nutritional Examination Surveys. J Gerontol A Biol Sci Med Sci. 2012; 67:1107–13. https://doi.org/10.1093/gerona/gls071 [PubMed]
- 11. Kitamura A, Seino S, Abe T, Nofuji Y, Yokoyama Y, Amano H, Nishi M, Taniguchi Y, Narita M, Fujiwara Y, Shinkai S. Sarcopenia: prevalence, associated factors, and the risk of mortality and disability in Japanese older adults. J Cachexia Sarcopenia Muscle. 2021; 12:30–8. https://doi.org/10.1002/jcsm.12651 [PubMed]
- 12. Chen LK, Woo J, Assantachai P, Auyeung TW, Chou MY, Iijima K, Jang HC, Kang L, Kim M, Kim S, Kojima T, Kuzuya M, Lee JSW, et al. Asian Working Group for Sarcopenia: 2019 Consensus Update on Sarcopenia Diagnosis and Treatment. J Am Med Dir Assoc. 2020; 21:300–7.e2. https://doi.org/10.1016/j.jamda.2019.12.012 [PubMed]
- 13. Curcio F, Testa G, Liguori I, Papillo M, Flocco V, Panicara V, Galizia G, Della-Morte D, Gargiulo G, Cacciatore F, Bonaduce D, Landi F, Abete P. Sarcopenia and Heart Failure. Nutrients. 2020; 12:211. https://doi.org/10.3390/nu12010211 [PubMed]
- 14. Gao K, Cao LF, Ma WZ, Gao YJ, Luo MS, Zhu J, Li T, Zhou D. Association between sarcopenia and cardiovascular disease among middle-aged and older adults: Findings from the China health and retirement longitudinal study. EClinicalMedicine. 2022; 44:101264. https://doi.org/10.1016/j.eclinm.2021.101264 [PubMed]
- 15. Xue Q, Wu J, Ren Y, Hu J, Yang K, Cao J. Sarcopenia predicts adverse outcomes in an elderly population with coronary artery disease: a systematic review and meta-analysis. BMC Geriatr. 2021; 21:493. https://doi.org/10.1186/s12877-021-02438-w [PubMed]
- 16. Xia MF, Chen LY, Wu L, Ma H, Li XM, Li Q, Aleteng Q, Hu Y, He WY, Gao J, Lin HD, Gao X. Sarcopenia, sarcopenic overweight/obesity and risk of cardiovascular disease and cardiac arrhythmia: A cross-sectional study. Clin Nutr. 2021; 40:571–80. https://doi.org/10.1016/j.clnu.2020.06.003 [PubMed]
- 17. Barbalho SM, Flato UAP, Tofano RJ, Goulart RA, Guiguer EL, Detregiachi CRP, Buchaim DV, Araújo AC, Buchaim RL, Reina FTR, Biteli P, Reina DOB, Bechara MD. Physical Exercise and Myokines: Relationships with Sarcopenia and Cardiovascular Complications. Int J Mol Sci. 2020; 21:3607. https://doi.org/10.3390/ijms21103607 [PubMed]
- 18. Pizzimenti M, Meyer A, Charles AL, Giannini M, Chakfé N, Lejay A, Geny B. Sarcopenia and peripheral arterial disease: a systematic review. J Cachexia Sarcopenia Muscle. 2020; 11:866–86. https://doi.org/10.1002/jcsm.12587 [PubMed]
- 19. Romero-Corral A, Montori VM, Somers VK, Korinek J, Thomas RJ, Allison TG, Mookadam F, Lopez-Jimenez F. Association of bodyweight with total mortality and with cardiovascular events in coronary artery disease: a systematic review of cohort studies. Lancet. 2006; 368:666–78. https://doi.org/10.1016/S0140-6736(06)69251-9 [PubMed]
- 20. Han A, Bokshan SL, Marcaccio SE, DePasse JM, Daniels AH. Diagnostic Criteria and Clinical Outcomes in Sarcopenia Research: A Literature Review. J Clin Med. 2018; 7:70. https://doi.org/10.3390/jcm7040070 [PubMed]
- 21. Cruz-Jentoft AJ, Baeyens JP, Bauer JM, Boirie Y, Cederholm T, Landi F, Martin FC, Michel JP, Rolland Y, Schneider SM, Topinková E, Vandewoude M, Zamboni M, and European Working Group on Sarcopenia in Older People. Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing. 2010; 39:412–23. https://doi.org/10.1093/ageing/afq034 [PubMed]
