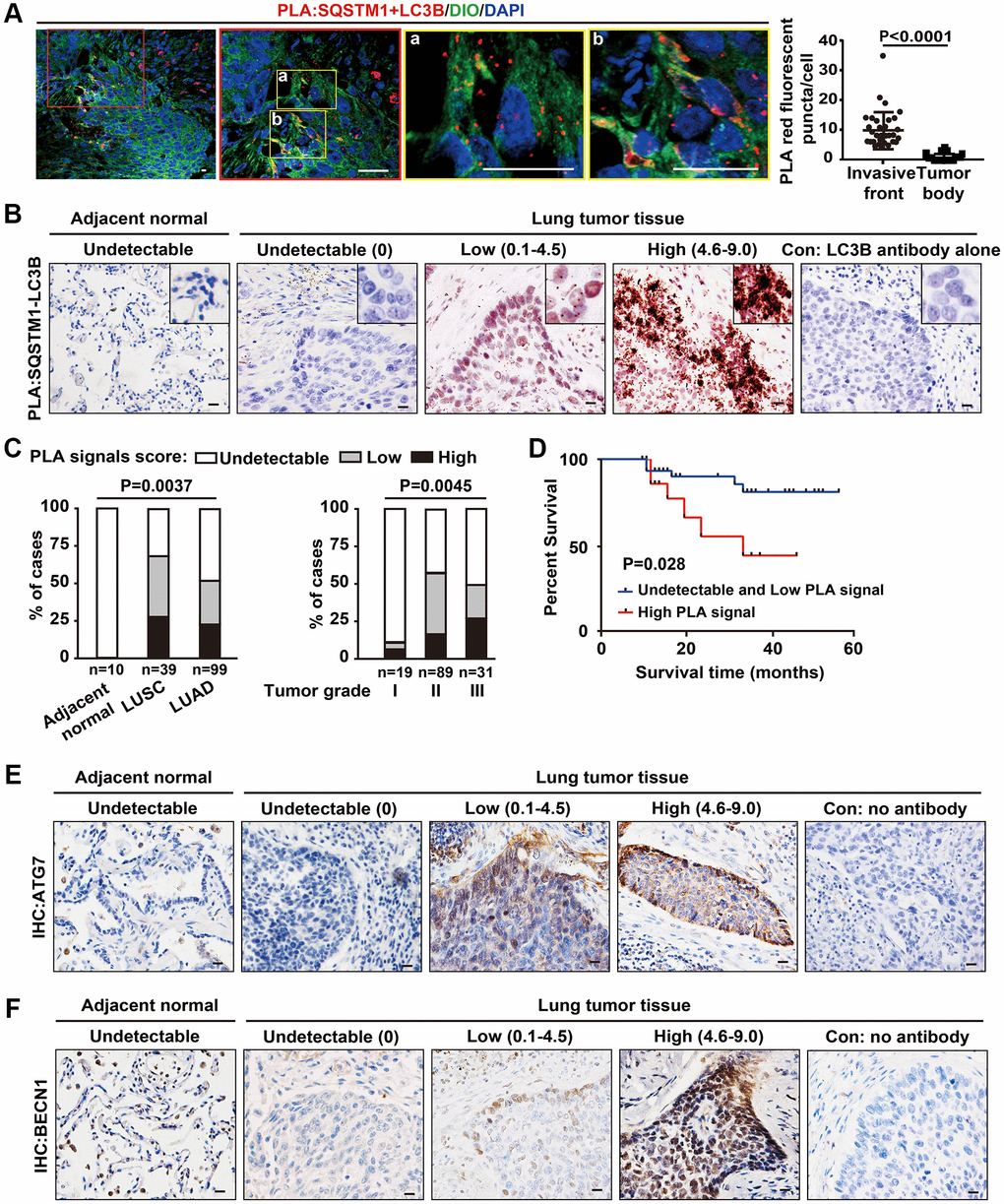
Figure 5.Increased SQSTM1-LC3B interaction at the tumor invasive front is correlated with poor outcome in NSCLC patients. (A) Endogenous physical interactions between SQSTM1 and LC3B in LUSC sections were detected by in situ PLA (indicated by red fluorescent puncta) and visualized by laser scan confocal microscopy. Membrane structures were counterstained with a DIO probe (green). The enlarged regions show positive PLA signals of SQSTM1-LC3B protein complexes at the tumor invasive front (a-b). Scale bars, 50 μm. The scatter diagram of the PLA results shows the number of red fluorescent puncta per cell at the tumor invasive front and inside the tumor body (P < 0.0001). Error bars, means ± SEM for 30 cells in a representative experiment. (B) Representative images of in situ bright-field PLA of SQSTM1-LC3B protein complexes in NSCLC specimens and tumor-adjacent tissues, counterstained by Hematoxylin. Scale bars, 50 μm. (C) in situ PLA of SQSTM1-LC3B protein interaction was performed on tumor-adjacent lung tissues (n = 10), LUSC (n = 39), and LUAD (n = 99) specimens. The frequency of patient with undetectable (0), low (< 4.5), or high (4.6–9.0) PLA signal scores stratified by IHC-defined lung cancer subtype (ANOVA, P = 0.003) (left panel) or by NSCLC grade (ANOVA, P = 0.0045) (right panel). (D) Survival rates in 64 of the 148 NSCLC patients determined by Kaplan-Meier analysis were compared between patients with undetectable and low (PLA signals score ≤ 4.5, n = 48, blue line) and high (PLA signals score > 4.6, n = 16, red line) PLA signal scores for the SQSTM1-LC3B interaction. Three-year survival rates were 85% (undetectable and low PLA signal scores of SQSTM1-LC3B interaction) versus 33% (high PLA signal scores of SQSTM1-LC3B interaction; P = 0.028). (E and F), Representative IHC images of ATG7 and BECN1 in NSCLC specimens and tumor-adjacent tissues. Scale bars, 50 μm.