Introduction
Even disregarding traumatic deaths due to violence and high-risk lifestyle [1], men have a shorter life span than women. In spite of a multitude of theories, the primary cause is unknown.
Hayflick's hypothesis, that each cell has its own limited division capacity [2], received fundamental support with the discovery of telomeric shortening during DNA replication in somatic cells [3]. In peripheral blood lymphocytes (PBL), a telomeric loss per year of 31 bp (between 2-95 years) [4], 41 bp (between 0-107 years) [5], and 60-66 bp (between 0 and 100 years) [6] is described. Thus, telomere erosion has been regarded as a biological clock (for review see [7]). Whether this also applies to aging at the organismic level is unclear.
There is, however, circumstantial evidence that this might be the case, as a prospective study showed that death due to cardiac und infectious diseases occurred earlier in those with relatively short telomeres [8]. In Mayer et al. [6], an age-dependent and gender-specific difference in average telomere lengths emerged, women being endowed with longer telomeres. We based our study on PBL from a total of 205 healthy persons, ranging from newborns up to the age of 100 years, and applied T/C- FISH [9] to quantify the telomere lengths of every single chromosome arm (Figure 1).
Results and discussion
Telomere erosion is faster in males
Confirming our previous results and that of others [4,8,10,11], we found an age-dependent decline in pan-genomic telomere lengths: this decline proved greater in males [6,9]. To date, the mechanism for this gender difference has proved to be elusive.
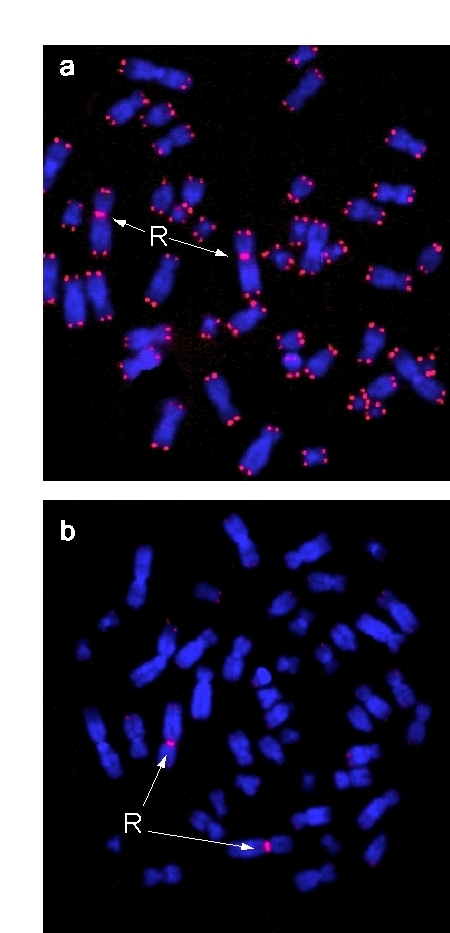
Figure 1. Metaphases of phytohemagglutinin-stimulated peripheral blood lymphocytes. Chromosomes were co-hybridized
with peptide nucleic acid (PNA) probes (red) for telomeric
sequences and the chromosome 2 centromere serving as
internal reference probe, and stained with DAPI (blue).
Centromere signals are marked by arrows. (a) Metaphase
derived from cord blood of a male newborn, (b) Metaphase
from blood of a centenarian male.
Skewed distribution of telomere length
When we plotted the frequency of a total of 235,266 single telomere values, the resulting curve was not Gaussian but was skewed to the right (Figure 2a, small insert). This is also shown mathematically by ascending skewness and kurtosis, the equation of the regression line of skewness being y=0.0106x+1.0359 with a regression coefficient of r=0.88 and that of kurtosis y=0.0559x+1.3705 (r=0.85) (Figure 2b). Since skew-ness characterizes the degree of asymmetry of a distri-bution around its mean, while kurtosis is a measure of the relative peakedness/flatness of a distribution, our ascending regression lines in Figure 2b indicate that the distribution of telomere lengths becomes more skewed to the right and, at the same time, more peaked with higher age.
