The prognostic value of YAP1 on clinical outcomes in human cancers
Abstract
Background: As an important downstream factor in the Hippo pathway, yes-associated protein 1(YAP1) has been detected to be elevated in various cancers and demonstrated to play a role in tumor development. Therefore, we evaluated by a meta-analysis the prognostic value of YAP1 in cancer patients.
Results: Sixty-eight studies with 8631 patients were identified. The results indicated that YAP1 overexpression predicted unfavorable patient prognosis in studies with overall survival (OS) (HR=1.76, 95%CI: 1.50-2.06, p<0.001) and disease-free survival (DFS) (HR=1.39, 95%CI: 1.22-1.59, p<0.001), as well as in studies with recurrence-free survival (RFS) (HR=2.38, 95%CI: 1.73-3.27, p<0.001), and disease-specific survival (DSS) (HR=2.04, 95%CI: 1.55-2.70, p<0.001). Meanwhile, YAP1 overexpression was also observed to be significantly associated with worse OS in GEPIA (HR=1.2, p<0.001).
Conclusions: Overexpression of YAP1 showed great association with poorer prognosis in patients with various cancers, particularly liver cancer. Therefore, YAP1 might be an important prognostic marker and a novel target of cancer therapy.
Methods: We searched for potential publications in several online databases and retrieved relevant data. Overall and subgroup analyses were performed. Begg’s and Egger’s tests were used to assess publication bias. Online dataset GEPIA was used to generate the survival curves and verify the prognostic role of YAP1 in patients with tumors.
Introduction
Cancer, a health challenge worldwide, is a dominating cause of death mainly in developed countries, with high incidence and low curative rate [1]. In 2018, there were 18.1 million people diagnosed with cancer and 9.6 million people died of cancer all over the world [2]. Until now, the general treatment includes surgery, chemotherapy, radiotherapy and the immune therapy, which is found to be effective in a multitude of cancers. Despite the dramatic advancements in medical treatment, there are still millions of people suffering from it.
Cancers are derived from the excessive growth of cancer cells, which activate the mechanisms to promote the cells growth and cancer progression [3]. A variety of studies had proved the important role of cellular signaling pathways in cancer initiation and development [4]. Hippo pathway, an important growth regulatory pathway, plays an integral role in controlling the size of organs and in the renewal of stem cells by regulating cell proliferation and apoptosis [4, 5]. The components of the Hippo pathway, including Warts, Salvador, mob-as-tumor suppressor, and Hippo, were first discovered in Drosophila by using genetic screens to search the tumor suppressor genes [6–15]. YAP1 is a transcription co-activator and main downstream effector of the Hippo pathway. Previously, YAP1 was thought to be an apoptosis-promoting protein. However, Huang et al. suggested that YAP was likely an oncogene rather than an apoptosis-promoting protein [16]. Meanwhile, some studies put forward that the function of YAP1 was different according to its location of expression. Nuclear YAP1 can promote cell growth and restrain apoptosis, whereas cytoplasmic YAP1 participate in cell apoptosis [16, 17]. Subsequently, many studies demonstrated that YAP1 was a negative prognostic marker in many tumors and promoted epithelial-mesenchymal transition, which indicated that YAP1 might induce cancer metastasis and invasion [18, 19]. In contrast, many researchers held opposite views. Matallanas et al. [20] reported that alleviating YAP cytoplasmic retention was related to p73-mediated apoptosis, indicating that YAP might play a role in tumor suppression. Consistent with it, Sun et al. [21] considered the cytoplasmic YAP expression was related with favorable prognosis.
Therefore, it is critical to investigate the relationship between YAP1 and cancer. The first meta-analysis regarding YAP1 was published three years ago, and reported that the positive expression of YAP1 was associated with poorer prognosis in various cancers [22]. In recent years, an increasing number of studies have been published which explored the correlation between YAP1 and patient prognosis with cancers. In this study, we gathered more studies to assess the prognostic value of YAP1 overexpression in patients with cancer.
Results
Characteristics of studies included in the meta-analysis
In total, 3208 studies were searched initially, and 101 were considered potentially relevant articles. Among them, 33 studies were excluded (6 without full text, 6 with unavailable data, 4 in which we were unable to extract relevant data, 13 with data from databases, and 4 with questionable data). In the process of selecting literature, some studies were excluded due to the questionable data, such as the studies of Han et al., Fan Ye et al., and Wang et al. [23–25]. The confidence interval of the study conducted by Salcedo Allende et al. [26] was too narrow for the studied to be included in our study. Considering the same patients were possibly included indifferent public databases, we excluded the 13 studies that used results from public databases. Finally, 69 studies [18, 21, 27–92] that met our criteria were selected in our meta-analysis, and their characteristics were summarized in Supplementary Table 3. The flow diagram of the literature selection is shown in Figure 1. Besides, a scoring summary of all high-quality studies considered for this analysis were presented in Supplementary Table 2.
