Introduction
Glioblastoma (GBM) is the most common and aggressive malignant brain tumor in the central nervous system. GBM treatment typically involves surgery, complemented by chemotherapy and radiotherapy [1, 2]. However, due to its multifocal nature and high invasiveness, complete surgical resection is often challenging, leading to a high recurrence rate and a median survival of only 14.6 months post-diagnosis [3]. Consequently, developing novel and effective therapeutic methods for GBM is imperative.
Calycosin (CA), a major bioactive compound extracted from Radix astragali, is an isoflavonoid and phytoestrogen [4]. It has demonstrated various pharmacological properties, including anti-inflammatory, neuroprotective, and cardiovascular effects [5–7]. Recent studies have highlighted its anti-tumor activities; for instance, calycosin suppresses breast cancer via the downregulation of the Foxp3/VEGF/MMP-9 signaling pathway [8]. Additionally, it induces apoptosis in osteosarcoma through the ERβ-mediated PI3K/Akt signaling pathways [9]. Notably, Nie et al. reported calycosin’s anti-GBM effects through TGFβ inhibition [10, 11]. However, the underlying mechanisms remain to be fully elucidated.
Chemokines, small secretory immunoregulatory proteins, regulate cell trafficking through interactions with seven-transmembrane G protein-coupled receptors (GPCRs) and are implicated in oncogenesis, including tumor development and metastatic spread. CXCL10, also known as interferon-gamma-induced protein 10 (IP-10), is a 10 kDa polypeptide of the CXC chemokine subfamily. It plays multiple roles, such as mediating immune cell chemotaxis, angiogenesis, and inflammation response [12–14]. Recent studies have linked CXCL10 to the progression of various cancers, including pancreatic, breast, ovarian, and colorectal cancer [15–18]. However, its role as either a tumor suppressor or contributor in glioma remains controversial.
Inflammation significantly promotes tumorigenesis onset and progression [19], as observed in GBM, where it facilitates cancer progression and treatment resistance [20]. The NOD-like receptor protein-3 (NLRP3) inflammasome, excessively activated in various cancers including glioma, and its suppression has been shown to reduce tumor growth and prolong survival in glioma-bearing mice. The NLRP3 downstream effectors, IL-1β and NF-κB, are abundantly present in the tumor microenvironment of glioblastomas, contributing to their development [21–23]. However, the relationship between CXCL10 and NLRP3 is not well understood, and whether CA can modulate CXCL10 to inhibit GBM remains unknown.
In this study, we demonstrated that high CXCL10 expression is associated with higher pathological grades and poorer prognosis. We also identified CXCL10 as a potential therapeutic target for CA in GBM suppression, via downregulating downstream molecules such as NLRP3, NF-κB, and IL-1β, both in vivo and in vitro. These findings suggest that CXCL10 is a novel biomarker in glioblastoma, and calycosin may serve as a potential therapeutic agent.
Results
CXCL10 was upregulated in glioma tissues and contributes to poor prognosis
Our study evaluated the mRNA expression of CXCL10 using public data from The Cancer Genome Atlas (TCGA) and the GTEx database, as well as 24 pairs of glioma tissue samples. Analysis of the TCGA database revealed a significant increase in CXCL10 mRNA expression correlating with higher WHO glioma grades (Figure 1A). Additionally, CXCL10 levels were notably higher in glioma tissues compared to normal tissues (Figure 1B). High CXCL10 expression was associated with markedly poorer overall survival (OS) in patients (Figure 1C). Similarly, in our 24 paired human glioma samples, CXCL10 expression increased with glioma grades (Figure 1D, 1E and Supplementary Table 1) and was more pronounced in glioma than in adjacent normal brain tissues (Figure 1F, 1G). Patients with high CXCL10 expression also exhibited shorter survival periods (Figure 1H). These findings suggest a positive correlation between high CXCL10 expression and poor prognosis in glioma patients.
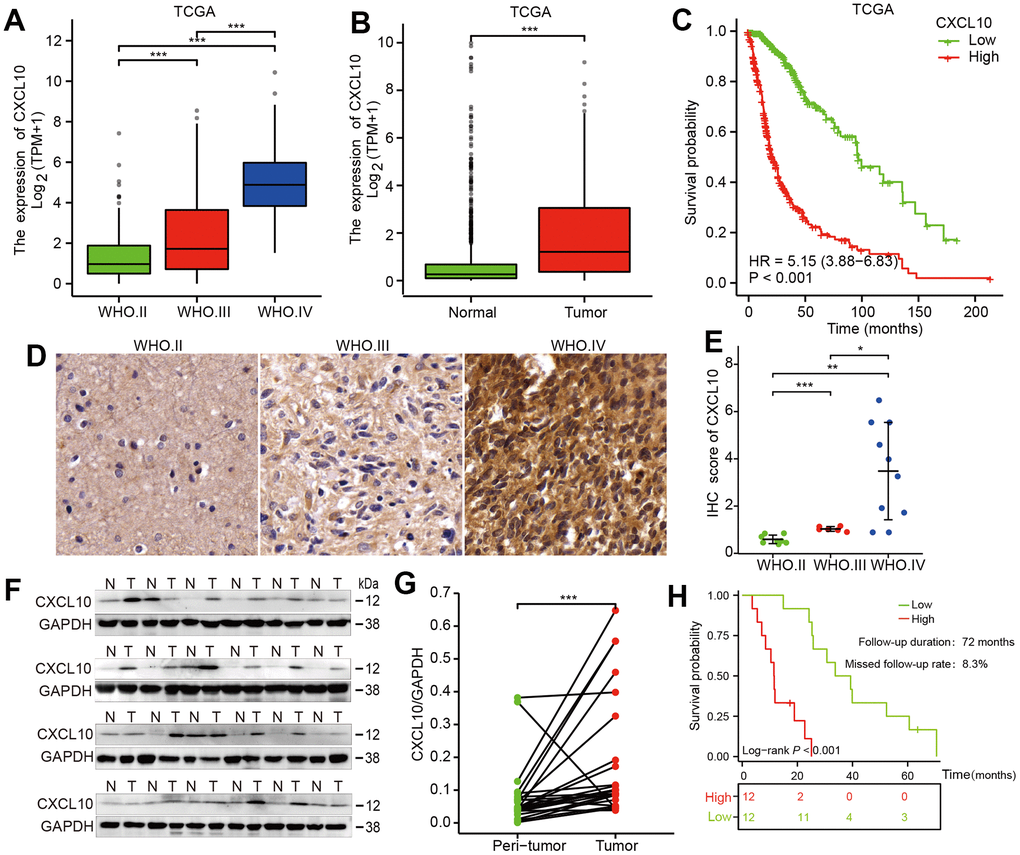
Figure 1. CXCL10 is upregulated in glioma tissues and positively associated with tumor progression and poor prognosis. (A) Statistical evaluation of CXCL10 expression across different grades of glioma in the TCGA database. (B) analysis of CXCL10 expression in GBM tumor tissues compared to normal brain tissues using data from the TCGA database. (C) Survival curves showing the overall survival (OS) stratified by CXCL10 expression levels in glioma patients, based on TCGA data. (D, E) Comparative analysis of CXCL10 expression in 24 paired tumor and peritumor tissues from glioma patients. (F, G) examination of CXCL10 expression in GBM tumor tissues compared to normal brain tissues in 24 glioma cases. (H) OS analysis in a cohort of 24 glioma patients stratified by CXCL10 expression.
