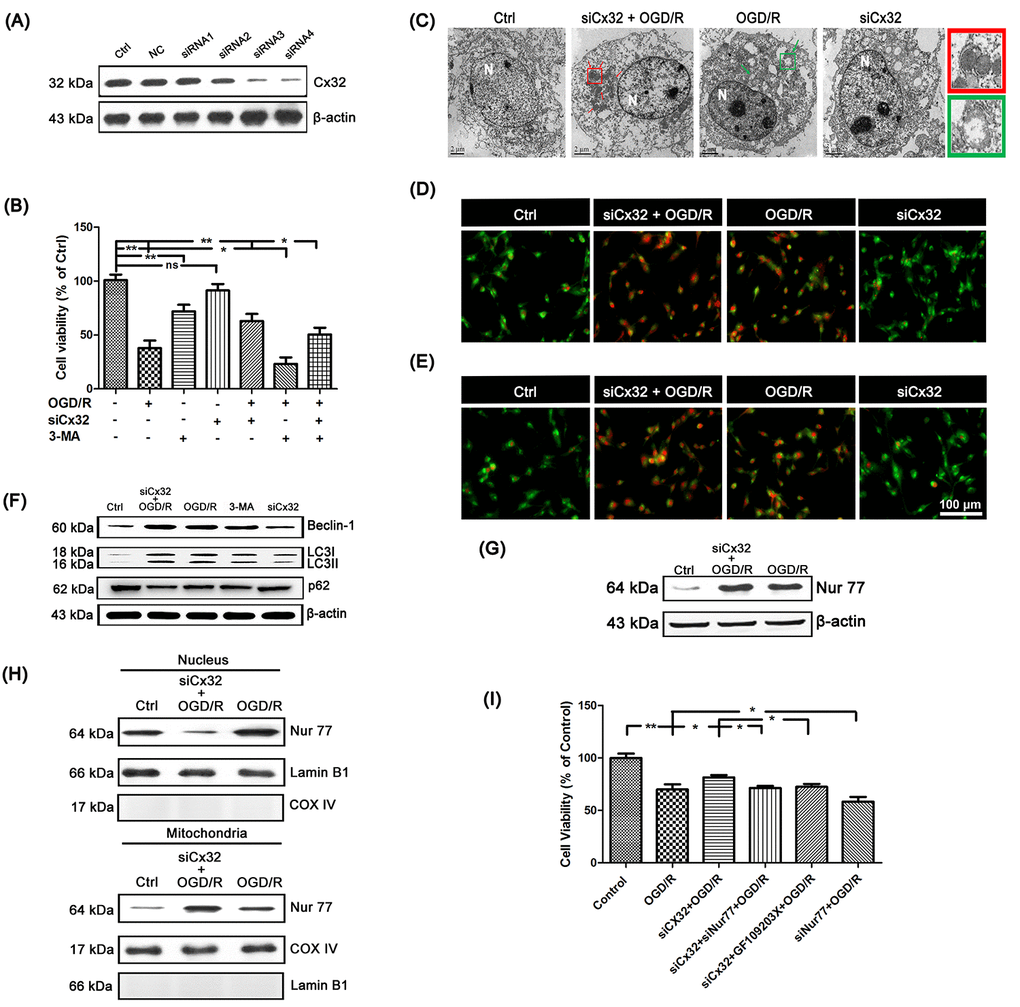
Figure 2.Inhibition of Cx32 activated autophagy and promoted Nur77 translocating from nucleus to mitochondria after OGD/R injury. (A) After transfection with control siRNA or Cx32 siRNA for 24 h, the cells were collected for Western blots of Cx32. Cells were transfected with Cx32 siRNA for 24 h followed by OGD/R (2 h OGD following by 24 h recovery) before harvested. (B) A MTT assay was used for cell viability. (C) Images acquired by transmission electron microscope. The arrow marks autophagic vacuoles. Scale bars, 2 μm. (D–E) Images of Monodansylcadaverine (MDC) staining and acridine orange (AO) staining under fluorescence microscope. Scale bars, 100 μm. (F) Representative bands of Beclin1, LC3 and p62 protein in siCx32 cells after OGD/R. (G) Representative bands of Nur77 protein in the cells after OGD/R. Variation in protein loading was determined by blotting with an anti-β-actin antibody (n = 3). (H) Mitochondrial translocation of Nur77 was examined after OGD/R. Mitochondria Nur77 expression was normalized against COX IV. Nucleus Nur77 expression was normalized against α-tubulin expression. (I) A MTT assay was applied for the cell viability. Data are expressed as means ± SD (n = 6). **p < 0.01, *p < 0.05.