Introduction
Intravenous thrombolysis with alteplase is a standard therapy for patients who present with ischemic stroke symptoms within the first 4.5 h of onset in guidelines [1]. Tenecteplase (TNK), which is modified variant form alteplase by genetic recombinant technology, with greater fibrin specificity and a longer serum half-life is administered as a single bolus rather than as an infusion [2, 3]. Accumulating evidences from clinical trials has suggested benefits of TNK compared with alteplase for AIS [4–6]. A meta-analysis for fourteen studies, including retrospective trials and randomized controlled trials (RCTs), suggested that TNK is as safe as alteplase and may provide more effectiveness than alteplase in AIS patients [7]. Additionally, previous meta-analysis of 5 RCTs indicated that TNK is as safe and effective to alteplase in the intravenous thrombolysis treatment for AIS [8]. However, the safe and effective dose of TNK for patients with AIS still has not been determined clearly yet. Recently, a randomized trial found that compared with alteplase, treated with TNK at a dose of 0.4 mg/kg result in worse functional and safety outcomes for patients with AIS [9]. The controversy led to several RCTs have been conducted recently. Thus, we intend to conduct a pooled analysis of data from newly published RCTs to compare the efficacy and safety of TNK at different doses (0.1, 0.25, 0.4 mg/kg) with alteplase for the intravenous thrombolysis treatment of AIS patients in this study.
Methods
This study was performed following the prespecified protocol according to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines [10].
Data sources, inclusion and exclusionE criteria
Two investigators (X.Z., T.-F.W.) performed the literature search independently in PubMed, Embase, and ClinicalTrials.gov inception to April 17, 2023, using search strategy “alteplase” and “tenecteplase” and “stroke”. Only studies published in English language were searched. To avoid missing eligible studies, we retrieved all of the articles and also manually checked the references. The inclusion criteria were as follows: (1) randomized clinical trial; (2) allocation to TNK versus alteplase; (3) compared outcomes of TNK vs. alteplase for patients with AIS; (4) efficacy outcomes were analyzed by modified Rankin scale (mRS); (5) patients (aged ≥18 years) enrolled with acute ischemic stroke (in the anterior circulation or posterior circulation, with a premorbid score of two or less on the mRS also were included); (6) all the participants could be treated with TNK or alteplase within 6 hours after symptom onset. Studies with unavailable data to get effect estimates were excluded. Non-English-language studies, non-RCTs, duplicate reports, commentaries, fundamental experiment studies, abstracts, meeting proceedings were also excluded. To determine whether the data presented in the articles had been duplicated in other publications, we checked the data presented in the articles carefully and we excluded the subgroup analysis studies.
Data extraction and outcomes
Data were extracted and documented by two authors independently (T.-F.W., L.L.). Details recorded from each study include the name of study, study period, sources of data, sample size, TNK dose(s), and primary outcome. Any disagreements were resolved by consensus review.
The primary efficacy end point was excellent functional outcome (defined as patients who had a score of 0 or 1 on the mRS or a return to baseline for patients with a pre-stroke and the mRS score was 2) at 90 days. The mRS score is a 7-point ordered categorical scale from 0 to 6 for functional neurological outcome, with a lower score indicating independent living and 6 indicating death. Secondary outcomes were the following: good functional outcome at 90 days (defined as a mRS ≤2), symptomatic intracranial hemorrhage (sICH, according to the definition used in each study) and mortality at 90 days.
Quality assessment
Two authors (T.-F.W., L.L.) conducted the quality assessment of this study independently. We evaluated the risk of bias of all of included RCTs according to the Cochrane Collaboration’s tool and the Jadad Scale [11, 12]. The following seven domains were evaluated based on Cochrane Collaboration’s tool, including sequence generation (selection bias), allocation concealment (selection bias), blinding of participants and personnel (performance bias), blinding of outcome assessors (detection bias), incomplete outcome data (attrition bias), reporting biases (reporting bias), and other potential sources of bias. According to established criteria, the risk of bias in each domain would be scored low, unclear, or high. The study was defined as having a high (>4 high-risk domains), moderate (2–3 high-risk domains) or low (0–2 high-risk domains) risk of bias.