Figure 1. The flow diagram of studies selection in this meta-analysis.
Of these 68 studies, 53 evaluated the relationship between YAP1 and OS, 19 for DFS, 7 for progression-free survival (PFS), 8 for RFS, and 5 for DSS. Collectively, these 68 included studies contained data for 8631 patients, from 12 countries, who were diagnosed with 18 different types of cancer, such as liver cancer, gastric cancer, and breast cancer, and so on.
Meta-analysis results
There was obvious heterogeneity of OS in 53 studies (I2=73.7%, p<0.001) and a random-effects model was applied. The result indicated that overexpression of YAP1 would lead to poorer OS (HR=1.76, 95%CI: 1.50-2.06, p<0.001) (Figure 2).
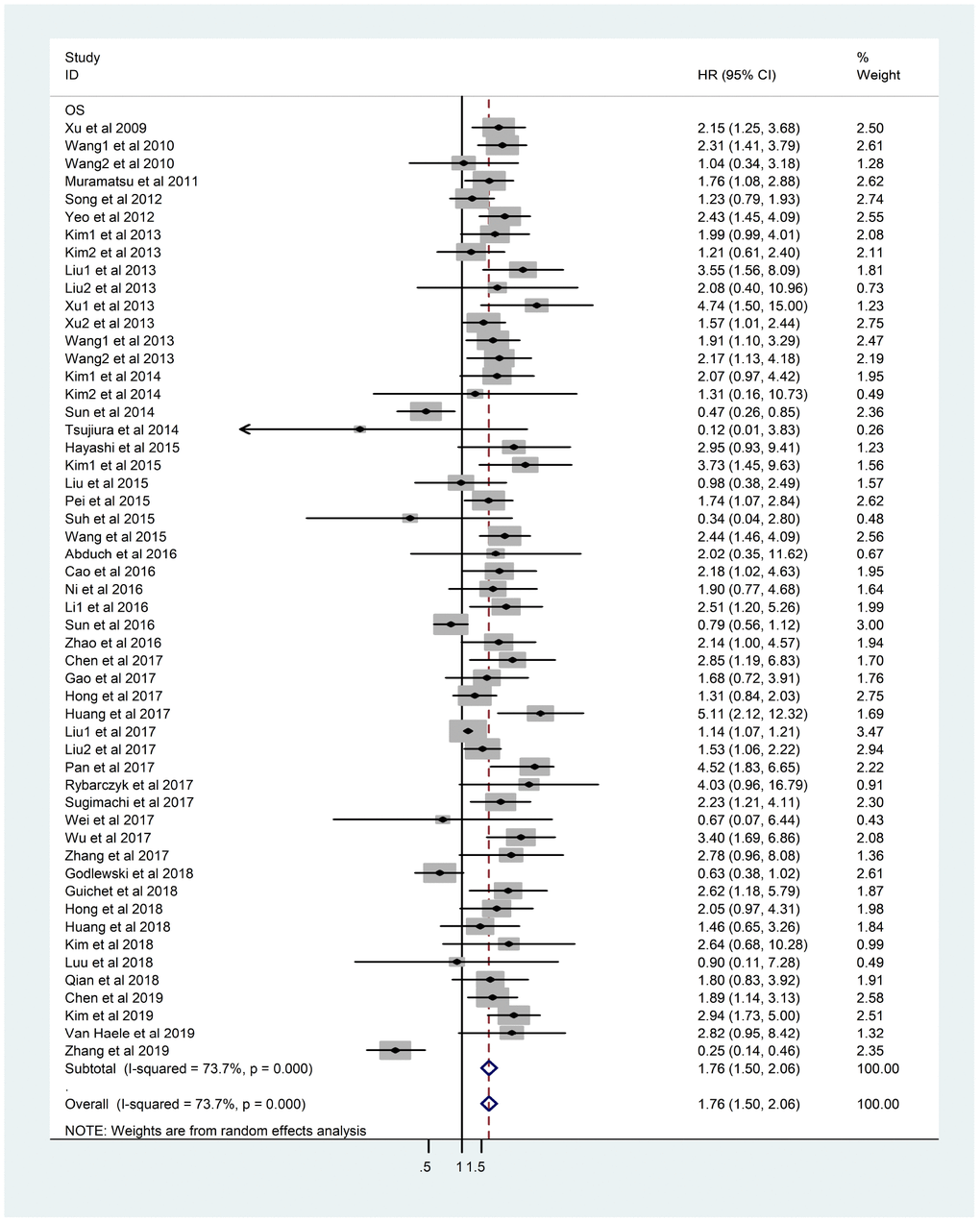
Figure 2. Forest plot of HR for association between YAP1 overexpression and OS. Note: Weights are from random-effects analysis. Abbreviations: CI confidence interval; HR hazard ratio; OS overall survival.
