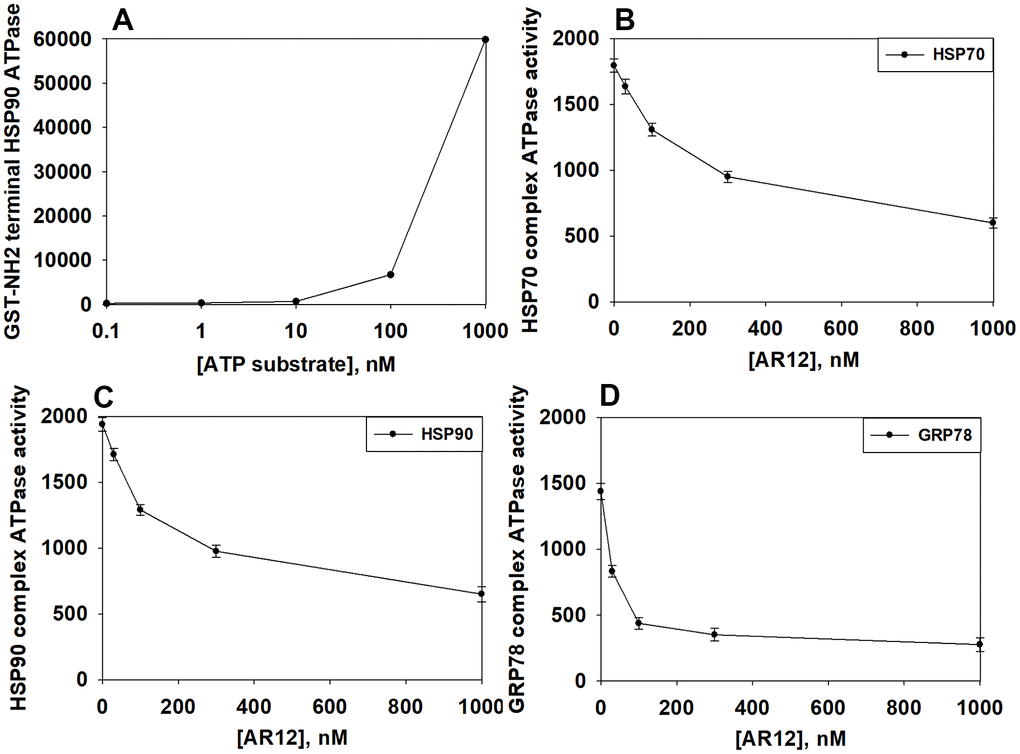
Figure 4.AR12 inhibits the chaperone ATPase activities of HSP90, HSP70 and GRP78. (A) A GST-HSP90 NH2-terminal fragment containing the ATP binding domain of the chaperone was synthesized in E. coli and purified from other bacterial proteins using glutathione Sepharose. The GST-HSP90 NH2-terminal fragment protein was not eluted off the Sepharose beads. Equal portions of beads were immediately aliquoted into individual wells in a 96 well plate. Beads were resuspended in kinase reaction buffer in triplicate, and incubated for 30 min at 37° C. The reaction was started by addition of increasing concentrations of the ATP-lite substrate. The plate was removed from the incubator and placed into a Vector 3 plate reader to determine the luminescence of the reactions under each condition (n = 3 (× 3) +/-SD). (B, C) GBM12 cells were transfected with a plasmid to express HSP70-GFP or to express FLAG-tagged HSP90. Twenty-four h after transfection chaperone proteins were immuno-precipitated using their tags in the presence of phosphatase inhibitors. Equal portions of precipitate Sepharose beads were immediately aliquoted into individual wells in a 96 well plate. Beads were resuspended in ATPase reaction buffer containing vehicle control or AR12 (30 nM; 100 nM; 300 nM; 1 μM) in triplicate, and incubated for 30 min at 37° C. The reaction was started by addition of ATP-lite substrate (2 μM). The plate was removed from the incubator and placed into a Vector 3 plate reader to determine the luminescence of the reactions under each treatment condition (n = 3 (× 3) +/-SD). (D) GBM12 cells were transfected with a plasmid to express GRP78. Twenty-four h after transfection GRP78 was immuno-precipitated using an antibody directed to the COOH terminal portion of the protein in the presence of phosphatase inhibitors. Equal portions of precipitate Sepharose beads were immediately aliquoted into individual wells in a 96 well plate. Beads were resuspended in ATPase reaction buffer containing vehicle control or AR12 (30 nM; 100 nM; 300 nM; 1 μM) in triplicate, and incubated for 60 min at 37° C. The reaction was started by addition of ATP-lite substrate (2 μM). The plate was removed from the incubator and placed into a Vector 3 plate reader to determine the luminescence of the reactions under each treatment condition (n = 3 (× 3) +/-SD).