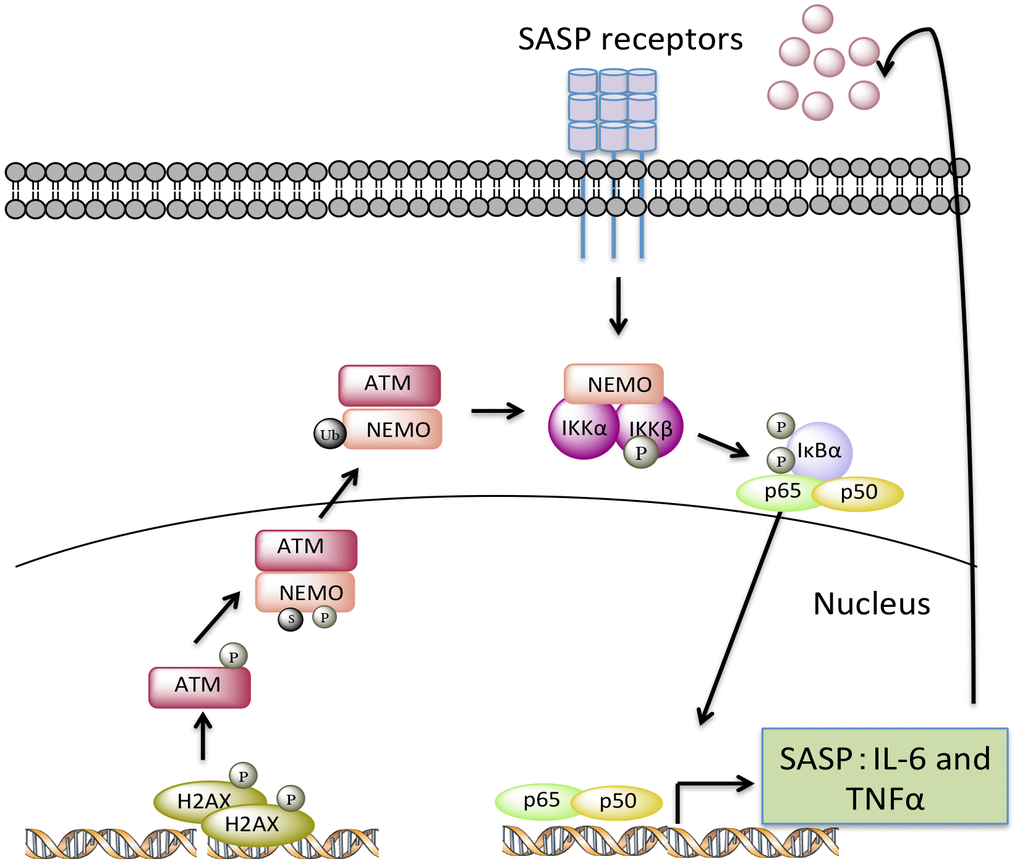
Figure 8.A model depicting how endogenous nuclear DNA damage activates NF-κB via an ATM- and NEMO-dependent mechanism to drive cellular senescence and senescence-associated secretory phenotype (SASP). In response to chronic accumulation of endogenous DNA damage, ATM undergoes autophosphorylation and promotes phosphorylation, SUMOylation, and monoubiquitylation of NEMO. As a result, monoubiquitylated NEMO along with ATM translocates to the cytoplasm, activating the IKK complex. Phosphorylation of IκB leads to the release of p65 so that it can translocate into nucleus upregulating a transcriptional program of certain SASP factors, such as TNFα and IL-6. Secreted SASP factors then trigger a second wave of NF-κB activation through cytokine receptors, further enhancing cell-autonomous pathway-mediated senescence and inducing non-cell-autonomous pathway-mediated senescence.