Introduction
Autophagy
(from the Greek, "auto" oneself, "phagy" to eat) involves the sequestration and
degradation by lysosomal enzymes of old, supernumerary, damaged or ectopic
organelles and/or portions of the cytoplasm [1]. At least
three forms of autophagy have been described - macroautophagy, microautophagy,
and chaperone-mediated autophagy - that differ with respect to the mode of
cargo delivery to lysosomes [2,3]. This
article will focus on macroautophagy (herein referred to as autophagy), the
most important catabolic pathway that cells employ for the turnover of
long-lived proteins and organelles and also one of the most prominent
cytoprotective mechanisms in eukaryotic cell biology [4].
During
macroautophagy, the cytoplasmic material targeted to degradation is delivered
to lysosomes upon sequestration within double-membraned vesicles that are
called autophagosomes. The generation of the autophagosome begins with the
nucleation and elongation of the so-called phagophore, an isolation membrane
that likewise originates from the endoplasmic reticulum. The edges of the
phagophore then merge, resulting in the formation of a bona fide
double-membraned autophagosome, which next fuses with a lysosome to generate an
auto(phago)lysosome. Finally, the luminal content as well as the inner membrane
of the auto(phago)lysosome (which together are known as "autophagic body") are
degraded by lysosomal hydrolases. The end products of these catabolic reactions
are exported to the cytoplasm, where they can re-enter anabolic and/or
bioenergetic metabolisms [2,3,5,6].
The biochemical cascade that executes
autophagy has originally been characterized at a molecular level in yeast (Saccharomyces
cerevisiae) [7,8]. Hundreds
of studies in different model organisms including mammals have confirmed that
the essential machinery of autophagic sequestration and execution is phylo-genetically
conserved, and hence involves the orthologs of a series of yeast genes that
have been designated a
ut
ophag
y-related (ATG) genes [7,8]. Autophagy
likewise occurs at low baseline levels in all cells to ensure the homeostatic
turnover of long-lived proteins and organelles [4]. Moreover,
autophagy is upregulated well beyond basal levels: (i) when cells need to
mobilize intracellular nutrients, as occurring during glucose and/or amino acid
deprivation, hypoxia or growth factor withdrawal [2,3]; and (ii)
when cells rid themselves of potentially noxious cytoplasmic materials
including damaged organelles, aggregates of misfolded proteins, or invading
microbes [9,10].
The
complex regulation of autophagy in response to stress
One of the key regulators of
autophagy in human and murine cells is the mammalian target of rapamycin (mTOR,
whose yeast ortholog is TOR) kinase, which suppresses autophagy in conditions
of nutrient and growth factor repletion. Signal transducers including class I
phoshatidylinositol-3-kinases (PI3Ks) and Akt link receptor tyrosine kinases to
mTOR activation, thereby repressing autophagy in response to insulin,
insulin-like growth factor (IGF) and other growth signals [11]. Activation
of the mTOR complex 1 (mTORC1) - and consequent repression of autophagy - can
also be mediated by mitogen-activated protein kinases (MAPKs) including
extracellular signal-regulated kinases (ERKs) [12], by
Ras-dependent activation of the p90 ribosomal S6 kinase [13], as well as
by the Wnt signaling pathway [14]. Other
prominent regulators of autophagy include (but are not limited to):
AMP-activated protein kinase (AMPK), which inhibits mTOR in response to reduced
ATP levels [15]; eukaryotic
translation initiation factor 2α (eIF2α), which responds to nutrient
deprivation as well as to double-stranded RNA (dsRNA) [16], ERN1 (whose
yeast ortholog is known as IRE1), an endoplasmic reticulum (ER)-associated
protein possessing intrinsic kinase and endoribonuclease activities and playing
an important role in the alteration of gene expression upon ER stress [10,17]; and
c-Jun N-terminal kinase (JNK), which is involved in multiple signaling cascades
activated by stressful conditions [18].
