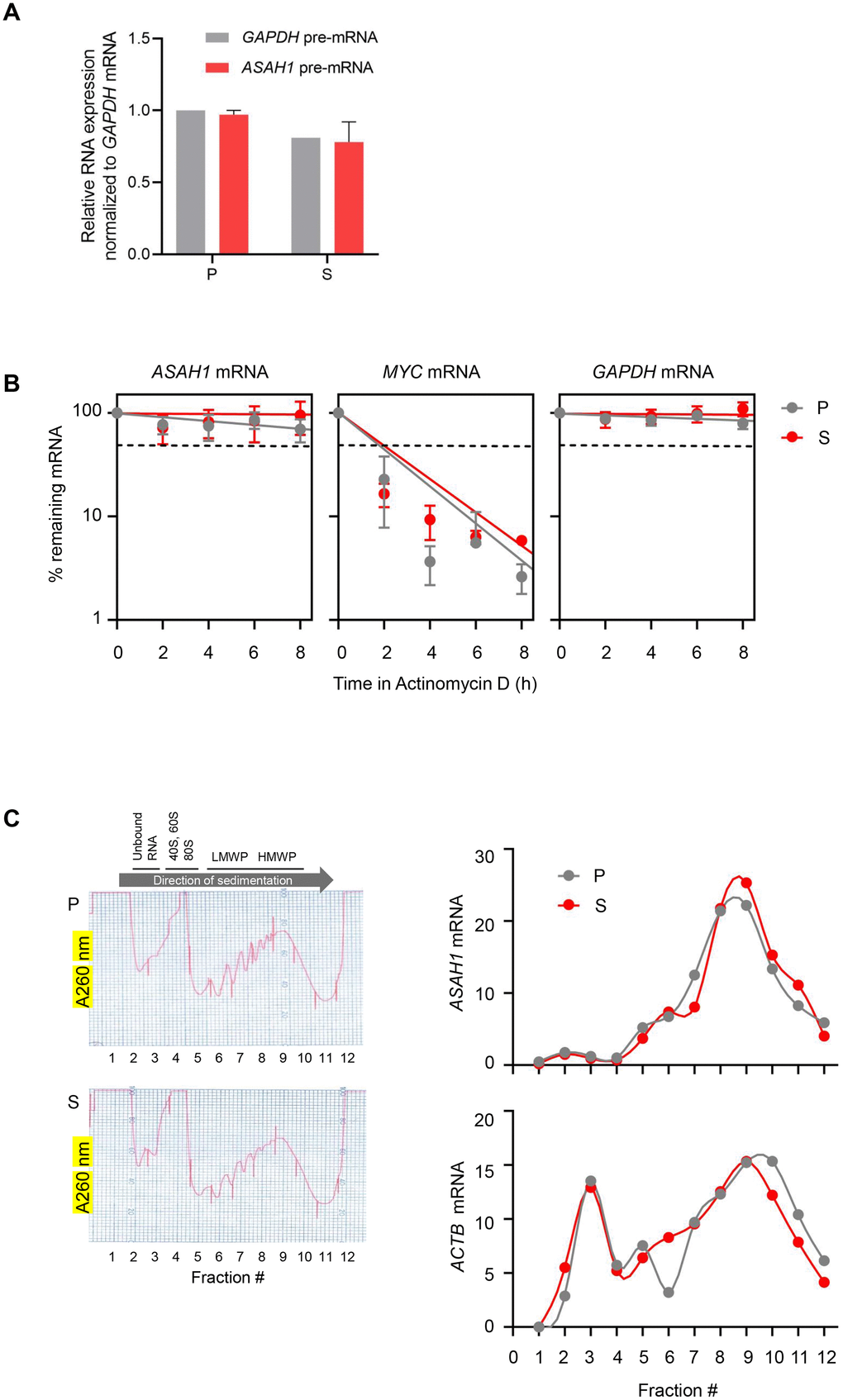
Figure 3.ASAH1 mRNA stability and translation in senescent cells. (A) RT-qPCR analysis of the levels of ASAH1 and GAPDH pre-mRNAs using total RNA from proliferating (P) and senescent (S) cells upon replicative exhaustion and normalized to GAPDH mRNA levels. (B) Proliferating and senescent cells were treated with Actinomycin D for the times indicated to block transcription; total RNA was then isolated, and RT-qPCR analysis was performed to assess the levels of ASAH1 mRNA. The labile MYC mRNA and the stable GAPDH mRNA were included as control transcripts. Discontinuous line indicates 50% of the original levels of mRNAs at time 0. (C) Cytoplasmic lysates obtained from P and S cells were fractionated through sucrose gradients to assess global polysome distribution profiles; ‘Unbound RNA’ fractions, fractions containing small ribosomal subunits (‘40S’), large ribosomal subunits (‘60S’), and monosomes (‘80S’), as well as polysomes of low and high molecular weight (LMWP and HMWP) are indicated. The relative distribution of ASAH1 mRNA and housekeeping ACTB (β-actin) mRNA was studied by RT-qPCR analysis of RNA in each of 12 gradient fractions. Data in (A) and (B) represent the means and S.D. from three independent experiments; data in C are representative of three independent experiments.