Age-related macular degeneration (AMD) is the leading cause of irreversible blindness in the elderly [1-2]. Considerable evidence supports the opinion that the retinal pigment epithelium (RPE), a monolayer of cells between the light sensitive photoreceptors and the vascular choroid, is a primary site of pathology in the disease [1-5]. The RPE provides support for the photoreceptors and plays a critical role in the visual cycle; thus, degeneration and loss of RPE lead to secondary degeneration of photoreceptor cells [3]. Early AMD is characterized by the presence of extracellular deposits, or drusen, beneath the RPE. Increasing numbers of large drusen predispose to the development of the late blinding forms of the disease which can manifest in two disparate ways. In late, "dry" AMD, geographic loss of RPE occurs in the macular region, while in the late neovascular or "wet" form of the disease, there is abnormal growth of choroidal vessels under the retina which leak fluid and may progress to form a disciform scar (Figure 1) [1-5]. Pathogenic mechanisms for AMD include both genetic and environmentalfactors related to primary RPE senescence, alterations in the complement pathway, increased inflammation, changes in the balance of growth factors, excessive lipofuscinaccumulation, and oxidative stress [5]. Major genetic risk factors for AMD, including Complement Factor H and HTRA1 variants, appear to predispose to both atrophic and neovascular AMD [6,7]; only recently has a genetic variant been identified that specifically predisposes to the atrophic form [8]. Consequently, there is considerable interest in further establishing the factors that mediate the "molecular switch" that may determine which late form of the disease an individual develops.
Recently, we reported that bone morphogenetic protein (BMP)4 is prominently expressed in the RPE and adjacent extracellular matrix of patients with the dry or atrophic form of AMD when compared to controls (Figure 2A, B). Here, we show that in the wet or neovascular form of the disease (5 patients with surgical excision of choroidal neovascular membranes due to neovascular AMD) there is almost no expression of BMP4 in the RPE and adjacent neovascular tissues (Figure 2C). Interestingly, in cases (3 patients) in which the neovascular lesion had progressed to a fibrous scar, the level of BMP4 expression increased in the RPE and adjacent tissues (Figure 2D). This has led us to the hypothesis that BMP4 may be a molecular switch participating in the pathway decision that determines which form of late AMD develops.
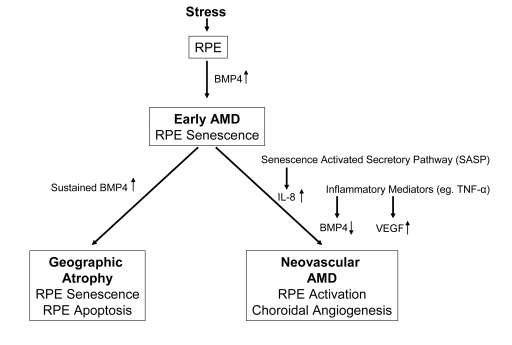
Figure 1. Diagram
illustrating the progression of early age related macular degeneration
(AMD) into 2 divergent late stages and the potential role of BMP4 as a
switch between these pathways. Chronic stressors such as oxidative stress
can promote the expression of BMP4 in the retinal pigment epithelium (RPE)
and induce RPE senescence as part of the phenotype of early AMD. If BMP4
expression is sustained, it could lead to RPE apoptosis and geographic
atrophy. In other individuals, activation of the senescence activated
secretory pathway and expression of pro-inflammatory mediators could result
in increased expression of interleukin (IL)-8, decreased expression of BMP4
and increased expression of vascular endothelial growth factor (VEGF)
resulting in neovascular AMD with choroidal angiogenesis.
BMP4 is an important regulator of differentiation, senescence and apoptosis in many different cells and tissues [9,10]. We reported that BMP4 can induce RPE senescence in vitro [11], and that RPE chronically exposed to sublethal doses of oxidative stress can increase their BMP4 expression and exhibit a senescent phenotype, thus supporting the contention that BMP4 mediates oxidative stress-induced RPE senescence. We further determined that BMP4 mediates RPE senescence via activation of Smad and p38 pathways to activate p53, and increase expression of p21WAF1/ cip1, and to decrease phospho-Rb. Importantly, BMP4-mediated RPE senescence can be inhibited by Chordin-like, a BMP4 antagonist, and SB203580, a phospho-p38 inhibitor. Our findings not only disclose a molecular pathway linking oxidative stress with RPE senescence, but also provide a novel therapeutic target for treatment of atrophic AMD.
