Introduction
L-arginine is a semi-essential amino acid which is not only involved in protein synthesis, is also a single substrate for endothelial nitric oxide synthase (eNOS) to produce the important vasoprotective molecule nitric oxide (NO) [1, 2]. Depletion of L-arginine causes eNOS dysfunction in cultured endothelial cells [3]. Hence, L-arginine supplementation has been widely used under many physiological and pathological conditions aiming to improve health status or to treat diseases including cardiovascular diseases [2, 4-7]. However, controversial results have been reported. Although many studies demonstrate that acute or short-term supplementation of L-arginine improves endothelium-dependent vaso-dilation or reduces blood pressure in diseased animal models or patients with cardiovascular diseases [8-15], numerous studies with L-arginine supplementation, however, show no sustained effects on endothelial function [16-20]. Most importantly, studies with long-term (6 months) L-arginine supplementation even show harmful effects in atherosclerotic animal models [21] as well as in patients with cardiovascular diseases for unknown reasons [22, 23]. It seems that the impact of L-arginine on cardiovascular function depends on duration of the amino acid supplementation.
The harmful effects of chronic L-arginine supplementation have been recently recapitalized on vascular endothelial cells by Scalera and colleagues [24]. They showed that chronic L-arginine supplementation is capable of accelerating endothelial cell senescence associated with enhanced expression of arginase-II (Arg-II) and decreased endothelial NO generation. This detrimental effect of L-arginine is prevented by Arg-II gene silencing, suggesting that chronic L-arginine supplementation causes endothelial dysfunction through up-regulation of Arg-II, an enzyme that metabolizes L-arginine and is predominantly involved in accelerating vascular endothelial cell senescence [25]. The mechanism of Arg-II gene up-regulation by L-arginine, however, remains unknown. Our recent study demonstrates that a persistent activation of the mammaliantargetofrapamycincomplex-1 (mTORC1) and its down-stream target S6K1 promotes endothelial senescence and dysfunction through up-regulation of Arg-II [25].
Given that mTORC1-S6K1 pathway can be activated by nutritional components including amino acids, which occurs through the Leucyl-tRNA synthetase-Rag [26, 27], we hypothesize that chronic L-arginine supplementation may cause endothelial dysfunction and senescence through mTORC1-S6K1 pathway and Arg-II.
Results
Chronic, but not acute L-arginine treatment promotes endothelial senescence phenotype
In young endothelial cells, when compared to cells treated with the physiological concentration of L-arginine (0.1 mmol/L), chronic treatment of the cells with a higher concentration of L-arginine (0.5 mmol/L) for 7 days enhanced the number of SA-β-gal positive cells (Fig. 1A), protein levels of intercellular adhesion molecule-1 (ICAM-1) and vascular cell adhesion molecule-1 (VCAM-1) (Fig. 1B). Moreover, treatment of the cells with L-arginine at the concentration 0.5 mmol/L for 7 days caused endothelial dysfunction as measured by enhanced superoxide