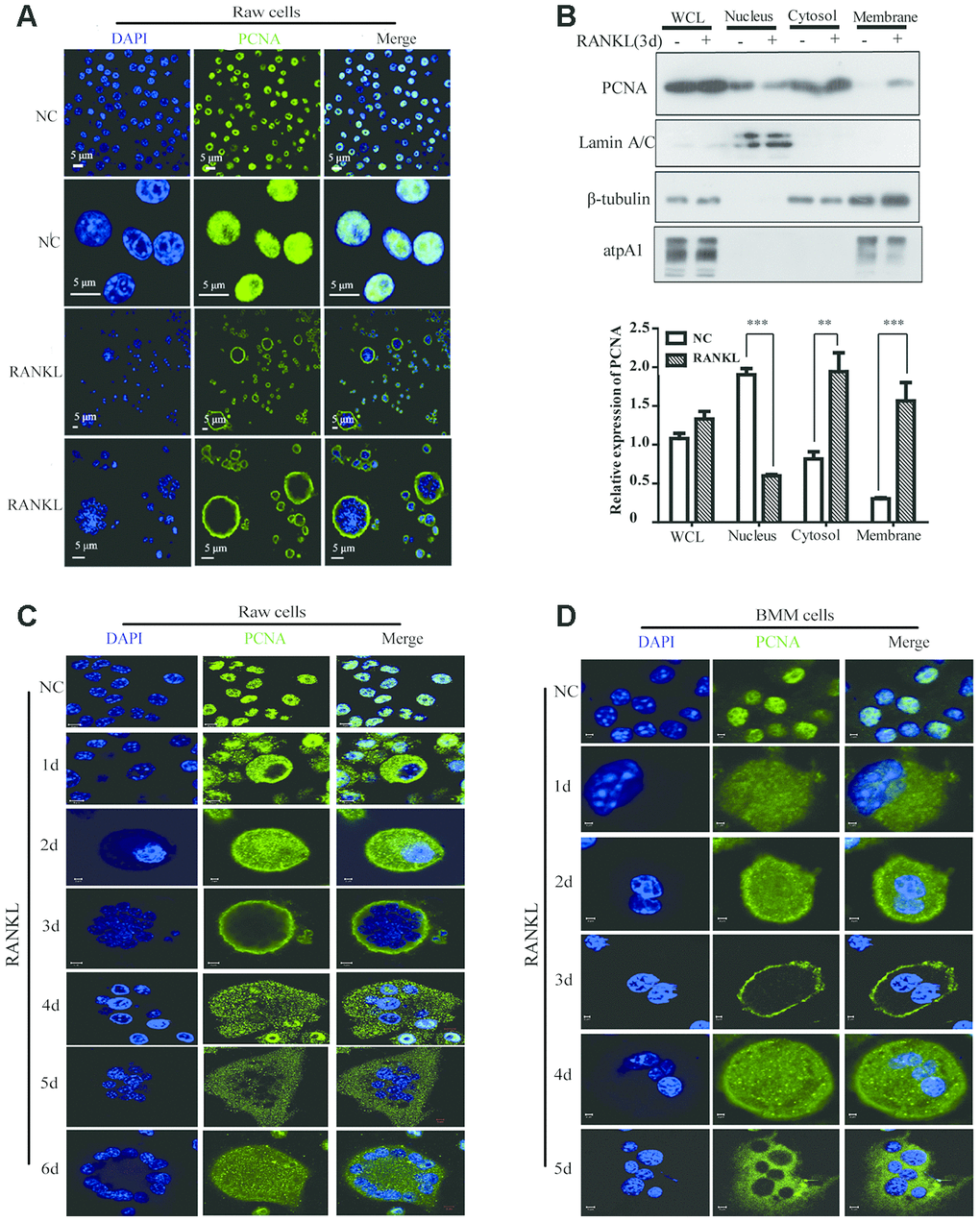
Figure 1.The subcellular localization of PCNA during the RANKL-induced osteoclast differentiation. (A) Nucleus-cytoplasm translocation of PCNA after 3-day RANKL treatment. RAW264.7 cells were treated with or without RANKL (100 ng/mL) for 3 days to induce osteoclast differentiation. Then, the cells were performed IF assay to test the cellular location of PCNA using primary PCNA antibody. DAPI staining was used to mark cell nucleus. Scale bar: 5μm. (B) PCNA subcellular distribution in differentiated osteoclast analyzed by cell fractionation assay. Upper panel: cell fractionation experiments were performed with RAW264.7 previously treated with or without RANKL (100 ng/mL) for 3 days to induce osteoclast differentiation. PCNA protein in various cell fractions was detected by western blotting. Lamin A/C, β-tubulin and atpA1 were respectively used as markers of nucleus, cytosol, and membrane fractions. Lower panel: the quantification of PCNA expression in WCL, Nucleus, Cytosol, and Membrane fractions of three independent experiments. (C) Tracking of PCNA subcellular localization during the 6-day RANKL treatment of RAW264.7 cells. IF assays were performed with RAW264.7 cells treated with or without RANKL (100 ng/mL) for the indicated times of treatment. Scar bar: 2-5μm. (D) Tracking of PCNA subcellular localization during the 5-day RANKL treatment of BMM cells. After isolation, BMM cells were cultivated with M-CSF (10 ng/mL) and RANKL (100 ng/mL) for 5 days to induce the osteoclast differentiation, and analyzed as described in (C). Scar bar: 2-5μm.