As the heterogeneity was not significant, the fixed-effects model was applied in 19 DFS studies comprising 3013 patients (I2=30.7%, p=0.10). The results showed that the overexpression of YAP1 was related to poorer DFS (HR=1.39, 95%CI: 1.22-1.59, p<0.001) (Figure 3).
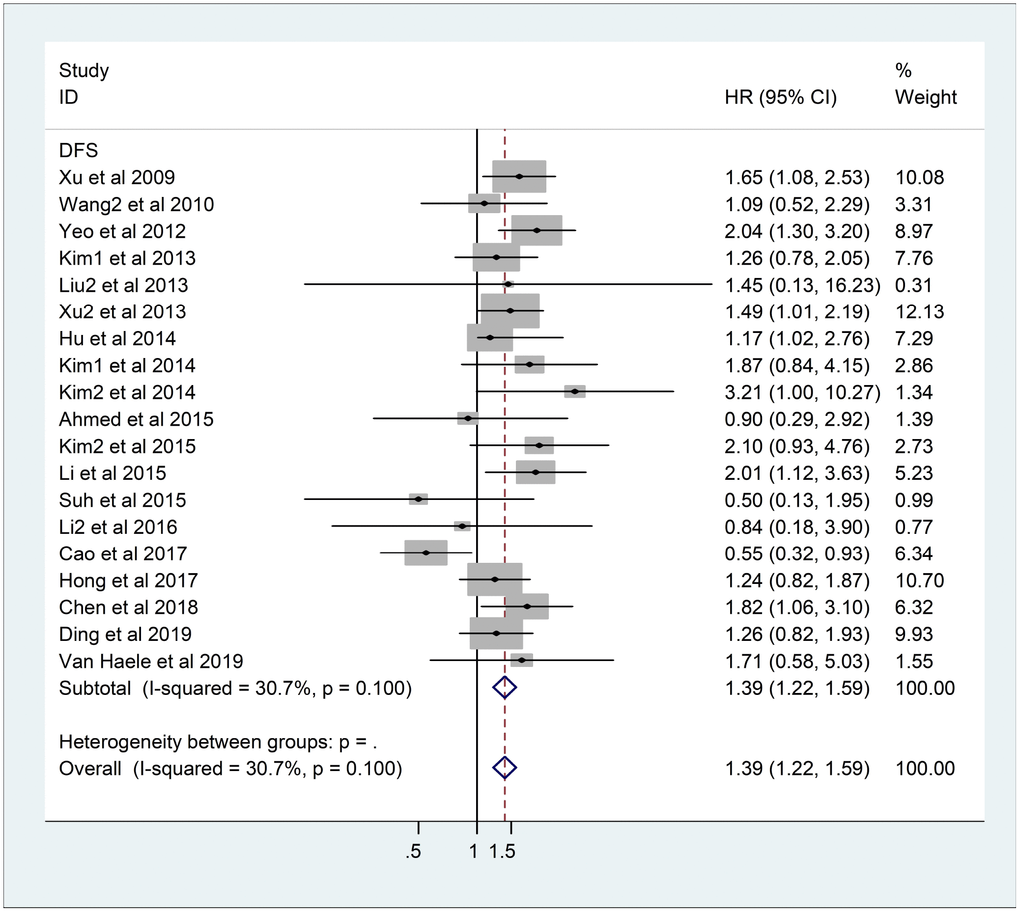
Figure 3. Forest plot of HR for association between YAP1 overexpression and DFS. Note: Weights are from fixed-effects analysis. Abbreviations: CI confidence interval; DFS disease-free survival; HR hazard ratio.