anion and decreased NO production (Fig. 1C). Inhibition of eNOS by L-NAME (1 mmol/L for 2 hours) abolished NO production as expected, but also reduced superoxide anion levels, demonstrating eNOS-uncoupling under the chronic L-arginine treatment (Fig. 1C). In addition, chronic treatment of the endothelial cells with L-arginine (0.5 mmol/L, 7 days) significantly decreased eNOS protein levels (Fig. 1D). In contrast to the chronic L-arginine treatment, acute treatment of the cells with L-arginine at the same concentration (0.5 mmol/L) for 30 minutes enhanced NO production but also increased superoxide anion generation while having no effects on protein levels of eNOS, VCAM-1 and ICAM-1 (Fig. 2).
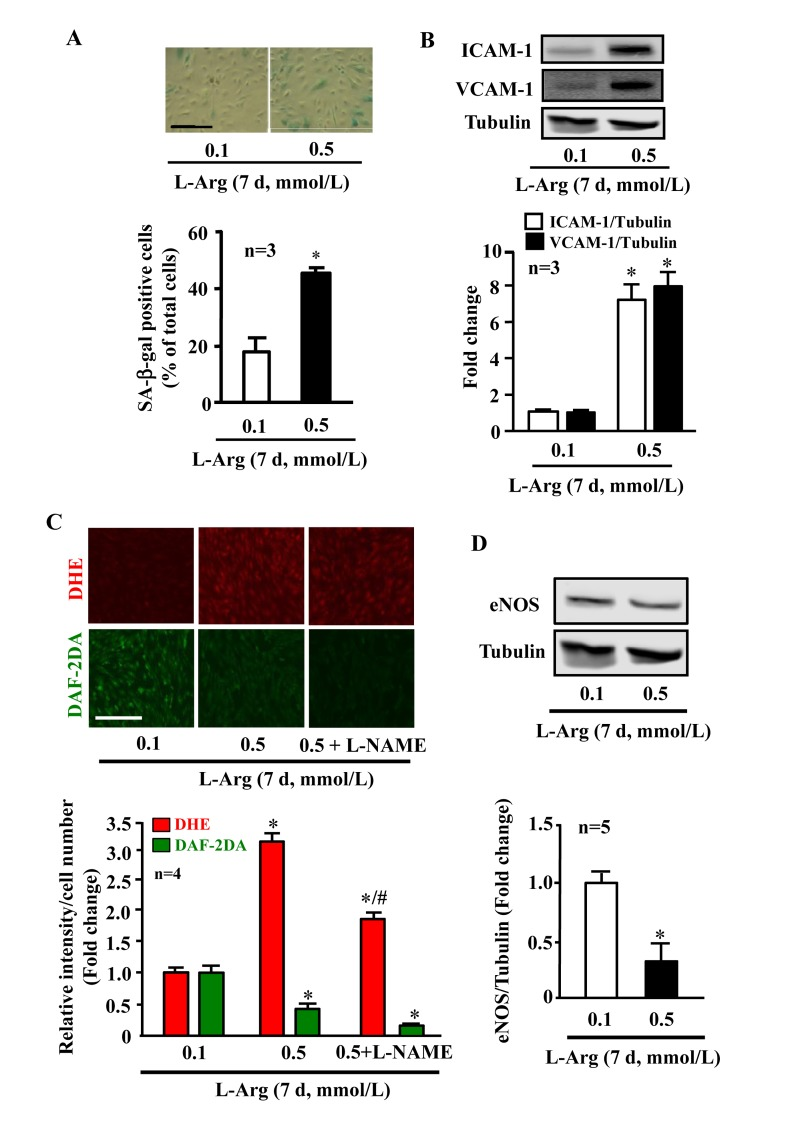
Figure 1. Chronic L-arginine supplementation promotes endothelial senescence, inflammation and eNOS-uncoupling. Confluent young HUVECs were L-arginine-starved overnight followed by treatment with 0.1 mmol/L or 0.5 mmol/L L-arginine for 7 days (7 d) with change of medium every 48 hours. Cells were serum starved overnight prior to experiment. (A)SA-β gal assay. (B)Western blot analysis of ICAM-1/VCAM-1. Tubulin served as loading control. (C) Detection of O2.- and NO by DHE and DAF-2DA staining, respectively. The eNOS inhibitor L-NAME (1 mmol/L) was added to the cells 2 hours prior to the DAF-2DA staining. (D) Western blot analysis of eNOS protein levels with tubulin as loading control. The Western blot analysis of expression of ICAM-1, VCAM-1 and eNOS shown in (B) and (D), respectively, was performed with the same membrane. The quantification of the signals is presented as graphics below the corresponding images or blots. n represents the number of the repeated independent experiments. *p < 0.05 vs 0.1 mmol/L; #p < 0.05 vs 0.5 mmol/L. Scale bar = 200 μm
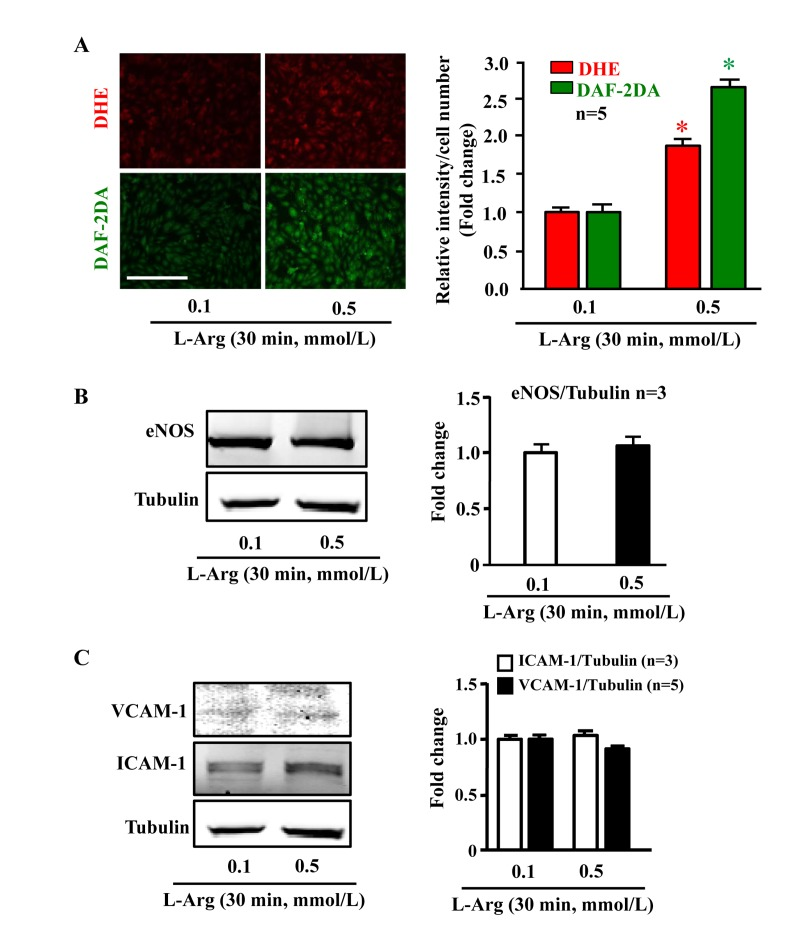
Figure 2. Acute L-arginine supplementation enhances production of both NO and superoxide anion Cells were serum- and L-arginine-starved overnight followed by treatment with 0.1 mmol/L or 0.5 mmol/L L-arginine for 30 minutes. (A) Detection of O2.− and NO by DHE and DAF-2DA staining, respectively. (B) Western blot analysis of eNOS protein level with tubulin as loading control. (C) Western blot analysis of ICAM-1/VCAM-1 levels. The Western blot analysis shown in (B) and (C) was performed with the same membrane. Tubulin served as loading control. The quantification of the signals is presented as graphics at the corresponding right panels. *p < 0.05 vs 0.1 mmol/L. Scale bar = 200 μm
Chronic L-arginine treatment increases Arg-II and mTORC1/S6K1 signaling in endothelial cells, while acute treatment activates mTORC1/S6K1 without increase in Arg-II
In young endothelial cells, chronic L-arginine treatment (0.5 mmol/L, 7 days) enhanced Arg-II expression and activity (Fig. 3A) as well as activation of mTORC1/S6K1 signaling as measured by enhanced ratio of S6K1-T389/S6K1 and that of S6-S235/236:S6, when compared to cells treated with 0.1 mmol/L of L-arginine (Fig. 3B). In contrast to chronic treatment, acute treatment of the cells with L-arginine (0.5 mmol//L) for 30 minutes enhanced mTORC1-S6K1 signaling without having an effect on Arg-II expression or activity (Fig. 4).