Greater skewness and kurtosis in females
A very obvious new fact emerged when we plotted all our data classified by sex. The skewness of the female distribution curve (1.322) resulting from 115,754 values was greater in extent than the male equivalent (1.099) resulting from 119,472 values (Figure 2a). The difference is significant at a level of a = 0.0098. This is all the more astonishing since the left sides of the curves are almost perfectly superimposed, meaning that the above sex-specific differences in telomere size are not due to differences in frequency of short telomeres but to higher frequencies of long ones in females. Figure 2c shows skewness and kurtosis classified by sex with skewness equations of y=0.013x+0.98 (r=0.78) for females and y=0.012x+0.85 (r=0.86) for males and the kurtosis equations of y=0.07x+1.25 (r=0.67) for females and y=0.06x+0.79 (r=0.82) for males. In both criteria, the female line runs above the male line, corroborating the histograms in Figure 2a and both lines run nearly parallel, meaning that skewness and kurtosis do not essentially differ between genders during a lifetime.
What was the morphologic correlate of the abnormally high T/C FISH medians? Re-examination of our basic data, i.e. the hybridized metaphases, revealed an overall infrequent occurrence of extremely long telomeres of single chromosome arms. This was most evident in the elderly with pangenomically shorter telomeres (Figure 3c). These extremely long, single telomeres appeared to be randomly distributed all over the genome and were singularities, since we could not relocate an identical event in other metaphases of the same individual. These elongations, however, often occurred in doublets, meaning that both chromatids of the same p or q arm were elongated. This strongly suggests that, once the elongation has been achieved in a single chromatid, this newly acquired long telomere is the template for the next generation of descendants of this single individual cell. We coined the term "erratic extensive elongation" (EEE) of single telomeres to define this phenomenon. Once identified, we also found EEE of telomeres in younger people (Figure 3b) and even in newborns, although it was hardly visible and only detectable by means of the software, (Figure 3a). It is conceivable that minor differences in telomere lengths of individual chromosome arms might be the result of EEE that occurred years earlier and was subsequently exposed to general pangenomic erosion. We think it highly likely that EEE is responsible for the skewness of the distribution of telomere values (Figure 2a).
Evidence that EEE is propagated to the cells' progeny came from an anecdotal observation in a middle-aged woman in whose PBL we encountered a considerable number of endoreduplications (ER), together with an increased frequency of EEE. ER is the result of a dysregulation in cell-cycle progression that ends not in normal mitosis but in re-entering of the next cycle, leading to tetraploidy with descendant chromosomes closely attached [12] (Figure 3d). Every "tetrad" of chromatids represents 3 generations. If EEE is detected on two of four telomeres of comparable length, it should be derived from the EEE of an ancestor chromatid. In Figure 3e, this is visible at the telomeres of chromosome 2p, 5p, 6q, and 10p.
A broadly accepted theory explains accelerated telomere shortening by oxidative stress (ROS), with men having the greater load [23-28]. Assuming that ROS causes DNA damage directly, forcing the cell either to repair or to die, the result should be telomere length distributions in two isomorphic curves with a parallel shift of the female curve to the right. As we show, this is not the case. Therefore, the curves we obtained are most likely the result of a mechanism counteracting telomeric loss which is more active/effective in females.
