Introduction
Aging has been defined as a progressive organic functional decline, with loss of homeostasis and increasing probability of illness and death [1]. Although applied research on aging had resulted in considerable scientific knowledge with regard to the related causes, it is still subject to numerous debates and contradictions. Indeed, in most cases, it is difficult to understand how a particular variable is a possible cause or consequence of aging. Some studies have suggested an age-related accumulation of macromolecular damage, which may cause progressive and irreversible physiological attrition and homeostasis loss, accelerating aging. In addition, considering the important role of DNA in living organisms and regarding the changes that occur in this macromolecule throughout life, the question arises, whether DNA modifications should be considered a central factor of aging. Moreover, it is not clear if these damages are a possible cause or an expression of aging. Since the first study of Failla [2], numerous other investigations have been performed and different assays to measure DNA changes have been developed. These assays allow the assessment of some age-related DNA changes, as well as other associated variables that could interfere with DNA damage accumulation.
Many theories of aging are based on DNA changes, including the Intrinsic Mutagenesis Theory, Somatic Mutations Theory and DNA Repair Theory [3, 4]. Other theories also explain the age-related changes in DNA as a consequence of stochastic events. The Oxidative Stress Theory is, perhaps, the main stochastic explanation of DNA and other macromolecular damage accumulating with age [4]. Despite the importance of the age-related DNA damage accumulation, some researchers argue that aging is not caused by the accumulation of damage but is the result of continued activity (cell hyperfunction) of pathways and processes during adulthood that evolved to optimize development to this life stage [5, 6]. According this theory, the same pathway, which drives developmental growth, later drives aging and associated diseases [7]. Cell hyperfunction is driven by the nutrient-sensitive signaling network that controls growth (and thereby, reproduction), and includes the insulin, insulin-like growth factor 1 (IGF-1), and in particular, the target of rapamycin (TOR) kinase pathways [6-8]. Thus, accumulation of DNA damage is a consequence of aging and not a leading cause.
Although DNA is not the only target changed with aging, taking account of the major role of this macromolecule in the regulation of all cellular structures and its own cell cycle, DNA damage has been studied with particular attention. The alterations could have several consequences for genome stability with repercussions on cellular component synthesis, cell cycle machinery and signaling pathways that control cell cycle arrest, and programed cell death or apoptosis [9]. The consequences of DNA damage will depend on the type of damage, genes affected and type of cell and tissue damaged.
The prevailing view is that there is a tendency for an age-related DNA damage accumulation. However, on examination, results of studies show inconsistency [10-13]; it is possible that confounding factors influence this relation and explain some of the inconsistency.
Considering the complexity of aging, it must be emphasized that aging does not happen in the same way in different individuals, nor in the same way in all cell types and tissues of the same individual. Moreover, aging is a life-long process, influenced continually by environmental conditions. Factors such as diet, lifestyle, exposure to radiation and genotoxic chemicals seem to have a significant influence on the relationship between cumulative DNA damage and age [14-16].
Methodological factors might have also influenced the observed results [14-16]. Indeed, different assays may be used to measure DNA damage. Furthermore, the measured DNA damage could reflect changes in the causative factors, and/or changes in DNA protection and/or changes in DNA repair capacity. It must also be noted that the type of cell and tissue used could reflect different aging rates within the organism.
Although there are several excellent narrative reviews on age-related nuclear DNA damage [17-20], they usually refer to individual animal and humans studies and, as far as we know, no meta-analytic technique has been used to estimate the extent of effect of potential moderators on age-related DNA damage in humans. Thus, the overall goal of this paper is to address this important gap in the literature. The first aim of this review is to provide a summary of age-related changes in nuclear DNA in humans. The second aim is to examine the effects of some moderators associated with DNA damage. The third aim is to discuss promising directions for future researches in the light of our findings.
Results
An initial search using the keywords described located 2953 studies. After reading titles and abstracts, the number of studies was reduced to 267. In the final refinement of the research, applying inclusion and exclusion criteria, only 36 studies fulfilled all necessary requirements.
