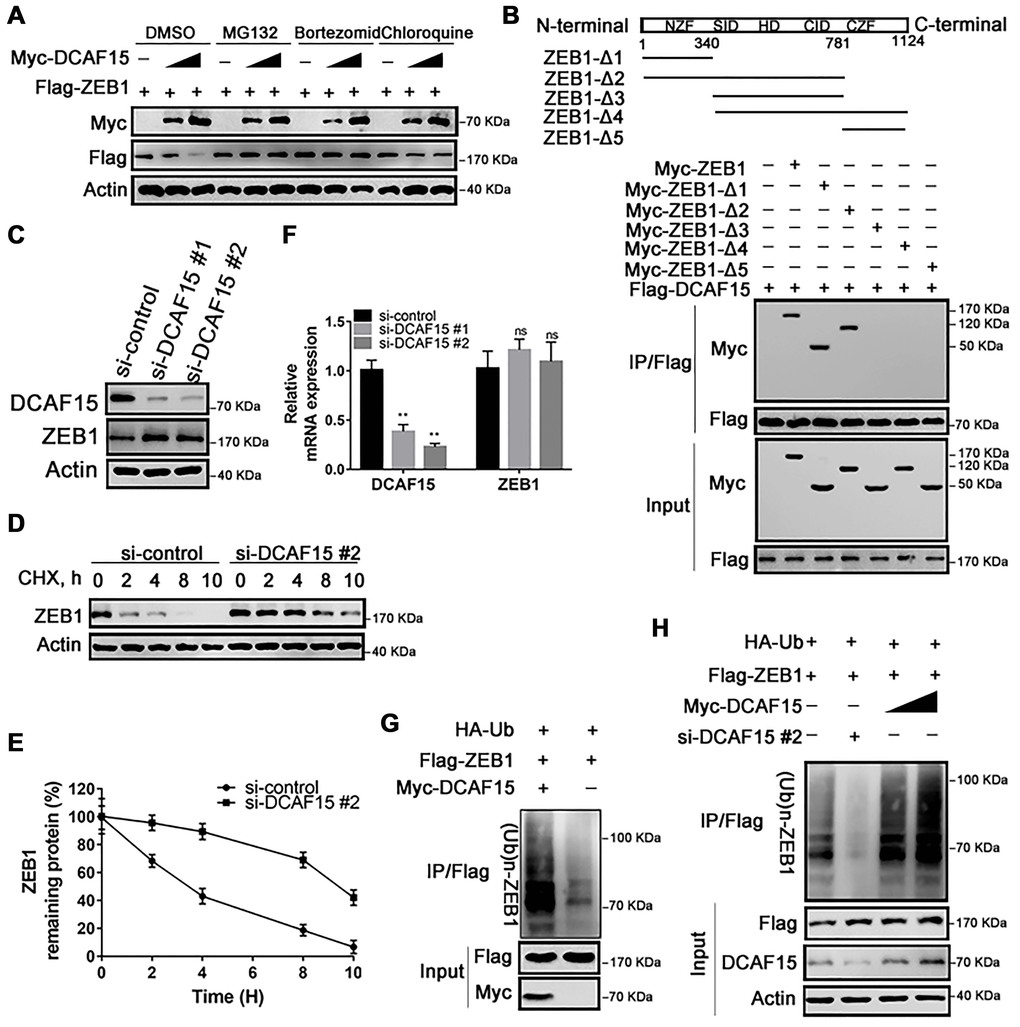
Figure 3.DCAF15 ubiquitinates and degrades ZEB1 protein. (A) DCAF15 regulates protein level of ZEB1 through the proteasome pathway. HEK293T cells were co-transfected with Flag-ZEB1 and increasing amounts of Myc-DCAF15. After 24h, cells were treated with DMSO, MG132, bortezomib or chloroquine respectively and detected by western blot. (B) HEK293T cells were transfected with Flag-DCAF15 and Myc-full-length or truncated ZEB1. After 24 h, cell lysates were immunoprecipitated using FLAG-M2 agarose beads. Immune complexes were analyzed by immunoblotting with the indicated antibodies (lower panel). Schematic diagram illustrating ZEB1 deletion mutants (upper panel). (C) SMMC-7721 cells were transfected with the control or two independent DCAF15 siRNAs, respectively. Cell lysates were analyzed via immunoblotting with the indicated antibodies. (D) SMMC-7721 cells were transfected with the control or DCAF15 siRNAs. After 24 h, cells were treated with cycloheximide (CHX) for indicated time-periods. Cell lysates were analyzed via immunoblotting with the indicated antibodies. (E) The relative intensities of ZEB1 protein were first normalized to the intensities of actin and then normalized to the value of the 0 h point. (F) qPCR validation of relative DCAF15 and ZEB1 mRNA levels in DCAF15 control and knockdown cells. GAPDH was used for normalization. (G) HEK293T cells were co-transfected with Flag-ZEB1, Myc-DCAF15 and HA-Ub. After 24 h, cells were treated with 10μM MG132 for 6 h. Cell lysates were immunoprecipitated using FLAG-M2 agarose beads and analyzed by immunoblotting with the indicated antibodies. (H) DCAF15 promotes ZEB1 ubiquitination. HEK293T cells were co-transfected with Flag-ZEB1, HA-Ub, si-DCAF15 #2 and increasing amounts of Myc-DCAF15. After 24h, cells were treated with MG132. The ZEB1 protein was immunoprecipitated using FLAG-M2 agarose beads. The ubiquitinated ZEB1 was analyzed by Western blotting.