Statistics
Data synthesis and statistical analysis were conducted by STATA software, version 12.0 (StataCorp, USA). The results of meta-analysis were expressed as odds ratios (ORs) with corresponding 95% confidence intervals (95% CIs). The random-effects (DerSimonian-Laird method) or fixed-effects (Mantel-Haenszel method) meta-analysis model was used to calculate the results, and which meta-analysis model would be used in line with the heterogeneity among the included RCTs. Equivalent Z test was used to determine the statistical significance of pooled ORs and 95% CIs [13]. The heterogeneity among the included RCTs was evaluated using the P value of Chi-square-based Q-tests and the I-squared (I2) statistic [14]. Publication bias of included RCTs was evaluated using the Egger regression method [15], and showed by funnel plots. P value less than 0.05 was defined to have a statistical significance.
Availability of data and materials
The datasets used and/or analyzed during the present study are available from the corresponding author on reasonable request.
Results
Study selection and study characteristics
We identified 1085 potentially relevant citations from PubMed, Embase and https://clinicaltrials.gov/ (Figure 1). Of these, 173 duplicated articles were excluded. And we screen abstract and title to exclude 887 unrelated articles. Among the remaining 25 studies, 15 studies were excluded after read the full article and 10 studies with 5097 patients met the selection criteria [4, 5, 9, 16–22]. All eligible RCTs were conducted between 2006 and 2022. In eight of the trials, intravenous thrombolysis was conducted within 4.5 h of symptom onset while in a trial it was within 6 h, and one trial it was within 3 h. Patients were randomly assigned to TNK (at doses of 0.1, 0.25, 0.32 mg/kg) or alteplase at a dose of 0.9 mg/kg. All of the included studies had evaluated the mRS score at 90 days after treatment. The characteristics of included RCTs in this study were presented in Table 1.
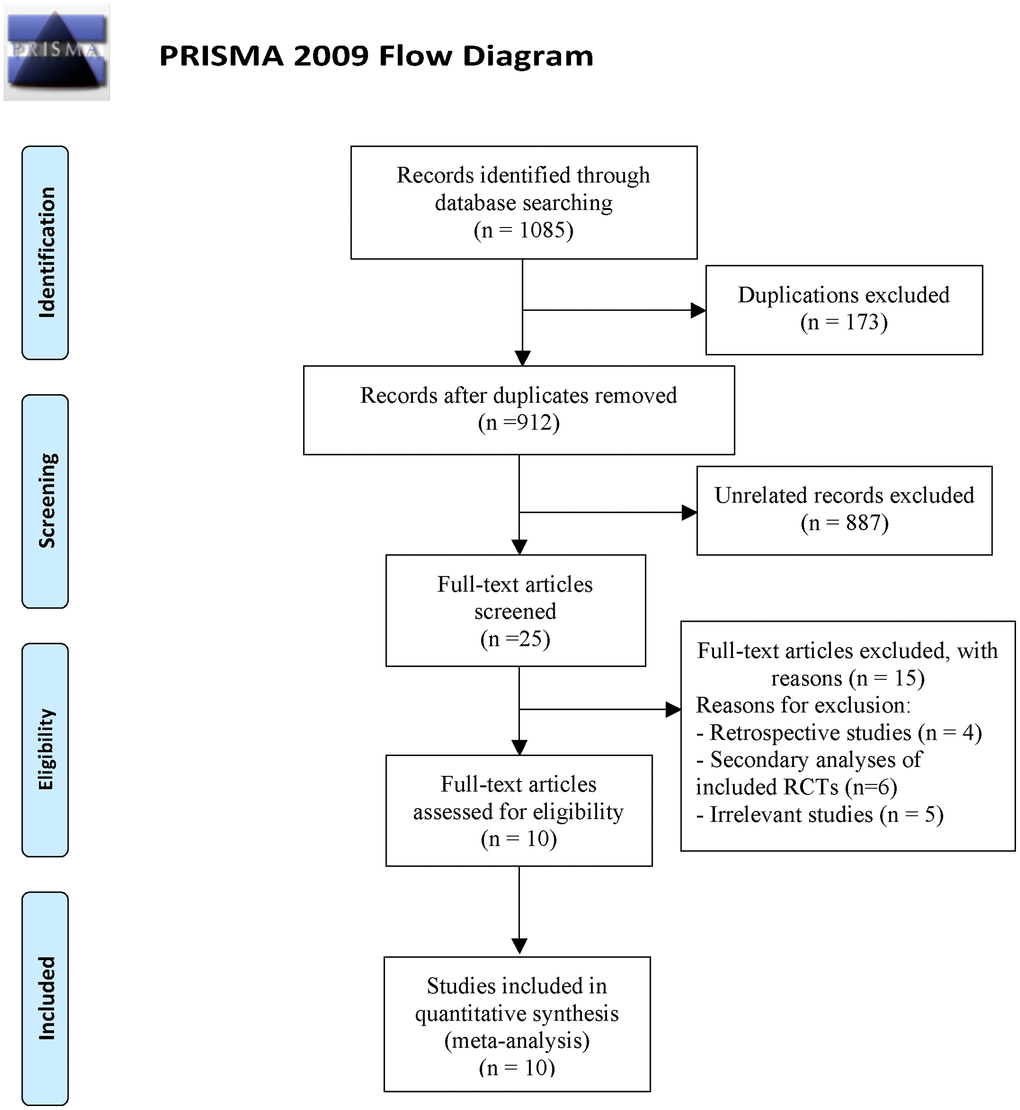
Figure 1. Flowchart of literature search and study selection.