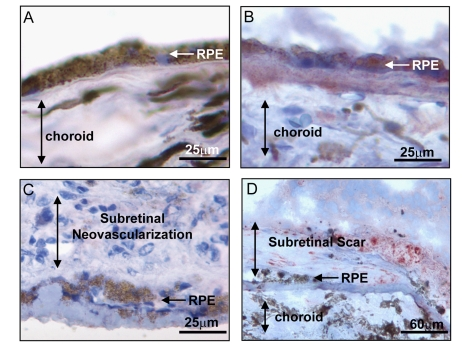
Figure 2.
Expression of BMP4 in late stages of age related macular degeneration
(AMD). Immunohistochemical stains for BMP4 (red chromogen) in retinal
pigment epithelium (RPE)/choroid tissue sections from donor eyes with
hematoxylin counterstain. In (A) a control individual without AMD
shows no apparent BMP4 staining in RPE or choroid. In (B) an
individual with late dry AMD, away from a region of geographic atrophy
shows prominent BMP4 immunoreactivity in RPE and in the accumulated drusen
material between the RPE and the choroid. In (C) an individual with
neovascular form of late AMD shows no apparent BMP4 staining in the RPE or
the neovascular lesion between the RPE and retina. In (D) an
individual with neovascular form of late AMD that further progressed to
scar with loss of neovascular channels shows re-expression of BMP4 staining
in cells within and adjacent to the lesion. Note loss of most cells in RPE
layer. The institutional review board (IRB) of
the University of Southern California approved our use of human donor eyes.
All procedures conformed to the Declaration of Helsinki forresearch
involving human subjects.
Recently, Demidenko et al. [12] evaluated the concept that duration of cell cycle arrest determines the progressive loss of proliferative capacity characteristic of cellular senescence [12]. Using a variety of cell lines including the spontaneously immortalized human RPE cell line, ARPE-19, they found that rapamycin, an inhibitor of the nutrient-sensor mammalian target of rapamycin (mTOR), partially prevented loss of proliferative potential induced by oxidative stress, or ectopic p21 or p16 exposure, leading to deceleration of cellular senescence [12]. This work supports the critical role of oxidative stress, and cell cycle arrest in induction of senescence and demonstrates a pharmacologic approach to suppression of RPE senescence [12].
Interestingly, BMP4 has been found to be involved in chemotherapeutic agent-induced premature senescence of cancer cells [13]. Adriamycin and BMP4 treatment can induce lung cancer cell senescence, and BMP4 expression is increased in Adriamycin-treated lung cancer cells. This BMP4-induced premature senescence is mediated through Smad signaling to up-regulate p16INK4a and p21WAF1/ cip1. BMP4 and other BMP signaling pathways were also found to participate in senescence of multiple cancer cell types or in the inhibition of tumor cell growth [14,15]. For example, BMP-2 and -4 inhibit prostate cancer cell growth through Smad-1 phosphorylation, p21WAF1/ cip1 up-regulation, and Rb dephosphorylation, while in glioblastoma, BMP4 and its cognate receptors can trigger the Smad signaling cascade to reduce the proliferation of tumor cells [16]. Together, these studies reveal that BMP4 induces and mediates the premature senescence of both malignant cells in tumors and aging RPE cells in dry AMD.
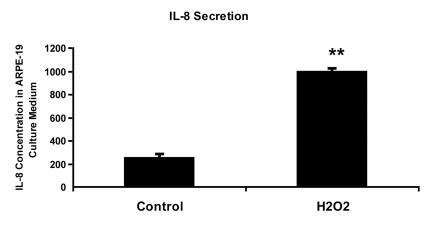
Figure 3.