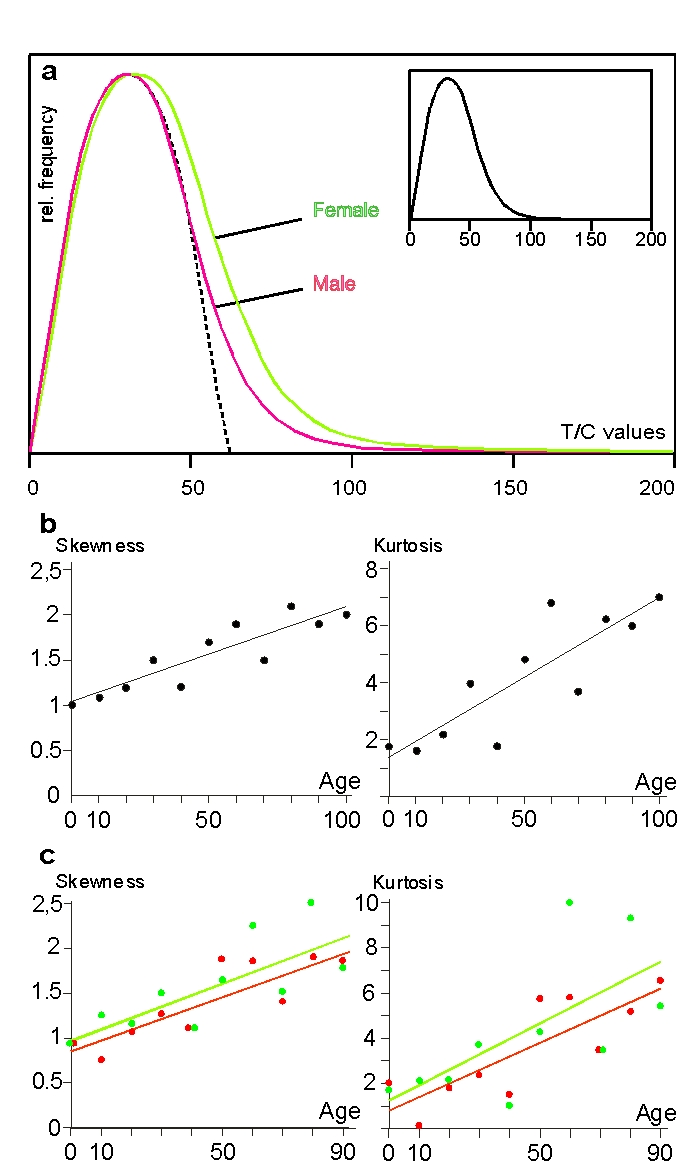
Figure 2. Erratic extensive elongation (EEE) of single telomeres in peripheral lymphocytes. (a) Histogram of telomere
lengths of all p and q arms of chromosomes of females (green)
and males (red). The dotted line represents the theoretical
Gaussian distribution, the histogram in the smaller insert
represents the curve of female and male values together. The
actual curves are skewed to the right. (b) Skewness and kurtosis
of telomere lengths of p and q arms of chromosomes of lymphocytes
in all age groups. Values are limited to 150 T/C values. (c)
Skewness and kurtosis of telomere lengths of p and q arms of
chromosomes of female (filled circles and green line) and
male probands (open circles and red line) ranging from newborns
up to 90 years. Centenarians are excluded because the male
group consists of only 2 persons. Values limited to 150 T/C
values as mentioned above. (see “Statistical analysis” for further details).