Table 1. Characteristics of studies included in meta-analysis.
| Study | Study period | Country | Patients, n | TNK dose(s), mg/kg | Primary outcome | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TNK Phase IIB | 2006–2008 | United States | 112 | 0.1/0.25/0.4 | Functional Handicap (mRS) at 90 days | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Australian TNK | 2008–2011 | Australia | 75 | 0.1/0.25 | Achieving functional independence (mRS score, 0–2) at 90 days | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ATTEST | 2012–2013 | Scotland | 96 | 0.25 | Favorable outcome defined as mRS score 0–2 at 90 days after stroke onset | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| NOR-TEST | 2012–2016 | Norway | 1100 | 0.4 | Excellent (mRS: 0–1 points) functional outcome at 3 months | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| EXTEND-IA TNK | 2015–2017 | Australia and New Zealand | 202 | 0.25 | Reperfusion of greater than 50% of the involved ischemic territory or an absence of retrievable thrombus at the time of the initial angiographic assessment. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TRACE | 2018–2020 | China | 236 | 0.1/0.25/0.32 | Improvement on NIHSS of ≥4 points or a score ≤1 at day 14 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| NOR-TEST 2, part A | 2019–2021 | Norway | 204 | 0.4 | A favourable functional outcome at 3 months, defined as an mRS score of 0–1 or a return to baseline if the prestroke mRS score was 2. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TASTE-A | 2019–2021 | Australia | 104 | 0.25 | The volume of the perfusion lesion on CT-perfusion imaging performed on arrival at the receiving hospital | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AcT | 2019–2022 | Canada | 1567 | 0.25 | A score of 0 or 1 on the mRS at 90 days, up to 120 days after randomisation. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TRACE-2 | 2021–2022 | China | 1401 | 0.25 | Excellent (mRS: 0–1 points) functional outcome at 3 months | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: TNK: tenecteplase; mRS: modified Rankin Scale; CT: computed tomography. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quality assessment and publication bias
Overall, the quality of included RCTs were assessed with a low risk of bias (Additional file 1: Supplementary Table 1) according to the Jadad scale and Cochrane Risk of Bias Tool. In the domain of the blinding of participants and personnel, all of the included RCTs showed a high risk of bias for these included trials were blinded for outcomes assessor but not researchers and participants. No significant publication bias was found in the included RCTs (Egger’s test: P = 0.402, Supplemental Figure 1).
Primary outcome
All of the included trials with 5097 patients were included to analysis the primary outcome of excellent outcome (mRS score ≤1) at 90 days after treatment. Significantly different results were found in primary outcome to favor the TNK at 0.25 mg/kg (OR, 1.21 (95% CI, 1.04–1.54); P = 0.014), but 0.1 mg/kg (OR, 0.91 (95% CI, 0.54–1.54); P = 0.734) and 0.32 mg/kg (OR, 0.75 (95% CI, 0.39–1.45); P = 0.393) TNK groups compared with alteplase, respectively (Table 2). The I2 values were estimated as 0 both in 0.1 mg/kg and 0.25 mg/kg TNK groups, which indicate no obvious heterogeneity were found. Obvious heterogeneity (I2 = 73.7%) was observed in 0.4 mg/kg TNK groups, so the random-effect model was performed.