Study Analysis
The results indicated a significant and positive association between age and DNA damage in 76 correlations of the 36 studies (N = 4676) r = 0.230, p = 0.000 (95% confidence interval = 0.111; 0.342). A test for heterogeneity of variance indicates that the results of the study are significantly higher than would be expected, Q (75) = 1754.831, p = 0.000. The effect size of each study can be seen in Figure 1.
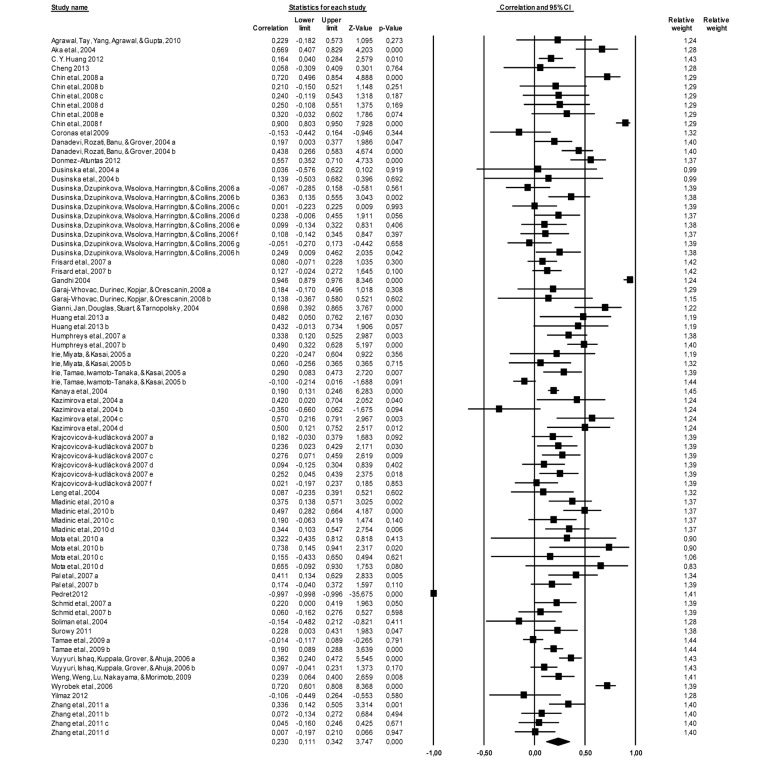
Figure 1. Forest plot, the effect size (r) of each study (relative weight of each study in the age-related DNA damage). IC=confidence interval. a, b, c, d, e, f, g, h – different measured endpoints from the same study [10-15, 21-48].
The analysis of the moderator variables is shown in Table 1. As can be seen, tobacco use, sample/tissue and technique, but not sex, are identified as moderator variables.
Table 1.
