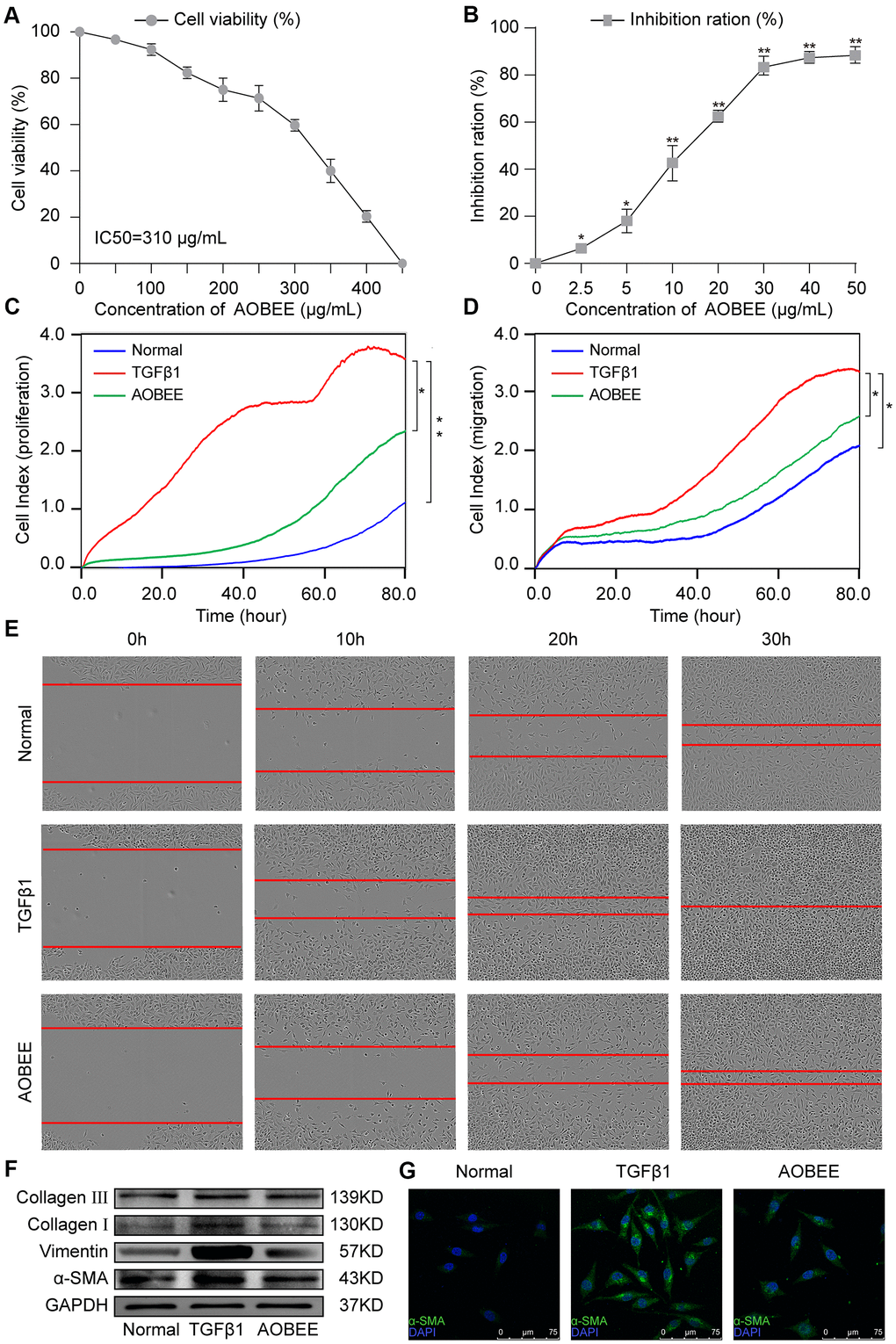
Figure 2.AOBEE cytotoxicity and anti-pulmonary fibrosis in vitro. (A) CCK-8 was used to test AOBEE toxicity in normal L929 cells. IC50 was approximately 310 μg/mL. (B) Inhibition of AOBEE on TGFβ1-treated cells in a dose-dependent manner. AOBEE had a significant inhibitory effect at a concentration of 10 μg/mL. (C) L929 cells were first administered with 5 ng/mL of TGFβ1 for 72 h and then cotreated with 24 μg/mL AOBEE. Using an xCELLigence RTCA instrument, the proliferation curves showed that AOBEE inhibited the activated-fibroblast proliferation compared with those in the TGFβ1 treatment group. (D) The migration curves revealed that AOBEE significantly repressed the activated-fibroblast migration compared with those in the TGFβ1 treatment group. (E) Images automatically monitored by an IncuCyte S3 instrument confirmed that AOBEE slowed the migration of activated fibroblasts at different time points. (F) AOBEE substantially reduced the expression of related fibrotic proteins including α-SMA, vimentin, and collagen I and III. (G) The immunofluorescence images showed that the TGFβ1-treated fibroblasts were spindle-shaped and had increased α-SMA. AOBEE improved cell state and reduced α-SMA expression. Each bar represents the mean ± SD (n = 6; *p < 0.05, **p < 0.01).