Our own work in this field has
added to this list of autophagy regulators: members of the Bcl-2 protein family
that contain a single Bcl-2 homology (BH) domain, the so-called BH3-only
proteins, which displace (and hence derepress) the essential autophagy
modulator Beclin 1 from inhibitory complexes with Bcl-2 or Bcl-XL[19,20]; Sirtuin
1, which responds to high NAD+ levels, de facto acting as a
sensor of nutrient availability [21]; the oncosuppressor
protein p53, which inhibits autophagy when present in the cytoplasm [22]; the
IκB kinase (IKK) complex, which is also essential for the activation of
NF-κB by stress [23,24]; as well
as the inositol 1,4,5-trisphosphate (IP3) receptor (IP3R)
at the level of the ER [20,25].
Finally, autophagy is positively regulated by the transcription factor activity
of E2F1 [26], FoxO3a [27,28],
NF-κB [29] and p53 [30,31], among
others. The apical events of the phylogenetically ancient molecular pathway for
autophagy involve ULK1 and ULK2 (the mammalian orthologs of Atg1) as well as
Beclin 1 (the human ortholog of Atg6). Beclin 1 functions as an allosteric
activator of the class III
PI3K hVps34 (which promotes phagophore nucleation/elongation
via its product phosphatidylinositol-3-phosphate), and is part of a highly
dynamic multiprotein complex that can incorporate various autophagic
stimulators (e.g., UVRAG, Bif-1/endophilin B1, Ambra 1) and/or
inhibitors (e.g., Bcl-2, RUBICON) [32-36].
In synthesis, autophagy is connected to multiple stress pathways. In
some cases, specific proteins and organelles are "tagged" for autophagic
sequestration, implying that intrinsic features of the cargo determine its
elimination by autophagy. This has been documented for proteotoxins [37-39], uncoupled mitochondria (a
process that has been dubbed "mitophagy") [40-42], peroxisomes (for which the term
"pexophagy" has been introduced) [43]; damaged ER (which is eliminated
by "reticulophagy") [44]; and invading pathogens (which
activate "xenophagy") [45]. In many other instances,
autophagy occurs in a rather unselective fashion and represents a general
response of the cell that transits from baseline to induced, yet limited
levels. It is tempting to speculate that this change from basal to upregulated
autophagy may involve the activation of a general "switch" that can respond to multiple distinct stress-responsive and damage-sensing pathways. Complex molecular switches regulate
the clear-cut separation between discrete cellular states including the
transition from an undifferentiated to a more differentiated state, the
advancement of the cell cycle, or the "decision" to activate the apoptotic
cascade [46,47]. Usually, such switches
integrate diverse signals that are transmitted through negative feedback loops
(which maintain homeostasis and keep cells in a defined state) and positive
feedback loops (which mark the rapid evolution between two states) [48]. There is abundant evidence that
the induction of autophagy involves positive feedback loops. For example, we
have documented (i) that autophagy induced by rapamycin (which inhibits mTOR)
is accompanied by the degradation of p53 and the activation of IKK; (ii) that
pharmacological inhibition of p53 with pifithrin-α leads to mTOR
inhibition and IKK activation; and that (iii) transgene-enforced activation of
IKK stimulates p53 degradation at the same time as it inhibits mTOR [22-24,49]. This implies that mTOR
inhibition, IKK activation and the degradation of cytoplasmic p53 are
cross-linked through a network of self-amplifying feedforward loops (Figure 1),
although it remains elusive how this occurs in molecular terms.
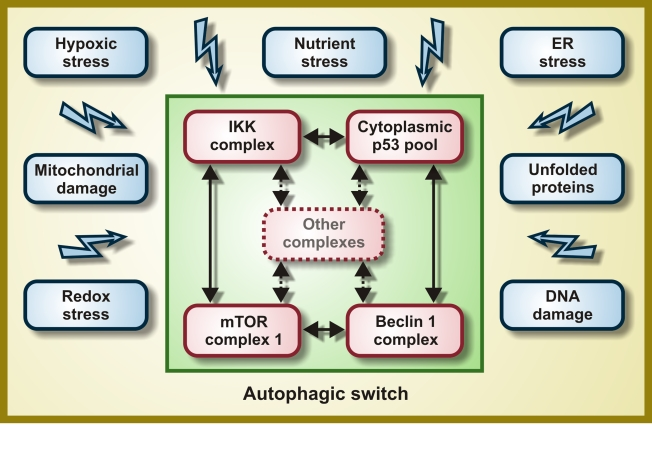
Figure 1. Molecular composition of the hypothetical autophagic switch. Irrespective of
the primary stress signal, a homogeneous response would be obtained by the
independent activation of molecular complexes organized around the IκB
kinase (IKK), cytoplasmic p53, mTOR and Beclin 1. Within these complexes
(and perhaps others), proteins would undergo reversible post-translational
modifications and/or shuttle from one complex to another, thereby
determining the function of the integrator/switch that activates autophagy.
