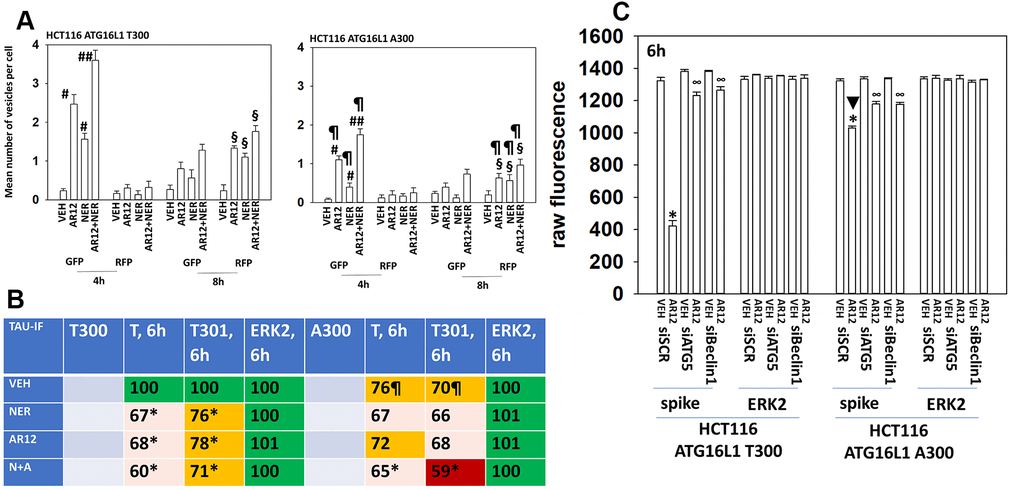
Figure 11.Autophagy is compromised in HCT116 ATG16L1 A300 cells; cells that are less capable of expressing Tau. (A) Isogenic HCT116 ATG16L1 T300 and HCT116 ATG16L1 A300 cells were transfected with a plasmid to express LC3-GFP-RFP. After 24h, cells were treated with vehicle control, AR12 (2 μM), neratinib (50 nM) or the drugs in combination for 4h or 8h. Randomly cells (> 50 per data point) were examined 4h and 8h after drug exposure and the mean number of GFP+ and RFP+ intense staining punctae determined under each condition (n = 3 +/-SD) # p < 0.05 greater than vehicle control; ## p < 0.05 greater than neratinib as a single agent; § greater than corresponding values at the 4h time point; ¶ p < 0.05 less than corresponding values in ATG16L1 T300 cells. (B) HCT116 cells (ATG16L1 T300 and ATG16L1 A300) were transfected with a plasmid to express Tau-GFP. After 24h, cells were treated with vehicle control, AR12 (2 μM), neratinib (50 nM) or the drugs in combination for 6h. Cells were fixed in place and in cell immunostaining performed to determine Tau expression (n = 3 +/-SD) * p < 0.05 less than vehicle control; ¶ p < 0.05 less than control value in ATG16L1 T300 cells. (C) HCT116 cells (ATG16L1 T300 and ATG16L1 A300) were transfected with a plasmid to express the SARS-CoV-2 spike protein and in parallel with a scrambled siRNA or with siRNA molecules to knock down expression of Beclin1 or ATG5. After 24h cells were treated with vehicle control or AR12 (2 μM) for 6h. Cells were fixed in place and in cell immunostaining performed to determine SARS-CoV-2 spike expression: raw fluorescence data is presented from a representative study performed in triplicate (n = 3 +/-SD) * p < 0.05 less than vehicle control; ∞ p < 0.05 greater than corresponding value in siSCR cells.