CA inhibited cells growth and downregulated CXCL10 signaling in GBM cells
To evaluate CA’s potential effects on GBM, CCK-8 assays were conducted. Results showed that CA treatment significantly inhibited cell proliferation in U87 and U251 GBM cells in a dose- and time-dependent manner, with cell viability decreasing from 90% to 40% as CA concentration increased from 100 to 400 μM. This effect was not observed in HNA (Human Normal Astrocytes) (Figure 2A–2C). Colony formation assays further confirmed CA’s inhibitory effect on U87 and U251 cell growth (Figure 2D, 2E). Cell migration and invasion assays demonstrated that calycosin treatment significantly reduced these activities in GBM cells in a dose-dependent manner (Figure 2F–2I). These results indicate that calycosin effectively inhibits GBM cell migration and invasion.
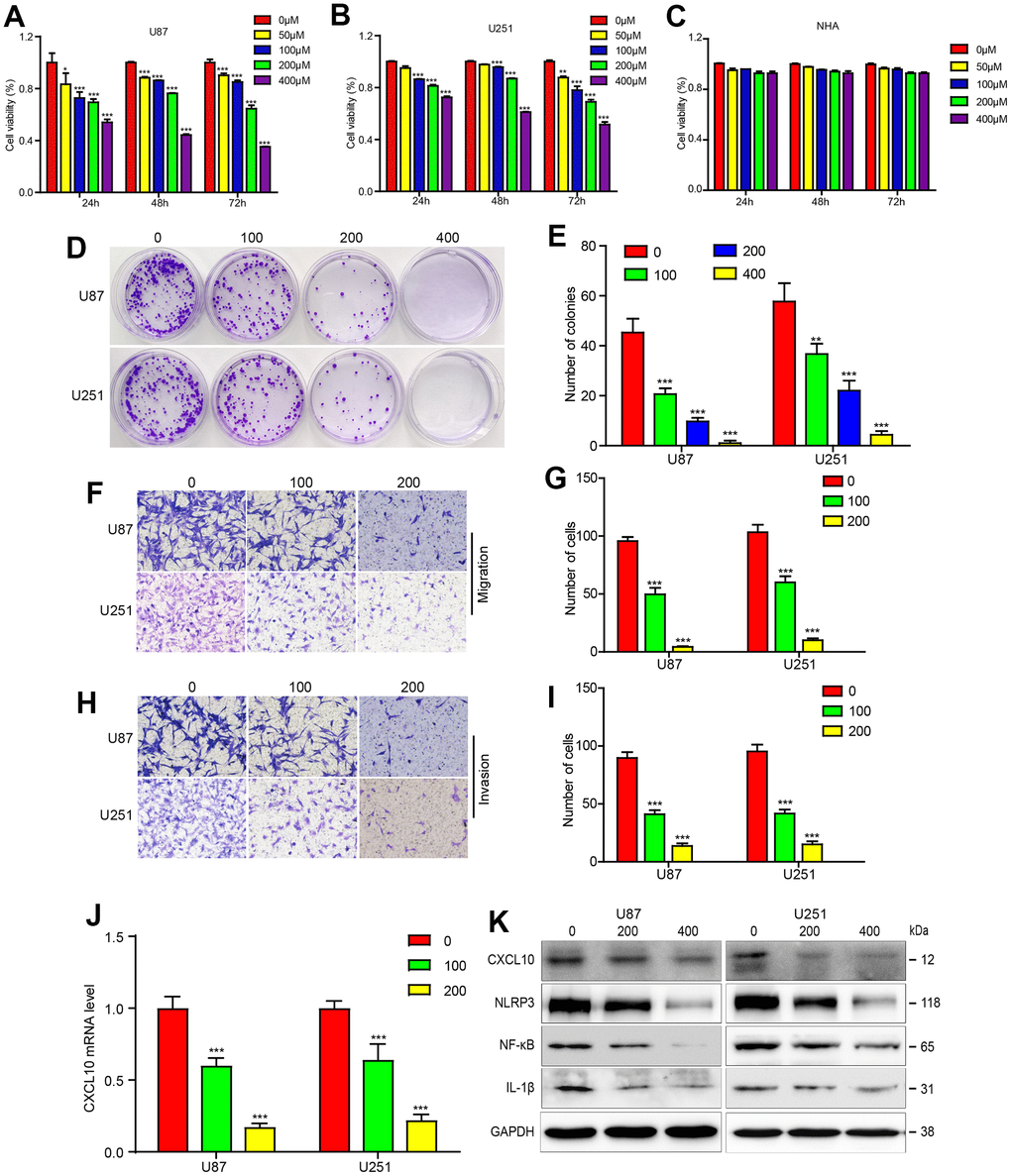
Figure 2. Calycosin suppressed GBM progression and downregulated CXCL10 pathway in U87 and U251 cells. (A–C) The CCK8 experiment were performed in U87, U251 and HNA cells with the different concentrations of calycosin. (D, E) Cell colony formation in glioblastoma cells with CA treatments as indicated dose. (F–I) U87 and U251 cells were treated with the indicated dose of CA for 24 hours, and transwell assay was applied to examine the migration and invasion. (J) RT-PCR was applied to detect the mRNA level after indicated dose of CA. (K) U87 and U251 cells were performed with the different dose of CA for 24 hours and western bolt was applied to detect the protein expression levels of CXCL10 pathway. *P<0.05, **P<0.01, ***P<0.001, compared with control (0μM).