Please note that the autophagic switch is expected to contain several
positive feedback loops that determine its (in)activation.
Autophagy
as a cytoprotective and anti-aging mechanism
Cells that are stressed and
on the verge of death frequently manifest the cytoplasmic accumulation of
autophagosomes and auto(phago)lysosomes, an observation that has been
(mis)interpreted as if autophagy would contribute to the cellular suicide [50]. Thus,
hundreds of papers have described "autophagic" (also dubbed "type 2") cell
death, a cell demise subroutine preceded by massive autophagic vacuolization
that is morphologically distinct from apoptosis ("type 1") and necrosis ("type
3") [51-54]. Although "autophagic cell death" undoubtedly exists as a
morphological entity [53], this only
exceptionally (at least in mammalian models) reflects the execution of cells by
autophagy [50]. Rather,
autophagy most frequently constitutes a (sometimes futile) mechanism of
cellular adaptation to a diverse range of adverse conditions including
hypoglycemia, hypoxia, lack of essential amino acids, absence of obligate
growth factors or sublethal damage to cytoplasmic organelles including
mitochondria and the ER [4,55,56].
Accordingly, the genetic inhibition of autophagy by knockout or knockdown of ATGgenes often precipitates the apoptotic or necrotic death of cells that
otherwise would survive nutrient depletion, growth factor withdrawal, hypoxia,
ionizing radiation or anticancer chemotherapy [11,50,57-60].
Deficient autophagy is directly involved in a number of pathologies including
neurodegenerative diseases, heart failure, hereditary myopathies,
steatosis/steatohepatitis and other chronic inflammatory states [6,61-64].
Genetic and pharmacological manipulations designed to induce autophagy have
been shown to protect cells against otherwise lethal damage in vitro[5,6]. Autophagyfavors the maintenance of high intracellular ATP levels [22,65],
increases the capacity of cells to resist metabolic stress (hypoxia combined
with nutrient deprivation) [22,66],
prevents genomic instability [60,67] and
limits the accumulation of potentially toxic proteins including proteotoxins
that are responsible for neurodegeneration [10,38]. From a
physiological point of view, aging can be viewed as a continuous decline in
cellular and organismal functions that (at least partially) reflects the
accumulation of misfolded proteins, oxidized lipids, as well as mutated
mitochondrial and nuclear DNA.
The sole regimen leading to lifespan
extension in every organism tested to date is dietary restriction, a reduction
of the organism's caloric intake not associated to malnutrition [68]. Dietary
restriction is a potent inducer of autophagy in virtually all species including
mammals [69-71]. In the
nematode Caenorhabditis elegans, autophagy is required for lifespan
prolongation mediated by caloric restriction [72-74] or p53
depletion [22,49,75-77].
Thus, worms undergoing dietary restricttion do not live longer than control
animals if concomitantly subjected to RNA interference (RNAi) against atg
genes [72-74].
Rapamycin,
which activates autophagy via inhibition of (m)TOR, has also been ascribed with
prominent anti-aging properties, in various model organisms. However, rapamycin
cannot extend the chronological lifespan (i.e., the time post-mitotic
cells survive during the stationary phase [78]) of yeast
mutants that lack functional Atg1, Atg7 or Atg11 [79]. In C.
elegans, the beneficial effects of rapamycin on longevity are lost when the
essential autophagy modulator BEC-1 (the worm ortholog of mammalian Beclin 1
and yeast Atg6) is knocked down [74]. Thus,
autophagy is required for rapamycin-mediated lifespan extension and delay of
chronological aging in yeast and nematodes. Although it has not been formally
demonstrated that rapamycin prolongs the lifespan of mice by inducing
autophagy, even the treatment of pre-aged, genetically heterogeneous (out-bred)
mice has been shown to increase longevity [80]. In mice,
rapamycin avoids the age-related decline in hematopoietic stem cells function [81], an
anti-senescence effect that has also been described in vitro[82,83].