As shown in Table 1, 7 studies with 652 patients, 8 studies with 954 patients, and 5 studies with 500 patients reported PFS, RFS, and DSS, respectively. Heterogeneity was found in PFS and RFS (PFS: I2=69.2%, p=0.003; RFS: I2=55.6%, p=0.027), so we used a random-effects model to evaluate the pooled HR and its 95% CI (PFS: HR=1.62, 95%CI: 0.96-2.73, p=0.069; RFS: HR=2.38, 95%CI: 1.73-3.27, p<0.001); for DSS, pooled HR (HR= 2.04, 95%CI: 1.55-2.70, p<0.001), for insignificant heterogeneity (I2=0%, p=0.82) was calculated using a fixed-effects model. The results suggested that YAP1 overexpression would predict poorer RFS and DSS, but not PFS (Supplementary Figure 1).
Table 1. Subgroup analysis of meta-analysis.
| Analysis | No of patients | No of studies | HR(95%CI) | P-value | Heterogeneity |
| I2(%) | P-value |
| OS | 6771 | 53 | 1.76(1.50-2.06) | <0.001 | 73.70% | <0.001 |
| Ethnicity |
| Asian | 6271 | 46 | 1.78(1.51-2.10) | <0.001 | 74.90% | <0.001 |
| Non-Asian | 500 | 7 | 1.51(0.69-3.30) | 0.304 | 66.70% | 0.006 |
| Tumor types |
| Liver cancer | 1273 | 11 | 1.93(1.62-2.31) | <0.001 | 0.00% | 0.707 |
| Lung cancer | 546 | 5 | 1.54(0.85-2.82) | 0.156 | 79.90% | 0.001 |
| Esophageal cancer | 404 | 3 | 2.12(1.51-2.97) | <0.001 | 0.00% | 0.590 |
| Gastric cancer | 1474 | 8 | 1.58(1.00-2.51) | 0.051 | 77.30% | 0.001 |
| Colorectal cancer | 838 | 7 | 1.48(0.75-2.90) | 0.256 | 86.10% | <0.001 |
| Bladder cancer | 268 | 2 | 3.20(1.76-5.83) | <0.001 | 0.00% | 0.719 |
| Breast cancer | 983 | 3 | 2.48(1.40-4.38) | 0.002 | 0.00% | 0.527 |
| Ovarian cancer | 236 | 3 | 1.49(0.89-2.49) | 0.132 | 0.00% | 0.571 |
| Renal cancer | 140 | 2 | 1.40(0.23-8.51) | 0.712 | 82.70% | 0.016 |
| Others | 609 | 9 | 1.69(0.975-2.93) | 0.062 | 68.20% | 0.001 |
| Method |
| IHC | 6559 | 50 | 1.73(1.47-2.03) | <0.001 | 74.60% | <0.001 |
| PCR | 212 | 3 | 3.01(1.35-6.69) | 0.007 | 0.00% | 0.835 |
| Staining location |
| YAP1 expression | 4131 | 34 | 1.90(1.54-2.33) | <0.001 | 76.90% | <0.001 |
| Nuclear YAP1 expression | 2516 | 19 | 1.63(1.29-2.07) | <0.001 | 51.20% | 0.005 |
| Cytoplasmic YAP1 expression | 535 | 5 | 1.55(0.71-3.40) | 0.275 | 81.00% | <0.001 |
| HR estimation |
| Univariate analysis | 2774 | 31 | 1.78(1.45-2.17) | <0.001 | 65.80% | <0.001 |
| Multivariate analysis | 3997 | 22 | 1.77(1.32-2.37) | <0.001 | 78.90% | <0.001 |
| DFS | 3013 | 19 | 1.39(1.22-1.59) | <0.001 | 30.70% | 0.100 |
| Ethnicity |
| China | 1295 | 10 | 1.32(1.11-1.57) | 0.001 | 43.90% | 0.066 |
| Korea | 1605 | 7 | 1.54(1.22-1.93) | <0.001 | 26.50% | 0.226 |
| Others | 113 | 2 | 1.26(0.57-2.77) | 0.562 | 0.00% | 0.427 |
| Tumor types |
| Liver cancer | 856 | 6 | 1.59(1.29-1.95) | <0.001 | 0.00% | 0.864 |
| Gastric cancer | 573 | 4 | 1.15(0.86-1.52) | 0.352 | 0.00% | 0.658 |
| Breast cancer | 1155 | 4 | 1.09(0.81-1.47) | 0.560 | 74.70% | 0.008 |
| Others | 429 | 5 | 1.79(1.25-2.56) | 0.001 | 0.00% | 0.589 |
| Staining location |
| YAP1 expression | 1818 | 14 | 1.32(1.13-1.54) | 0.001 | 37.90% | 0.074 |
| Nuclear YAP1 expression | 1324 | 7 | 1.67(1.32-2.12) | <0.001 | 16.90% | 0.301 |
| HR estimation |
| Univariate analysis | 1188 | 11 | 1.40(1.17-1.69) | <0.001 | 0.00% | 0.537 |
