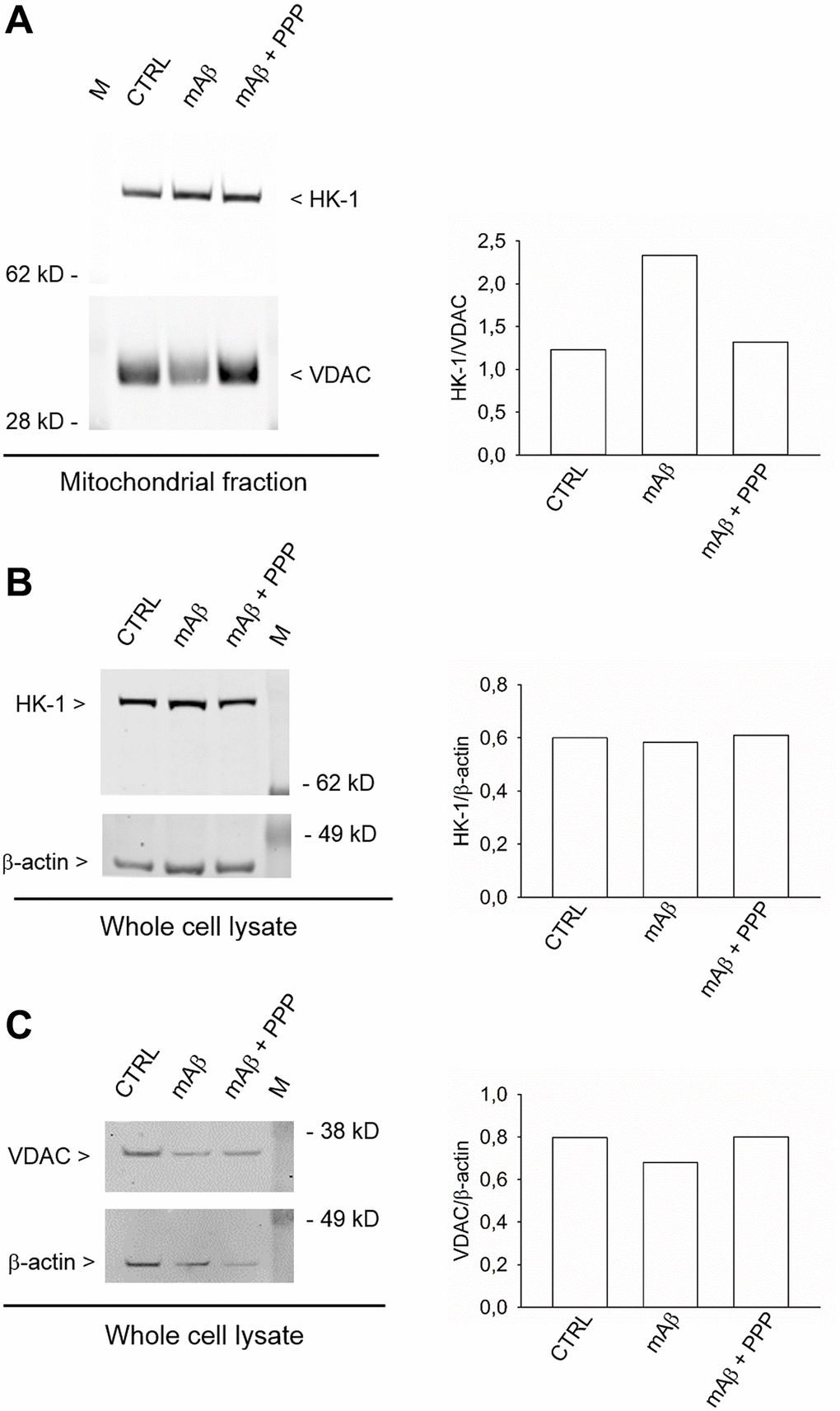
Figure 6.Synthetic Aβ42 monomers enhanced the mitochondrial abundance of HK-1 without altering the total protein content. In (A), the western blot analysis of HK-1 in the mitochondrial fraction of neurons that, following glucose deprivation and replenishing, were exposed to Aβ42 monomers in the absence (mAβ, 100 nM for 40 min) and in the presence of 500 nM PPP (mAβ ± PPP). Densitometric values of HK-1, normalized on VDAC signals, are represented in the graph bars (right). In (B and C), the western blot analysis of HK-1 and VDAC, respectively, in the whole neuronal lysate. Densitometric values of HK-1 or VDAC, normalized on β-actin signals, are represented in the respective graph bars (right). The whole cell lysate and the mitochondrial fraction were derived from the same experiment, but proteins were loaded in different amounts/gel (15 μg in (A), 20 μg in (B) and 10 μg in (C)) to avoid the saturation of hybridization signals. The experiment was repeated twice with similar results. Hybridization signals were detected with the Odyssey infrared imaging system in their original green or red colors and automatically converted into greyscale. M = protein marker.