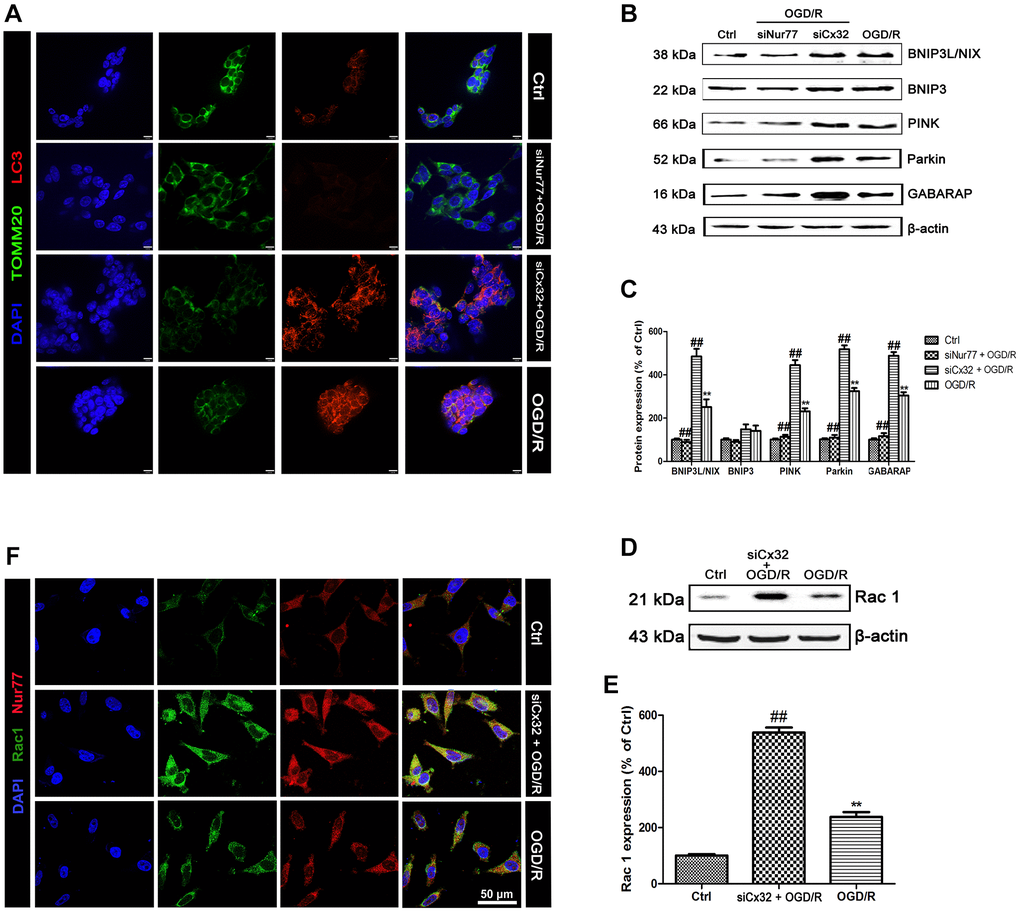
Figure 4.Nur77 activated mitophagy after the inhibition of Cx32 following OGD/R injury. Cells were transfected with Cx32 siRNA or Nur77 siRNA for 24 h followed by OGD/R (2 h OGD following by 24 h recovery) before harvested. (A) The autophagosome was marked with LC3 (red); mitochondria was marked by TOMM20 (green). Scale bars, 25 μm. (B–C) Representative bands of BNIP3L/NIX, BNIP3, PINK, Parkin, and GABARAP protein in the cells after OGD/R. (D–E) Representative bands of Rac1 protein after OGD/R. Variation in protein loading was determined by blotting with an anti-β-actin antibody. Densitometric scanning of band intensities were calculated as means ± SD (n = 3). **p < 0.01 vs. Ctrl group, ##p < 0.01 vs. OGD/R. (F) Double labeling of Nur77 and Rac1 using fixed cells of Control, OGD/R or siCx32 group. Scale bars, 50 μm.