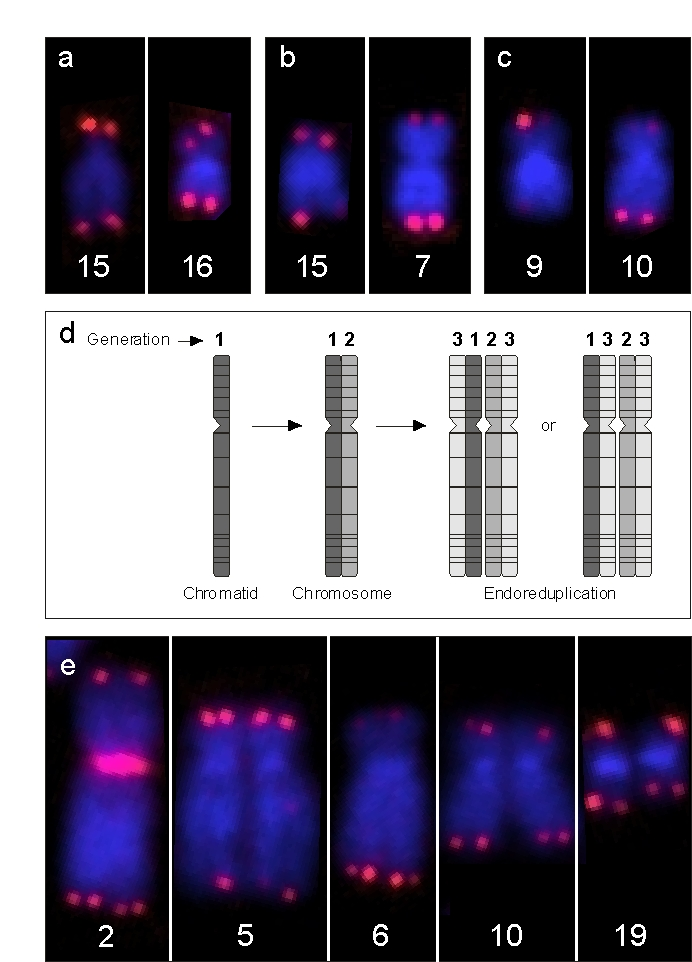
Figure 3. Examples of EEE. (a) Depicts chromosomes of a newborn male
with enhanced signal intensity at one p arm of chromosome 15 and at both
chromatids of chromosome 16, (b). This is also the case at a single
chromatid of 15q and on both chromatids of chromosome 7q in a 50-year-old
male and, (c) at a single chromatid of chromosome 9p and at both
arms of chromosome 10q in a male centenarian. (d) Schematic view of
endoreduplication (ER), the result being a group of four homologous
chromatids which emerged from one single chromatid. Note that the
juxtapositions of the sister and descendant chromatids may be variable,
since their three-dimensional packing is broken up by chromosome spreading.
(e) Single chromosomes of a sporadic ER observed in a 40-year-old
female with features of EEE. Since telomeric EEEs at 2p, 5q, 6q, and 10p
are doublets and not quadruplets, EEE at these positions must have occurred
one cell cycle prior to the ER event. In addition, there is a single
telomeric EEE at 19q, which must have occurred during the S-phase directly
preceding this ER.
Phenomenologically, EEE is more likely to be a repair of accidental individual telomere damage than a mechanism operating continuously. Following this line of argument, it is very unlikely that telomerase is responsible for EEE, since telomerase-stabilized telomeres show constant lengths of around 4-10 kb in humans (for review, see [13]). A fitting candidate mechanism is the alternative pathway (ALT). ALT is involved in maintenance or lengthening of telomeres, uses enzymes operative in DNA recombination and rep- lication, and can create extreme heterogeneity in telomere lengths [13] by using so-called telomeric circles. Theoretically, these circles can arise from telomeres by means of homologous recombination [14] and might be used by telomerase as a template to elongate telomeres as has been proposed [15].
ALT was found to be active in neoplastic cells and immortalized cell lines with or without telomerase [14]. A physiological role of ALT is suggested by the observation that telomerase-null mice show some degree of telomere elongation in B-lymphocytes during germinal center reaction [16]. Furthermore, Rad54-null mice have significantly shorter telomeres than wild animals, in spite of unimpaired telomerase activity [17]. Rad54 is involved in DNA recombination and may be part of ALT. We frequently found telomeric EEE of around 150 T/C values (corresponding to 33 kb) and, in rare cases, even up to 300 T/C value (63 kb). Our observations thus match data of mean telomere lengths of over 50 kb, as measured by terminal restriction fragment analysis in so-called human ALT-positive cell lines [18]. Finally, if ALT is indeed the key player in EEE, which still has to be proven, the question remains whyfemales use ALT more effectively.