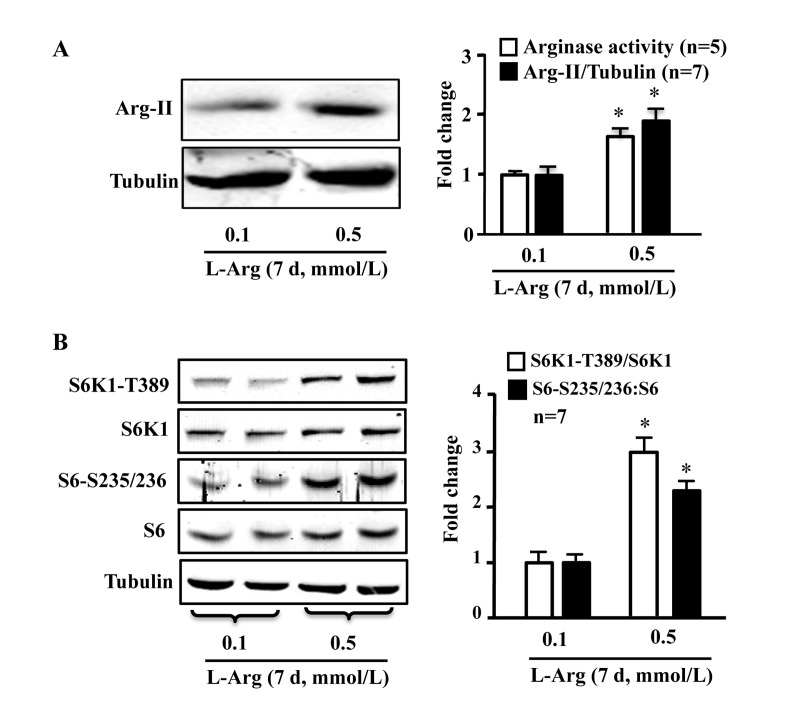
Figure 3. Chronic L-arginine supplementation induces Arg-II expression/activity and mTORC1-S6K1 signaling Cells were treated as described in Fig.1. (A) Western blot analysis of Arg-II expression and arginase activity assay. (B) Western blots showing activation of mTORC1 and S6K1 as monitored by phosphorylation of S6K1-T389 and S6-S235/236, respectively. The graphics at the corresponding right panels present the quantification of the signals. *p < 0.05 vs 0.1 mmol/L
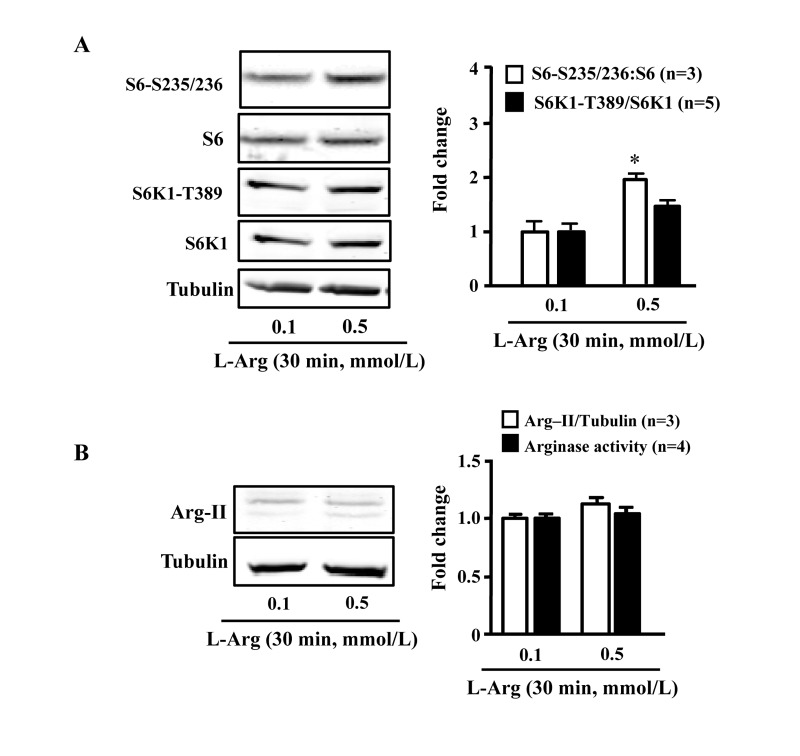
Figure 4. Acute L-arginine supplementation activates mTORC1-S6K1 signaling, but not Arg-II expression/activity Endothelial cells were treated as described in Fig. 2. (A) Western Blots showing activation of mTORC1 and S6K1 as monitored by phosphorylation of S6K1-T389 and S6-S235/236, respectively. (B) Western Blot analysis of Arg-II expression and arginase activity assay. The graphics at the corresponding right panels present the quantification of the signals. *p < 0.05 vs 0.1 mmol/L
Positive crosstalk between Arg-II and mTORC1/S6K1 mediates the chronic effect of L-arginine on cell senescence
To evaluate the role of Arg-II and S6K1 in the chronic effects of L-arginine on cell senescence, Arg-II and S6K1 were knocked down by transducing the cells with rAd/U6-shRNA against Arg-II or S6K1, after overnight L-arginine-starvation and prior to addition of L-arginine 0.1 or 0.5 mmol/L, 7 days). The up-regulation of Arg-II and sustained activation of mTORC1-S6K1 pathway by chronic treatment of L-arginine (0.5 mmol/L, 7 days) depended on each other, as silencing either Arg-II or S6K1 prevented up-regulation/activation of the other (Fig. 5A). Since S6K1 is activated by acute treatment of L-arginine (0.5 mmol/L, 30 minutes) without Arg-II up-regulation (Fig. 4), we wish to determine whether the basal Arg-II level is required for L-arginine-induced acute S6K1 activation. As revealed by Fig. 5B, silencing Arg-II did not affect the S6K1 activation by acute L-arginine treatment (0.5 mmol/L, 30 minutes).
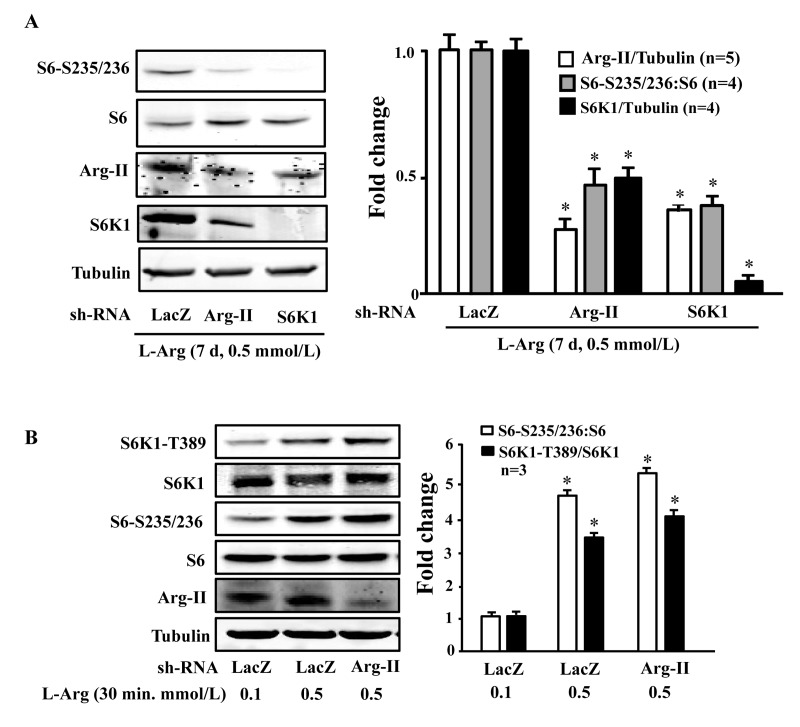
Figure 5. S6K1 and Arg-II form a positive regulatory circuit under the condition of chronic L-arginine supplementation (A) Endothelial cells were treated as described in Fig. 1, except that the cells were transduced with the rAd/U6-LacZshRNA as control, rAd/U6-Arg-IIshRNA or -S6K1shRNA after L-arginine-starvation and prior to the treatment with only 0.5 mmol/L of L-arginine for 7 days. Shown are Western blot analyses of S6-S235/236, total S6, Arg-II and S6K1, and the quantification of the signals. *p < 0.05 vs rAd/U6-LacZshRNA+0.5 mmol/L L-arginine control. (B) Endothelial cells were treated as described in Fig. 1, except that the cells were transduced with the rAd/U6-LacZshRNA as control or rAd/U6-Arg-IIshRNA48 hours prior to serum- and L-arginine-starvation. Shown are Western blot analyses of S6K1-T389, S6K1, S6-S235/236, total S6, Arg-II, tubulin, and the quantification of the signals. *p < 0.05 vs rAd/U6-LacZshRNA+0.1 mmol/L L-arginine
Along with the observation presented in Fig. 5A, silencing either Arg-II or S6K1 significantly reduced the SA-β-gal positive cell number (Fig. 6A), enhanced eNOS protein level while decreasing ICAM-1 and VCAM-1 levels (Fig. 6B), inhibited superoxide anion and enhanced NO production (Fig. 6C) under condition of chronic L-arginine treatment (0.5 mmol/L, 7 days). Similar results were obtained when cells were treated with the mTORC1 inhibitor rapamycin. As shown in Fig. 7, the cell senescence induced by chronic L-arginine treatment (0.5 mmol/L, 7 days) as analyzed by the positive SA-β-gal cells was significantly reduced by rapamycin (20 ng/ml, 7 days, Fig. 7A). Moreover, rapamycin (20 ng/ml, 7 days) reduced superoxide anion production (DHE staining) and increased NO generation (DAF-2DA staining, Fig. 7B), inhibited expression levels of VCAM-1, ICAM-1, Arg-II, while enhanced eNOS levels in the endothelial cells under the condition of chronic exposure to L-arginine (0.5 mmol/L, 7 days, Fig. 7C). As expected, activation of S6K1 signaling (measured by S6K1-T389 phosphorylation levels and S6-S235/236 levels) was inhibited by rapamycin under this condition (Fig. 7C).