Effect size of the moderator variables (Sex, Tobacco, Sample/Tissue, and Technique) on age-related DNA damage
| Moderator | Value | K | R | 95%CI | P | p between groups | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sex | Female | 25 | 0.116 | 0.05; 0.18 | 0.000 | 0.114 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Male | 14 | 0.177 | 0.13; 0.22 | 0.000 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tobacco | Non-Smokers | 26 | −0.043 | −0.10; −0.02 | 0.162 | 0.026 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smokers | 2 | 0.176 | −0.01; 0.35 | 0.058 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sample/Tissue | Buccal | 4 | 0.229 | 0.14; 0.32 | 0.000 | 0.000 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mammary | 1 | −0.106 | −0.45; 0.26 | 0.580 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PBMC | 57 | 0.235 | 0.20; 0.27 | 0.000 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spermatozoa | 3 | 0.392 | 0.28; 0.49 | 0.000 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Urine | 11 | −0.007 | −0.05; 0.03 | 0.711 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Technique | SCSA | 1 | 0.720 | 0.60; 0.81 | 0.000 | 0.000 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SCGE–CA | 47 | 0.208 | 0.17; 0.24 | 0.000 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ELISA | 7 | −0.083 | −0.14; −0.03 | 0.002 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HPLC | 8 | 0.072 | 0.02; 0.12 | 0.007 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MN | 11 | 0.268 | 0.19; 0.34 | 0.000 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SCE | 2 | 0.831 | 0.73; 0.90 | 0.000 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PBMC – peripheral blood mono-nuclear cells; SCSA – sperm chromatin structure assay; SCGE–CA – Single cell gel electrophoresis- Comet Assay; ELISA - enzyme-linked immunosorbent assay; HPLC – High Performance/Pressure Liquid Chromatography; MN- micronucleus; SCE – sister chromatid exchange. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Discussion
DNA changes associated with age have been claimed as one of the main possible causes of aging. These alterations may result in genetic instability, mutagenesis, disease, and cell death. Despite its popularity, this association has a lack of consensus in the literature apparently due to several factors such as sample characteristics, technique and methods used. To clarify this relation, we conducted a meta-analysis to investigate the association between age and DNA damage in humans. Our main finding is a positive association between age and DNA damage in humans, both in males and females. So, aging in humans is accompanied by an increase in general DNA damage.
However, the association found is weak and as the Cochran's Q statistic and the I2 statistic revealed a high heterogeneity between studies. To better understand this association, there are some points that must be taken into consideration. First of all, this weak association implies that there are other variables which may influence age-related DNA damage. Variables suggested in the literature include sex, smoking, alcohol consumption, physical exercise, nutrition, psychological stress, etc. Secondly, the sample's characteristics, technique used, tissue and the type of damage analyzed could influence results. Considering this, some variables were analyzed here as possible moderator variables influencing age-related DNA damage. Several works have studied the effect of sex on DNA damage. Despite the fact that some studies have identified differences between sexes in DNA damage [23, 26, 36], our results based on analysis of 36 studies have shown no such differences. This means that both sexes show increased DNA damage with age, even though the absolute values could be different. In short, sex as a moderator variable has no influence on age-related DNA damage.
Although in the literature several lifestyle variables have been related with DNA damage, according to the results from the studies analyzed here, only smoking habit could be considered as a moderator variable; at the time of our analysis, there were not enough studies concerning the remaining variables. Since tobacco smoke contains known carcinogens, it seems plausible that smokers could accumulate more DNA damage with age, compared with non-smokers. Our analysis confirms this hypothesis, showing that smokers demonstrate more age-related DNA damage. Our results clearly suggest that smoking should be considered as a moderator variable in the age-related DNA damage studies.
As mentioned before, it is well established that aging does not occur at the same rate in the different organs. Accordingly, it might be expected that different sample tissues might show different age-related DNA damage. Considering the sample tissue studied, we found significant differences between them. Unexpectedly, mammary cells and urine samples have shown no there was only one study of mammary tissue cells and the urine sample results show high heterogeneity remaining cell types studied (buccal cells, peripheral blood cells and spermatozoa), less heterogeneity between studies was seen, and a positive correlation with age-related DNA damage was found.
Our results have shown that technique is a moderator variable when age-related DNA damage is studied, so that depending on the technique used, we might expect different results. Studies using ELISA have shown a weak negative association between age and DNA damage; HPLC, SCGE - CA and MN have shown a positive association but also weak; and a strong association for SCSA and SCE has been found, even though these last two were based on only one and two studies, respectively. Regarding these techniques, it is important to mention that ELISA and HPLC are the only two techniques which are used in urine and/or in tissues cells. This is of major importance because results outcomes are clearly different, since in the case of urine samples DNA damage is not analyzed directly, but rather the result of that damage in the overall body system. So our results illustrate the importance of careful interpretation, especially in comparisons of results from different studies.