Table 2. Summary of pooled analyses for primary and secondary outcomes.
| Outcomes | TNK dose | Studies | Test of association | Heterogeneity | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OR (95% CI) | P value | Model | Z | Χ2 | P value | I2 (%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Primary outcome | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Excellent outcome | 0.1 mg | 3 | 0.91 (0.54–1.54) | 0.734 | FE | 0.34 | 0.26 | 0.880 | 0.0% | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 0.25 mg | 8 | 1.21 (1.04–1.54) | 0.014 | FE | 2.47 | 5.73 | 0.572 | 0.0% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 0.4 mg | 3 | 0.75 (0.39–1.45) | 0.393 | RE | 0.85 | 7.6 | 0.022 | 73.7% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Secondary outcomes | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Functional independence | 0.1 mg | 2 | 1.07 (0.56–2.04) | 0.839 | FE | 0.20 | 1.53 | 0.217 | 34.4% | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 0.25 mg | 7 | 1.13 (0.98–1.30) | 0.091 | FE | 1.77 | 9.64 | 0.141 | 37.8% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 0.4 mg | 2 | 0.65 (0.31–1.36) | 0.250 | RE | 1.15 | 5.23 | 0.022 | 80.9% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| sICH, n (%) | 0.1 mg | 3 | 0.81 (0.23–2.87) | 0.739 | FE | 0.33 | 2.27 | 0.322 | 11.7% | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 0.25 mg | 8 | 0.98 (0.79–1.20) | 0.817 | FE | 0.23 | 8.48 | 0.293 | 17.4% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 0.4 mg | 3 | 2.23 (1.04–4.80) | 0.040 | FE | 2.05 | 2.49 | 0.288 | 19.6% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mortality at 90 days, n (%) | 0.1 mg | 3 | 0.63 (0.23–1.70) | 0.359 | FE | 0.92 | 2.73 | 0.255 | 26.8% | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 0.25 mg | 8 | 1.04 (0.68–1.52) | 0.946 | FE | 0.07 | 2.12 | 0.908 | 0.0% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 0.4 mg | 3 | 1.37 (0.56–3.39) | 0.494 | RE | 0.68 | 5.15 | 0.076 | 61.2% | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: TNK: tenecteplase; OR: odds ratio; CI: confidence interval; RE: random effects; FE: fixed effects; sICH: symptomatic intracranial hemorrhage. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
In subgroup analysis according to geographic regions, we found that compared with alteplase, no significantly different results were found in the primary outcome (mRS score ≤1) to favor the 0.25 mg/kg TNK in different regions (including China, America, Europe and Australia. Additional file 1: Supplementary Table 2).
Secondary outcomes
The proportion of good outcome (mRS score 0–2) at 90 days after treatment were not significantly different between participants receiving TNK and alteplase (0.1 mg/kg TNK: OR, 1.07 (95% CI, 0.56–2.04); P = 0.839; 0.25 mg/kg TNK: OR, 1.13 (95% CI, 0.98–1.30); P = 0.091; 0.4 mg/kg TNK: OR, 0.65 (95% CI, 0.31–1.36); P = 0.250) (Table 2). No treatment group differences were found in the incidence of sICH between 0.1 mg/kg or 0.25 mg/kg TNK groups and the alteplase group (0.1 mg/kg TNK: OR, 0.81 (95% CI, 0.23–2.87); P = 0.739; 0.25 mg/kg TNK: OR, 0.98 (95% CI, 0.79–1.20); P = 0.817) (Table 2). However, it may be important to note that patients receiving TNK at a dose of 0.4 mg/kg with significantly higher incidence of sICH compared with alteplase group (OR, 2.23 (95% CI, 1.04–4.80); P = 0.040) (Table 2). Moreover, no significant differences were found between the groups in the rates of 90-day mortality (0.1 mg/kg TNK: OR, 0.63 (95% CI, 0.23–1.70); P = 0.359; 0.25 mg/kg TNK: OR, 1.04 (95% CI, 0.68–1.52); P = 0.946; 0.4 mg/kg TNK: OR, 1.37 (95% CI, 0.56–3.39); P = 0.494) (Table 2).
Discussion
In this meta-analysis of 10 RCTs, we comprehensively compared the effectiveness and safety between TNK and alteplase in patients with AIS eligible for thrombolysis. There was a significant difference were found in excellent outcome (mRS ≤ 1) as compared TNK at dose of 0.25 mg/kg with alteplase but TNK at dose of 0.1 mg/kg or 0.4 mg/kg. However, no significant differences were found in the rates of functional independence (mRS 0–2), or mortality between patients receiving TNK and alteplase. Moreover, compared with alteplase, TNK at dose of 0.1 mg/kg and 0.25 mg/kg did not appear to increased risk of sICH. Importantly, we noticed that the dose of 0.4 mg/kg TNK increase the incidence of sICH for patients with AIS compared with alteplase.
