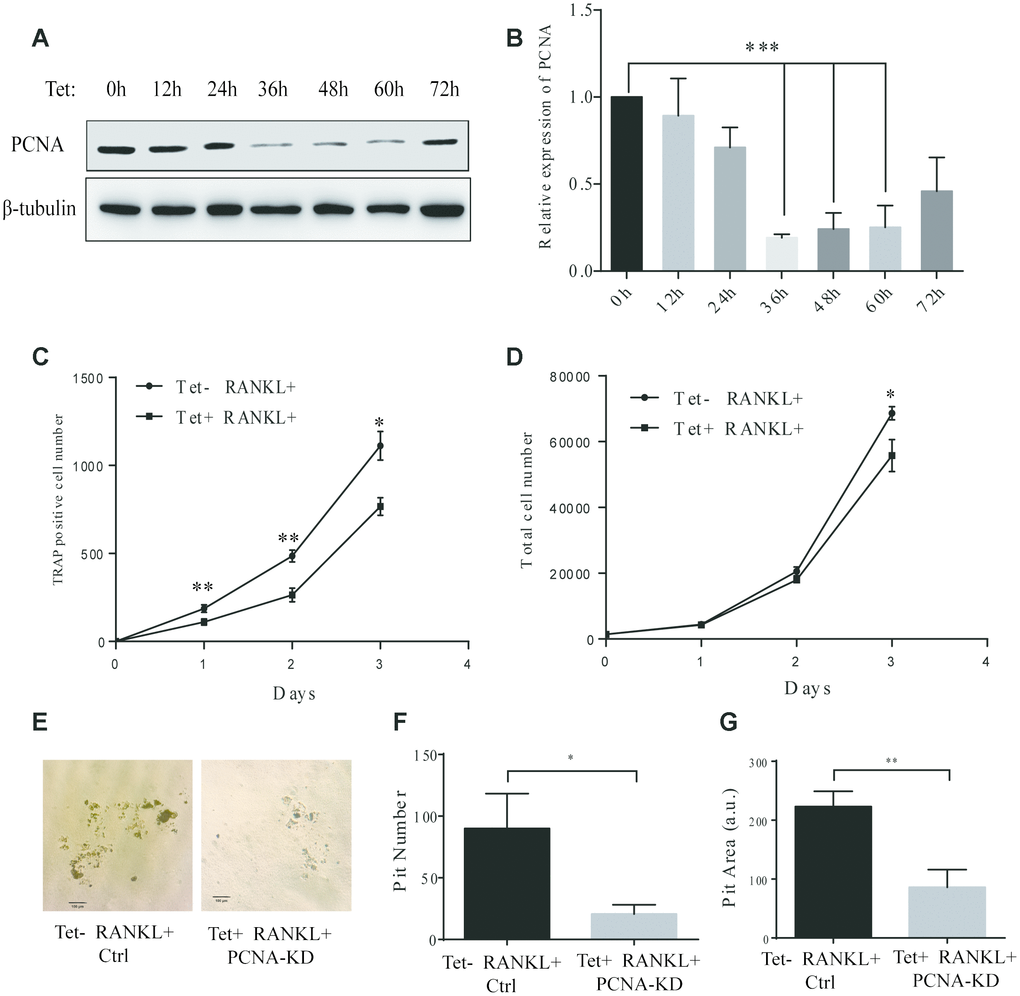
Figure 3.Effect of inducible knockdown of PCNA on RANKL-induced osteoclast differentiation. (A) The time course of PCNA knockdown effect after Tet-induction. 20 μg/mL of Tet was added Tet-PCNA KD cells to induce the knockdown of PCNA. Cells were harvested at different time points as indicated (0-72 hours) and subjected to western blotting to assess the effect of PCNA knockdown. (B) The quantification of PCNA expression level presented in (A). ***: p<0.001. (C) The impact of PCNA knockdown after its nuclear-cytoplasmic translocation on RANKL-induced osteoclast differentiation. Tet-PCNA KD cells were simultaneously treated with both Tet and RANKL for 3 days. TRAP-positive cells (considered as differentiated cells) were counted each day. (D) The effect of knockdown of cytoplasmic PCNA on the proliferation of RAW264.7 cells. The total cell number of cells after same treatments as in (C) was counted. *: p<0.05, **: p<0.01. (E) In vitro bone-resorption assays. Tet-PCNA KD cells were seeded at Corning Osteo Assay Surface multiple well plates and treated with Tet (20 μg/mL), RANKL (100 ng/mL) and M-CSF (50 ng/mL) for 10 days to induce the knockdown of PCNA, differentiation of osteoclasts and osteoclast-mediated bone resorption. Tet-PCNA KD cells treated without Tet were set as control. Resorption pits were observed using an Olympus microscope at 25x magnification. Scale bar: 100 μm. (F) Quantification of the resorption pits. Resorption pits on each well were counted. The results presented are the means ± SD of three independent experiments. *: p<0.05. (G) Quantification of the area of total pits. The area of total pits on each well was analyzed by ImageJ software. The results presented are the means ± SD of three independent experiments. **: p<0.01.