As CXCL10 is upregulated in GBM tissues and contributes to tumor progression and poor prognosis, inhibiting CXCL10 could be a viable treatment approach. Treatment with varying concentrations of CA downregulated CXCL10 expression at both mRNA and protein levels, reducing downstream molecules such as NLRP3, NF-κB, and IL-1β (Figure 2J, 2K).
Overexpression of CXCL10 reduced the effects of calycosin in GBM cells
To determine if calycosin’s suppression of GBM cells is mediated through downregulating CXCL10, U87 and U251 cells were infected with a lentiviral vector carrying CXCL10 cDNA. Overexpression of CXCL10 enhanced cell proliferation, migration, and invasion, and reduced the inhibitory effects of CA (Figure 3A–3H). Overexpressed CXCL10 also promoted NLRP3, NF-κB, and IL-1β expression, countering the suppressive effects of calycosin treatment (Figure 3I).
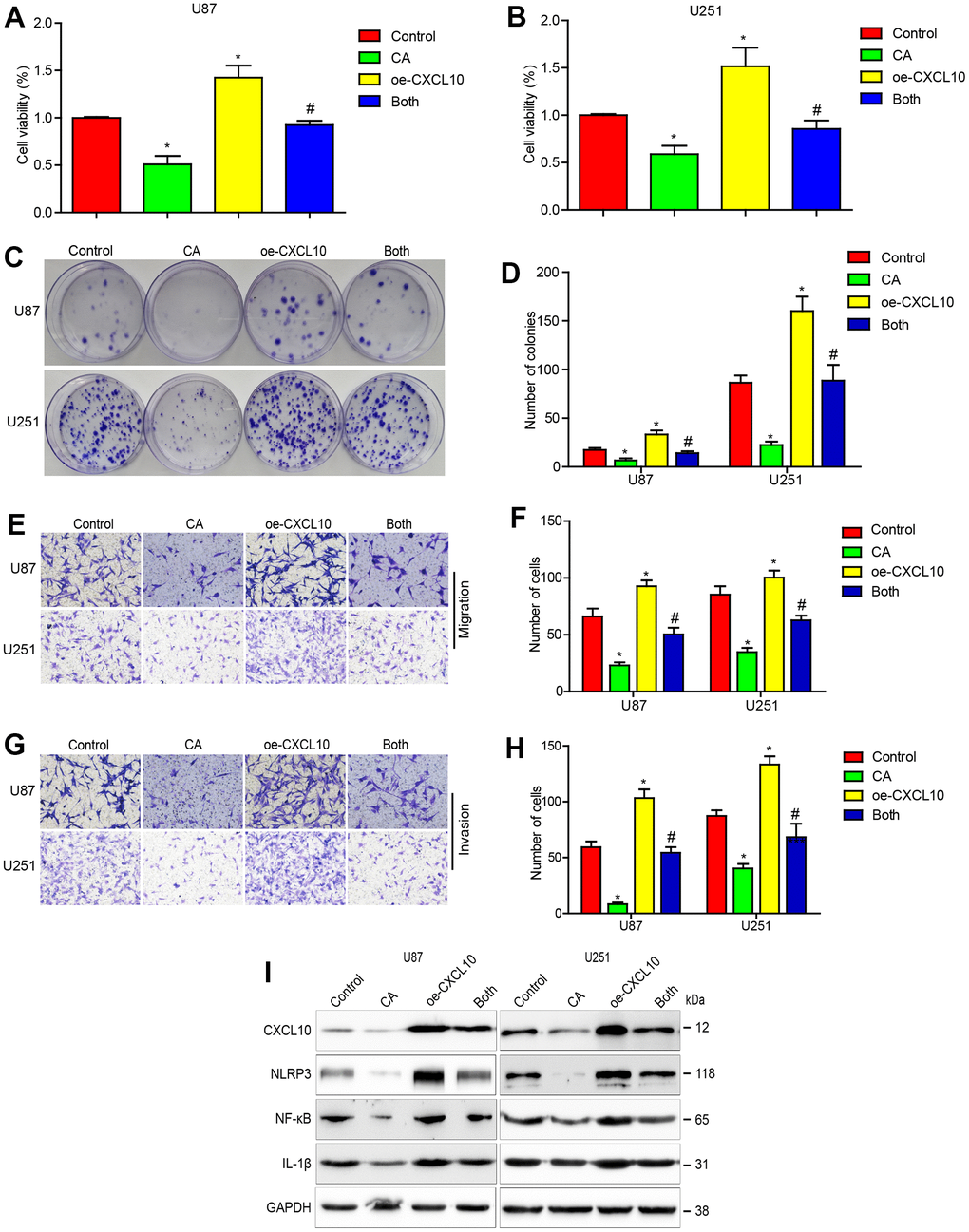
Figure 3. CXCL10 overexpression reduces the effects of calycosin on cells proliferation, migration, invasion and CXCL10 signaling in GBM. (A–D) Effect of CXCL10 overexpression on calycosin in U87 and U251 cell proliferation. (E–H) Effect of CXCL10 overexpression on calycosin in U87 and U251 cell migration and invasion. (I) Overexpressing CXCL10 rescues calycosin-induced CXCL10, NLRP3, NF-κB and IL-1β downregulation. Control: GFP lentivirus transfection; CA: GFP lentivirus transfection + 200μM calycosin; oe-CXCL10: lentivirus transfection CXCL10; Both: lentivirus transfection CXCL10 + 200μM calycosin. *P < 0.05 vs control. #P < 0.05, compared with either CA treatment or CXCL10 transfection alone.
Down-regulation of CXCL10 enhanced the effects of calycosin in GBM cells
To further confirm CXCL10’s oncogenic role in CA-mediated anticancer effects, CXCL10 was knocked down in glioblastoma cells using specific siRNA. CCK-8 and colony formation assays showed enhanced suppression of cell proliferation following CXCL10 knockdown in calycosin-treated cells (Figure 4A–4D). Cells treated with CXCL10 siRNA or calycosin exhibited potent inhibition of migration and invasion (Figure 4E–4H). This combination treatment showed greater efficacy than either CXCL10 siRNA or calycosin alone. Calycosin treatment also decreased the expression of NLRP3, NF-κB, and IL-1β, with further suppression observed upon CXCL10 downregulation (Figure 4I). These results confirm calycosin’s anticancer function, partly through downregulating CXCL10.