As we and others have clearly demonstrated a linear decline in telomere length [4,6,8-11], it seems at least unlikely that sex hormones play a role. It is, however, well documented that exposure to reactive oxygen species (ROS) leads to damage in nuclear DNA (see [19] and reviews [20,21]), and to telomere shortening [22], and a growing body of evidence indicates that males are subject to higher levels of cellular stress than females [23-28]. A direct influence of the extracellular environment, the biosphere, on telomere length was recently shown in a pair of twins of different gender with blood chimerism [29]: Compared with their "normal" length (i.e. female cells in the woman and male cells in the man), the telomeres within the male lymphocytes in the female twin were 33% longer than those in the male twin. By contrast, comparison of the telomeres within the female lymphocytes in the female with their counterparts in the male revealed that the latter were shortened to 87% of their "normal" length.
Apart from direct damaging influences, ROS can serve as messengers to control other physiological processes not directly involved in ROS defense [20,30,31]. For example, ROS reacts with plasma thiol to form disulfides. This extracellular thiol/disulfide equilibrium has signaling functions on different cell membrane receptors, e.g. EGFR, and regulates poly ADP-ribose polymerase (PARP) [32,33]. EGFR is involved in the expression of hTERT, which controls telomerase activity [34], and PARP is a key DNA repair enzyme and is involved in DNA replication, recombination [35], telomere maintenance (for review, see [36]), and DNA histone modifications and double-strand break repair [37].
Against this background, it is tempting to speculate on an interplay between ROS, the thiol/disulfide system and the control of telomere length. All the more, since telomere erosion can be slowed by N-acetylcysteine, which changes the thiol/disulfide system and inhibits ROS formation [20,38,39]. In conclusion, this study provides evidence that the gender-specific differences in telomere attrition depend on factors leading, in the female, to enhanced repair/recombination processes in the lymphocytes (visible by EEE) and, finally, to longer telomeres (evident by greater skewness and kurtosis of the female histogram). Time will show whether it contributes to the gender differences in life expectancy.
Materials and methods
Cell sources. Informed written consent was obtained from each participant and from the parents of the newborns and children. Blood samples from a total of 205 donors were used: 108 umbilical cord blood samples from 55 male and 53 female newborns and 97 peripheral blood samples from donors ranging from 10 to 100 years of age (five female and five male, each in every life decade of life), with the exception of male centenarians, of which we had only two.
Cell culture and metaphase preparation. Heparinized blood was cultured as described [9] to obtain lymphocytes which were then phytohemagglutinin-stimulated to obtain metaphases. Twenty metaphases from each individual were analyzed in a Zeiss Axioscope microscope (Jena, Germany) equipped with a CCD camera and linked to the Isis and telomere software (MetaSystems, Altlussheim, Germany).
Peptide nucleic acid (PNA) probes. The PNA probe for telomeric sequences is a ready-to-use probe included in the Telomere PNA FISH Kit/Cy3 (K5326 from DakoCytomation A/S, Glostrup, Denmark). The PNA centromeric probe for chromosome 2 was generated by Dako and is available upon request. The PNA probe for the centromere 2 was added as 1 μl to 10 μl ready-to-use telomere probe solution leading to a final concentration of 2 ng/μl. The hybridization was performed according to the manufacturer's instructions and is described in detail in reference 9.
Statistical analysis of T/C data . Normalized data were derived by calculating the ratio of the absolute telomere intensities and the reference signal intensities of each metaphase (T/C value). For each individual, about 20 metaphases were analyzed. These primary data were compiled by the software, and mean telomere intensities of the p- and q-arms of each chromosome were obtained. These data were used for further statistics.
