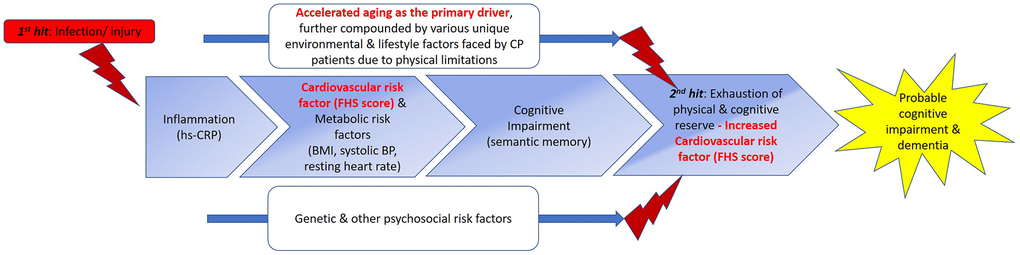
Figure 2.Proposed double-hit model for the early/ “premature” development of cognitive impairment and ultimately dementia in CP through shared biomarkers and phenotypes with MCI. We propose a double-hit model that hypothesizes the “kick-starter” effects of injury/ infections suffered at birth or during early childhood that result in persistent low-grade systemic inflammation in individuals with CP. (From left to right) With low-grade systematic inflammation mediated by hs-CRP, metabolic syndrome (MetS) could develop, as demonstrated by the association between the hs-CRP and delta FHS score in CP. MetS has been shown to be a prominent risk factor for the development of cognitive impairment, evidenced in our study by semantic memory scores in CP comparable to those of MCI, and association of delta FHS score with semantic fluency scores. The effects of accelerated aging, further compounded by various environmental, psychosocial, and lifestyle factors uniquely faced by adults with CP, due to physical limitations, further exacerbate the progression of CP to develop clinical symptoms of cognitive impairments, eventually culminating in clinical syndrome of dementia. Apart from these aforementioned factors, genetic and other psychosocial risk factors may plausibly influence the progression of this proposed continuum of dementia development by accelerating or decelerating the progression in this trajectory. Beyond what we have examined in this study, eventually, the influences of all the above-mentioned factors (main boxes and two lines) intertwine, tipping the homeostasis and eventual allostasis of the body, resulting in the progression to a phase represented by box number 4. This stage represents the second hit of our proposed double hit model, cumulating in the “breaking point”. We hypothesize that this phase is where both physical and cognitive reserve run out, causing the biomarkers, cognitive functions, and various phenotypes to further deteriorate, causing the early/ “premature” development of cognitive impairment severe enough, and coupled with the physical impairments, the adult with CP thus fulfil the clinical criteria for dementia. Based on our data, we speculate that once the reserves are exhausted in this process, CVS and metabolic risk factors play a more prominent effect (Table 4A, model 3 versus model 4, without and with aging as covariate), once aging is taken into account, delta FHS score became significantly associated with hs-CRP in patients with CP, supporting the penultimate role of aging in this trajectory. Interestingly, although BDNF levels may be lower in CP patients, symptoms of cognitive impairment have not manifested yet in CP. This could be due to the buffering from reserves [20]. But once reserves were run out (second hit and beyond), plausibly due to increased CVS risk factors, BDNF could not be further buffered and symptoms of cognitive impairment manifest. Picture adapted from Harding A, Robinson S, Crean S, Singhrao SK. “Can Better Management of Periodontal Disease Delay the Onset and Progression of Alzheimer’s Disease?” J Alzheimers Dis. 2017; 58:337-348. https://doi.org/10.3233/JAD-170046. Abbreviations: FHS=Framingham Heart Study; BDNF=Brain-Derived Neurotropic Factor; BP=Blood pressure; BMI=body-mass index; hs-CRP=high-sensitivity c-reactive protein; MetS=metabolic syndrome.