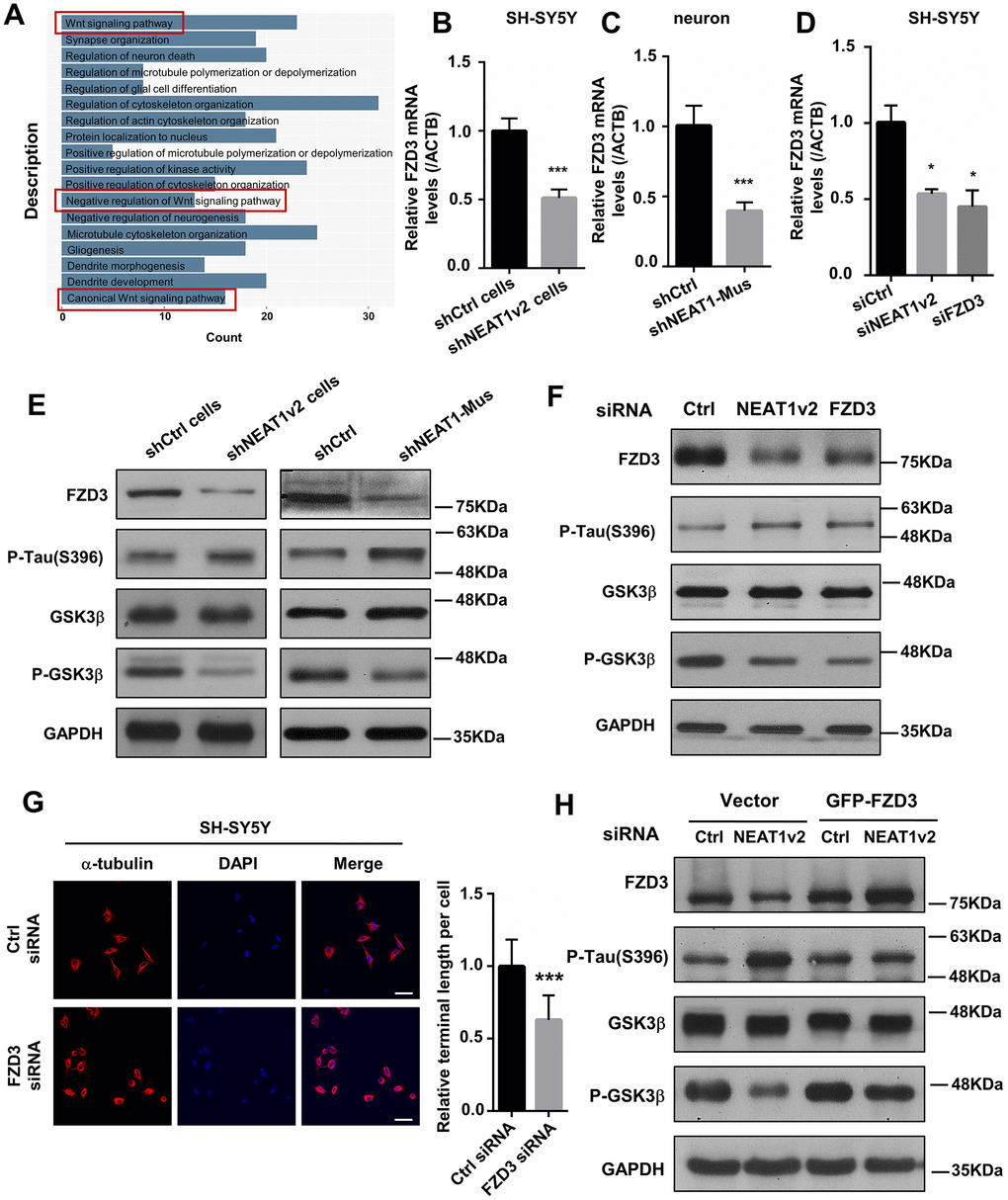
Figure 3.NEAT1 silencing mediates de-polymerization of MTs via FZD3/GSK3β/p-tau signaling pathway. (A) GO analyses were performed using the NEAT1-associated genes obtained from expression profile of braak stage 1/2 AD patients in GSE84422. (B) The FZD3 mRNA level was measured by quantitative PCR in shNEAT1v2 cells and shCtrl cells. (C) The mRNA level of FZD3 in shNEAT1-Mus and shCtrl transfected murine neurons. (D) The mRNA level of FZD3 in SH-SY5Y after being transfected with NEAT1 siRNA, FZD3 siRNA and Ctrl siRNA. (E) The expression levels of the FZD3, GSK3β, p-GSK3β, p-Tau(s396), Ace-tubulin and GAPDH were analyzed with immunoblotting in shNEAT1v2 cells, shCtrl cells and shNEAT1-Mus, shCtrl transfected murine neurons, respectively. (F) The expression levels of the FZD3, GSK3β, p-GSK3β, p-Tau(s396), Ace-tubulin and GAPDH were analyzed with immunoblotting in NEAT1v2 siRNA, FZD3 siRNA transiently transfected SH-SY5Y. (G) Immunofluorescence staining of α-tubulin (red) in FZD3 siRNA transiently transfected SH-SY5Y. DAPI (blue) was used to stain the nuclei. Scale bars, 20μm. Image J software was used to analyze the cell dendritic length. (H) The expression levels of the FZD3, GSK3β, p-GSK3β, p-Tau(s396) and GAPDH were analyzed with immunoblotting in GFP-FZD3 vector and control vector, while transfected NEAT1 siRNA (mean ± s.d, *P < 0.05, **P < 0.01, ***P < 0.001, Student 2-tailed t test).