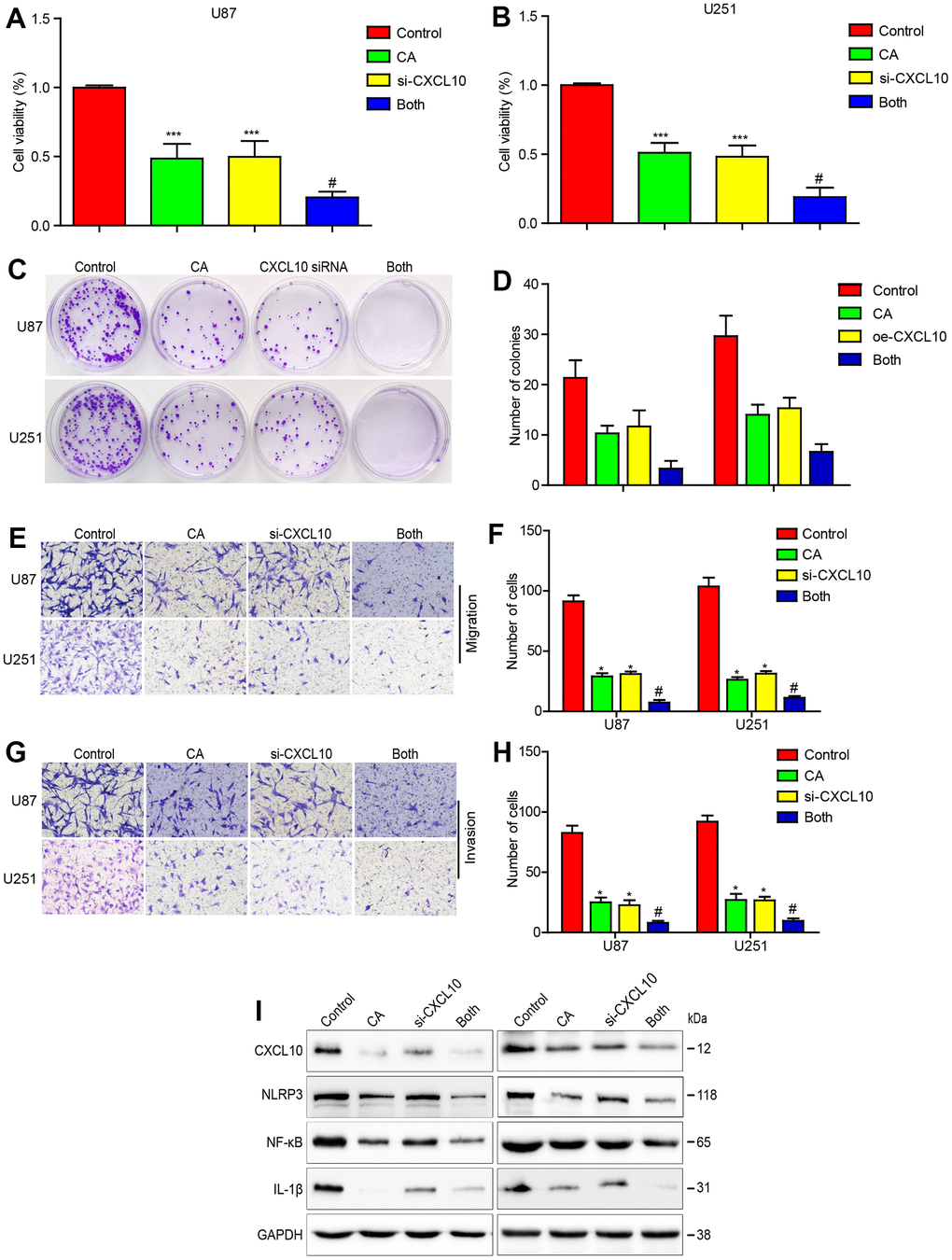
Figure 4. Downregulating CXCL10 enhances the effects of calycosin on cell proliferation, migration, invasion and CXCL10 signaling in GBM. (A–D) Effects of CXCL10 knockdown on calycosin in U87 and U251 cell proliferation. (E–H) Effects of downregulating CXCL10 on calycosin in U87 and U251 cell migration and invasion. (I) Downregulating CXCL10 promotes calycosin-induced CXCL10, NLRP3, NF-κB and IL-1β downregulation. Control: siRNA negative control transfection; CA: siRNA negative control transfection + 200μM calycosin; CXCL10: CXCL10 siRNA transfection; both: CXCL10 siRNA transfection + 200μM calycosin. *P < 0.05 vs control. #P < 0.05, compared with either CA treatment or CXCL10 siRNA alone.
Calycosin inhibited GBM growth in a U87 xenograft mouse model
The inhibitory effects of calycosin on glioblastoma progression were further explored in a U87 xenograft mouse model. Calycosin-treated mice exhibited significantly smaller tumor volumes compared to the vehicle-treated group, without changes in body weight (Figure 5A–4D). CXCL10 expression and its pathway components, NLRP3, NF-κB, and IL-1β, were markedly suppressed in tumor tissues from the calycosin-treated mice (Figure 5E). These in vivo results corroborate our in vitro findings, demonstrating calycosin’s critical role in suppressing glioblastoma growth through CXCL10 inhibition.
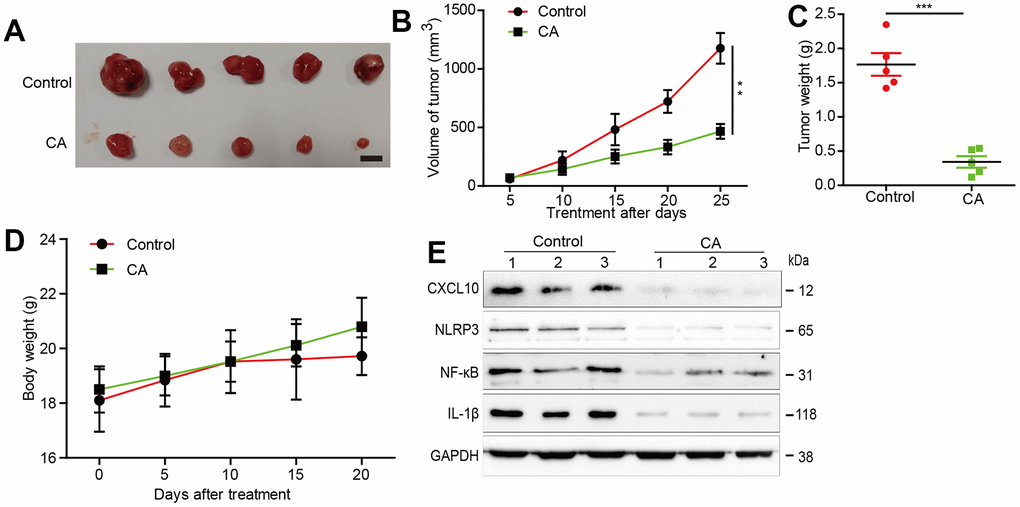
Figure 5. Effects of calycosin in xenograft mouse models. (A) Inhibition in the size of the xenograft U87 tumors were photographed. (B) The tumor volume and body weight were measured per 5 days. (C) At the end of the experiments, tumor was excised from the mice and the weigh is measured. (D) The mice’s body weight was measure at the indicated time point. (E) At the end of the experiment or after the mice dead, tumor tissues were excised from the mice and the protein lysates were performed to estimated protein expression *P<0.05, compared with control. Scale =1cm.