At present, although intravenous thrombolysis with alteplase remains the only approved choice for patients with AIS eligible for thrombolysis, current guidelines recommended that TNK might be considered as an alternative to alteplase [1, 23]. Our findings are in line with evidences from previous meta-analyses of RCTs, suggesting that no differences are found between any dose of TNK and alteplase for functional independence (0–2) and mortality at 90 days in patients with AIS [8, 24]. However, previous meta-analyses included fewer trials and comparisons of different dose tiers were very limited. In this study, total 10 RCTs were included and is the first to demonstrate that the dose of 0.4 mg/kg TNK appears unsafe for patients with AIS compared with alteplase when the accumulated evidences were collated. Importantly, our results showed that compared with alteplase, TNK at doses of 0.25 mg/kg may associated with the higher odds to achieve 90-day excellent independence (mRS score ≤1). Meanwhile, compared with alteplase, the pooled results from nonrandomized trials found that intravenous thrombolysis with TNK was associated with higher odds of good functional outcome (mRS 0–2) and early neurologic improvement for patients with AIS [9]. In addition, previous meta-analysis which included RCTs and nonrandomized trials showed that TNK might improve early neurologic function compared with alteplase for patients with AIS [7, 25]. Consistently, no statistical differences were found between the TNK and alteplase groups in the proportion of 3-month good functional outcome, sICH and mortality in these previous meta-analyses. Given the fact that absence of randomization and selection bias would be confounded the results of these studies, pooled data from RCTs in our study provided further evidences to resolve this controversy.
Although 0.9 mg/kg was the standard dose of intravenous alteplase in all studies, the dose of intravenous TNK was varied (0.1 mg/kg, 0.25 mg/kg, 0.32 mg/kg and 0.4 mg/kg) and has not been clearly determined. Total 8 RCTs included in this meta-analysis have compared TNK at dose of 0.25 mg/kg with alteplase in patients with AIS, and pooled results provide robust evidences for the comparative efficacy and safety of TNK at a dose of 0.25 mg/kg. Previous network meta-analysis, which found that TNK at a dose of 0.25 mg/kg showed better efficacy and imaging-based outcomes without increased risk of safety outcomes [26]. Moreover, the results of TRACE trial also showed that the 0.25 mg/kg dose tier showed better on excellent functional outcomes than 0.1 mg/kg and 0.32 mg/kg of TNK groups [19]. Among AIS patients with large vessel occlusion whom endovascular treatment is planned, EXTEND-IA TNK Part 2 trial demonstrated that TNK at a dose of 0.4 mg/kg did not provide any additional benefits compared with 0.25 mg/kg [27]. Importantly, NOR-TEST 2 part A trial was designed to demonstrate the non-inferiority of TNK at a dose of 0.4 mg/kg to alteplase for patients with AIS, but was stopped early for safety reasons [9]. This prematurely terminated study found that TNK at a dose of 0.4 mg/kg resulted in worse safety and less frequency of good functional outcomes compared with alteplase. Given the above evidences, TNK at a dose of 0.25 mg/kg might reduce risk of bleeding compared with the dose of 0.4 mg/kg, and showed better outcomes compared with a dose of 0.1 mg/kg, which could be the dose of choice for patients with AIS eligible for intravenous thrombolysis. Especially, CERTAIN trial has found that patients with AIS intravenous thrombolysis with TNK at a dose of 0.25 mg/kg showed lower risk of sICH than alteplase [28]. Currently, an ongoing trial, ATTEST 2 (ClinicalTrials.gov number, NCT02814409), were set to compare 0.25 mg/kg of TNK with alteplase 0.9 mg/kg with excellent functional outcome, which could provide further insight into the efficacy and safety of TNK at a dose of 0.25 mg/kg.
We must acknowledge that this study has limitations. First, the dose of intravenous TNK varied both within and between studies (0.1 mg/kg, 0.25 mg/kg, 0.32 mg/kg and 0.4 mg/kg). Fewer trials have evaluated the dose of 0.1 and 0.4 mg/kg of TNK, compared with the dose of 0.25 mg/kg. Thus, power to detect differences was constrained for 0.1 and 0.4 mg/kg dose. Second, we could not conduct some subgroup analysis for the lack of some of essential data in part of included trials (e.g., baseline NIHSS scores, occlusion site, cause of stroke, stroke onset to needle time, endovascular thrombectomy alone or combined IVT and endovascular thrombectomy and etc.). Third, the description of all adverse reactions mentioned in the included articles were various. Thus, we just analyzed the risk of sICH after intravenous thrombolysis with TNK or alteplase. Lastly, the time window for intravenous thrombolysis varied between studies, including patients with AIS eligible for intravenous thrombolysis within the first 3.0, 4.5 or 6.0 hours from stroke onset. Only one study which has reported the safety and efficacy of intravenous thrombolysis with TNK outside 4.5 hours from stroke onset [5]. Currently, three ongoing trials (TIMELESS trial: NCT03785678 [29], TEMPO-2 trial: NCT02398656 and CHABLIS-T: NCT04086147) are intended to investigate the efficacy and safety of TNK in patients with AIS in extended time window.