Firstly, an attempt was made to characterize the statistical distribution of all individual T/C values as a whole. To this end, the T/C data of all 46 chromosome arms from 102 males (n1 = 119,472) and 103 females (n2 = 115,754) were used. The skewness of the empirical frequency distribution of the individual T/C values was estimated for the male and the female group using standard software (SAS Institute 2000). The skewness estimates amounted to 1.099 in the male group and to 1.322 in the female group. To test these values for a significant difference, a randomization test based on independent random resampling without replacement was performed in the following manner [40-42]: All data were united to a single univariate set consisting of 235,226 T/C values. From this set, 9999 pairs of random samples with sizes n1 = 115,754 and n2 = 119,472 were drawn independently without replacement. For each pair of samples, the difference D* between the two estimated skewness values was calculated. The resulting 9999 D*-values of the skewness estimates from the virtual random samples with T/C values from both genders, and the difference D between the skewness estimates originating from the two real samples (male and female only), were sorted by size. A result was considered as significant at the 5%-level if D occupied rank 1-250, or rank 9751-10000 in the whole series of difference values. The D-value from the real sample pair, D = 0.223, occupied rank 9951/10000, hence it was considered significant at a level of α= 0.0098.
Furthermore, the functional relationship between telomere size and age was studied for both sexes. The medians of the T/C values were determined in the 205 individuals separately for the telomeres of all 46 chromosome regions. This part of the study yielded 4896 median T/C values in the male group and 4738 median T/C values in the female group. Linear regression analysis led to slope estimates ofg = -0.302 for the female group and g = -0.328 in the male group, i.e. the estimated regression line showed negative gradients with a slightly steeper descent for the male gender. A randomization test with resampling without replacement, analogous to the approach described above, was performed to test the slopes of the two regression lines for a significant difference. To this end, the two bivariate samples were united to a single bivariate sample with 9634 elements. From this united sample, 9999 pairs of bivariate samples (with age and T/C values from both genders) of sizes n1 = 4896 and n2 = 4738 were drawn at random without replacement. For both virtual samples of each pair, regression lines of T/C value on age were fitted by standard methods. The difference D* between the two slope estimates was recorded for each virtual sample pair. Thereafter, the 999 D*-values and the difference value D = 0.026 from the pair of regression lines corresponding to the true female and male sample were ranked by size. The true D-value occupied rank 10000/10000 in this series, hence it was considered significant at a level of α ≤ 0.0001.
Acknowledgments
We would like to thank C. Hasel for her participation in this study and C. Higginson for editorial work. The corresponding author had full access to all the data in this study and has the final responsibility of submission.
Conflicts of Interest
The authors in this manuscript have no conflict of interests to declare.
References
- 1. Anderson RN and Smith BL. Deaths: leading causes for 2001. Natl Vital Stat Rep. 2003; 52: 1 -85. .
- 2. Hayflick L The limited in vitro lifetime of human diploid cell strains. Exp Cell Res. 1965; 37: 614 -636. [PubMed] .
- 3. Harley CB , Futcher AB and Greider CW. Telomeres shorten during ageing of human fibroblasts. Nature. 1990; 345: 458 -460. [PubMed] .
- 4. Slagboom PE , Droog S and Boomsma DI. Genetic determination of telomere size in humans: a twin study of three age groups. Am J Hum Genet. 1994; 55: 876 -882. [PubMed] .
- 5. Allsopp RC , Vaziri H , Patterson C , Goldstein S , Younglai EV , Futcher AB , Greider CW and Harley CB. Telomere length predicts replicative capacity of human fibroblasts. Proc Natl Acad Sci USA. 1992; 89: 10114 -10118. [PubMed] .
- 6. Mayer S , Brüderlein S , Perner S , Waibel I , Holdenried A , Ciloglu N , Hasel C , Mattfeldt T , Nielsen KV and Möller P. Sex-specific telomere length profiles and age-dependent erosion dynamics of individual chromosome arms in humans. Cytogenet Genome Res. 2006; 112: 194 -201. [PubMed] .
- 7. Blackburn EH Switching and signaling at the telomere. Cell. 2001; 106: 661 -673. [PubMed] .
- 8. Cawthon RM , Smith KR , O'Brien E , Sivatchenko A and Kerber RA. Association between telomere length in blood and mortality in people aged 60 years or older. Lancet. 2003; 361: 393 -395. [PubMed] .