Calycosin’s interaction with CXCL10
In order to evaluate the potential interaction between calycosin and CXCL10, we conducted molecular docking experiments. These experiments demonstrated that calycosin exhibits a notable binding affinity to CXCL10, characterized by a binding energy of -6.8 kcal/mol. The interaction is primarily mediated through the formation of hydrogen bonds with amino acids LEU-65, VAL-68, and LEU-24 (Figure 6). These findings indicate a stable binding conformation and suggest that calycosin may have an inhibitory effect on CXCL10 activity.
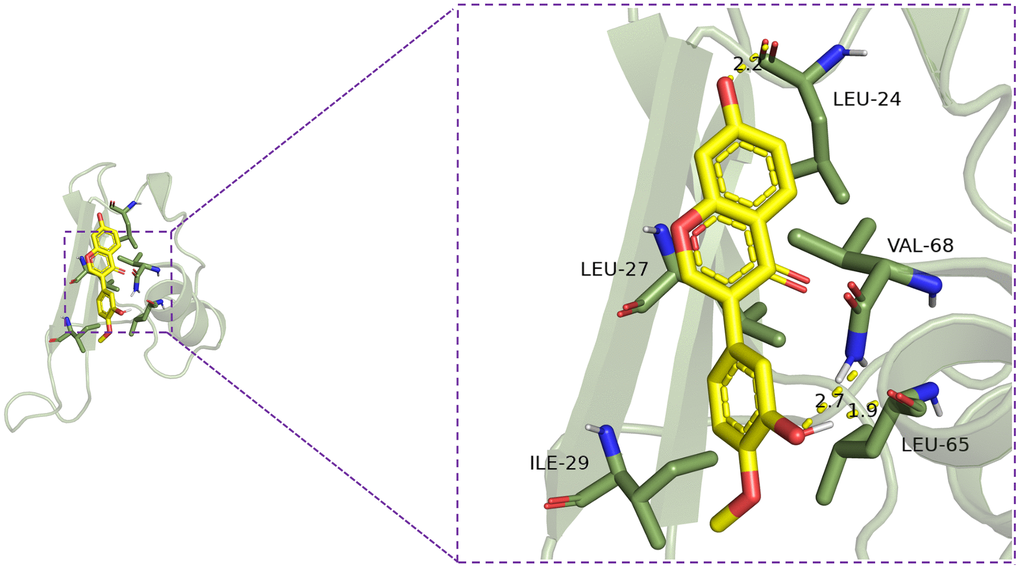
Figure 6. Calycosin’s interaction with CXCL10.
Discussion
In this study, we demonstrated that overexpressing CXCL10 promotes GBM progression. Furthermore, we showed that calycosin inhibits growth, migration, and invasion in GBM by downregulating the CXCL10 pathway in vitro. We also confirmed the anti-tumor effect of calycosin in a GBM xenograft mouse model, aligning with our in vitro findings.
The current standard therapy for GBM primarily involves maximal safe surgical resection, supplemented by chemoradiotherapy. However, complete eradication of glioblastoma is challenging due to its multifocal and aggressive invasion [1]. Therefore, the development of new therapeutic agents is urgently needed. In this context, we established that calycosin, a bioactive compound extracted from Radix astragali, exerts its anti-tumor function by downregulating CXCL10 expression in GBM.
Calycosin has been recognized for its anti-tumor effects in various cancers, including glioma [8–10]. Previous studies have shown that calycosin inhibits GBM progression by downregulating TGFβ and c-Met [10, 11]. Additionally, Ni Q et al. reported that combining calycosin with temozolomide enhances anti-glioma effects [24]. However, the precise molecular target of calycosin in GBM remains unclear. Our research reveals that calycosin suppresses GBM growth both in vitro and in vivo by inhibiting the CXCL10 signaling pathway, suggesting CXCL10 as a potential target for GBM treatment.
CXCL10 plays a crucial role in regulating multiple inflammatory signaling pathways and is intimately linked to the onset and development of various tumors. However, its role in GBM progression is debated. Maru, SV et al. reported that overexpressing CXCL10 significantly influences the proliferation of glioma cells [25]. Additionally, previous studies using immunohistochemical staining have shown CXCL10 overexpression in GBM [26]. Kenji Shono et al. found that CXCL10 downregulation contributes to anti-tumor effects in a malignant glioma mouse model. These findings suggest that CXCL10 is instrumental in GBM growth and progression, and its inhibition may serve as a potential therapeutic target for glioblastoma [27]. Contrarily, other studies have indicated that upregulating CXCL10 could inhibit glioma progression [28, 29]. In our current study, we demonstrated high CXCL10 expression in glioma, negatively correlating with patient prognosis. This supports the notion of CXCL10 as an oncogene in glioma. Furthermore, we identified CXCL10 as a potential target for calycosin, which downregulates the CXCL10-related inflammatory signaling pathway to suppress GBM progression.
Numerous agents show anti-GBM effects in vitro but are ineffective in GBM orthotopic xenograft mouse models due to the blood-brain barrier (BBB). Previous research has shown calycosin’s therapeutic effects in cerebral ischemic and brain reperfusion injury models, along with a favorable safety profile [5]. In this study, we found that calycosin inhibited glioblastoma growth by downregulating the CXCL10 signaling pathway in a subcutaneous GBM model, consistent with our in vitro findings. Significantly, calycosin markedly reduced tumor volume without affecting body weight, validating its safety and efficacy in vivo.