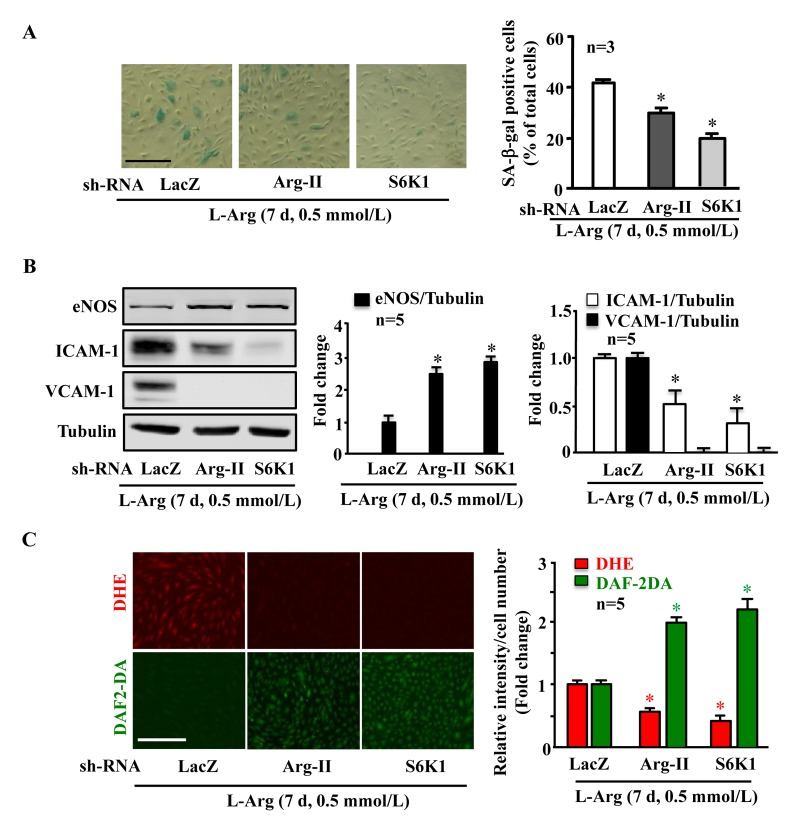
Figure 6. Arg-II and S6K1 mediate the chronic effect of L-arginine on cell senescence Endothelial cells were treated as in Fig. 5. (A) SA-β gal assay. (B) Western blot analysis of eNOS, ICAM-1, VCAM-1. Tubulin served as loading control. (C) DHE staining for detection of O2.−, DAF-2DA staining for detection of NO. The graphics at the corresponding right panels present the quantification of the signals. *p < 0.05 vs rAd/U6-LacZshRNA control+0.5 mmol/L L-arginine. Scale bar = 200 μm
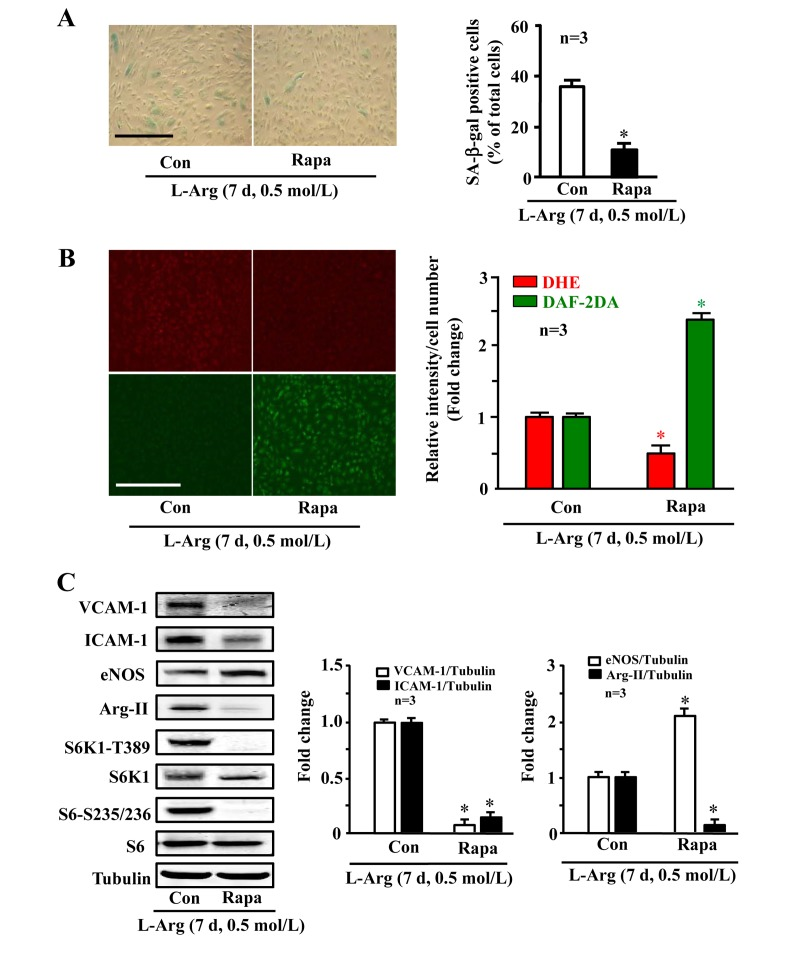
Figure 7. Arg-II and S6K1 mediate the chronic effect of L-arginine on cell senescence Endothelial cells were treated as in Fig. 1 except that rapamycin (20 ng/ml) was added at same time as L-arginine (0.5 mmol/L). (A) SA-β gal assay. (B) DHE staining for detection of O2.−, DAF-2DA staining for detection of NO. (C) Western blot analysis of VCAM-1, ICAM-1, eNOS, Arg-II, S6K1-T389, S6K1, S6-S235/236 and S6. Tubulin served as loading control. The bar graphs are the quantifications of signals from the corresponding experiments shown in the left panels. *p < 0.05 vs L-arginine alone as the control (Con). Scale bar = 200 μm
Discussion
Since discovery that L-arginine is the only substrate for endothelial cells to produce the vasoprotective molecule NO via eNOS [3], L-arginine supplementation is widely investigated for decades to boost NO production in different models including human subjects in vivo. However, results from paramount numbers of published studies in the literature are very confusing. Beneficial, no sustained effects, and harmful effects including enhanced mortality rate in clinical studies have been reported with L-arginine supplementation therapy [28]. Importantly, a randomized, double-blinded, placebo-controlled study in patients with acute myocardial infarction, the VINTAGEMI study, demonstrates that 6-month oral L-arginine supplementation (3 g 3 times a day on top of standard post infarction therapy) does not have any benefits on vascular stiffness and left ventricular ejection fraction, but increases mortality [22]. A second clinical trial in patients with peripheral artery disease, the so-called NO-PAIN study with the same protocol, shows decreased NO production and shortened walking distance in patients receiving L-arginine supplementation as compared to the placebo group [23]. The impact of L-arginine supplementation, particularly chronic supplementation for treatment of cardiovascular diseases seems detrimental. However, the mechanisms underlying these findings remain obscure. A recent study in cultured cells suggests that Arg-II could be involved in the detrimental effects of chronic L-arginine supplementation [24]. This group shows that chronic L-arginine supplementation causes endothelial senescence which can be prevented by silencing Arg-II gene [24].
Arg-II has been shown to play an important role in various diseases including atherosclerosis, type-II diabetes and vascular aging [25, 28-31]. The capability of L-arginine to induce Arg-II expression was first shown by Crody et. al. in 1989 in the renal epithelial HEK cell line [32]. Our present study further confirms this result and demonstrates that supplementation of L-arginine to endothelial cells for 7 days causes Arg-II up-regulation along with endothelial senescence phenotype as demonstrated by increased SA-β-gal staining, inflammatory adhesion molecule expression, eNOS-uncoupling, i.e., decreased NO production and enhanced superoxide anion generation which is sensitive at least in part to the eNOS inhibitor L-NAME. Our results are fully in line with the findings by Scalera and colleagues [24]. Moreover, our study has explored the underlying mechanisms of the detrimental effects of chronic L-arginine-induced endothelial Arg-II up-regulation in the endothelial cells and demonstrates that this is mediated by mTORC1-S6K1. Among other regulators, amino acids are potent activators of mTORC1-S6K1 pathway [33]. Indeed, we show that in endothelial cells, L-arginine at a modest