Altogether,
these results suggest that whole-body induction of autophagy by pharmacological
agents may prolong the healthy lifespan, at least in laboratory conditions,
supporting the idea that autophagy does not only confer cytoprotection but that
it also has anti-aging effects at the organismal level.
Autophagy
mediates lifespan extension by resveratrol
Driven by
the aforementioned considerations, we launched the working hypothesis that
autophagy constitutes (one of) the major mechanism(s) through which
longevity-extending drugs operate. We thus studied whether resveratrol, a
well-studied anti-aging agent [84], would
extend the lifespan of model organisms via the induction of autophagy. Although
it also affects mitochondrial functions [85],
resveratrol prominently acts as an allosteric activator of Sirtuin 1, a
phylogenetically conserved deacetylase that senses the NAD+/NADH
ratio [84].
Resveratrol increases the longevity of yeast, nematodes, and flies (Drosophila
melanogaster) and also exerts anti-aging effects on mice kept on a high-fat
diet [84,86]. Circumstantial evidence indicates that resveratrol
can induce autophagy in yeast (although this was attributed to the oxidation of
mitochondrial lipids [87]) and in
human cancer cells (in which resveratrol-induced autophagy often precedes cell
death [88]). Sirtuin 1
is the first protein that has been demonstrated to prolong lifespan in yeast
(and then in animals including C. elegans and flies) [89], and has
also been shown to trigger autophagy in human and murine cultured cells [90].
We
confirmed that Sirtuin 1 overexpression increased the autophagic flux in human
cancer cells in vitro, and that this effect was abolished by the
addition of EX527, a pharmacological inhibitor of its catalytic activity [91,92].
Similarly, a transgene coding for SIR-2.1 (the C. elegans ortholog
of human Sirtuin 1) caused autophagy in nematodes, suggesting that the link
between Sirtuin 1 activation and autophagy is evolutionarily conserved [91,92].
Importantly, Sirtuin 1 was required for the induction of autophagy by nutrient
deprivation (that was achieved by culturing cells in the absence of serum,
amino acids and glucose) but not by other stimuli. Thus, in human cells, the
depletion (by RNAi) or inhibition (with EX527) of Sirtuin 1 fully prevented the
proautophagic effects of nutrient starvation, yet failed to affect the
stimulation of autophagy by mTOR inhibition (with rapamycin), p53 inhibition
(with pifithrin-α) or ER stress (triggered by the addition of
tunicamycin). Similarly, loss-of-function
mutations of sir-2.1 abolished
autophagy induced by caloric restriction but not that promoted by rapamycin or
tunicamycin in C. elegans [91,92].
Transgenic overexpression of sir-2.1 increased the median and maximum
lifespan of nematodes as compared to non-transgenic control strains with the
same genetic background. This gain in longevity was lost when the essential
autophagic modulator BEC-1 was depleted by RNAi [91,92].
RNAi-mediated knockdown of the C. elegans p53 ortholog CEP-1, a
manipulation that extends longevity through the stimulation of autophagy [77], failed
to further ameliorate the beneficial effects of sir-2.1 overexpression
on longevity [91,92]. This
epistatic analysis suggests that SIR-2.1 accumulation and CEP-1 depletion
extend lifespan through a common final pathway that relies on the induction of
autophagy.
Another genetic intervention designed to
indirectly activate Sirtuin 1 (or its worm ortholog SIR-2.1) consists in the
transgenic overexpression of the gene coding for the pyrazinamidase/nicotinamidase
PNC-1, which depletes nicotinamide, a negative regulator of Sirtuin 1/SIR-2.1.