There are some questions underlying age-related accumulation of DNA damage which must be taken into account. Firstly, it was our proposal to study the set of variables that might be associated with the DNA damage and aging. Though we were only able to consider smoking habits, other lifestyle variables should be evaluated in studies with human samples. Also, the methodology is important for understanding the results and interpreting heterogeneity. Sample characteristics as well as inclusion criteria are relevant to understand the results achieved. The range of ages included in a study is likely to influence the results – a wide range likely leading to more pronounced effects: in this meta-analysis age range was not considered due the variability of the studies designs and to the lack of information presented in the articles. Further, the weak association between DNA damage and age found in this meta-analysis raises the question of if accumulation of damage is determinative for the aging process. As mentioned above, it is not clear if age-associated damage is a cause or an expression of aging. The molecular damage theory has postulated that aging is caused by the progressive accumulation of damage; however according to the hyperfunction theory this damage is a consequence of aging and [7] thus does not necessarily limit lifespan [5, 7, 49]. Instead, the observed increased levels of damage are important for some pathologies, such as cancer, and are the result of hyperfunction. The hyperfunction theory even suggests that repair of molecular damage is important for increased longevity, but the involvement of any process for viability does not imply its role in aging [7]. In summary, there are emerging explanations concerning our understanding of aging, which provide a novel perspective on aging and the DNA accumulation of damage.
In conclusion, independent of the perspective theory of aging, our meta-analysis results show an age-related increase in DNA damage in humans. Furthermore, smoking habits, the sample/tissue and the analysis technique used are important moderator variables. There is a set of lifestyle variables which should be more carefully studied in longitudinal studies, since it seems that age-related DNA damage only explains a small part of aging. Future studies may also rely on the relevance of the age-related DNA damage and its possible role as a marker of biological aging.
Methods
We have conducted a meta-analysis based on the criteria described for meta-analyses and systematic reviews by Moher et al. [50].
Data Source and search strategy
In order to achieve the largest number of publications, MEDLINE PubMed and Web of science (Web of ScienceSM; current Contents Connect) databases were used with the combination of the English key terms: “DNA”, “damage” and “age”, and all eligible studies between 2004 and “March 2013” were selected.
In addition, the reference lists in these articles were searched manually to find other relevant publications.
Selection Criteria and Identification of Studies
The following inclusion criteria were used to select the articles to our study: articles in English, studies on nuclear DNA, male and/or female human studies, healthy subjects or studies with healthy control subjects, papers clearly describing the sources of cases and controls, and information given on the size of the sample and statistical values.
The exclusion criteria were: post-mortem studies, in vitro studies, studies of newborns, children and puberty, studies in exposed subjects without control group, and studies in non-healthy subjects without healthy control group.
Data extraction
The following data were collected from each study: first author's surname; year of publication; cells and tissues analyzed; evaluation technique used; total number of cases and controls; age groups, DNA damage data.
Selection of moderator variables
Moderator variables were largely based on the models presented by Cook-Cottone et al. (2009) [51].
Two authors of the present study were responsible for separately encoding each of the moderator variables, which were then compared to ascertain the percentage of agreement. The description of the criteria for coding is presented in the following section.
Many biomonitoring studies have shown that some indicators of genetic damage in different cells depend on various internal factors (such as sex and age) and external factors (such as smoke).
Sex
Sex seems to be an important factor to be taken into account when conducting epidemiological studies. There is a lack of consensus on its influence on DNA damage. Adult women were reported to have lower levels of damage than men but others studies contradict this trend. In addition, it is not known whether sex might influence the age-related DNA damage.
Smoking
It is well-known that some lifestyle behaviors have an influence on the stability and integrity of the DNA. In particular, smoking is a source of carcinogens which could have a significant effect on DNA damage. Despite that, the relationship between smoking and DNA damage as measured with the comet assay in PBMN cells is still inconsistent in the literature.
Sample/Tissue
Aging does not occur in all biological structures in the same way. Indeed, some conflicting results could be explained by the different sample/tissue studied.
Technique
Some of the inconsistency of results in the literature can be attributed to the different techniques used in different laboratories; these might vary in sensitivity, or might emphasize different kinds of lesion (for instance, SBs or oxidized bases).