- 22. Lu YW, Tsai YL, Chou RH, Kuo CS, Chang CC, Huang PH, Lin SJ. Serum creatinine to cystatin C ratio is associated with major adverse cardiovascular events in patients with obstructive coronary artery disease. Nutr Metab Cardiovasc Dis. 2021; 31:1509–15. https://doi.org/10.1016/j.numecd.2021.01.024 [PubMed]
- 23. Kashani KB, Frazee EN, Kukrálová L, Sarvottam K, Herasevich V, Young PM, Kashyap R, Lieske JC. Evaluating Muscle Mass by Using Markers of Kidney Function: Development of the Sarcopenia Index. Crit Care Med. 2017; 45:e23–9. https://doi.org/10.1097/CCM.0000000000002013 [PubMed]
- 24. Fujita K, Ohkubo H, Nakano A, Takeda N, Fukumitsu K, Fukuda S, Kanemitsu Y, Uemura T, Tajiri T, Maeno K, Ito Y, Oguri T, Ozawa Y, et al. Serum creatinine/cystatin C ratio is a surrogate marker for sarcopenia in patients with idiopathic pulmonary fibrosis. BMC Pulm Med. 2022; 22:203. https://doi.org/10.1186/s12890-022-02000-3 [PubMed]
- 25. Tang T, Xie L, Hu S, Tan L, Lei X, Luo X, Yang L, Yang M. Serum creatinine and cystatin C-based diagnostic indices for sarcopenia in advanced non-small cell lung cancer. J Cachexia Sarcopenia Muscle. 2022; 13:1800–10. https://doi.org/10.1002/jcsm.12977 [PubMed]
- 26. Hyun YY, Lee KB, Kim H, Kim Y, Chung W, Park HC, Han SH, Oh YK, Park SK, Oh KH. Serum creatinine to cystatin C ratio and clinical outcomes in adults with non-dialysis chronic kidney disease. Front Nutr. 2022; 9:996674. https://doi.org/10.3389/fnut.2022.996674 [PubMed]
- 27. Shin JY. Low serum creatinine to cystatin C ratio is independently associated with sarcopenia and high carotid plaque score in patients with type 2 diabetes. Nutr Metab Cardiovasc Dis. 2022; 32:1454–62. https://doi.org/10.1016/j.numecd.2022.02.005 [PubMed]
- 28. Hashimoto Y, Takahashi F, Okamura T, Osaka T, Okada H, Senmaru T, Majima S, Ushigome E, Nakanishi N, Asano M, Hamaguchi M, Yamazaki M, Fukui M. Relationship between serum creatinine to cystatin C ratio and subclinical atherosclerosis in patients with type 2 diabetes. BMJ Open Diabetes Res Care. 2022; 10:e002910. https://doi.org/10.1136/bmjdrc-2022-002910 [PubMed]
- 29. Cai X, Hu J, Wen W, Wang M, Zhu Q, Liu S, Yang W, Dang Y, Hong J, Li N. Association between the geriatric nutritional risk index and the risk of stroke in elderly patients with hypertension: A longitudinal and cohort study. Front Nutr. 2022; 9:1048206. https://doi.org/10.3389/fnut.2022.1048206 [PubMed]
- 30. Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF
3rd , Feldman HI, Kusek JW, Eggers P, Van Lente F, Greene T, Coresh J, and CKD-EPI (Chronic Kidney Disease Epidemiology Collaboration). A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009; 150:604–12. https://doi.org/10.7326/0003-4819-150-9-200905050-00006 [PubMed] - 31. Sundararajan V, Henderson T, Perry C, Muggivan A, Quan H, Ghali WA. New ICD-10 version of the Charlson comorbidity index predicted in-hospital mortality. J Clin Epidemiol. 2004; 57:1288–94. https://doi.org/10.1016/j.jclinepi.2004.03.012 [PubMed]