Conclusions
The pooled data from RCTs in this study provides supporting evidences that compare with alteplase, intravenous thrombolysis with TNK at dose of 0.25 mg/kg shows a better safety and similar efficacy profile and is a reasonable option for patients with AIS. Given the TNK at a dose of 0.4 mg/kg appears unsafe versus alteplase, a lower dose of TNK should be used for patients with AIS. However, these findings are mainly based on smaller sample size and due to several limitations of this study, additional multi-center RCTs to definitively address these issues are warranted.
Author Contributions
L.L. conceived the study. X.Z., T.F.W., and J.C. collected the data and drafted the manuscript. L.L. and X.Z. revised the manuscript and language.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
Funding
The work was supported by grants from the National Nature Science Foundation of China (No. 81901217) and China Postdoctoral Science Foundation (No. 2023M734297).
References
- 1. Powers WJ, Rabinstein AA, Ackerson T, Adeoye OM, Bambakidis NC, Becker K, Biller J, Brown M, Demaerschalk BM, Hoh B, Jauch EC, Kidwell CS, Leslie-Mazwi TM, et al. Guidelines for the Early Management of Patients With Acute Ischemic Stroke: 2019 Update to the 2018 Guidelines for the Early Management of Acute Ischemic Stroke: A Guideline for Healthcare Professionals From the American Heart Association/American Stroke Association. Stroke. 2019; 50:e344–418. https://doi.org/10.1161/STR.0000000000000211 [PubMed]
- 2. Tanswell P, Modi N, Combs D, Danays T. Pharmacokinetics and pharmacodynamics of tenecteplase in fibrinolytic therapy of acute myocardial infarction. Clin Pharmacokinet. 2002; 41:1229–45. https://doi.org/10.2165/00003088-200241150-00001 [PubMed]
- 3. Llevadot J, Giugliano RP, Antman EM. Bolus fibrinolytic therapy in acute myocardial infarction. JAMA. 2001; 286:442–9. https://doi.org/10.1001/jama.286.4.442 [PubMed]
- 4. Campbell BCV, Mitchell PJ, Churilov L, Yassi N, Kleinig TJ, Dowling RJ, Yan B, Bush SJ, Dewey HM, Thijs V, Scroop R, Simpson M, Brooks M, et al, and EXTEND-IA TNK Investigators. Tenecteplase versus Alteplase before Thrombectomy for Ischemic Stroke. N Engl J Med. 2018; 378:1573–82. https://doi.org/10.1056/NEJMoa1716405 [PubMed]
- 5. Parsons M, Spratt N, Bivard A, Campbell B, Chung K, Miteff F, O'Brien B, Bladin C, McElduff P, Allen C, Bateman G, Donnan G, Davis S, Levi C. A randomized trial of tenecteplase versus alteplase for acute ischemic stroke. N Engl J Med. 2012; 366:1099–107. https://doi.org/10.1056/NEJMoa1109842 [PubMed]
- 6. Bivard A, Huang X, McElduff P, Levi CR, Campbell BC, Cheripelli BK, Kalladka D, Moreton FC, Ford I, Bladin CF, Davis SM, Donnan GA, Muir KW, Parsons MW. Impact of Computed Tomography Perfusion Imaging on the Response to Tenecteplase in Ischemic Stroke: Analysis of 2 Randomized Controlled Trials. Circulation. 2017; 135:440–8. https://doi.org/10.1161/CIRCULATIONAHA.116.022582 [PubMed]
- 7. Ma P, Zhang Y, Chang L, Li X, Diao Y, Chang H, Hui L. Tenecteplase vs. alteplase for the treatment of patients with acute ischemic stroke: a systematic review and meta-analysis. J Neurol. 2022; 269:5262–71. https://doi.org/10.1007/s00415-022-11242-4 [PubMed]
- 8. Burgos AM, Saver JL. Evidence that Tenecteplase Is Noninferior to Alteplase for Acute Ischemic Stroke: Meta-Analysis of 5 Randomized Trials. Stroke. 2019; 50:2156–62. https://doi.org/10.1161/STROKEAHA.119.025080 [PubMed]