- 9. Perner S , Brüderlein S , Hasel C , Waibel I , Holdenried A , Ciloglu N , Chopurian H , Nielsen KV , Plesch A , Högel J and Möller P. Quantifying telomere lengths of human individual chromosome arms by centromere-calibrated fluorescence in situ hybridization and digital imaging. Am J Pathol. 2003; 163: 1751 -1756. [PubMed] .
- 10. Iwama H , Ohyashiki K , Ohyashiki JH , Hayashi S , Yahata N , Ando K , Toyama K , Hoshika A , Takasaki M , Mori M and Shay JW. Telomeric length and telomerase activity vary with age in peripheral blood cells obtained from normal individuals. Hum Genet. 1998; 102: 397 -402. [PubMed] .
- 11. Nawrot TS , Staessen JA , Gardner JP and Aviv A. Telomere length and possible link to X chromosome. Lancet. 2004; 363: 507 -510. [PubMed] .
- 12. Grafi G Cell cycle regulation of DNA replication: the endoreduplication perspective. Exp Cell Res. 1998; 244: 372 -378. [PubMed] .
- 13. Scheel C and Poremba C. Telomere lengthening in telomerase-negative cells: the ends are coming together. Virchows Arch. 2002; 440: 573 -582. [PubMed] .
- 14. Reddel RR Alternative lengthening of telomeres, telomerase, and cancer. Cancer Lett. 2003; 194: 155 -162. [PubMed] .
- 15. Natarajan S and McEachern MJ. Recombinational telomere elongation promoted by DNA circles. Mol Cell Biol. 2002; 22: 4512 -4521. [PubMed] .
- 16. Herrera E , Martinez A and Blasco MA. Impaired germinal center reaction in mice with short telomeres. EMBO J. 2000; 19: 472 -481. [PubMed] .
- 17. Jaco I , Munoz P , Goytisolo F , Wesoly J , Bailey S , Taccioli G and Blasco MA. Role of mammalian Rad54 in telomere length maintenance. Mol Cell Biol. 2003; 23: 5572 -5580. [PubMed] .
- 18. Henson JD , Neumann AA , Yeager TR and Reddel RR. Alternative lengthening of telomeres in mammalian cells. Oncogene. 2002; 21: 598 -610. [PubMed] .
- 19. Von Zglinicki T , Burkle A and Kirkwood TB. Stress, DNA damage and ageing -- an integrative approach. Exp Gerontol. 2001; 36: 1049 -1062. [PubMed] .
- 20. Dröge W Free radicals in the physiological control of cell function. Physiol Rev. 2002; 82: 47 -95. [PubMed] .
- 21. Chen JH , Hales CN and Ozanne SE. DNA damage, cellular senescence and organismal ageing: causal or correlative. Nucleic Acids Res. 2007; 35: 7417 -7428. [PubMed] .
- 22. Von Zglinicki T , Pilger R and Sitte N. Accumulation of single-strand breaks is the major cause of telomere shortening in human fibroblasts. Free Radic Biol Med. 2000; 28: 64 -74. [PubMed] .
- 23. Lacy F , Kailasam MT , O'Connor DT , Schmid-Schonbein GW and Parmer RJ. Plasma hydrogen peroxide production in human essential hypertension: role of heredity, gender, and ethnicity. Hypertension. 2000; 36: 878 -884. [PubMed] .
- 24. Tsutsui T , Kumakura S , Tamura Y , Tsutsui TW , Sekiguchi M , Higuchi T and Barrett JC. Greater oxidative stress in healthy young men compared with premenopausal women. Arterioscler Thromb Vasc Biol. 2002; 22: 438 -442. [PubMed] .
- 25. Proteggente AR , England TG , Rehman A , Rice-Evans CA and Halliwell B. Gender differences in steady-state levels of oxidative damage to DNA in healthy individuals. Free Radic Res. 2002; 36: 157 -162. [PubMed] .
- 26. Dantas AP , Franco MC , Silva-Antonialli MM , Tostes RC , Fortes ZB , Nigro D and Carvalho MH. Gender differences in superoxide generation in microvessels of hypertensive rats: role of NAD(P)H-oxidase. Cardiovasc Res. 2004; 61: 22 -29. [PubMed] .
