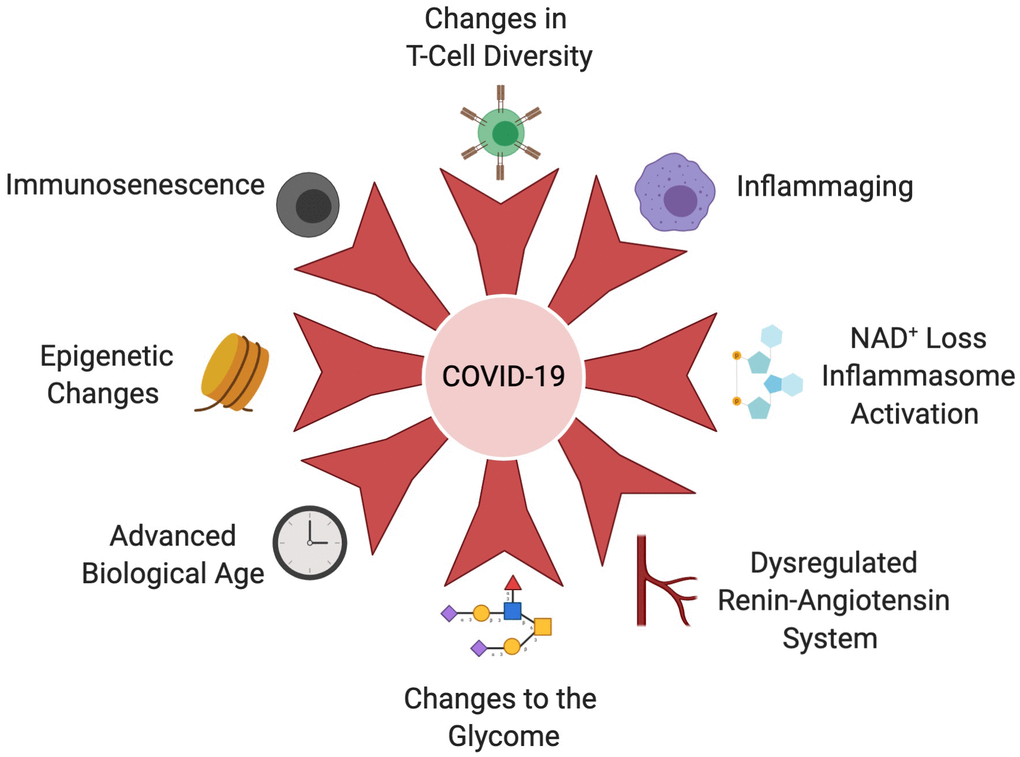
Figure 3.Age-related changes that increase COVID-19 susceptibility. The aging immune system undergoes immunosenescence, T-cell diversity alterations and chronic activation of the innate immune system, known as inflammaging. These hallmarks of the aging immune system cripple the body’s ability to clear the SARS-CoV-2 virus, initiate and sustain cytokine storms, than cause acute organ injury, DIC and multi-organ failure. An age-associated decline in NAD+ results in derepression of NLRP3 and inflammasome in older people, further exacerbating the cytokine storm. Coronaviruses also possess an ADP-ribosylhydrolase that further depletes already-low NAD+ levels in older people. Leveling of the epigenetic landscape during aging results in changes in immune cell composition and function that decrease the immune system’s ability to mount a response to infection. Epigenetic dysregulation of ACE2 may also impact increased viral loads in older people. Dysregulation of the RAS during aging and in the context of age-associated disease, such as cardiovascular disease, hypertension, COPD and obesity, contributes to severity of COVID-19 infection. The glycome which controls a variety of immune signaling pathways changes during aging and in the context of metabolic diseases. For example, decreases in IgG galactosylation contribute to chronic inflammation. Biological clocks that measure different biomarkers of biological age may explain increased COVID-19 susceptibility more accurately than advanced chronological age. Created with BioRender.