IL-8 protein concentration in culture medium measured by ELISA. ARPE-19
cells were treated with 150 uM H2O2 in culture medium
with 10% fetal bovine serum for 2 hours and allowed to recover in stressor-free
ARPE medium for 22 hours. The procedure was repeated to generate the next
treatment cycle. The
twice treated cells were allowed to stay in 1% serum ARPE medium for 72
hours after stress before proceeding to further analytic assays.
The culture media from control and senescent RPE cells were collected and
used directly for ELISA measurement. IL-8 secretion level was measured in pg/ml using
human IL-8 ELISA Kit (BioLegend, Inc., San Diego, CA) according to
manufacturer's instructions. The level of IL-8 secretion shown here was
averaged from a triplicate of each sample and from 3 independent repeats of
H2O2 treatments. Student's t test was used for
statistical analysis (**; p < 0.0005).
Transforming growth factor (TGF)-β has been extensively reported to be involved in mediating oxidative stress induced premature senescence of fibroblasts [17-19]. Recently it has been reported that TGF-β mediates oxidative stress induced RPE cell senescence through the up-regulation of p21 WAF1/ cip1 and the down-regulation of phosphorylated Rb and that blockade of TGF-β signaling by specific TGF-β antibody can impede RPE senescence [20]. This finding is similar to our finding for BMP4 mediated oxidative stress-induced RPE senescence. We suggest that TGF-β and BMP4 may have a synergistic effect in mediating the oxidative stress-induced RPE senescence, because neither TGF-β antibodies nor BMP4 antagonist alone can completely block the expression of senescence marker genes to baseline in the oxidative stress treated RPE cells. More investigations are needed to elucidate the interactions between TGF-β and BMP signaling cascades in oxidative stress-induced RPE senescence.
A variety of intrinsic and extrinsic stress signals can activate the p53 pathway, which then triggers either cellular senescence or apoptosis [21,22]. We found that both BMP4 and oxidant treatment can increase p53 protein level in RPE cells. A microarray analysis of the RPE transcriptome from the maculas of six healthy, elderly human donors revealed a statistically significant overrepresentation of genes associated with stress, with the p53 gene listed in the top 30 most highly expressed RPE genes [23]. Although little is known about how p53 regulates cellular senescence and how p53 interacts with the BMP-Smad pathway, the fact that p53 levels were increased in RPE cells after BMP4 treatment and Smad1/5 could bind to p53 [24], raises the possibility that Smad1/5 activates p53 dependent transcription through the regulation of post-translational modifications of p53, such as phosphorylation and acetylation.
It remains unanswered why some patients develop atrophic AMD while others develop the neovascular form of the disease. The switch between dry and wet AMD may be related to differences in the microenvironment created by senescent RPE cells, which secrete a number of cytokines and growth factors [25]. The defined components of this "senescence associated secretory phenotype" (SASP) include elements associated with inflammation, and angiogenesis, such as interleukin (IL)-6 and IL-8 [26-28]. We have found that RPE cells induced into senescence by chronic oxidative stress secrete 4 times higher IL-8 than non-senescent RPE cells (Figure 3). IL-8 promotes angiogenesis by increasing the proliferation, survival and migration of endothelial cells and promotes inflammation by increasing neutrophil chemotaxis and degranulation [29-31]. Together these findings suggest that chronic oxidative stress increases the premature senescence of RPE. If RPE do not go down the cell death pathway to atrophic AMD, the senescent RPE may secrete high levels of IL-8, which in turn stimulate inflammation and angiogenesis. But what about the finding that neovascular AMD lesions show minimal levels of BMP4? In other cell types, pro-inflammatory mediators such as tumor necrosis factor (TNF)-alpha have been shown to downregulate BMP4 expression [32]. In the absence of BMP4, neovascular endothelial cells, stimulated by increased expression of vascular endothelial growth factor, and without the growth inhibitory senescence and cell death effects mediated by BMP4, would be in a permissive environ-ment for angiogenesis [5]. This idea is further supported by the finding that when neovascular AMD lesions undergo subsequent scar formation, with degeneration and loss of neovascular endothelial cells, there is a concomitant increase in BMP4 expression (Figure 2).