- 32. Bao J, Zhou L, Liu G, Tang J, Lu X, Cheng C, Jin Y, Bai J. Current state of care for the elderly in China in the context of an aging population. Biosci Trends. 2022; 16:107–18. https://doi.org/10.5582/bst.2022.01068 [PubMed]
- 33. Tu WJ, Zeng X, Liu Q. Aging tsunami coming: the main finding from China's seventh national population census. Aging Clin Exp Res. 2022; 34:1159–63. https://doi.org/10.1007/s40520-021-02017-4 [PubMed]
- 34. Ko BJ, Chang Y, Jung HS, Yun KE, Kim CW, Park HS, Chung EC, Shin H, Ryu S. Relationship Between Low Relative Muscle Mass and Coronary Artery Calcification in Healthy Adults. Arterioscler Thromb Vasc Biol. 2016; 36:1016–21. https://doi.org/10.1161/ATVBAHA.116.307156 [PubMed]
- 35. Foley RN, Wang C, Ishani A, Collins AJ, Murray AM. Kidney function and sarcopenia in the United States general population: NHANES III. Am J Nephrol. 2007; 27:279–86. https://doi.org/10.1159/000101827 [PubMed]
- 36. Chen Z, Li WY, Ho M, Chau PH. The Prevalence of Sarcopenia in Chinese Older Adults: Meta-Analysis and Meta-Regression. Nutrients. 2021; 13:1441. https://doi.org/10.3390/nu13051441 [PubMed]
- 37. Xia X, Yue W, Chao B, Li M, Cao L, Wang L, Shen Y, Li X. Prevalence and risk factors of stroke in the elderly in Northern China: data from the National Stroke Screening Survey. J Neurol. 2019; 266:1449–58. https://doi.org/10.1007/s00415-019-09281-5 [PubMed]
- 38. Le Couteur DG, Solon-Biet SM, Cogger VC, Ribeiro R, de Cabo R, Raubenheimer D, Cooney GJ, Simpson SJ. Branched chain amino acids, aging and age-related health. Ageing Res Rev. 2020; 64:101198. https://doi.org/10.1016/j.arr.2020.101198 [PubMed]
- 39. Nascimento CM, Ingles M, Salvador-Pascual A, Cominetti MR, Gomez-Cabrera MC, Viña J. Sarcopenia, frailty and their prevention by exercise. Free Radic Biol Med. 2019; 132:42–9. https://doi.org/10.1016/j.freeradbiomed.2018.08.035 [PubMed]
- 40. Hong SH, Choi KM. Sarcopenic Obesity, Insulin Resistance, and Their Implications in Cardiovascular and Metabolic Consequences. Int J Mol Sci. 2020; 21:494. https://doi.org/10.3390/ijms21020494 [PubMed]
- 41. Kitada M, Koya D. Autophagy in metabolic disease and ageing. Nat Rev Endocrinol. 2021; 17:647–61. https://doi.org/10.1038/s41574-021-00551-9 [PubMed]
- 42. Gümüşsoy M, Atmış V, Yalçın A, Bahşi R, Yiğit S, Arı S, Dokuyan HC, Gözükara MG, Silay K. Malnutrition-sarcopenia syndrome and all-cause mortality in hospitalized older people. Clin Nutr. 2021; 40:5475–81. https://doi.org/10.1016/j.clnu.2021.09.036 [PubMed]
- 43. Shu X, Lin T, Wang H, Zhao Y, Jiang T, Peng X, Yue J. Diagnosis, prevalence, and mortality of sarcopenia in dialysis patients: a systematic review and meta-analysis. J Cachexia Sarcopenia Muscle. 2022; 13:145–58. https://doi.org/10.1002/jcsm.12890 [PubMed]
- 44. Kashani K, Sarvottam K, Pereira NL, Barreto EF, Kennedy CC. The sarcopenia index: A novel measure of muscle mass in lung transplant candidates. Clin Transplant. 2018; 32:e13182. https://doi.org/10.1111/ctr.13182 [PubMed]
- 45. Barreto EF, Poyant JO, Coville HH, Dierkhising RA, Kennedy CC, Gajic O, Nystrom EM, Takahashi N, Moynagh MR, Kashani KB. Validation of the sarcopenia index to assess muscle mass in the critically ill: A novel application of kidney function markers. Clin Nutr. 2019; 38:1362–7. https://doi.org/10.1016/j.clnu.2018.05.031 [PubMed]