concentration 0.5 mmol/L significantly activates mTORC1/S6K1 over 7 days as compared to a lower concentration 0.1 mmol/L, which is associated with increased Arg-II up-regulation and activity. Silencing S6K1 not only inhibits Arg-II up-regulation, but also prevents endothelial cell senescence, enhancement of adhesion molecule expression, as well as eNOS-uncoupling under chronic L-arginine supplementation condition, demonstrating the important role of mTORC1-S6K1 in up-regulation of Arg-II and endothelial senescence. This conclusion is further supported by the experiment showing that pharmacological inhibition of mTORC1-S6K1 signaling with rapamycin reveals the same effects as S6K1 silencing. Our results are in line with the concept that mTORC1-S6K1 pathway is involved in loss of regenerative/replicative potential and drives geroconversion, a conversion from reversible cell cycle arrest to irreversible cell senescence [34-39].
Our previous study showed a positive crosstalk between S6K1 and Arg-II in the replicative endothelial cell senescence [25]. Here, we show that this crosstalk also exists in L-arginine-induced cell senescence, since silencing either S6K1 or Arg-II inhibits the activity of each other and is able to prevent endothelial dysfunction, inflammation and senescence. Taking into account that acute L-arginine-treatment (30 minutes) causes S6K1 activation without having any changes in Arg-II protein level and that silencing Arg-II does not affect S6K1 activation by the acute L-arginine treatment (30 minutes), it is likely that initial acute S6K1 activation by L-arginine, which is mediated by Leucyl-tRNA - Rag GTPase [26, 27], does not require Arg-II, but leads to up-regulation of Arg-II. The enhanced Arg-II in turn positively modulates mTORC1-S6K1 activity resulting in sustained activation of S6K1, which may involve either inhibition of negative regulatory mechanisms of mTORC1-S6K1 pathways or activation of positive regulators of the signaling pathway. The detailed mechanisms of the positive regulatory circuit between S6K1 and Arg-II remain to be investigated.
There are studies demonstrating that NO production from eNOS is primarily determined by extracellular L-arginine bioavailability which directly channels to the cell membrane-associated eNOS [40-43]. Our present study with acute L-arginine supplementation further supports this concept by showing that short term L-arginine supplementation for 30 minutes enhances NO production. As expected, acute L-arginine supplementation does not alter eNOS expression, levels of adhesion molecules and Arg-II. It is, however, accompanied with superoxide anion generation, which obviously does not decrease the bioavailability of NO. The L-arginine-induced superoxide generation may be linked to acute activation of mTORC1/S6K1 pathway which is capable to produce the free radical from mitochondrion [44, 45]. If this signaling is persistently activated such as under the condition of chronic L-arginine supplementation, an oxidative stress, i.e., eNOS-uncoupling then occurs as shown in our present study. It is obvious that endothelial dysfunction under chronic L-arginine supplementation could not be attributed to L-arginine deficiency, because there is a constant concentration of L-arginine (0.5 mmol/L) available in the culture medium. We cannot exclude the possibility that intracellular specific L-arginine pool is depleted because of up-regulation of Arg-II. However, this concept is not supported by studies showing that continuous cellular exposure to L-arginine for 7 days does not affect L-arginine uptake and generation of the endogenous eNOS inhibitor ADMA [46]. Our previous study showed that Arg-II causes eNOS-uncoupling requiring L-arginine metabolizing enzymatic activity [25]. Taking into account of a recent study demonstrating that the metabolic products of arginase such as urea and L-ornithine, or downstream enzymes do not contribute to eNOS inhibition [47], it still remains elusive how Arg-II causes eNOS-uncoupling in an enzymatic activity-dependent manner.
