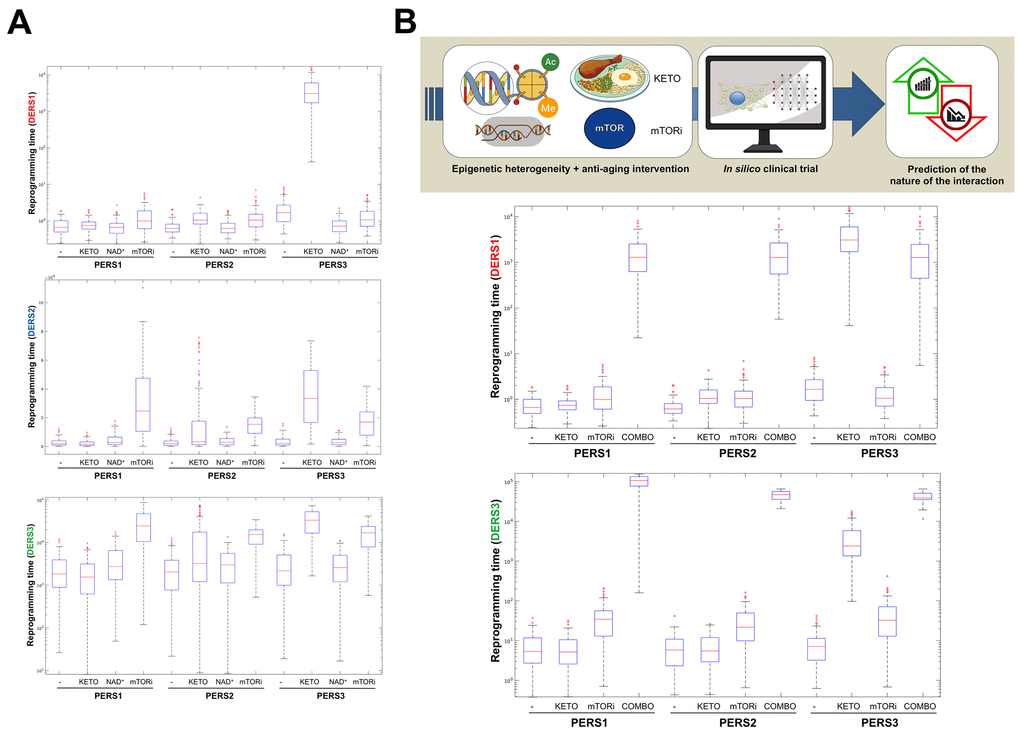
Figure 3.Baseline epigenetic heterogeneity and efficacy of anti-aging strategies: A proof-of-concept in silico trial. We have recently presented a stochastic biomathematical modeling and computational simulation strategy that might be incorporated as a valuable tool for assessing the benefit/risk ratio of therapeutic approaches aimed to target the aging/cancer-related perturbations of the epigenome [35]. Briefly, we quantified the heterogeneity and robustness of differentiation epigenetic and pluripotency regulatory systems (DERS and PERS, respectively) in terms of the average reprogramming time associated with differentiation-primed (benefit) and pluripotency-locked (risk) states. Such calculation can therefore be employed as a proxy of the expected efficacy of a given anti-aging strategy (i.e., longer reprogramming times associate with more efficient anti-aging outcomes). Regarding DERS heterogeneity, we observed three different clusters associated with a differentiated-primed behavior (DERS1), a differentiation-refractory (stem-like) behavior (DERS2), and an indecision behavior (DERS3). Several kinetic parameters associated with histone deacetylase (HDAC, c11) and histone acetylase (HAC, c15 and c16) activities sufficed to account for the distinction between DERS clusters [35]. Regarding PERS heterogeneity, we observed three different clusters defined by the large (PERS1), intermediate (PERS2), and small (PERS3) values of the average waiting time needed for transitioning from closed to open ER states [35]. As a proof of concept, here we considered nine combinatorial scenarios corresponding to all the possible combinations between three different differentiation DERS and PERS challenged with three different anti-aging strategies (A), namely ketogenic diet (mimicked by reducing by 50% the kinetic parameter associated with HDAC activity [i.e., c11]), NAD+ boosters (mimicked by increasing by 50% the kinetic parameter associated with HDAC activity [i.e., c11]), and mTOR inhibition (mimicked by reducing by 25% the two parameters associated with HAC activity [i.e., c15 and c16]). Whereas ketogenic diet- and mTOR inhibition-like strategies were notably found to be more efficient than NAD+ boosting-like approaches across all the baseline epigenetic scenarios, it was noteworthy that the most sensitive one was that defined by the combination of PERS3 with DERS1 (A). Based on these results, we decided to assess the direction and intensity of the average reprogramming time when combining ketogenic diet- and mTOR inhibition-like strategies (B). With the exception of baseline scenarios where the strong single-agent positive outcome left little room for additional gains, we observed that strong, synergistic interactions tend to be more specific to particular epigenetic states than were single strategies (B). Our in silico approach exemplifies how baseline epigenetic heterogeneity might dictate not only the positive outcomes of single aging-targeted therapies such as ketogenic diet and mTOR inhibition but also the degree of positive synergistic effects that were predicted to occur upon concurrent targeting of an apparently existing HDAC-mTOR cross-link.