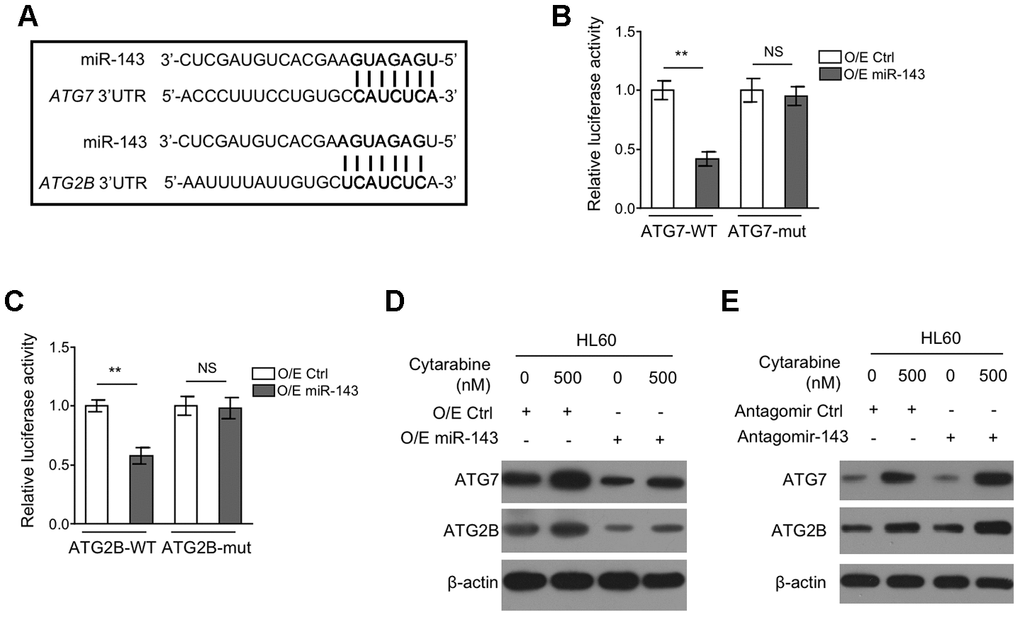
Figure 4.miR-143 decreases expression of ATG7 and ATG2B by directly targeting in HL60 cells. (A) Schematic illustration of the complementary sequence between miR-143 and the 3’-UTRs of ATG7 and ATG2B mRNAs. This information is provided by the computational and bioinformatics-based approach using TargetScan. (B) HEK293 cells were co-transfected pMIR-LUC-3’-UTR-ATG7-wt (ATG7-wt) or pMIR-LUC-3’-UTR-ATG7-mut (ATG7-mu) with 100 nM O/E miR-143 or 100 nM O/E Ctrl for 48 h, and then luciferase activity was measured. The results relative to O/E Ctrl group are shown. (C) HEK293 cells were co-transfected pMIR-LUC-3’-UTR-ATG2B-wt (ATG2B-wt) or pMIR-LUC-3’-UTR-ATG2B-mut (ATG2B-mu) with 100 nM O/E miR-143 or 100 nM O/E Ctrl for 48 h, and then luciferase activity was measured. The results relative to O/E Ctrl group are shown. (D) HL60 cells were transfected with 100 nM O/E miR-143 or 100 nM O/E Ctrl for 48 h, and then treated with or without 500 nM cytarabine for 24 h. The protein expression of ATG7 and ATG2B was measured by immunoblotting. β-actin was used as a loading control. (E) HL60 cells were transfected with 100 nM Antagomir-143 or 100 nM Antagomir Ctrl for 48 h, and then treated with or without 500 nM cytarabine for 24 h. The protein expression of ATG7 and ATG2B was analyzed as in (D). All data were from 3 independent experiments and expressed as mean ± SD. Data were compared using Student t-test. **, P<0.01; NS, not significant.