Statistical analyses
This meta-analysis included 36 studies. The strength of the association between DNA damage and age was assessed by the correlation values (r) described in the studies. In comparative studies the p values or t tests were analyzed to estimate the r value. In other works the correlation coefficient was calculated from mean and standard deviation values.
Analyses of results were performed using subgroups based on the moderator variables described above.
The statistical analyses were performed with the software Comprehensive Meta-analysis (CMA, version 2.2.048) [52]. Both the Cochran's Q statistic and the I2 statistic, to test for heterogeneity and to quantify the proportion of the total variation due to heterogeneity, were calculated. We chose the random-effects model due the great variability of the samples and techniques used.
Several methods were used to assess the potential publication bias. Visual inspection of funnel plot asymmetry was conducted. The Begg's rank correlation method [53] and the Egger's weighted regression method [54] were used to statistically assess publication bias (P<0.05 was considered statistically significant). The funnel plot shows a slightly asymmetrical distribution of points; however the rank-correlation test of Begg's (p=0.095) and Egger's (p=0.112) showed a non-significant publication bias.
Funding
This work was supported by Foundation of Science and Technology (FCT) for the research grant SFRH/BD/66438/2009 and for the project entitled “Physical exercise role on Human' lymphocyte DNA damage reduction: possible influence of oxidative stress and DNA repair capacity” PTDC/DES/121575/2010.
Conflicts of Interest
Authors declare no conflict of interests.
References
- 1. Schumacher B, Garinis GA, Hoeijmakers JH. Age to survive: DNA damage and aging. Trends Genet. 2008; 24: 77 -85. [PubMed] .
- 2. Failla G. The aging process and cancerogenesis. Annals of the New York Academy of Sciences. 1958; 71: 1124 -1140. [PubMed] .
- 3. Weinert BT and Timiras PS. Invited review: Theories of aging. J Appl Physiol. 2003; 95: 1706 -1716. [PubMed] .
- 4. Rattan SI. Theories of biological aging: genes, proteins, and free radicals. Free radical research. 2006; 40: 1230 -1238. [PubMed] .
- 5. Gems D and de la Guardia Y. Alternative Perspectives on Aging in Caenorhabditis elegans: Reactive Oxygen Species or Hyperfunction? Antioxidants & redox signaling. 2013; 19: 321 -329. [PubMed] .
- 6. Leontieva OV, Paszkiewicz GM, Blagosklonny MV. Mechanistic or mammalian target of rapamycin (mTOR) may determine robustness in young male mice at the cost of accelerated aging. Aging-Us. 2012; 4: 899 -916. .
- 7. Blagosklonny MV. Answering the ultimate question “what is the proximal cause of aging?” Aging. Albany NY 2012; 4: 861 -877. [PubMed] .
- 8. Blagosklonny MV and Hall MN. Growth and aging: a common molecular mechanism. Aging-Us. 2009; 1: 357 -362. .
- 9. Lombard DB, Chua KF, Mostoslavsky R, Franco S, Gostissa M, Alt FW. DNA repair, genome stability, and aging. Cell. 2005; 120: 497 -512. [PubMed] .
- 10. Coronas MV, Pereira TS, Rocha JA, Lemos AT, Fachel JM, Salvadori DM, Vargas VM. Genetic biomonitoring of an urban population exposed to mutagenic airborne pollutants. Environ Int. 2009; 35: 1023 -1029. [PubMed] .
- 11. Pedret A, Valls RM, Fernandez-Castillejo S, Catalan U, Romeu M, Giralt M, Lamuela-Raventos RM, Medina-Remon A, Arija V, Aranda N, Espinel A, Delgado MA, Sola R. Polyphenol-rich foods exhibit DNA antioxidative properties and protect the glutathione system in healthy subjects. Mol Nutr Food Res. 2012; 56: 1025 -1033. [PubMed] .