In conclusion, our present study provides evidence demonstrating that L-arginine exerts either beneficial or detrimental effects on endothelial cells which is dependent on the duration of the supplementation. We show that short term L-arginine supplementation enhances endothelial NO production, while long-term supplementation causes endothelial senescence through stimulation of mTORC1-S6K1 signaling and up-regulation of Arg-II. The findings may not only explain the contradictory or inconsistent results on the vascular effects of L-arginine supplementation reported in the literature, but also provide the mechanistic insight into the detrimental clinical outcomes in patients with cardiovascular diseases. The L-arginine supplementation in combination of mTORC1-S6K1 inhibition may preserve the beneficial effect on NO production while diminishing the detrimental effects of chronic L-arginine supplementation on endothelium, which may represent a novel therapeutic option for L-arginine supplementation therapy.
Materials and Methods
All chemicals, if not specifically indicated, including those used for Western blotting, mouse anti-tubulin monoclonal antibodies were obtained from Sigma (Buchs, Switzerland). Rabbit antibodies against phospho-S6-S235/236 (#2211s), S6 (#2217s) and anti-VCAM-1 (#12367S) were from Cell Signaling (Allschwil, Switzerland); mouse anti-eNOS (#610297) and anti-S6K1 (#611260) were from BD Transduction laboratories (Allschwil, Switzerland); rabbit anti-Arg-II (sc-20151) and mouse anti-ICAM-1 (sc-8439) were purchased from Santa-Cruz Biotech (Nunningen, Switzerland); Alexa Fluor680-conjugated anti-mouse IgG (A21057) and dihydroethidium (DHE) were from Molecular Probes/Invitrogen (Lucerne, Switzerland); X-gal was from Promega (Dübendorf, Switzerland); the membrane-permeable 4,5-diaminofluoresceine acetate (DAF-2DA) was from VWR international SA (Dietikon, Switzerland); QuickTiter™ Adenovirus Titer Immunoassay Kit (VPK-109, Cell Biolabs, Inc) were purchased from LuBioScience GmbH (Luzern, Switzerland); endothelial cell growth supplement (ECGS) pack was from PromoCell GmbH (Allschwil, Switzerland); all cell culture media and materials were purchased from Gibco BRL (Lucerne, Switzerland).
Recombinant adenovirus (rAd)
Generation of rAd expressing shRNA targeting human Arg-II and S6K1 driven by the U6 promoter (rAd/U6-hArg-IIshRNAand -hS6K1shRNA, respectively) was described previously [48] The control rAd expressing shRNA targeting LacZ (rAd/U6-LacZshRNA) was from Invitrogen life Technologies. The rAd titer (infectious unit: ifu/ml) was determined by staining the largest and most abundant structural proteins “hexon proteins” in the adenovirus capsid using QuickTiter™ Adenovirus Titer Immunoassay Kit after infecting HER911 cells, derived from human retina cells by Adenovirus E1 transformation [49], for 48 hours.
Western blot
Cell lysate preparation, SDS-PAGE and Western blot, antibody incubation and signal detection were carried out as described previously [30]. Quantification of signals was performed using NIH Image J (NIH, Bethesda, MD, USA).
Arginase activity assay
Arginase activity in cells lysates was measured by colorimetric determination of urea formed from L-arginine as described previously [31].
Activity of mTORC1 and S6K1
mTORC1 and S6K1 activity were analyzed by monitoring phosphorylation of S6K1 at threonine-389 (S6K1-T389) and its substrate S6 at serine-235/serine-236 (S235/S236), respectively, by Western blotting.
Senescence-associated β-galactosidase (SA-β-gal) staining
Cells were initially washed twice with PBS followed by fixation with 3.7% formaldehyde solution in PBS for 10–15 min. After washing twice with PBS, cells were then incubated with the SA-β-gal staining solution (1 mg/ml X-gal, 40 mmol/L citric acid, 5 mmol/L potassium ferrocyanide, 5 mmol/L potassium ferricyanide, 150 mmol/L sodium chloride, 2 mmol/L magnesium chloride dissolved in phosphate buffer, pH 6.0) for 16 hours at 37°C in a CO2-free atmosphere. The stained senescent cells were detected by conventional microscopy.
Detection of nitric oxide (NO) and superoxide levels in cultured endothelial cells
For detection of NO, HUVECs were gently washed twice with Ca2+-free PBS, and incubated in a modified Krebs-Ringer bicarbonate solution (in mmol/L, NaCl 118, KCl 4.7, CaCl2 2.5, MgSO4 1.2, KH2PO4 1.2, NaHCO3 25, EDTA 0.026, and glucose 5.5) containing 5 μmol/L of DAF-2DA for 30 minutes. For measurement of superoxide, cells were incubated with 5 μmol/L DHE in culture medium for 20 minutes. For functional eNOS-uncoupling study, cells were pre-treated with the eNOS inhibitor L-NAME (1 mmol/L) for 2 hours before DHE was added. After staining, the cells were washed three times and images were obtained with Zeiss fluorescence microscopy. The intensity of the fluorescence was quantified by Image J software (U. S. National Institutes of Health).
Statistics
Data are given as mean±SEM. In all experiments, n represents the number of independent sets of experiments. Statistical analysis was performed with Student's unpaired t-test or analysis of variance (ANOVA) with Bonferroni post test. Differences in mean values were considered significant at two tailed p ≤ 0.05.
Acknowledgments
We are grateful to Mr. Jean Ruffieux for technical assistance with endothelial cell isolation and culture. This work was supported by the Swiss National Science Foundation (310030_141070/1) and Swiss Heart Foundation. Yuyan Xiong and Yi Yu were supported by the Chinese Scholarship Council.
Conflicts of Interest
The authors declare no conflict of interests.
References
- 1. Morris SM Jr. Recent advances in arginine metabolism: roles and regulation of the arginases. Br J Pharmacol. 2009; 157: 922 -930. [PubMed] .
- 2. Wu G and Meininger CJ. Arginine nutrition and cardiovascular function. J Nutr. 2000; 130: 2626 -2629. [PubMed] .
- 3. Palmer RM, Ashton DS, Moncada S. Vascular endothelial cells synthesize nitric oxide from L-arginine. Nature. 1988; 333: 664 -666. [PubMed] .
- 4. Nieves C Jr and Langkamp-Henken B. Arginine and immunity: a unique perspective. Biomed Pharmacother. 2002; 56: 471 -482. [PubMed] .
- 5. McKnight JR, Satterfield MC, Jobgen WS, Smith SB, Spencer TE, Meininger CJ, McNeal CJ, Wu G. Beneficial effects of L-arginine on reducing obesity: potential mechanisms and important implications for human health. Amino Acids. 2010; 39: 349 -357. [PubMed] .