It has been previously observed that tissues in aged individuals may exhibit the paradoxical juxtaposition of atrophy and hyperplasia within the same tissue or even within the same cell type [33]. This response may be explained in part by senescent heterogeneity [34,35]. In vitro culture of human fibroblasts results in a fraction of cells senescing at every population doubling. The senescent cells have shorter telomeres than their cycling counterparts. Thus, it was concluded that the main cause of intrinsic heterogeneity of senescent fibroblasts was the cell to cell variation of telomere shortening [36]. Using pulse-chase 5-bromodeoxyuridine-labeling assay, Gonzalez and colleagues revealed that the senescent heart contained functionally competent cardiac progenitor cells (CPCs) with longer telomeres, and these stem cell-like CPCs can be activated and migrate to the damaged regions to generate a population of young cardiomyocytes and partly reverse the aging myopathy [37].
Much remains to be learned about the genetic and environmental factors mediating the progression of early AMD to its late forms. Our finding of differential expression of BMP4 in geographic atrophy and neovascular AMD and the interactive roles of oxidative stress, inflammation and senescence in the regulation and functional effects of this growth factor, suggests the possibility that BMP4 may be playing a part in the molecular switch determining which phenotypic pathway is taken in the progression of AMD.
Acknowledgments
This work was supported by National Institutes of Health grants EY01545 and EY03040 and by the Arnold and Mabel Beckman Foundation.
Conflicts of Interest
The authors of this manuscript have no conflict of interest to declare.
References
- 1. Zarbin MA Current concepts in the pathogenesis of age-related macular degeneration. Arch Ophthalmol. 2004; 122: 598 -614. [PubMed] .
- 2. Ambati J , Ambati BK , Yoo SH , Ianchulev S and Adamis AP. Age-related macular degeneration: etiology, pathogenesis, and therapeutic strategies. Surv Ophthalmol. 2003; 48: 257 -293. [PubMed] .
- 3. Holz FG , Pauleikhoff D , Klein R and Bird AC. Pathogenesis of lesions in late age-related macular disease. American journal of ophthalmology. 2004; 137: 504 -510. [PubMed] .
- 4. Klein ML , Ferris FL 3rd , Armstrong J , Hwang TS , Chew EY , Bressler SB and Chandra SR. Retinal precursors and the development of geographic atrophy in age-related macular degeneration. Ophthalmology. 2008; 115: 1026 -1031. [PubMed] .
- 5. Ding X , Patel M and Chan CC. Molecular pathology of age related macular degeneration. Prog Retin Eye Res. 2009; 28: 1 -18. [PubMed] .
- 6. Cameron DJ , Yang Z , Gibbs D , Chen H , Kaminoh Y , Jorgensen A , Zeng J , Luo L , Brinton G , Brand JM , Bernstein PS , Zabriskie NA , Tang S , Constantine R , Tong Z and Zhang K. HTRA1 variant confers similar risks to geographic atrophy and neovascular age-related macular degeneration. Cell Cycle. 2007; 6: 1122 -1125. [PubMed] .
- 7. Sepp T , Khan JC , Thurlby DA , Shahid H , Clayton DG , Moore AT , Bird AC and Yates JR. Complement factor H variant Y402H is a major risk determinant for geographic atrophy and choroidal neovascularization in smokers and nonsmokers. Invest Ophthalmol Vis Sci. 2006; 47: 536 -540. [PubMed] .
- 8. Yang Z , Stratton C , Francis P , Kleinman ME , Tan PL , Gibbs D , Tong Z , Chen H , Constantine R , Yang X , Chen Y , Zeng J , Davey L , Ma X , Hau VS , Wang C , Harmon J , Buehler J , Pearson E , Patel S , Kaminoh Y , Watkins S , Luo L , Zabriskie NA , Berstein PS , Cho W , Schwager A , Hinton DR , Klein ML , Hamon SC , Simmons E , Sunness JS , Campochiaro P , Jorde L , Parmigiani G , Zack DL , Katsanis N , Ambati J and Zhang K. Toll-Like Receptor-3 and Geographic Atrophy in Age-Related Macular Degeneration. New Engl J Med. 2008; 359: 1456 -63. [PubMed] .