- 12. Wyrobek AJ, Eskenazi B, Young S, Arnheim N, Tiemann-Boege I, Jabs EW, Glaser RL, Pearson FS, Evenson D. Advancing age has differential effects on DNA damage, chromatin integrity, gene mutations, and aneuploidies in sperm. Proceedings of the National Academy of Sciences of the United States of America. 2006; 103: 9601 -9606. [PubMed] .
- 13. Gandhi G and Kumar N. DNA damage in peripheral blood lymphocytes of individuals residing near a wastewater drain and using underground water resources. Environmental and molecular mutagenesis. 2004; 43: 235 -242. [PubMed] .
- 14. Kazimirova A, Barancokova M, Volkovova K, Staruchova M, Krajcovicova-Kudlackova M, Wsolova L, Collins AR, Dusinska M. Does a vegetarian diet influence genomic stability? Eur J Nutr. 2004; 43: 32 -38. [PubMed] .
- 15. Tamae K, Kawai K, Yamasaki S, Kawanami K, Ikeda M, Takahashi K, Miyamoto T, Kato N, Kasai H. Effect of age, smoking and other lifestyle factors on urinary 7-methylguanine and 8-hydroxydeoxyguanosine. Cancer Sci. 2009; 100: 715 -721. [PubMed] .
- 16. Sellappa S, Prathyumnan S, Balachandar V. DNA damage induction and repair inhibition among building construction workers in South India. Asian Pacific journal of cancer prevention : APJCP. 2010; 11: 875 -880. [PubMed] .
- 17. Freitas AA and de Magalhaes JP. A review and appraisal of the DNA damage theory of ageing. Mutation research. 2011; 728: 12 -22. [PubMed] .
- 18. Fenech M and Bonassi S. The effect of age, gender, diet and lifestyle on DNA damage measured using micronucleus frequency in human peripheral blood lymphocytes. Mutagenesis. 2011; 26: 43 -49. [PubMed] .
- 19. Seviour EG and Lin SY. The DNA damage response: Balancing the scale between cancer and ageing. Aging (Albany NY). 2010; 2: 900 -907. [PubMed] .
- 20. Wilson DM 3rd, Bohr VA, McKinnon PJ. DNA damage, DNA repair, ageing and age-related disease. Mech Ageing Dev. 2008; 129: 349 -352. [PubMed] .
- 21. Aka P, Mateuca R, Buchet JP, Thierens H, Kirsch-Volders M. Are genetic polymorphisms in OGG1, XRCC1 and XRCC3 genes predictive for the DNA strand break repair phenotype and genotoxicity in workers exposed to low dose ionising radiations? Mutation research. 2004; 556: 169 -181. [PubMed] .
- 22. Huang CY, Su CT, Chung CJ, Pu YS, Chu JS, Yang HY, Wu CC, Hsueh YM. Urinary total arsenic and 8-hydroxydeoxyguanosine are associated with renal cell carcinoma in an area without obvious arsenic exposure. Toxicol Appl Pharmacol. 2012; 262: 349 -354. [PubMed] .
- 23. Cheng X, Zhang T, Zhao J, Zhou J, Shao H, Zhou Z, Kong F, Feng N, Sun Y, Shan B, Xia Z. The association between genetic damage in peripheral blood lymphocytes and polymorphisms of three glutathione S-transferases in Chinese workers exposed to 1,3-butadiene. Mutation research. 2013; 750: 139 -146. [PubMed] .
- 24. Chin SF, Hamid NA, Latiff AA, Zakaria Z, Mazlan M, Yusof YA, Karim AA, Ibahim J, Hamid Z, Ngah WZ. Reduction of DNA damage in older healthy adults by Tri E Tocotrienol supplementation Nutrition. Burbank, Los Angeles County, Calif 2008; 24: 1 -10. [PubMed] .
- 25. Danadevi K, Rozati R, Banu BS, Grover P. Genotoxic evaluation of welders occupationally exposed to chromium and nickel using the Comet and micronucleus assays. Mutagenesis. 2004; 19: 35 -41. [PubMed] .