- 6. Clemmensen C, Smajilovic S, Smith EP, Woods SC, Brauner-Osborne H, Seeley RJ, D'Alessio DA, Ryan KK. Oral L-arginine stimulates GLP-1 secretion to improve glucose tolerance in male mice. Endocrinology. 2013; 154: 3978 -3983. [PubMed] .
- 7. Alvares TS, Meirelles CM, Bhambhani YN, Paschoalin VM, Gomes PS. L-Arginine as a potential ergogenic aid in healthy subjects. Sports Med. 2011; 41: 233 -248. [PubMed] .
- 8. Girerd XJ, Hirsch AT, Cooke JP, Dzau VJ, Creager MA. L-arginine augments endothelium-dependent vasodilation in cholesterol-fed rabbits. Circ Res. 1990; 67: 1301 -1308. [PubMed] .
- 9. Creager MA, Gallagher SJ, Girerd XJ, Coleman SM, Dzau VJ, Cooke JP. L-arginine improves endothelium-dependent vasodilation in hypercholesterolemic humans. J Clin Invest. 1992; 90: 1248 -1253. [PubMed] .
- 10. Clarkson P, Adams MR, Powe AJ, Donald AE, McCredie R, Robinson J, McCarthy SN, Keech A, Celermajer DS, Deanfield JE. Oral L-arginine improves endothelium-dependent dilation in hypercholesterolemic young adults. J Clin Invest. 1996; 97: 1989 -1994. [PubMed] .
- 11. Adams MR, McCredie R, Jessup W, Robinson J, Sullivan D, Celermajer DS. Oral L-arginine improves endothelium-dependent dilatation and reduces monocyte adhesion to endothelial cells in young men with coronary artery disease. Atherosclerosis. 1997; 129: 261 -269. [PubMed] .
- 12. Boger RH, Bode-Boger SM, Thiele W, Creutzig A, Alexander K, Frolich JC. Restoring vascular nitric oxide formation by L-arginine improves the symptoms of intermittent claudication in patients with peripheral arterial occlusive disease. J Am Coll Cardiol. 1998; 32: 1336 -1344. [PubMed] .
- 13. Sydow K, Schwedhelm E, Arakawa N, Bode-Boger SM, Tsikas D, Hornig B, Frolich JC, Boger RH. ADMA and oxidative stress are responsible for endothelial dysfunction in hyperhomocyst(e)inemia: effects of L-arginine and B vitamins. Cardiovasc Res. 2003; 57: 244 -252. [PubMed] .
- 14. Palloshi A, Fragasso G, Piatti P, Monti LD, Setola E, Valsecchi G, Galluccio E, Chierchia SL, Margonato A. Effect of oral L-arginine on blood pressure and symptoms and endothelial function in patients with systemic hypertension, positive exercise tests, and normal coronary arteries. Am J Cardiol. 2004; 93: 933 -935. [PubMed] .
- 15. Dong JY, Qin LQ, Zhang Z, Zhao Y, Wang J, Arigoni F, Zhang W. Effect of oral L-arginine supplementation on blood pressure: a meta-analysis of randomized, double-blind, placebo-controlled trials. Am Heart J. 2011; 162: 959 -965. [PubMed] .
- 16. Wennmalm A, Edlund A, Granstrom EF, Wiklund O. Acute supplementation with the nitric oxide precursor L-arginine does not improve cardiovascular performance in patients with hypercholesterolemia. Atherosclerosis. 1995; 118: 223 -231. [PubMed] .
- 17. Chin-Dusting JP, Alexander CT, Arnold PJ, Hodgson WC, Lux AS, Jennings GL. Effects of in vivo and in vitro L-arginine supplementation on healthy human vessels. J Cardiovasc Pharmacol. 1996; 28: 158 -166. [PubMed] .
- 18. Blum A, Hathaway L, Mincemoyer R, Schenke WH, Kirby M, Csako G, Waclawiw MA, Panza JA, Cannon RO III. Oral L-arginine in patients with coronary artery disease on medical management. Circulation. 2000; 101: 2160 -2164. [PubMed] .
- 19. Oomen CM, van Erk MJ, Feskens EJ, Kok FJ, Kromhout D. Arginine intake and risk of coronary heart disease mortality in elderly men. Arterioscler Thromb Vasc Biol. 2000; 20: 2134 -2139. [PubMed] .
- 20. Alvares TS, Conte-Junior CA, Silva JT, Paschoalin VM. Acute L-Arginine supplementation does not increase nitric oxide production in healthy subjects. Nutr Metab (Lond). 2012; 9: 54 [PubMed] .
- 21. Chen J, Kuhlencordt P, Urano F, Ichinose H, Astern J, Huang PL. Effects of chronic treatment with L-arginine on atherosclerosis in apoE knockout and apoE/inducible NO synthase double-knockout mice. Arterioscler Thromb Vasc Biol. 2003; 23: 97 -103. [PubMed] .
- 22. Schulman SP, Becker LC, Kass DA, Champion HC, Terrin ML, Forman S, Ernst KV, Kelemen MD, Townsend SN, Capriotti A, Hare JM, Gerstenblith G. L-arginine therapy in acute myocardial infarction: the Vascular Interaction With Age in Myocardial Infarction (VINTAGE MI) randomized clinical trial. JAMA. 2006; 295: 58 -64. [PubMed] .
- 23. Wilson AM, Harada R, Nair N, Balasubramanian N, Cooke JP. L-arginine supplementation in peripheral arterial disease: no benefit and possible harm. Circulation. 2007; 116: 188 -195. [PubMed] .
- 24. Scalera F, Closs EI, Flick E, Martens-Lobenhoffer J, Boissel JP, Lendeckel U, Heimburg A, Bode-Boger SM. Paradoxical effect of L-arginine: acceleration of endothelial cell senescence. Biochem Biophys Res Commun. 2009; 386: 650 -655. [PubMed] .
- 25. Yepuri G, Velagapudi S, Xiong Y, Rajapakse AG, Montani JP, Ming XF, Yang Z. Positive crosstalk between arginase-II and S6K1 in vascular endothelial inflammation and aging. Aging Cell. 2012; 11: 1005 -1016. [PubMed] .
- 26. Bonfils G, Jaquenoud M, Bontron S, Ostrowicz C, Ungermann C, De VC. Leucyl-tRNA Synthetase Controls TORC1 via the EGO Complex. Mol Cell. 2012; 46: 105 -110. [PubMed] .
- 27. Han JM, Jeong SJ, Park MC, Kim G, Kwon NH, Kim HK, Ha SH, Ryu SH, Kim S. Leucyl-tRNA Synthetase Is an Intracellular Leucine Sensor for the mTORC1-Signaling Pathway. Cell. 2012; 149: 410 -424. [PubMed] .