Transgenic overexpression of pnc-1 did indeed induce autophagy in worms,
and this response was abolished by RNAi-mediated depletion of SIR-2.1. Accordingly,
the longevity-extending effects of PNC-1 were lost upon the knockdown of
SIR-2.1, as well as upon that of either of the two essential autophagy
modulators BEC-1 or ATG-5 [77]. Thus, both
the overexpression and the metabolic activation of Sirtuin 1/SIR-2.1 increase
lifespan through the induction of autophagy.
Next, we
investigated whether resveratrol would induce autophagy in C. elegans
via the activation of SIR-2.1. Addition of resveratrol to the worm culture
medium did indeed stimulate autophagy, and this effect was lost upon
RNAi-mediated depletion of SIR-2.1. Similarly, resveratrol reduced the
aging-associated mortality of C. elegans, unless the products of sir-2.1or bec-1 were knocked down [77]. We
concluded from these experiments that resveratrol prolongs lifespan in human
and nematode cells by inducing autophagy, which results from
resveratrol-mediated activation of Sirtuin 1/SIR-2.1 (rather than from an
off-target effect).
Autophagy
mediates lifespan extension by spermidine
Driven by
the fact that the intracellular level of polyamines declines in (otherwise
healthy) aging humans [93], we
investigated whether the polyamine spermidine display anti-aging properties. To
address this question, we first took advantage of a yeast strain that is
deficient in the ornithine decarboxylase SPE1, which catalyzes the first step
of polyamine biosynthesis. In chronological aging experiments, Δspe1 yeast
cells exhibited an increased mortality (and hence a shortened lifespan), which
could be restored to normal levels by supplementation with low doses (0.1 mM)
of spermidine or its precursor putrescine [94]. Surprisingly,
we found that higher concentrations of spermidine were able to increase the
lifespan of wild type yeast cells with different genetic backgrounds. Thus,
both chronological aging (which constitutes a model of post-mitotic aging) and
replicative aging (which constitutes a model of stem cell aging) of yeast cells
were significantly inhibited by spermidine supplementation. Lifespan
prolongation in spermidine-treated yeast cells could be correlated with the
reduced acetylation of several lysine residues located at the N-terminal tail
of histone H3 (i.e., Lys9, Lys14 and Lys18) [94]. Deletion ofsir2 (the yeast ortholog of Sirtuin 1) or any other sirtuin did
not affect the ability of spermidine to extend chronological lifespan. Instead,
epistatic analyses revealed that the anti-aging effect of spermidine was
phenocopied by the knockout of histone acetylases, which hence were shown to
regulate the same longevity-increasing pathway than spermidine does [94]. Moreover,
spermidine efficiently inhibited general histone acetylase activity in extracts
from purified yeast and mammalian nuclei in an in vitro assay [94]. These
results suggest that spermidine acts differently from resveratrol. Thus, while
the former inhibits histone acetylase(s), the latter stimulates the deacetylase
activity of Sirtuin 1. However, formal evidence that the (de)acetylation of
histones rather than that of other proteins (either in the nucleus or in the
cytoplasm) account for the anti-aging properties of spermidine is still
missing.
Microarray
profiling of spermidine-treated yeast cells revealed the transcriptional
activation of several autophagy genes including atg7, atg11 and atg15,
and we indeed found that spermidine induces autophagy in yeast cells.
Similarly, spermidine was highly efficient in upregulating the autophagic
pathways when it was added to the culture medium or solid food of C. elegansor D. melanogaster, respectively. The same concentrations of
spermidine that exerted pro-autophagic effects also had a marked
lifespan-extending effect on yeast, nematodes and flies. The genetic inhibition
of essential ATG genes (i.e., knockout of atg7 in yeast
and flies, RNAi-mediated silencing of bec-1 in nematodes) abrogated
longevity extension induced by spermidine, indicating this polyamine can
prolong lifespan by the induction of autophagy [94].
Open
questions
The
aforementioned results indicate that resveratrol and spermidine can prolong the
lifespan of model organisms through the induction of autophagy (Figure 2). In
addition, our work raises at least three issues that must be addressed by
future investigation.