- 26. Donmez-Altuntas H and Bitgen N. Evaluation of the genotoxicity and cytotoxicity in the general population in Turkey by use of the cytokinesis-block micronucleus cytome assay. Mutation research. 2012; 748: 1 -7. [PubMed] .
- 27. Dusinska M, Collins A, Kazimirova A, Barancokova M, Harrington V, Volkovova K, Staruchova M, Horska A, Wsolova L, Kocan A, Petrik J, Machata M, Ratcliffe B, et al. Genotoxic effects of asbestos in humans. Mutation research. 2004; 553: 91 -102. [PubMed] .
- 28. Dusinska M, Dzupinkova Z, Wsolova L, Harrington V, Collins AR. Possible involvement of XPA in repair of oxidative DNA damage deduced from analysis of damage, repair and genotype in a human population study. Mutagenesis. 2006; 21: 205 -211. [PubMed] .
- 29. Frisard MI, Broussard A, Davies SS, Roberts LJ 2nd, Rood J, de Jonge L, Fang X, Jazwinski SM, Deutsch WA, Ravussin E. Aging, resting metabolic rate, and oxidative damage: results from the Louisiana Healthy Aging Study. J Gerontol A Biol Sci Med Sci. 2007; 62: 752 -759. [PubMed] .
- 30. Garaj-Vrhovac V, Durinec M, Kopjar N, Orescanin V. A survey on the cytogenetic status of the Croatian general population by use of the cytokinesis-block micronucleus assay. Mutation research. 2008; 649: 91 -100. [PubMed] .
- 31. Gianni P, Jan KJ, Douglas MJ, Stuart PM, Tarnopolsky MA. Oxidative stress and the mitochondrial theory of aging in human skeletal muscle. Exp Gerontol. 2004; 39: 1391 -1400. [PubMed] .
- 32. Huang Y, Zhang M, Zou H, Li X, Xing M, Fang X, He J. Genetic damage and lipid peroxidation in workers occupationally exposed to organic bentonite particles. Mutation research. 2013; 751: 40 -44. [PubMed] .
- 33. Humphreys V, Martin RM, Ratcliffe B, Duthie S, Wood S, Gunnell D, Collins AR. Age-related increases in DNA repair and antioxidant protection: a comparison of the Boyd Orr Cohort of elderly subjects with a younger population sample. Age Ageing. 2007; 36: 521 -526. [PubMed] .
- 34. Irie M, Miyata M, Kasai H. Depression and possible cancer risk due to oxidative DNA damage. J Psychiatr Res. 2005; 39: 553 -560. [PubMed] .
- 35. Irie M, Tamae K, Iwamoto-Tanaka N, Kasai H. Occupational and lifestyle factors and urinary 8-hydroxydeoxyguanosine. Cancer Sci. 2005; 96: 600 -606. [PubMed] .
- 36. Kanaya S, Ikeya M, Yamamoto K, Moriya T, Furuhashi K, Sonoda M, Goto K, Ochi H. Comparison of an oxidative stress biomarker “urinary8-hydroxy-2'-deoxyguanosine,” between smokers and non-smokers. BioFactors (Oxford, England). 2004; 22: 255 -258. .
- 37. Krajcovicova-Kudlackova M, Valachovicova M, Paukova V, Dusinska M. Effects of diet and age on oxidative damage products in healthy subjects. Physiol Res. 2008; 57: 647 -651. [PubMed] .
- 38. Leng S, Dai Y, Niu Y, Pan Z, Li X, Cheng J, He F, Zheng Y. Effects of genetic polymorphisms of metabolic enzymes on cytokinesis-block micronucleus in peripheral blood lymphocyte among coke-oven workers. Cancer Epidemiol Biomarkers Prev. 2004; 13: 1631 -1639. [PubMed] .
- 39. Mladinic M, Kopjar N, Milic M, Dasovic AB, Huzak M, Zeljezic D. Genomic instability in a healthy elderly population: a pilot study of possible cytogenetic markers related to ageing. Mutagenesis. 2010; 25: 455 -462. [PubMed] .
