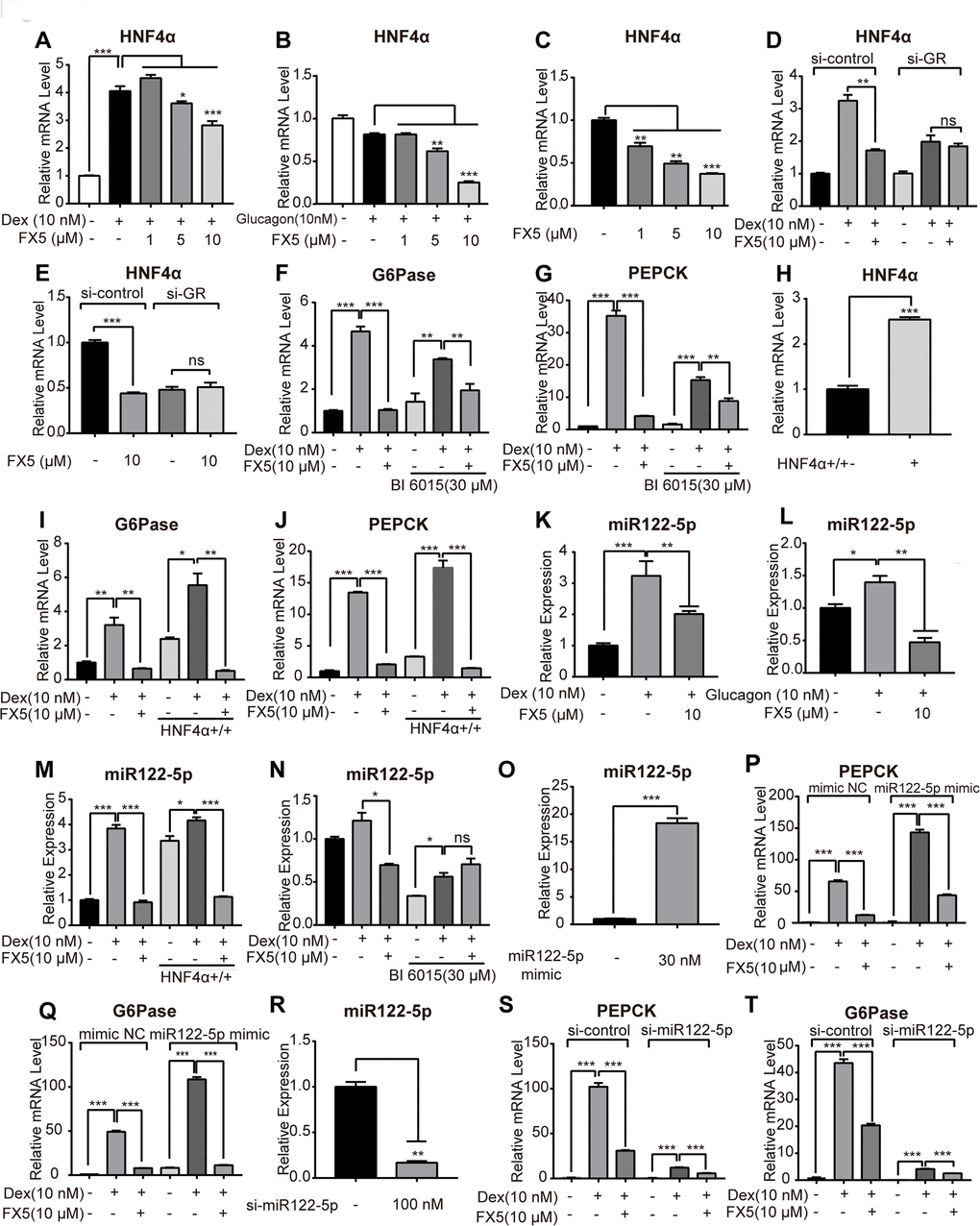
Figure 4.FX5 suppressed gluconeogenesis via GR/HNF4α/miR122-5p pathway in hepatocytes. Mouse primary hepatocytes were treated with different concentrations of FX5 (1, 5, 10 μM) and (A) 10 nM Dex or (B) 10 nM glucagon for 6 h, and mRNA level of HNF4α was investigated by qRT-PCR assay. (C) Mouse primary hepatocytes were treated with FX5 (1, 5, 10 μM) for 6 h, HNF4α mRNA level was then detected. (D, E) Mouse primary hepatocytes were transfected with si-control or si-GR for 48h. Hepatocytes were then treated with 10 nM Dex and 10 μM FX5 or 10 μM FX5 for 6 h. mRNA level of HNF4α was measured. (F, G) Mouse primary hepatocytes were treated with 30 μM BI 6015 for 24 h, then incubated with 10 nM Dex and 10 μM FX5 for 6 h. The suppression mRNA levels of (F) G6Pase (77.8% to 44.3%) and (G) PEPCK (88.2% to 44.9%) in the presence of BI6015 were measured by quantitative RT-PCR. (H–J) Mouse primary hepatocytes were transfected with HNF4α overexpression plasmid for 48h. Hepatocytes were then treated with 10 nM Dex and 10 μM FX5 for 6 h. mRNA level of (H) HNF4α, (I) G6Pase and (J) PEPCK were measured by quantitative RT-PCR. (K, L) Mouse primary hepatocytes were incubated in the presence of (K) 10 nM Dex or (L) 10 nM glucagon with 10 μM FX5 and miR122-5p level was measured by quantitative RT-PCR. (M) Mouse primary hepatocytes were transfected with HNF4α overexpression plasmid and then treated with 10 nM Dex and 10 μM FX5 for 6 h. Expression level of miR122-5p was measured by quantitative RT-PCR. (N) Mouse primary hepatocytes were treated with 30 μM BI6015 for 24 h, then incubated with 10 nM Dex and 10 μM FX5 for 6 h. Expression level of miR122-5p was quantified. (O-Q) Mouse primary hepatocytes were transiently transfected with micrON mimic NC #22 or micrON mimic NC #22 (5 FAM) for 48h and then were treated with 10 nM Dex and 10 μM FX5 for 6 h. Expression levels of (O) miR122-5p, (P) PEPCK and (Q) G6Pase were measured by qRT-PCR. (R–T) Mouse primary hepatocytes were transiently transfected with si-control or si-miR122-5p for 48h. Hepatocytes were then treated with 10 nM Dex and 10 μM FX5 for 6 h. Expression levels of (R) miR122-5p (83.2% interference efficiency), (S) PEPCK (70% to 52.5%) and (T) G6Pase (53.3% to 38.7%) were measured by quantitative RT-PCR in either absence or presence of si-miR122-5p. Values were mean ± S.E.M (n=3/group) (*P<0.05, **P<0.01 and ***P<0.001).