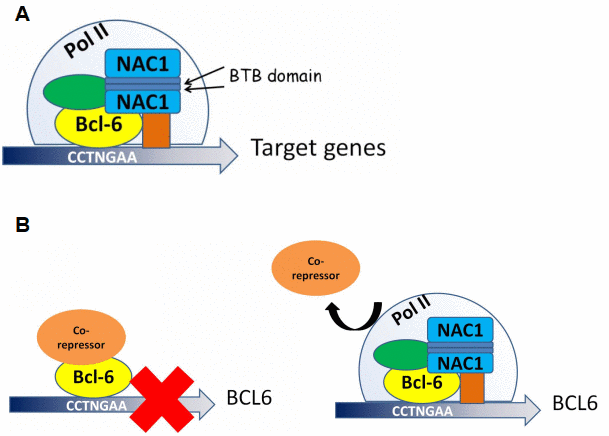
Figure 8.Proposed models for NAC1 and BCL6 activation of transcription and NAC1 attenuation of BCL6 negative autoregulation. (A) NAC1 forms a homodimer by interaction of its BTB domains. NAC1 homodimer interacts with BCL6 protein through the 186 amino acids from its C-terminus. NAC1-BCL6 interaction forms a higher magnitude complex that regulates transcription of target genes. (B) When NAC1 is absent, BCL6 protein is complexed with its co-repressors, which inhibits BCL6 transcription. When upregulated, NAC1 displaces BCL6 corepressors, and forms a regulatory complex with BCL6, which allows BCL6 expression.