| Multivariate analysis | 1825 | 8 | 1.38(1.13-1.68) | 0.001 | 58.90% | 0.017 |
| PFS | 652 | 6 | 1.62(0.96-2.73) | 0.069 | 69.20% | 0.003 |
| RFS | 954 | 8 | 2.38(1.73-3.27) | <0.001 | 55.60% | 0.027 |
| DSS | 500 | 5 | 2.04(1.55-2.70) | <0.001 | 0.00% | 0.820 |
| Abbreviations: CI confidence interval; DFS disease-free survival; DSS disease-specific survival; HR hazard ratio; IHC immunohistochemistry; M multivariate; NA no applicable; OS overall survival; PFS progression-free survival; RFS recurrence-free survival; U univariate. |
Subgroup analysis of OS
To explore the source of heterogeneity, we further carried out subgroup analysis according to four possible factors (ethnicity, tumor type, method, staining location and analysis types of studies), summarized in Table 1. The subgroup analysis by ethnicity suggested that overexpression of YAP1 was a negative prognosticator in Asian patients (HR=1.78, 95%CI: 1.51-2.10, p<0.001), but not in non-Asian patients. In the tumor type subgroup, significant associations were found in liver cancer (HR=1.93, 95%CI: 1.62-2.31, p<0.001), esophageal cancer (HR=2.12, 95%CI: 1.51-2.97, p<0.001), breast cancer (HR=2.48, 95%CI: 1.40-4.38, p=0.002), and bladder cancer (HR=3.20, 95%CI: 1.76-5.83, p<0.001). When stratified by method, IHC (HR=1.73, 95%CI: 1.47-2.03, p<0.001) and PCR (HR=3.01, 95%CI: 1.35-6.69, p=0.007), both showed a significant relationship with poorer OS. When analyzed by YAP1 staining location, total YAP1 expression and nuclear YAP1 expression showed great significance with dismal prognosis in tumor patients (YAP1 expression: HR= 1.90, 95%CI: 1.54-2.33, p<0.001; nuclear YAP1 expression: HR=1.63, 95%CI: 1.29-2.07, p<0.001). Regarding the type of analysis, univariate analysis (HR=1.78, 95%CI: 1.45-2.17, p<0.001) indicated that overexpression of YAP1 was significantly related with elevated OS, as did the multivariate analysis (HR=1.77, 95%CI: 1.32-2.37, p<0.001).
Subgroup analysis of DFS
Based on ethnicity, tumor type, staining location, and analysis type, subgroup analysis was also performed in the studies reporting DFS. YAP1 overexpression in Korean studies (HR=1.54, 95%CI: 1.22-1.93, p<0.001), as well as in Chinese (HR=1.32, 95%CI: 1.11-1.57, p=0.001), was associated with poor DFS. In the subgroup analysis by tumor type, a significant relationship was found between patients with liver cancer (HR=1.59, 95%CI: 1.29-1.95, p<0.001) and DFS. Regarding staining location, subgroup analysis suggested that both total YAP1 expression (HR=1.32, 95%CI: 1.13-1.54, p=0.001) and nuclear YAP1 expression (HR=1.67, 95%CI: 1.32-2.12, p<0.001) were predictors of poorer DFS. According to the subgroup result of analysis type, univariate analysis (HR=1.40, 95%CI: 1.17-1.69, p<0.001) and multivariate analysis (HR=1.38, 95%CI: 1.13-1.68, p=0.001) showed significant associations with poorer DFS.
Sensitivity analysis
Each study was omitted consecutively to explore its impact on the entire result. As shown in Figure 4, no individual study substantially altered the pooled HR of studies reporting OS, which implied the result was stable. Same result was observed in studies reporting DFS (Supplementary Figure 2).
