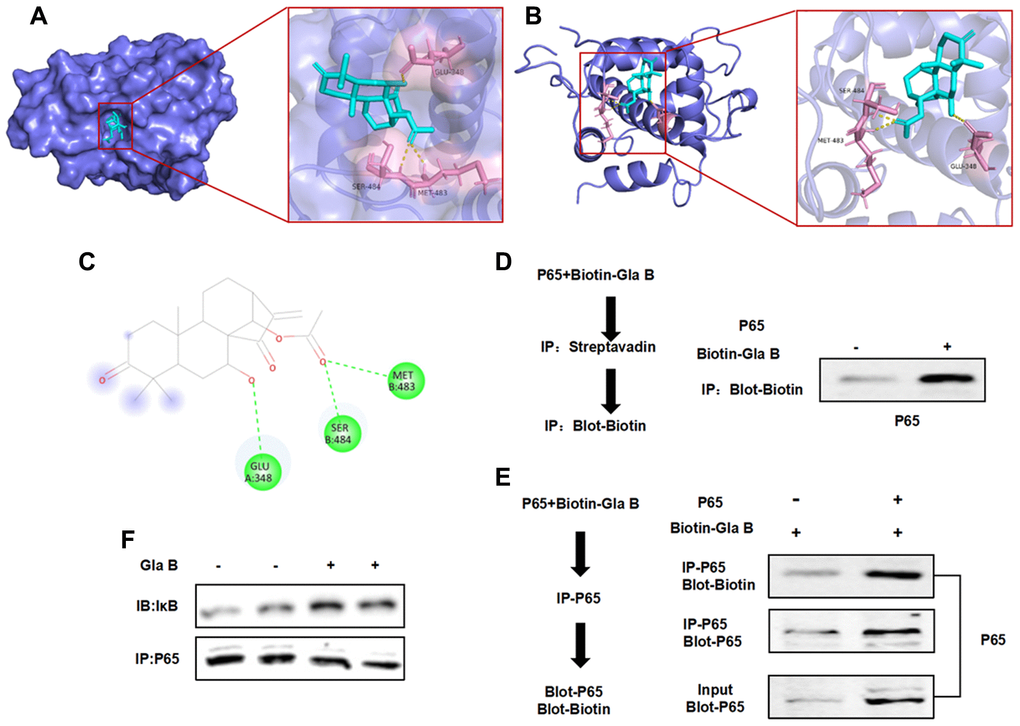
Figure 7.The targeted binding relationship between Gla B and P65. (A–C) 3D and 2D images of the binding between Gla B and P65. Gla B bound to the protein pocket of P65, and Gla B had hydrogen bond with Glu, Ser and Met. The binding energy was −8.4 kcal/mol, which could affect the phosphorylation and isomerization of P65. (D–F) Pull-down assay showed that Biotin-Gla B had a binding relationship with P65, and Gla B was an effective inhibitor of P65.