- 9. Yokouchi Y , Sakiyama J , Kameda T , Iba H , Suzuki A , Ueno N and Kuroiwa A. BMP-2/-4 mediate programmed cell death in chicken limb buds. Development. 1996; 122: 3725 -3734. [PubMed] .
- 10. Trousse F , Esteve P and Bovolenta P. Bmp4 mediates apoptotic cell death in the developing chick eye. J Neurosci. 2001; 21: 1292 -1301. [PubMed] .
- 11. Zhu D , Wu J , Spee C , Ryan SJ and Hinton DR. BMP4 mediates oxidative stress-induced retinal pigment epithelial cell senescence and is overexpressed in age-related macular degeneration. J Biol Chem. 2009; 284: 9529 -9539. [PubMed] .
- 12. Demidenko ZN , Zubova SG , Bukreeva EI , Pospelov VA , Pospelova TV and Blagosklonny MV. Rapamycin decelerates cellular senescence. Cell Cycle. 2009; 8: 1888 -1895. [PubMed] .
- 13. Su D , Zhu S , Han X , Feng Y , Huang H , Ren G , Pan L , Zhang Y , Lu J and Huang B. BMP4-Smad signaling pathway mediates adriamycin-induced premature senescence in lung cancer cells. J Biol Chem. 2009; 284: 12153 -12164. [PubMed] .
- 14. Brubaker KD , Corey E , Brown LG and Vessella RL. Bone morphogenetic protein signaling in prostate cancer cell lines. J Cell Biochem. 2004; 91: 151 -160. [PubMed] .
- 15. Fukuda N , Saitoh M , Kobayashi N and Miyazono K. Execution of BMP-4-induced apoptosis by p53-dependent ER dysfunction in myeloma and B-cell hybridoma cells. Oncogene. 2006; 25: 3509 -3517. [PubMed] .
- 16. Piccirillo SG , Reynolds BA , Zanetti N , Lamorte G , Binda E , Broggi G , Brem H and Olivi A. Bone morphogenetic proteins inhibit the tumorigenic potential of human brain tumour-initiating cells. Nature. 2006; 444: 761 -765. [PubMed] .
- 17. Frippiat C , Chen QM , Zdanov S , Magalhaes JP , Remacle J and Toussaint O. Subcytotoxic H2O2 stress triggers a release of transforming growth factor-beta 1, which induces biomarkers of cellular senescence of human diploid fibroblasts. J Biol Chem. 2001; 276: 2531 -2537. [PubMed] .
- 18. Chretien A , Dierick JF , Delaive E , Larsen MR , Dieu M , Raes M , Deroanne CF and Roepstorff P. Role of TGF-beta1-independent changes in protein neosynthesis, p38alphaMAPK, and cdc42 in hydrogen peroxide-induced senescence-like morphogenesis. Free Radic Biol Med. 2008; 44: 1732 -1751. [PubMed] .
- 19. Zdanov S , Remacle J and Toussaint O. Establishment of H2O2-induced premature senescence in human fibroblasts concomitant with increased cellular production of H2O2. Ann N Y Acad Sci. 2006; 1067: 210 -216. [PubMed] .
- 20. Yu AL , Fuchshofer R , Kook D , Kampik A , Bloemendal H and Welge-Lussen U. Subtoxic oxidative stress induces senescence in retinal pigment epithelial cells via TGF-beta release. Invest Ophthalmol Vis Sci. 2009; 50: 926 -935. [PubMed] .
- 21. Vousden KH and Lane DP. p53 in health and disease. Nat Rev Mol Cell Biol. 2007; 8: 275 -283. [PubMed] .
- 22. Harris SL and Levine AJ. The p53 pathway: positive and negative feedback loops. Oncogene. 2005; 24: 2899 -2908. [PubMed] .