- 46. Fu X, Tian Z, Wen S, Sun H, Thapa S, Xiong H, Liu H, Li L, Yu S. A new index based on serum creatinine and cystatin C is useful for assessing sarcopenia in patients with advanced cancer. Nutrition. 2021; 82:111032. https://doi.org/10.1016/j.nut.2020.111032 [PubMed]
- 47. Barreto EF, Kanderi T, DiCecco SR, Lopez-Ruiz A, Poyant JO, Mara KC, Heimgartner J, Gajic O, Rule AD, Nystrom EM, Kashani KB. Sarcopenia Index Is a Simple Objective Screening Tool for Malnutrition in the Critically Ill. JPEN J Parenter Enteral Nutr. 2019; 43:780–8. https://doi.org/10.1002/jpen.1492 [PubMed]
- 48. Ding P, Guo H, Sun C, Chen S, Yang P, Tian Y, Lowe S, Zhao Q. Serum creatinine/cystatin C ratio is a systemic marker of sarcopenia in patients with gastrointestinal stromal tumours. Front Nutr. 2022; 9:963265. https://doi.org/10.3389/fnut.2022.963265 [PubMed]
- 49. Osaka T, Hamaguchi M, Hashimoto Y, Ushigome E, Tanaka M, Yamazaki M, Fukui M. Decreased the creatinine to cystatin C ratio is a surrogate marker of sarcopenia in patients with type 2 diabetes. Diabetes Res Clin Pract. 2018; 139:52–8. https://doi.org/10.1016/j.diabres.2018.02.025 [PubMed]
- 50. Lin YL, Chen SY, Lai YH, Wang CH, Kuo CH, Liou HH, Hsu BG. Serum creatinine to cystatin C ratio predicts skeletal muscle mass and strength in patients with non-dialysis chronic kidney disease. Clin Nutr. 2020; 39:2435–41. https://doi.org/10.1016/j.clnu.2019.10.027 [PubMed]
- 51. Tabara Y, Kohara K, Okada Y, Ohyagi Y, Igase M. Creatinine-to-cystatin C ratio as a marker of skeletal muscle mass in older adults: J-SHIPP study. Clin Nutr. 2020; 39:1857–62. https://doi.org/10.1016/j.clnu.2019.07.027 [PubMed]
- 52. Ulmann G, Kaï J, Durand JP, Neveux N, Jouinot A, De Bandt JP, Goldwasser F, Cynober L. Creatinine-to-cystatin C ratio and bioelectrical impedance analysis for the assessement of low lean body mass in cancer patients: Comparison to L3-computed tomography scan. Nutrition. 2021; 81:110895. https://doi.org/10.1016/j.nut.2020.110895 [PubMed]
- 53. Lin YL, Chang IC, Liou HH, Wang CH, Lai YH, Kuo CH, Hsu BG. Serum indices based on creatinine and cystatin C predict mortality in patients with non-dialysis chronic kidney disease. Sci Rep. 2021; 11:16863. https://doi.org/10.1038/s41598-021-96447-9 [PubMed]
- 54. Romeo FJ, Chiabrando JG, Seropian IM, Raleigh JV, de Chazal HM, Garmendia CM, Smietniansky M, Cal M, Agatiello CR, Berrocal DH. Sarcopenia index as a predictor of clinical outcomes in older patients undergoing transcatheter aortic valve replacement. Catheter Cardiovasc Interv. 2021; 98:E889–96. https://doi.org/10.1002/ccd.29799 [PubMed]
- 55. Tang T, Zhuo Y, Xie L, Wang H, Yang M. Sarcopenia index based on serum creatinine and cystatin C is associated with 3-year mortality in hospitalized older patients. Sci Rep. 2020; 10:1260. https://doi.org/10.1038/s41598-020-58304-z [PubMed]
- 56. Kalyani RR, Corriere M, Ferrucci L. Age-related and disease-related muscle loss: the effect of diabetes, obesity, and other diseases. Lancet Diabetes Endocrinol. 2014; 2:819–29. https://doi.org/10.1016/S2213-8587(14)70034-8 [PubMed]