- 27. Ali SS , Xiong C , Lucero J , Behrens MM , Dugan LL and Quick KL. Gender differences in free radical homeostasis during aging: shorter-lived female C57BL6 mice have increased oxidative stress. Aging Cell. 2006; 5: 565 -574. [PubMed] .
- 28. Malorni W , Campesi I , Straface E , Vella S and Franconi F. Redox features of the cell: a gender perspective. Antioxid Redox Signal. 2007; 9: 1779 -1801. [PubMed] .
- 29. Brüderlein S , Müller K , Melzner J , Högel J , Wiegand P and Möller P. Different rates of telomere attrition in peripheral lymphocytes in a pair of dizygotic twins with hematopoietic chimerism. Aging Cell. 2008; 7: 663 -666. [PubMed] .
- 30. Go YM and Jones DP. Intracellular proatherogenic events and cell adhesion modulated by extracellular thiol/disulfide redox state. Circulation. 2005; 111: 2973 -2980. [PubMed] .
- 31. Jones DP Redefining oxidative stress. Antioxid Redox Signal. 2006; 8: 1865 -1879. [PubMed] .
- 32. Banne AF , Amiri A and Pero RW. Reduced level of serum thiols in patients with a diagnosis of active disease. J Anti Aging Med. 2003; 6: 327 -334. [PubMed] .
- 33. Hartwig A , Asmuss M , Blessing H , Hoffmann S , Jahnke G , Khandelwal S , Pelzer A and Burkle A. Interference by toxic metal ions with zinc-dependent proteins involved in maintaining genomic stability. Food Chem Toxicol. 2002; 40: 1179 -1184. [PubMed] .
- 34. Pendino F , Tarkanyi I , Dudognon C , Hillion J , Lanotte M , Aradi J and Segal-Bendirdjian E. Telomeres and telomerase: Pharma-cological targets for new anticancer strategies. Curr Cancer Drug Targets. 2006; 6: 147 -180. [PubMed] .
- 35. Petermann E , Keil C and Oei SL. Importance of poly(ADP-ribose) polymerases in the regulation of DNA-dependent processes. Cell Mol Life Sci. 2005; 62: 731 -738. [PubMed] .
- 36. Schreiber V , Dantzer F , Ame JC and de Murcia G. Poly(ADP-ribose): novel functions for an old molecule. Nat Rev Mol Cell Biol. 2006; 7: 517 -528. [PubMed] .
- 37. Dantzer F , Mark M , Quenet D , Scherthan H , Huber A , Liebe B , Monaco L , Chicheportiche A , Sassone-Corsi P , de Murcia G and Menissier-de Murcia J. Poly(ADP-ribose) polymerase-2 contributes to the fidelity of male meiosis I and spermiogenesis. Proc Natl Acad Sci USA. 2006; 103: 14854 -14859. [PubMed] .
- 38. Hoffmann J , Diehl JF , Vasa M , Spyridopoulos I , Zeiher AM and Dimmeler S. Antioxidants inhibit nuclear export of telomerase reverse transcriptase and delay replicative senescence of endothelial cells. Circ Res. 2004; 94: 768 -775. [PubMed] .
- 39. Liu L , Trimarchi JR , Navarro P , Blasco MA and Keefe DL. Oxidative stress contributes to arsenic-induced telomere attrition, chromosome instability, and apoptosis. J Biol Chem. 2003; 278: 31998 -32004. [PubMed] .
- 40. Noreen EW New York Wiley Computer intensive methods for testing hypothesis: An introduction. 1989; .
- 41. Manly BF Randomization and MonteCarlo methods in biology. London: Chapman and Hall. London: Chapman and Hall. 1991; 1505 -1509. .
- 42. Ludbrook J Computer-intensive statistical procedures. Crit Rev Biochem Mol Biol. 2000; 35: 339 -358. [PubMed] .