- 28. Yang Z and Ming XF. Arginase: the emerging therapeutic target for vascular oxidative stress and inflammation. Front Immunol. 2013; 4: 149 [PubMed] .
- 29. Ming XF, Barandier C, Viswambharan H, Kwak BR, Mach F, Mazzolai L, Hayoz D, Ruffieux J, Rusconi S, Montani JP, Yang Z. Thrombin stimulates human endothelial arginase enzymatic activity via RhoA/ROCK pathway: implications for atherosclerotic endothelial dysfunction. Circulation. 2004; 110: 3708 -3714. [PubMed] .
- 30. Ming XF, Rajapakse AG, Yepuri G, Xiong Y, Carvas JM, Ruffieux J, Scerri I, Wu Z, Popp K, Li J, Sartori C, Scherrer U, Kwak BR, Montani JP, Yang Z. Arginase II Promotes Macrophage Inflammatory Responses Through Mitochondrial Reactive Oxygen Species, Contributing to Insulin Resistance and Atherogenesis. J Am Heart Assoc. 2012; 1: e000992 [PubMed] .
- 31. Xiong Y, Yu Y, Montani JP, Yang Z, Ming XF. Arginase-II Induces Vascular Smooth Muscle Cell Senescence and Apoptosis Through p66Shc and p53 Independently of Its l-Arginine Ureahydrolase Activity: Implications for Atherosclerotic Plaque Vulnerability. J Am Heart Assoc. 2013; 2: e000096 [PubMed] .
- 32. Grody WW, Argyle C, Kern RM, Dizikes GJ, Spector EB, Strickland AD, Klein D, Cederbaum SD. Differential expression of the two human arginase genes in hyperargininemia. Enzymatic, pathologic, and molecular analysis. J Clin Invest. 1989; 83: 602 -609. [PubMed] .
- 33. Efeyan A, Zoncu R, Sabatini DM. Amino acids and mTORC1: from lysosomes to disease. Trends Mol Med. 2012; 18: 524 -533. [PubMed] .
- 34. Demidenko ZN, Zubova SG, Bukreeva EI, Pospelov VA, Pospelova TV, Blagosklonny MV. Rapamycin decelerates cellular senescence. Cell Cycle. 2009; 8: 1888 -1895. [PubMed] .
- 35. Korotchkina LG, Leontieva OV, Bukreeva EI, Demidenko ZN, Gudkov AV, Blagosklonny MV. The choice between p53-induced senescence and quiescence is determined in part by the mTOR pathway. Aging. 2010; 2: 344 -352. [PubMed] .
- 36. Kolesnichenko M, Hong L, Liao R, Vogt PK, Sun P. Attenuation of TORC1 signaling delays replicative and oncogenic RAS-induced senescence. Cell Cycle. 2012; 11: 2391 -2401. [PubMed] .
- 37. Nakano M, Nakashima A, Nagano T, Ishikawa S, Kikkawa U, Kamada S. Branched-chain amino acids enhance premature senescence through mammalian target of rapamycin complex I-mediated upregulation of p21 protein. PloS One. 2013; 8: e80411 [PubMed] .
- 38. Luo Y, Li L, Zou P, Wang J, Shao L, Zhou D, Liu L. Rapamycin enhances long-term hematopoietic reconstitution of ex vivo expanded mouse hematopoietic stem cells by inhibiting senescence. Transplantation. 2014; 97: 20 -29. [PubMed] .
- 39. Leontieva OV, Demidenko ZN, Blagosklonny MV. S6K in geroconversion. Cell Cycle. 2013; 12: 3249 -3252. [PubMed] .
- 40. Hallemeesch MM, Lamers WH, Deutz NE. Reduced arginine availability and nitric oxide production. Clin Nutr. 2002; 21: 273 -279. [PubMed] .
- 41. Hardy TA and May JM. Coordinate regulation of L-arginine uptake and nitric oxide synthase activity in cultured endothelial cells. Free Radic Res Commun. 2002; 32: 122 -131. .
- 42. Rajapakse NW and Mattson DL. Role of L-arginine in nitric oxide production in health and hypertension. Clin Exp Pharmacol Physiol. 2009; 36: 249 -255. [PubMed] .
- 43. Shin S, Mohan S, Fung HL. Intracellular L-arginine concentration does not determine NO production in endothelial cells: implications on the ‘L-arginine paradox’. Biochem Biophys Res Commun. 2011; 414: 660 -663. [PubMed] .
- 44. Cunningham JT, Rodgers JT, Arlow DH, Vazquez F, Mootha VK, Puigserver P. mTOR controls mitochondrial oxidative function through a YY1-PGC-1alpha transcriptional complex. Nature. 2007; 450: 736 -740. [PubMed] .
- 45. Chen C, Liu Y, Liu R, Ikenoue T, Guan KL, Liu Y, Zheng P. TSC-mTOR maintains quiescence and function of hematopoietic stem cells by repressing mitochondrial biogenesis and reactive oxygen species. J Exp Med. 2008; 205: 2397 -2408. [PubMed] .
- 46. Mohan S, Wu CC, Shin S, Fung HL. Continuous exposure to L-arginine induces oxidative stress and physiological tolerance in cultured human endothelial cells. Amino acids. 2012; 43: 1179 -1188. [PubMed] .
- 47. Elms S, Chen F, Wang Y, Qian J, Askari B, Yu Y, Pandey D, Iddings J, Caldwell RB, Fulton DJ. Insights into the arginine paradox: evidence against the importance of subcellular location of arginase and eNOS. Am J Physiol Heart Circ Physiol. 2013; 305: H651 -H666. [PubMed] .
- 48. Ming XF, Rajapakse AG, Carvas JM, Ruffieux J, Yang Z. Inhibition of S6K1 accounts partially for the anti-inflammatory effects of the arginase inhibitor L-norvaline. BMC Cardiovasc Disord. 2009; 9: 12 [PubMed] .
- 49. Fallaux FJ, Kranenburg O, Cramer SJ, Houweling A, van OH, Hoeben RC, van Der Eb AJ. Characterization of 911: a new helper cell line for the titration and propagation of early region 1-deleted adenoviral vectors. Hum Gene Ther. 1996; 7: 215 -222. [PubMed] .
- 50. Ming XF, Viswambharan H, Barandier C, Ruffieux J, Kaibuchi K, Rusconi S, Yang ZH. Rho GTPase/Rho kinase negatively regulates endothelial nitric oxide synthase phosphorylation through the inhibition of protein kinase B/Akt in human endothelial cells. Mol Cell Biol. 2002; 22: 8467 -8477. [PubMed] .



