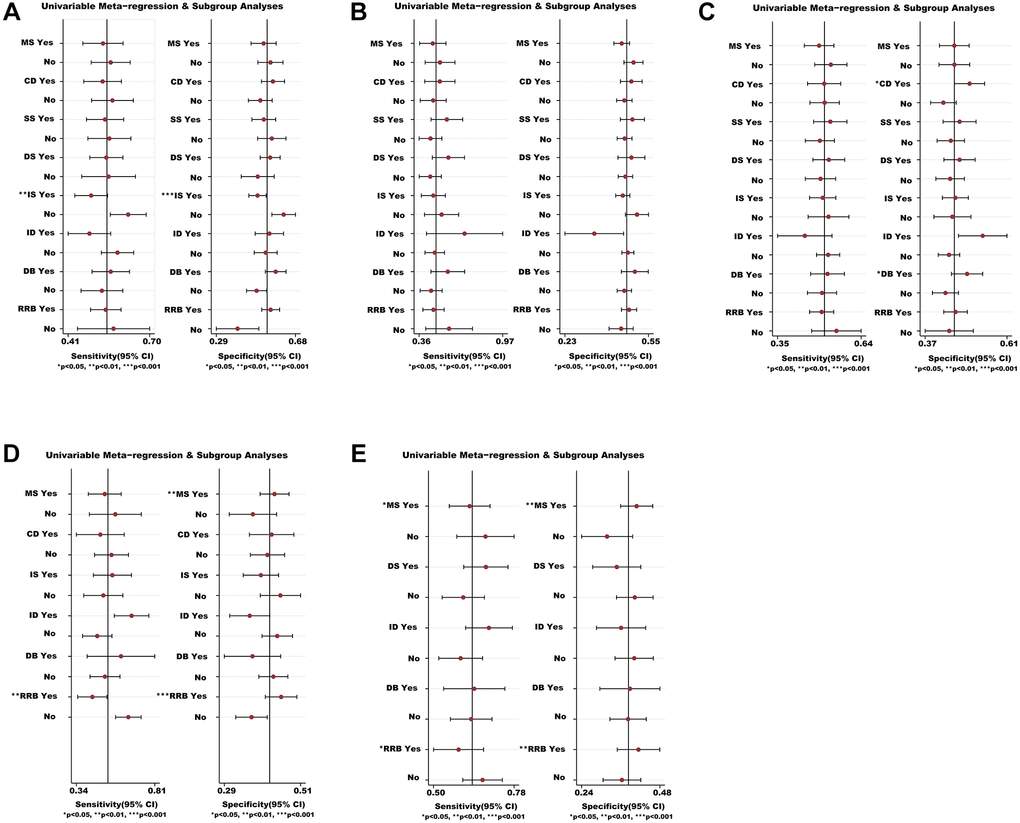
Figure 6.Meta-regression and sensitivity analyses for the efficacy and safety of medications. (A) The VC ratio of non-severe COVID-19 patients. (B) All-cause mortality of non-severe COVID-19 patients. (C) The TEAEs ratio of non-severe COVID-19 patients. (D) All-cause mortality of severe COVID-19 patients. (E) The TEAEs ratio of severe COVID-19 patients. *We conducted meta-regression and sensitivity analyses to estimate the impact of variable for each outcome. The potential modifiers (variables) for meta-regression we select are listed below: MS, DS, DB, CD, SS, IS, ID, and RRB. Abbreviations: COVID-19: coronavirus disease 2019; VC: virological cure; TEAEs: treatment-emergent adverse events; MS: multicenter study; DS: duration of study; DB: double blind; CD: crossover design; SS: sample size; IS: industry sponsorship; ID: inequalities in doses; RRB: risk of reported bias.