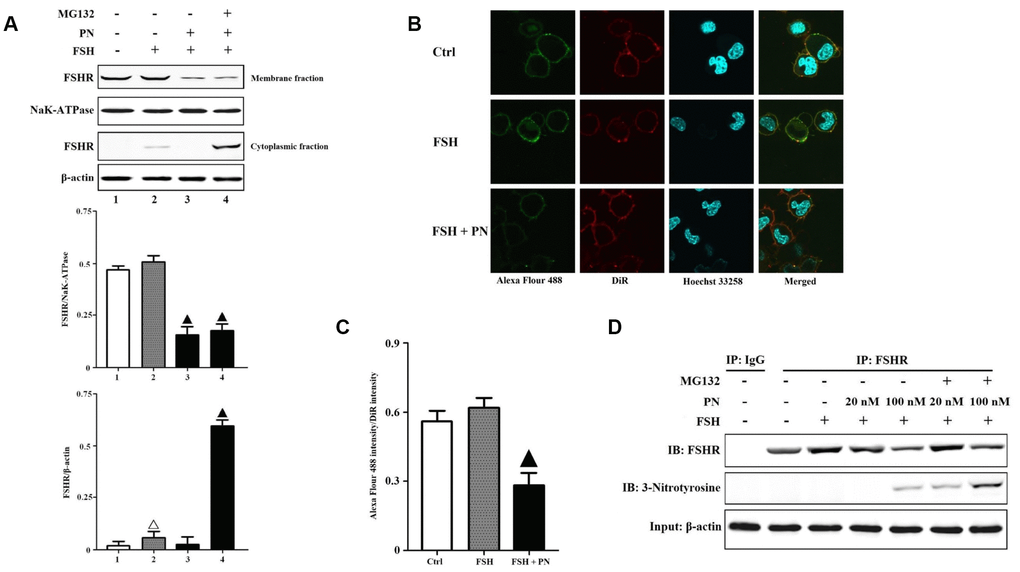
Figure 2.PN-mediated tyrosine nitrations of FSHR abolished the membrane anchoring of FSHR and induced its degradation. (A) PN impaired the membrane expression of FSHR and induced its degradation. KGN cells were incubated with or without PN (100 nM, 12 hrs), MG132 (30 μM, 4 hrs) followed by treatment with FSH (1 nM, 4 hrs). Relative protein expressions of FSHR in membrane fractions (NaK-ATPase as internal standard of membrane proteins) and cytoplasmic fractions (β-actin as internal standard of cytoplasmic proteins) were determined by immunoblots. (B) PN impaired the membrane anchoring of FSHR. KGN cells were incubated with or without PN (100 nM, 12 hrs), followed by treatment with FSH (1 nM, 4 hrs). Representative photo-micrographs of Alexa Flour 488 staining in KGN cells were examined by confocal microscopy, where Green fluorescence indicated Alexa Flour 488-positive FSHR proteins, red fluorescence indicated DiR-positive membrane, and blue fluorescence indicated Hoechst 33258-positive nuclei (Scale bar: 10 μm). (C) Relative ratios of FSHR to membrane were determined as the ratios of Alexa Flour 488 density to DiR intensity. (D) PN induced the protein nitrations of FSHR. KGN cells were incubated with or without PN (20 nM/100 nM, 12 hrs) and MG132 (30 μM, 4 hrs) followed by treatment with FSH (1 nM, 4 hrs). FSHR proteins were purified from cell lysates. The endogenous FSHR complex were analyzed by immunoblots (β-actin as internal standard). Open triangle: p<0.05 vs. Ctrl; Bold triangle: p<0.05 vs. FSH (n = 3–6).