- 57. Wang C, Bai L. Sarcopenia in the elderly: basic and clinical issues. Geriatr Gerontol Int. 2012; 12:388–96. https://doi.org/10.1111/j.1447-0594.2012.00851.x [PubMed]
- 58. Abbatecola AM, Paolisso G, Fattoretti P, Evans WJ, Fiore V, Dicioccio L, Lattanzio F. Discovering pathways of sarcopenia in older adults: a role for insulin resistance on mitochondria dysfunction. J Nutr Health Aging. 2011; 15:890–5. https://doi.org/10.1007/s12603-011-0366-0 [PubMed]
- 59. Petersen MC, Shulman GI. Mechanisms of Insulin Action and Insulin Resistance. Physiol Rev. 2018; 98:2133–223. https://doi.org/10.1152/physrev.00063.2017 [PubMed]
- 60. Laakso M, Kuusisto J. Insulin resistance and hyperglycaemia in cardiovascular disease development. Nat Rev Endocrinol. 2014; 10:293–302. https://doi.org/10.1038/nrendo.2014.29 [PubMed]
- 61. Wilmore JH, Stanforth PR, Gagnon J, Rice T, Mandel S, Leon AS, Rao DC, Skinner JS, Bouchard C. Heart rate and blood pressure changes with endurance training: the HERITAGE Family Study. Med Sci Sports Exerc. 2001; 33:107–16. https://doi.org/10.1097/00005768-200101000-00017 [PubMed]
- 62. Pedersen BK, Febbraio MA. Muscles, exercise and obesity: skeletal muscle as a secretory organ. Nat Rev Endocrinol. 2012; 8:457–65. https://doi.org/10.1038/nrendo.2012.49 [PubMed]
- 63. Parmacek MS. Myocardin-related transcription factors: critical coactivators regulating cardiovascular development and adaptation. Circ Res. 2007; 100:633–44. https://doi.org/10.1161/01.RES.0000259563.61091.e8 [PubMed]
- 64. Severinsen MCK, Pedersen BK. Muscle-Organ Crosstalk: The Emerging Roles of Myokines. Endocr Rev. 2020; 41:594–609. https://doi.org/10.1210/endrev/bnaa016 [PubMed]
- 65. Lee HS, Park KW, Kang J, Ki YJ, Chang M, Han JK, Yang HM, Kang HJ, Koo BK, Kim HS. Sarcopenia Index as a Predictor of Clinical Outcomes in Older Patients with Coronary Artery Disease. J Clin Med. 2020; 9:3121. https://doi.org/10.3390/jcm9103121 [PubMed]
- 66. Ferrucci L, Fabbri E. Inflammageing: chronic inflammation in ageing, cardiovascular disease, and frailty. Nat Rev Cardiol. 2018; 15:505–22. https://doi.org/10.1038/s41569-018-0064-2 [PubMed]
- 67. Stojanović SD, Fiedler J, Bauersachs J, Thum T, Sedding DG. Senescence-induced inflammation: an important player and key therapeutic target in atherosclerosis. Eur Heart J. 2020; 41:2983–96. https://doi.org/10.1093/eurheartj/ehz919 [PubMed]
- 68. Childs BG, Li H, van Deursen JM. Senescent cells: a therapeutic target for cardiovascular disease. J Clin Invest. 2018; 128:1217–28. https://doi.org/10.1172/JCI95146 [PubMed]
- 69. Ferrucci L, Penninx BW, Volpato S, Harris TB, Bandeen-Roche K, Balfour J, Leveille SG, Fried LP, Md JM. Change in muscle strength explains accelerated decline of physical function in older women with high interleukin-6 serum levels. J Am Geriatr Soc. 2002; 50:1947–54. https://doi.org/10.1046/j.1532-5415.2002.50605.x [PubMed]
- 70. Batsis JA, Mackenzie TA, Jones JD, Lopez-Jimenez F, Bartels SJ. Sarcopenia, sarcopenic obesity and inflammation: Results from the 1999-2004 National Health and Nutrition Examination Survey. Clin Nutr. 2016; 35:1472–83. https://doi.org/10.1016/j.clnu.2016.03.028 [PubMed]


