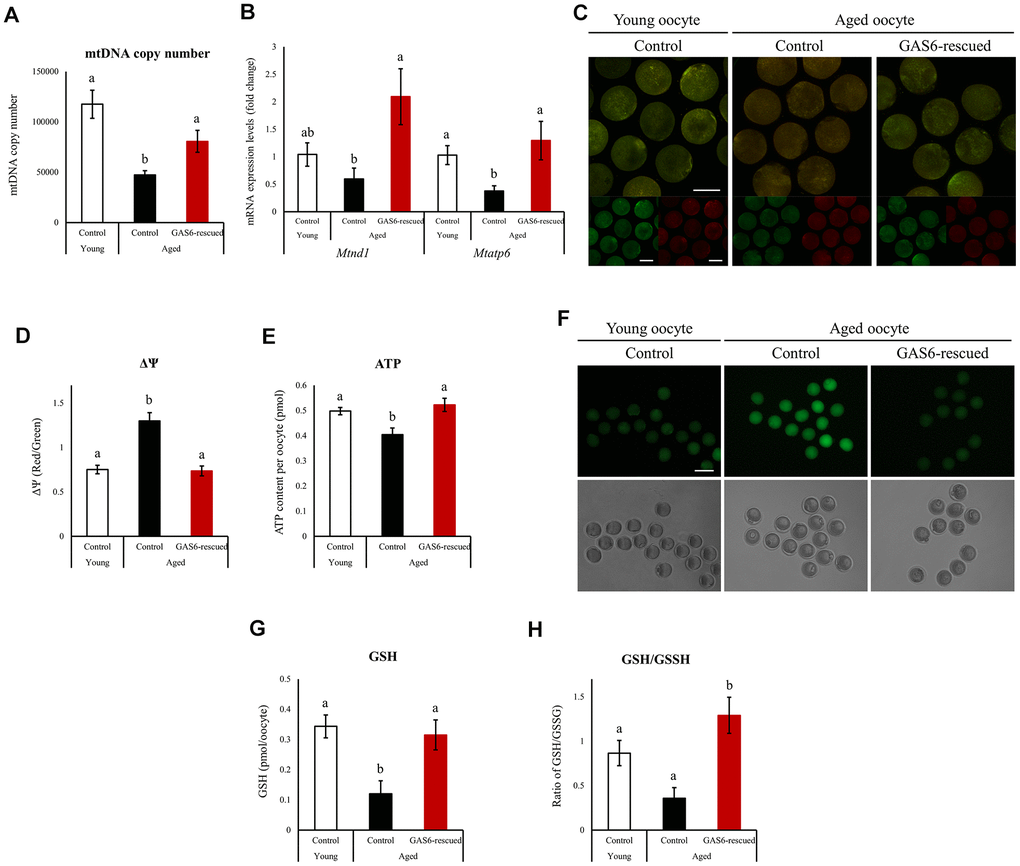
Figure 3.Improvements in mitochondrial function in oocytes during aging after GAS6 supplementation. (A) Measurements of the mtDNA copy numbers in aged oocytes expressing GAS6. The mtDNA copy number was higher in GAS6-rescued aged oocytes than in control aged oocytes. Different letters indicate significant differences at p<0.05. Control, nontreated oocyte; GAS6-rescued, oocyte treated with the GAS6 protein; Young, oocytes obtained from 3-week-old female mice; Aged, oocytes obtained from 12-month-old female mice. (B) Expression of Mtnd1 and Mtatp6 in aged MII oocytes from the control and GAS6-rescued groups. Different letters indicate significant differences at p<0.05. (C) Representative images of ΔΨm in aged MII oocytes after GAS6 supplementation. ΔΨm indicates the ratio of RITC (J-aggregate, high membrane potential) to FITC (J-monomer, low membrane potential) intensity in GAS6-rescued aged MII oocytes. Scale bars represent 50 μm. (D) Graphic representation of the results shown in C. The data are presented as the means ± SEM. Different letters indicate significant differences at p<0.001. (E) Effects of GAS6 restoration on ATP levels in aging oocytes. Increasing GAS6 expression resulted in elevated mitochondrial ATP production. Different letters indicate significant differences at p<0.05. (F) Treatment with GAS6 reduced ROS levels in young and aged MII oocytes. After treatment, MII oocytes were cultured in M16 medium supplemented with DCFH-DA to evaluate ROS levels. Scale bars represent 100 μm. (G) GAS6 treatment increased oocyte GSH levels with aging. The data are presented as the means ± SEM. Different letters indicate significant differences at p<0.05. (H) The ratio of GSH to GSSG in GAS6-rescued aged MII oocytes was calculated. The GSH/GSSG ratio is a biochemical marker of oxidative stress. The GSH/GSSG ratio was increased in aged MII oocytes after the restoration of GAS6. Thus, GAS6 reduces the oxidative stress caused by maternal aging. Different letters indicate significant differences at p<0.05.