- 23. Booij JC , van Soest S , Swagemakers SM , Essing AH , Verkerk AJ and van der Spek PJ. Functional annotation of the human retinal pigment epithelium transcriptome. BMC Genomics. 2009; 10: 164 [PubMed] .
- 24. Takebayashi-Suzuki K , Funami J , Tokumori D , Saito A , Watabe T , Miyazono K , Kanda A and Suzuki A. Interplay between the tumor suppressor p53 and TGF beta signaling shapes embryonic body axes in Xenopus. Development. 2003; 130: 3929 -3939. [PubMed] .
- 25. Zender L and Rudolph KL. Keeping your senescence cells under control. Aging. 2009; 1: 1 -4. [PubMed] .
- 26. Koch AE , Polverini PJ , Kunkel SL , Harlow LA , DiPietro LA , Elner SG and Strieter RM. Interleukin-8 as a macrophage-derived mediator of angiogenesis. Science. 1992; 258: 1798 -1801. [PubMed] .
- 27. Yoshimura T , Matsushima K , Tanaka S , Robinson EA , Appella E , Oppenheim JJ and Leonard EJ. Purification of a human monocyte-derived neutrophil chemotactic factor that has peptide sequence similarity to other host defense cytokines. Proc Natl Acad Sci U S A. 1987; 84: 9233 -9237. [PubMed] .
- 28. Kuilman T , Michaloglou C , Vredeveld LC , Douma S , van Doorn R , Desmet CJ , Aarden LA , Mooi WJ and Peeper DS. Oncogene-induced senescence relayed by an interleukin-dependent inflammatory network. Cell. 2008; 133: 1019 -1031. [PubMed] .
- 29. Coppe JP , Patil CK , Rodier F , Sun Y , Munoz DP , Goldstein J , Nelson PS , Desprez PY and Campisi J. Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS biology. 2008; 6: 2853 -2868. [PubMed] .
- 30. Malgorzata Goczalik I , Raap M , Weick M , Milenkovic I , Heidmann J , Enzmann V , Wiedemann P , Reichenbach A and Francke M. The activation of IL-8 receptors in cultured guinea pig Muller glial cells is modified by signals from retinal pigment epithelium. Journal of neuroimmunology. 2005; 161: 49 -60. [PubMed] .
- 31. Waugh DJ and Wilson C. The interleukin-8 pathway in cancer. Clin Cancer Res. 2008; 14: 6735 -6741. [PubMed] .
- 32. Zhu NL , Li C , Huang HH , Sebald M , Londhe VA , Heisterkamp N , Warburton D , Bellusci S and Minoo P. TNF-alpha represses transcription of human bone morphogenetic protein-4 in lung epithelial cells. Gene. 2007; 393: 70 -80. [PubMed] .
- 33. Martin GM The genetics and epigenetics of altered proliferative homeostasis in ageing and cancer. Mech Ageing Dev. 2007; 128: 9 -12. [PubMed] .
- 34. Kill IR , Faragher RG , Lawrence K and Shall S. The expression of proliferation-dependent antigens during the lifespan of normal and progeroid human fibroblasts in culture. Journal of cell science. 1994; 107(Pt 2): 571 -579. [PubMed] .
- 35. Kirkwood TB and Finch CE. Ageing: the old worm turns more slowly. Nature. 2002; 419: 794 -795. [PubMed] .
- 36. Martin-Ruiz C , Saretzki G and Petrie J. Stochastic variation in telomere shortening rate causes heterogeneity of human fibroblast replicative life span. J Biol Chem. 2004; 279: 17826 -17833. [PubMed] .
- 37. Gonzalez A , Rota M , Nurzynska D , Misao Y , Tillmanns J , Ojaimi C , Padin-Iruegas ME , Muller P , Esposito G , Bearzi C , Vitale S , Dawn B , Sanganalmath SK , Baker M , Hintze TH , Bolli R , Urbanek K , Hosoda T , Anversa P , Kajstura J and Leri A. Activation of cardiac progenitor cells reverses the failing heart senescent phenotype and prolongs lifespan. Circ Res. 2008; 102: 597 -606. [PubMed] .