The molecular docking data suggest a potential interaction between calycosin and CXCL10, marked by a strong binding affinity. Such an interaction implies that calycosin could modulate the activity of CXCL10, which may have therapeutic implications. However, these in silico findings necessitate further empirical validation. Subsequent experiments, including in vitro and in vivo studies, are essential to confirm the binding and to understand the impact of calycosin on CXCL10’s biological functions.
Conclusions
Overall, we have demonstrated that CXCL10 functions as an oncogene and identified it as a potential therapeutic target for calycosin in glioma treatment.
Materials and Methods
Reagents
Calycosin was obtained from Tianjin Wanxiang Hengyuan Science and Technology Ltd., Tianjin, China. Dulbecco’s Modified Eagle Medium (DMEM) and Fetal Bovine Serum (FBS) were procured from Gibco™. Antibodies against CXCL10, NLRP3, and IL-1β were sourced from Cell Signaling Technology (MA, USA), while antibodies for NF-κB were acquired from Wuhan Servicebio Technology Co. Ltd., Wuhan, China.
Patients and sample
Specimens of tumor and adjacent tissues were collected from 24 patients in the Second Affiliated Hospital of Guangzhou Medical University, who had undergone curative surgery from 2015 to 2018 in our hospital, which was approved by the Institutional Ethics Committee in the Second Affiliated Hospital of Guangzhou Medical University. Proteins (24 pairs) were isolated from frozen tumor tissues and adjacent tissues for western blotting assay to assess the expression of CXCL10. Written informed consents were acquired from each patient relying on guidelines of the Declaration of Helsinki. The inclusion and exclusion criteria for collecting glioma samples may include the following:
Inclusion criteria:
Confirmed diagnosis of glioma;
Patient consent to participate in the study;
Not having received radiation, chemotherapy, or surgical treatment, or a certain time interval after treatment.
Exclusion criteria:
Presence of other diseases or history of diseases, such as autoimmune diseases, malignant tumors, etc.;
Use of drugs that may affect the study results, such as steroids, anti-inflammatory drugs, etc.;
Have significant organ diseases such as heart, liver, kidney, etc.;
Pregnant or lactating women.
Cell culture
Human GBM cell lines U87, U251, and HNA were obtained from iCell Bioscience Inc., Shanghai, China. These lines were cultured in DMEM supplemented with 10% FBS and maintained at 37° C in a humidified 5% CO2 atmosphere.
Cell viability assay
4000 cells per well were seeded in a 96-well plate and treated with varying calycosin concentrations for 24 hours. Subsequently, 10 μl of CCK-8 solution (Beyotime, Shanghai, China) was added, and incubation continued for 1 hour at 37° C. Absorbance was measured using a Multimode Reader.
Colony formation assay
500 cells per well were plated in 60mm dishes and treated with different calycosin concentrations for two weeks. Colonies were fixed with methanol and stained using Crystal Violet Staining Solution (Beyotime, Shanghai, China).
Cell invasion assay
For the cell migration and invasion assay, a transwell system (Corning, NY, USA) was used. 2×105 cells per well in 200 μl DMEM (1% FBS) were seeded in the upper chamber (8 μm Pore Polycarbonate Membrane) coated with 100 μl Matrigel (BD Biosciences, CA, USA). The lower chamber was filled with 600 μl DMEM (20% FBS) and different calycosin concentrations. After 24 hours, cells in the lower chamber were fixed with methanol and stained using Crystal Violet Staining Solution (Beyotime, Shanghai, China). Cells were photographed in five independent fields per well at 100× magnification and counted.
Reverse transcription polymerase chain reaction (RT-PCR)
Total cellular RNA was extracted using Trizol reagent (Sigma-Aldrich). First-strand cDNA synthesis was performed using a PrimeScripTM RT reagent Kit, followed by PCR with Taq DNA polymerase (Takara, Dalian, China) and specific primers: human CXCL10: 5’-GTGGCATTCAAGGAGTACCTC-3’ (forward) and 5’-GCCTTCGATTCTGGATTCAGACA-3’ (reverse); human GAPDH: 5’-GGAGCGAGATCCCTCCAAAAT-3’ (forward) and 5’-GGCTGTTGTCATACTTCTCATGG-3’ (reverse).
Western blotting
Western blotting was conducted on glioblastoma cell lysates, clinical normal or tumor tissues, and xenograft glioblastoma tissue homogenates. Protein extraction was performed using PRO-PREP™ Protein Extraction Solution (Cell/Tissue) (iNtRON Biotechnology, Korea), following the manufacturer’s instructions. Equal protein amounts were separated by 10–12% SDS-PAGE and transferred onto polyvinylidene difluoride membranes (Merck, KGaA, Darmstadt, Germany). Membranes were blocked with 5% BSA for 1 hour at room temperature, then incubated with primary antibodies overnight at 4° C. Secondary antibodies conjugated with HRP were applied for 1 hour at room temperature, and signals detected using Immobilon Western HRP Substrate (Merck, KGaA, Darmstadt, Germany).
Transfection
To overexpress CXCL10, glioblastoma cell lines were infected with a lentiviral vector carrying CXCL10-flag or its control vector (OBiO Technology Corp., Ltd, Shanghai, China). For CXCL10 knockdown, cells were transfected with CXCL10 siRNA or scrambled siRNA (GenePharma, Shanghai, China) using jetPRIME® (Polyplus Transfection, France), according to the manufacturer's protocol. The specific siRNA sequence targeting CXCL10: sense 5ʹ-CCUUAUCUUUUCUGACUCUATT-3ʹ; antisense 3ʹ-UAGAGUCAGAAAGAUAAGGTT-5ʹ.
U87 xenograft mouse model calycosin treatment
Female BALB/c nude mice were obtained from Guangdong Medical Laboratory Animal Center (Guangdong, China). Mice were aged 6–8 weeks and kept under a standard protocol approved by the Laboratory Animal Center of the Second Affiliated Hospital of Guangzhou Medical University. All procedures performed in studies involving animals were in accord with the ethical standards of the Ethics Committee and the IRB number is 2021-ks-15. Each mouse was injected subcutaneously with cultured U87 cells (5×106 cells/mouse) into the dorsum. The tumor size was measured in two orthogonal directions using calipers, and the tumor volume (mm3) was calculated using the equation: 1/2×length×width2. When the tumors grew to about 150 mm3, the tumor-bearing mice were distributed into two groups (n=5 each) and orally fed with calycosin (10mg/kg/day) or vehicle (equivalent amount of PBS) Tumor sizes and body weights were measured once every 5 days. At the end of these experiments, the mice were sacrificed and the tumors were resected and homogenized for western blotting.
