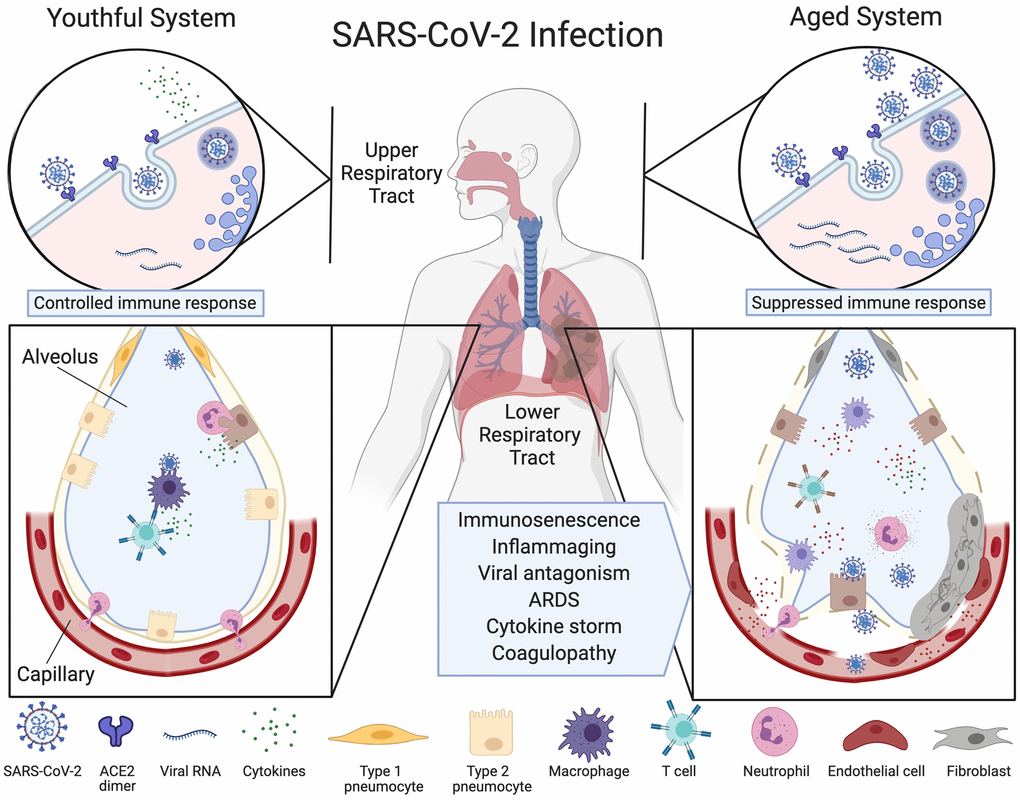
Figure 1.Ineffective clearance of SARS-CoV-2 infection in the aged respiratory system. The SARS-CoV-2 virus binds to ACE2 enzymes on airway epithelial cells in the upper respiratory tract where they are endocytosed and replicated (top left), alerting the immune system. Viruses then travel to the alveoli and infect type 2 pneumocytes which, in the youthful system (lower left), are recognized by alveolar macrophages (AMs) or dendritic cells (not pictured) that release cytokines and present antigens to T cells and other adaptive immune cells. T cells with the appropriate receptors activate other lymphocytes or directly kill infected cells, preventing the spread of the virus. Neutrophils migrate to the sites of infection to clear infected cell debris. In the aged system (top right), viral alert signals are initially slow, resulting in greater viral replication. Defective macrophages and T cells with a limited repertoire of receptors are less effective (lower right). More cells are infected, inducing high levels of inflammatory cytokine signaling. The endothelial cell lining of the capillary becomes inflamed, fibroblasts are activated, and SARS-CoV-2 viral components and cytokines enter the bloodstream. Fluid fills the alveolus, reducing lung capacity and the virus infects microvascular pericytes in other organs. A cytokine storm initiates microvasculature clotting, causing severe hypoxia, coagulopathy and organ failure. Created with BioRender.