- 40. Mota MP, Peixoto FM, Soares JF, Figueiredo PA, Leitao JC, Gaivao I, Duarte JA. Influence of aerobic fitness on age-related lymphocyte DNA damage in humans: relationship with mitochondria respiratory chain and hydrogen peroxide production. Age (Dordr). 2010; 32: 337 -346. [PubMed] .
- 41. Pal D, Banerjee S, Indra D, Mandal S, Dum A, Bhowmik A, Panda CK, Das S. Influence of regular black tea consumption on tobacco associated DNA damage and HPV prevalence in human oral mucosa. Asian Pac J Cancer Prev. 2007; 8: 263 -266. [PubMed] .
- 42. Schmid TE, Eskenazi B, Baumgartner A, Marchetti F, Young S, Weldon R, Anderson D, Wyrobek AJ. The effects of male age on sperm DNA damage in healthy non-smokers. Hum Reprod. 2007; 22: 180 -187. [PubMed] .
- 43. Soliman AS, Vulimiri SV, Kleiner HE, Shen J, Eissa S, Morad M, Taha H, Lukmanji F, Li D, Johnston DA, Lo HH, Lau S, Digiovanni J, et al. High levels of oxidative DNA damage in lymphocyte DNA of premenopausal breast cancer patients from Egypt. Int J Environ Health Res. 2004; 14: 121 -134. [PubMed] .
- 44. Surowy H, Rinckleb A, Luedeke M, Stuber M, Wecker A, Varga D, Maier C, Hoegel J, Vogel W. Heritability of baseline and induced micronucleus frequencies. Mutagenesis. 2011; 26: 111 -117. [PubMed] .
- 45. Vuyyuri SB, Ishaq M, Kuppala D, Grover P, Ahuja YR. Evaluation of micronucleus frequencies and DNA damage in glass workers exposed to arsenic. Environmental and molecular mutagenesis. 2006; 47: 562 -570. [PubMed] .
- 46. Yilmaz B, Sandal S, Ayvaci H, Tug N, Vitrinel A. Genotoxicity profiles in exfoliated human mammary cells recovered from lactating mothers in Istanbul; relationship with demographic and dietary factors. Mutation research. 2012; 749: 17 -22. [PubMed] .
- 47. Zhang XH, Zhang X, Wang XC, Jin LF, Yang ZP, Jiang CX, Chen Q, Ren XB, Cao JZ, Wang Q, Zhu YM. Chronic occupational exposure to hexavalent chromium causes DNA damage in electroplating workers. BMC Public Health. 2011; 11: 224 [PubMed] .
- 48. Weng H, Weng Z, Lu Y, Nakayama K, Morimoto K. Effects of cigarette smoking, XRCC1 genetic polymorphisms, and age on basal DNA damage in human blood mononuclear cells. Mutation research. 2009; 679: 59 -64. [PubMed] .
- 49. Blagosklonny MV. Damage-induced aging and perpetual motion. Cell Cycle. 2013; 12: 2709 -2710. [PubMed] .
- 50. Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ. Clinical research ed. 2009; 339: b2535 .
- 51. Stice E, Shaw H, Marti CN. A meta-analytic review of obesity prevention programs for children and adolescents: the skinny on interventions that work. Psychological bulletin. 2006; 132: 667 -691. [PubMed] .
- 52. Borenstein M, Hedges L, Higgins J, Rothstein H. 2008; Comprehensive meta-analysis version 2.2.048 Englewood .
- 53. Begg CB and Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics. 1994; 50: 1088 -1101. [PubMed] .
- 54. Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. Clinical research ed. 1997; 3157109: 629 -634. .
- 55. Agrawal A, Tay J, Yang GE, Agrawal S, Gupta S. Age-associated epigenetic modifications in human DNA increase its immunogenicity Aging. Albany NY 2010; 2: 93 -100. [PubMed] .