Molecular docking
Calycosin was designed using ChemBio3D Ultra 14.0, with an initial structure obtained from PubChem based on its CAS number. Energy minimization was performed, and the structure was saved in “mol2” and subsequently “pdbqt” format after processing with AutodockTools-1.5.6. The CXCL10 protein was prepared by removing crystallization water and original ligands using Pymol 2.3.0, followed by charge computation and atom typing with AutoDocktools, and saved in “pdbqt” format. Binding sites were predicted using POCASA 1.1, and molecular docking was executed with AutoDock Vina 1.1.2, where the search space dimensions were set to a grid of 60x60x60 with a grid spacing of 0.375Å and an exhaustiveness of 10. Finally, the docking result was analyzed for interaction mode using PyMOL 2.3.0, allowing for a detailed visualization and understanding of the molecular interactions between calycosin and CXCL10 protein.
Statistical analysis
Data represent three independent experiments and were analyzed using SPSS 20.0 software. Simple comparisons between two groups were performed using independent T-tests, while multiple group comparisons utilized one-way ANOVA, followed by post hoc Dunnett’s T3 or Tukey’s tests. A p-value < 0.05 was considered statistically significant.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplementary Materials
Author Contributions
Rong Xu, Lu Chen and Maoying Zhang designed the study, Xiaoyu Zheng performed the western blotting, CCK8 assay, cell culture and RT-PCR, Danmin Chen conducted the colony formation assay, cell invasion assay, transfection and collected the patient’s samples, Menghui Li performed the molecular docking, Jianchen Liao and Liqun He performed the in vivo experiments; all authors contributed to drafting the manuscript and reviewed the manuscript.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Ethical Statement and Consent
Animal Experiments: This study involves experiments on animals and has been reviewed and approved by the Institutional Animal Care and Use Committee (IACUC) of The Second Affiliated Hospital of Guangzhou Medical University. The approval number for these experiments is B2019-001. This approval confirms that the research project adheres to ethical standards and humane treatment of animals in research. The animal use permit number is SYXK2018-0192, indicating compliance with relevant regulations and ethical guidelines for animal welfare.
Human Participants: Research involving human participants was conducted following the ethical standards of the institutional and national research committee. It adheres to the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The project was reviewed and approved by the Clinical Research and Application Ethics Committee of The Second Affiliated Hospital of Guangzhou Medical University. The acceptance number for this study is 2021-ks-15. Informed consent was obtained from all individual participants included in the study. For those participants where obtaining informed consent was not possible, the research Ethics Committee provided a waiver due to the non-invasive nature of the study and the public interest in the research outweighing the need for individual consent.
Funding
This work was supported by the National Natural Science Foundation of China (81500985), the Specialized Research and Cultivation Fund of Affiliated Shunde Hospital of Jinan University (202101006), Guangzhou Health Science and Technology Project (20191A010063), Guangzhou TCM and Integrated Traditional Chinese and Western Medicine project (20192A010017) and Natural Science Foundation of Guangdong Province (2019A1515010926).
References
- 1. Batash R, Asna N, Schaffer P, Francis N, Schaffer M. Glioblastoma Multiforme, Diagnosis and Treatment; Recent Literature Review. Curr Med Chem. 2017; 24:3002–9. https://doi.org/10.2174/0929867324666170516123206 [PubMed]
- 2. Anjum K, Shagufta BI, Abbas SQ, Patel S, Khan I, Shah SAA, Akhter N, Hassan SS. Current status and future therapeutic perspectives of glioblastoma multiforme (GBM) therapy: A review. Biomed Pharmacother. 2017; 92:681–9. https://doi.org/10.1016/j.biopha.2017.05.125 [PubMed]
- 3. Taylor OG, Brzozowski JS, Skelding KA. Glioblastoma Multiforme: An Overview of Emerging Therapeutic Targets. Front Oncol. 2019; 9:963. https://doi.org/10.3389/fonc.2019.00963 [PubMed]
- 4. Gao J, Liu ZJ, Chen T, Zhao D. Pharmaceutical properties of calycosin, the major bioactive isoflavonoid in the dry root extract of Radix astragali. Pharm Biol. 2014; 52:1217–22. https://doi.org/10.3109/13880209.2013.879188 [PubMed]
- 5. Guo C, Tong L, Xi M, Yang H, Dong H, Wen A. Neuroprotective effect of calycosin on cerebral ischemia and reperfusion injury in rats. J Ethnopharmacol. 2012; 144:768–74. https://doi.org/10.1016/j.jep.2012.09.056 [PubMed]
- 6. Dong L, Yin L, Chen R, Zhang Y, Hua S, Quan H, Fu X. Anti-inflammatory effect of Calycosin glycoside on lipopolysaccharide-induced inflammatory responses in RAW 264.7 cells. Gene. 2018; 675:94–101. https://doi.org/10.1016/j.gene.2018.06.057 [PubMed]
- 7. Pan Q, Ban Y, Khan S. Antioxidant activity of calycosin against α-synuclein amyloid fibrils-induced oxidative stress in neural-like cells as a model of preventive care studies in Parkinson’s disease. Int J Biol Macromol. 2021; 182:91–7. https://doi.org/10.1016/j.ijbiomac.2021.03.186 [PubMed]
- 8. Li S, Wang Y, Feng C, Wu G, Ye Y, Tian J. Calycosin Inhibits the Migration and Invasion of Human Breast Cancer Cells by Down-Regulation of Foxp3 Expression. Cell Physiol Biochem. 2017; 44:1775–84. https://doi.org/10.1159/000485784 [PubMed]