First, do resveratrol and spermidine
extend longevity by acting on the same molecular pathway? While resveratrol can
prolong lifespan through the activation of the deacetylase activity of Sirtuin
1 (or its non-mammalian equivalents SIR2 in yeast and SIR-2.1 in C. elegans),
spermidine inhibits the general histone acetylase activity of yeast and mouse
liver extracts. Clearly, histone (de)acetylation has been recognized as an
important epigenetic regulator of longevity [95,96].
However, a fraction of Sirtuin 1 is present in the cytoplasm, from where it can
directly deacetylate essential autophagic proteins (including ATG5, ATG7 and
ATG8/LC3) [90], suggesting
that (at least part of) the pro-autophagic effects of resveratrol derive from
extranuclear, transcription-independent events. It will be important to know
whether polyamines (like spermidine) and Sirtuin 1 activators (including
resveratrol) can exert additive or synergistic effects on autophagy and
longevity or whether these agents exactly activate the same molecular pathway.
Moreover, the precise mechanisms by which spermidine and resveratrol control
the autophagic switch awaits further exploration.
Careful mechanistic and epistatic analyses are required to address this
problem.
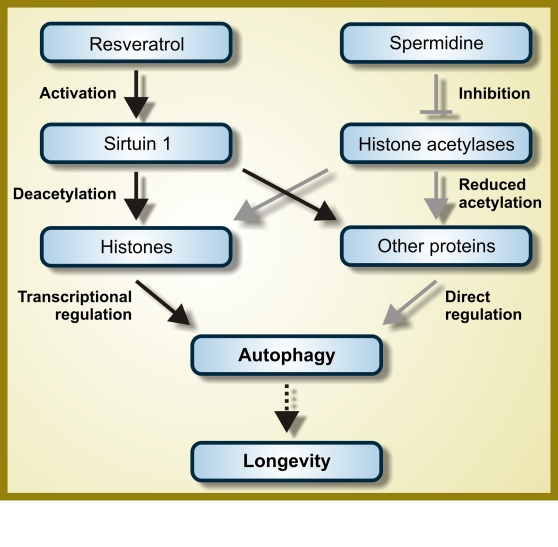
Figure 2. Hypothetical mode of action of resveratrol and spermidine as autophagy inducers. While
resveratrol functions as an activator of the deacetylase Sirtuin 1,
spermidine inhibits one or several histone acetylases. Therefore, both
resveratrol and spermidine are expected to favor protein hypoacetylation.
However, the autophagy-relevant substrates whose deacetylation is induced
by resveratrol and spermidine are not fully characterized and it is even
not known if they are completely distinct, partially overlapping or
identical. For further details, please consult the main text.
Second, do
all longevity-prolonging manipulations induce autophagy? And is autophagy
required for all such intervention to extend lifespan? Current results clearly
indicate that autophagy is indispensable for the anti-aging action of
rapamycin, resveratrol and spermidine. Moreover, it has been suggested that
autophagy is required for longevity extension by dietary restriction in C.
elegans, although this has not been tested for all caloric restriction
protocols [73]. It remains
an ongoing conundrum whether an increased level of autophagy is required in C.
elegans for longevity extension conferred by the deficiency of GTPase
RHEB-1 [97], the
transcription faction hypoxia-inducible factor 1 (HIF-1) [98] and its
negative regulator VHL-1 [99], the
ubiquitin ligase WWP-1 [100], as well
as the chaperones CCT4 and CCT6 [101]. A
positive response to this question could establish a new paradigm in longevity
research.
Third, and
most important, can the data that we discuss here, which have mostly been
obtained in simple model organisms and in laboratory conditions (where, for
instance, the immunosuppressive side effects of resveratrol are certainly less
incisive), be extrapolated to humans and to real life? Although rapamycin and
polyamines can increase lifespan in mice [80,102],
resveratrol only extends the longevity of mice that are kept on a high-caloric
diet [86]. Clearly, rapamycin and resveratrol can induce autophagy in vivo,
in mice [23,24,103].
However, it is thus far unknown whether there is indeed a cause-effect relationship
between increased autophagy and healthy aging in mammals and in particular in
humans. Such a causal relationship would revolutionize the entire field of
aging research.
The authors of this
manuscript have no conflict of interests to declare.