- 9. Kvistad CE, Næss H, Helleberg BH, Idicula T, Hagberg G, Nordby LM, Jenssen KN, Tobro H, Rörholt DM, Kaur K, Eltoft A, Evensen K, Haasz J, et al. Tenecteplase versus alteplase for the management of acute ischaemic stroke in Norway (NOR-TEST 2, part A): a phase 3, randomised, open-label, blinded endpoint, non-inferiority trial. Lancet Neurol. 2022; 21:511–9. https://doi.org/10.1016/S1474-4422(22)00124-7 [PubMed]
- 10. Moher D, Liberati A, Tetzlaff J, Altman DG, and PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ. 2009; 339:b2535. https://doi.org/10.1136/bmj.b2535 [PubMed]
- 11. Higgins JP, Altman DG, Gøtzsche PC, Jüni P, Moher D, Oxman AD, Savovic J, Schulz KF, Weeks L, Sterne JA, and Cochrane Bias Methods Group, and Cochrane Statistical Methods Group. The Cochrane Collaboration's tool for assessing risk of bias in randomised trials. BMJ. 2011; 343:d5928. https://doi.org/10.1136/bmj.d5928 [PubMed]
- 12. Jadad AR, Moore RA, Carroll D, Jenkinson C, Reynolds DJ, Gavaghan DJ, McQuay HJ. Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials. 1996; 17:1–12. https://doi.org/10.1016/0197-2456(95)00134-4 [PubMed]
- 13. Borenstein M, Higgins JP. Meta-analysis and subgroups. Prev Sci. 2013; 14:134–43. https://doi.org/10.1007/s11121-013-0377-7 [PubMed]
- 14. Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003; 327:557–60. https://doi.org/10.1136/bmj.327.7414.557 [PubMed]
- 15. Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997; 315:629–34. https://doi.org/10.1136/bmj.315.7109.629 [PubMed]
- 16. Haley EC
Jr , Thompson JL, Grotta JC, Lyden PD, Hemmen TG, Brown DL, Fanale C, Libman R, Kwiatkowski TG, Llinas RH, Levine SR, Johnston KC, Buchsbaum R, et al, and Tenecteplase in Stroke Investigators. Phase IIB/III trial of tenecteplase in acute ischemic stroke: results of a prematurely terminated randomized clinical trial. Stroke. 2010; 41:707–11. https://doi.org/10.1161/STROKEAHA.109.572040 [PubMed] - 17. Huang X, Cheripelli BK, Lloyd SM, Kalladka D, Moreton FC, Siddiqui A, Ford I, Muir KW. Alteplase versus tenecteplase for thrombolysis after ischaemic stroke (ATTEST): a phase 2, randomised, open-label, blinded endpoint study. Lancet Neurol. 2015; 14:368–76. https://doi.org/10.1016/S1474-4422(15)70017-7 [PubMed]
- 18. Logallo N, Novotny V, Assmus J, Kvistad CE, Alteheld L, Rønning OM, Thommessen B, Amthor KF, Ihle-Hansen H, Kurz M, Tobro H, Kaur K, Stankiewicz M, et al. Tenecteplase versus alteplase for management of acute ischaemic stroke (NOR-TEST): a phase 3, randomised, open-label, blinded endpoint trial. Lancet Neurol. 2017; 16:781–8. https://doi.org/10.1016/S1474-4422(17)30253-3 [PubMed]
- 19. Li S, Pan Y, Wang Z, Liang Z, Chen H, Wang D, Sui Y, Zhao X, Wang Y, Du W, Zheng H, Wang Y. Safety and efficacy of tenecteplase versus alteplase in patients with acute ischaemic stroke (TRACE): a multicentre, randomised, open label, blinded-endpoint (PROBE) controlled phase II study. Stroke Vasc Neurol. 2022; 7:47–53. https://doi.org/10.1136/svn-2021-000978 [PubMed]
- 20. Bivard A, Zhao H, Churilov L, Campbell BCV, Coote S, Yassi N, Yan B, Valente M, Sharobeam A, Balabanski AH, Dos Santos A, Ng JL, Yogendrakumar V, et al, and TASTE-A collaborators. Comparison of tenecteplase with alteplase for the early treatment of ischaemic stroke in the Melbourne Mobile Stroke Unit (TASTE-A): a phase 2, randomised, open-label trial. Lancet Neurol. 2022; 21:520–7. https://doi.org/10.1016/S1474-4422(22)00171-5 [PubMed]