- 9. Tian W, Wang ZW, Yuan BM, Bao YG. Calycosin induces apoptosis in osteosarcoma cell line via Erβ-mediated PI3K/Akt signaling pathways. Mol Med Rep. 2020; 21:2349–56. https://doi.org/10.3892/mmr.2020.11039 [PubMed]
- 10. Nie XH, Ou-yang J, Xing Y, Li DY, Liu RE, Xu RX. Calycosin inhibits migration and invasion through modulation of transforming growth factor beta-mediated mesenchymal properties in U87 and U251 cells. Drug Des Devel Ther. 2016; 10:767–79. https://doi.org/10.2147/DDDT.S90457 [PubMed]
- 11. Nie X, Zhou Y, Li X, Xu J, Pan X, Yin R, Lu B. Calycosin down-regulates c-Met to suppress development of glioblastomas. J Biosci. 2019; 44:96. https://doi.org/10.1007/s12038-019-9904-4 [PubMed]
- 12. Lee EY, Lee ZH, Song YW. CXCL10 and autoimmune diseases. Autoimmun Rev. 2009; 8:379–83. https://doi.org/10.1016/j.autrev.2008.12.002 [PubMed]
- 13. Yates-Binder CC, Rodgers M, Jaynes J, Wells A, Bodnar RJ, Turner T. An IP-10 (CXCL10)-derived peptide inhibits angiogenesis. PLoS One. 2012; 7:e40812. https://doi.org/10.1371/journal.pone.0040812 [PubMed]
- 14. Zhao Q, Kim T, Pang J, Sun W, Yang X, Wang J, Song Y, Zhang H, Sun H, Rangan V, Deshpande S, Tang H, Cvijic ME, et al. A novel function of CXCL10 in mediating monocyte production of proinflammatory cytokines. J Leukoc Biol. 2017; 102:1271–80. https://doi.org/10.1189/jlb.5A0717-302 [PubMed]
- 15. Hirth M, Gandla J, Höper C, Gaida MM, Agarwal N, Simonetti M, Demir A, Xie Y, Weiss C, Michalski CW, Hackert T, Ebert MP, Kuner R. CXCL10 and CCL21 Promote Migration of Pancreatic Cancer Cells Toward Sensory Neurons and Neural Remodeling in Tumors in Mice, Associated With Pain in Patients. Gastroenterology. 2020; 159:665–81.e13. https://doi.org/10.1053/j.gastro.2020.04.037 [PubMed]
- 16. Goldberg-Bittman L, Neumark E, Sagi-Assif O, Azenshtein E, Meshel T, Witz IP, Ben-Baruch A. The expression of the chemokine receptor CXCR3 and its ligand, CXCL10, in human breast adenocarcinoma cell lines. Immunol Lett. 2004; 92:171–8. https://doi.org/10.1016/j.imlet.2003.10.020 [PubMed]
- 17. Bronger H, Singer J, Windmüller C, Reuning U, Zech D, Delbridge C, Dorn J, Kiechle M, Schmalfeldt B, Schmitt M, Avril S. CXCL9 and CXCL10 predict survival and are regulated by cyclooxygenase inhibition in advanced serous ovarian cancer. Br J Cancer. 2016; 115:553–63. https://doi.org/10.1038/bjc.2016.172 [PubMed]
- 18. Chen J, Chen QL, Wang WH, Chen XL, Hu XQ, Liang ZQ, Cao YB, Cao YM, Su SB. Prognostic and predictive values of CXCL10 in colorectal cancer. Clin Transl Oncol. 2020; 22:1548–64. https://doi.org/10.1007/s12094-020-02299-6 [PubMed]
- 19. Karki R, Kanneganti TD. Diverging inflammasome signals in tumorigenesis and potential targeting. Nat Rev Cancer. 2019; 19:197–214. https://doi.org/10.1038/s41568-019-0123-y [PubMed]
- 20. Cho A, McKelvey KJ, Lee A, Hudson AL. The intertwined fates of inflammation and coagulation in glioma. Mamm Genome. 2018; 29:806–16. https://doi.org/10.1007/s00335-018-9761-8 [PubMed]
- 21. Shang S, Wang L, Zhang Y, Lu H, Lu X. The Beta-Hydroxybutyrate Suppresses the Migration of Glioma Cells by Inhibition of NLRP3 Inflammasome. Cell Mol Neurobiol. 2018; 38:1479–89. https://doi.org/10.1007/s10571-018-0617-2 [PubMed]
- 22. Yin XF, Zhang Q, Chen ZY, Wang HF, Li X, Wang HX, Li HX, Kang CM, Chu S, Li KF, Li Y, Qiu YR. NLRP3 in human glioma is correlated with increased WHO grade, and regulates cellular proliferation, apoptosis and metastasis via epithelial-mesenchymal transition and the PTEN/AKT signaling pathway. Int J Oncol. 2018; 53:973–86. https://doi.org/10.3892/ijo.2018.4480 [PubMed]
- 23. Sharma BR, Kanneganti TD. NLRP3 inflammasome in cancer and metabolic diseases. Nat Immunol. 2021; 22:550–9. https://doi.org/10.1038/s41590-021-00886-5 [PubMed]
- 24. Ni Q, Fan Y, Zhang X, Fan H, Li Y. In vitro and in vivo Study on Glioma Treatment Enhancement by Combining Temozolomide with Calycosin and Formononetin. J Ethnopharmacol. 2019; 242:111699. https://doi.org/10.1016/j.jep.2019.01.023 [PubMed]
- 25. Maru SV, Holloway KA, Flynn G, Lancashire CL, Loughlin AJ, Male DK, Romero IA. Chemokine production and chemokine receptor expression by human glioma cells: role of CXCL10 in tumour cell proliferation. J Neuroimmunol. 2008; 199:35–45. https://doi.org/10.1016/j.jneuroim.2008.04.029 [PubMed]
- 26. Sharma I, Siraj F, Sharma KC, Singh A. Immunohistochemical expression of chemokine receptor CXCR3 and its ligand CXCL10 in low-grade astrocytomas and glioblastoma multiforme: A tissue microarray-based comparison. J Cancer Res Ther. 2016; 793–7. https://doi.org/10.4103/0973-1482.153657 [PubMed]
- 27. Shono K, Yamaguchi I, Mizobuchi Y, Kagusa H, Sumi A, Fujihara T, Nakajima K, Kitazato KT, Matsuzaki K, Saya H, Takagi Y. Downregulation of the CCL2/CCR2 and CXCL10/CXCR3 axes contributes to antitumor effects in a mouse model of malignant glioma. Sci Rep. 2020; 10:15286. https://doi.org/10.1038/s41598-020-71857-3 [PubMed]
- 28. Wang P, Peng X, Zhang J, Wang Z, Meng J, Cen B, Ji A, He S. LncRNA-135528 inhibits tumor progression by up-regulating CXCL10 through the JAK/STAT pathway. Apoptosis. 2018; 23:651–66. https://doi.org/10.1007/s10495-018-1482-7 [PubMed]
- 29. Takano S, Ishikawa E, Matsuda M, Yamamoto T, Matsumura A. Interferon-β inhibits glioma angiogenesis through downregulation of vascular endothelial growth factor and upregulation of interferon inducible protein 10. Int J Oncol. 2014; 45:1837–46. https://doi.org/10.3892/ijo.2014.2620 [PubMed]