- 21. Menon BK, Buck BH, Singh N, Deschaintre Y, Almekhlafi MA, Coutts SB, Thirunavukkarasu S, Khosravani H, Appireddy R, Moreau F, Gubitz G, Tkach A, Catanese L, et al, and AcT Trial Investigators. Intravenous tenecteplase compared with alteplase for acute ischaemic stroke in Canada (AcT): a pragmatic, multicentre, open-label, registry-linked, randomised, controlled, non-inferiority trial. Lancet. 2022; 400:161–9. https://doi.org/10.1016/S0140-6736(22)01054-6 [PubMed]
- 22. Wang Y, Li S, Pan Y, Li H, Parsons MW, Campbell BCV, Schwamm LH, Fisher M, Che F, Dai H, Li D, Li R, Wang J, et al, and TRACE-2 Investigators. Tenecteplase versus alteplase in acute ischaemic cerebrovascular events (TRACE-2): a phase 3, multicentre, open-label, randomised controlled, non-inferiority trial. Lancet. 2023; 401:645–54. https://doi.org/10.1016/S0140-6736(22)02600-9 [PubMed]
- 23. Berge E, Whiteley W, Audebert H, De Marchis GM, Fonseca AC, Padiglioni C, de la Ossa NP, Strbian D, Tsivgoulis G, Turc G. European Stroke Organisation (ESO) guidelines on intravenous thrombolysis for acute ischaemic stroke. Eur Stroke J. 2021; 6:I–LXII. https://doi.org/10.1177/2396987321989865 [PubMed]
- 24. Huang X, MacIsaac R, Thompson JL, Levin B, Buchsbaum R, Haley EC
Jr , Levi C, Campbell B, Bladin C, Parsons M, Muir KW. Tenecteplase versus alteplase in stroke thrombolysis: An individual patient data meta-analysis of randomized controlled trials. Int J Stroke. 2016; 11:534–43. https://doi.org/10.1177/1747493016641112 [PubMed] - 25. Oliveira M, Fidalgo M, Fontão L, Antão J, Marques S, Afreixo V, Gregório T. Tenecteplase for thrombolysis in stroke patients: Systematic review with meta-analysis. Am J Emerg Med. 2021; 42:31–7. https://doi.org/10.1016/j.ajem.2020.12.026 [PubMed]
- 26. Kheiri B, Osman M, Abdalla A, Haykal T, Ahmed S, Hassan M, Bachuwa G, Al Qasmi M, Bhatt DL. Tenecteplase versus alteplase for management of acute ischemic stroke: a pairwise and network meta-analysis of randomized clinical trials. J Thromb Thrombolysis. 2018; 46:440–50. https://doi.org/10.1007/s11239-018-1721-3 [PubMed]
- 27. Campbell BCV, Mitchell PJ, Churilov L, Yassi N, Kleinig TJ, Dowling RJ, Yan B, Bush SJ, Thijs V, Scroop R, Simpson M, Brooks M, Asadi H, et al, and EXTEND-IA TNK Part 2 investigators. Effect of Intravenous Tenecteplase Dose on Cerebral Reperfusion Before Thrombectomy in Patients With Large Vessel Occlusion Ischemic Stroke: The EXTEND-IA TNK Part 2 Randomized Clinical Trial. JAMA. 2020; 323:1257–65. https://doi.org/10.1001/jama.2020.1511 [PubMed]
- 28. Warach SJ, Ranta A, Kim J, Song SS, Wallace A, Beharry J, Gibson D, Cadilhac DA, Bladin CF, Kleinig TJ, Harvey J, Palanikumar L, Doss VT, et al. Symptomatic Intracranial Hemorrhage With Tenecteplase vs Alteplase in Patients With Acute Ischemic Stroke: The Comparative Effectiveness of Routine Tenecteplase vs Alteplase in Acute Ischemic Stroke (CERTAIN) Collaboration. JAMA Neurol. 2023; 80:732–8. https://doi.org/10.1001/jamaneurol.2023.1449 [PubMed]
- 29. Albers GW, Campbell BC, Lansberg MG, Broderick J, Butcher K, Froehler MT, Schwamm LH, Nouh AM, Liebeskind DS, Toy F, Yang M, Massaro L, Schoeffler M, Purdon B. A Phase III, prospective, double-blind, randomized, placebo-controlled trial of thrombolysis in imaging-eligible, late-window patients to assess the efficacy and safety of tenecteplase (TIMELESS): Rationale and design. Int J Stroke. 2023; 18:237–41. https://doi.org/10.1177/17474930221088400 [PubMed]