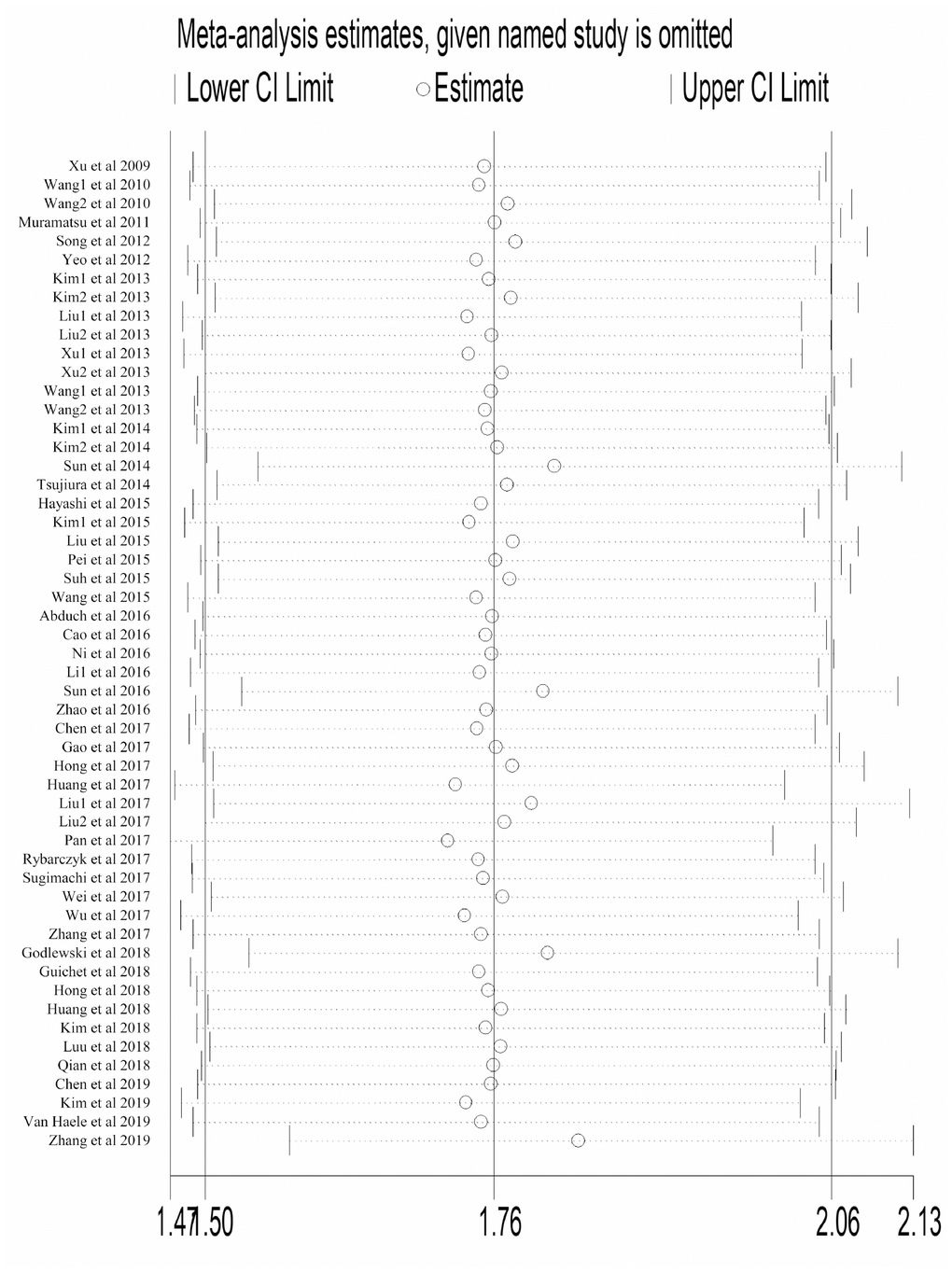
Figure 4. Sensitivity analysis to evaluate the influence of every study reporting OS in our meta-analysis. Abbreviation: OS overall survival; CI confidence interval.
Publication bias
We generated the funnel plot via Begg’s test and investigated the potential publication bias using Egger’s test. As shown in the funnel plot of OS, the points which represented the results of each study distributed asymmetrically on both sides of the axis (Figure 5). However, publication bias was not found according to Begg’s test (p=0.395), but there was significant bias per Egger’s test (p<0.05). Therefore, considering the efficiency of two tests, we believed that there was publication bias among studies reporting OS. Publication bias was not detected in studies reporting DFS (Begg’s test: p=0.675; Egger’s test: p=0.812) (Supplementary Figure 3).
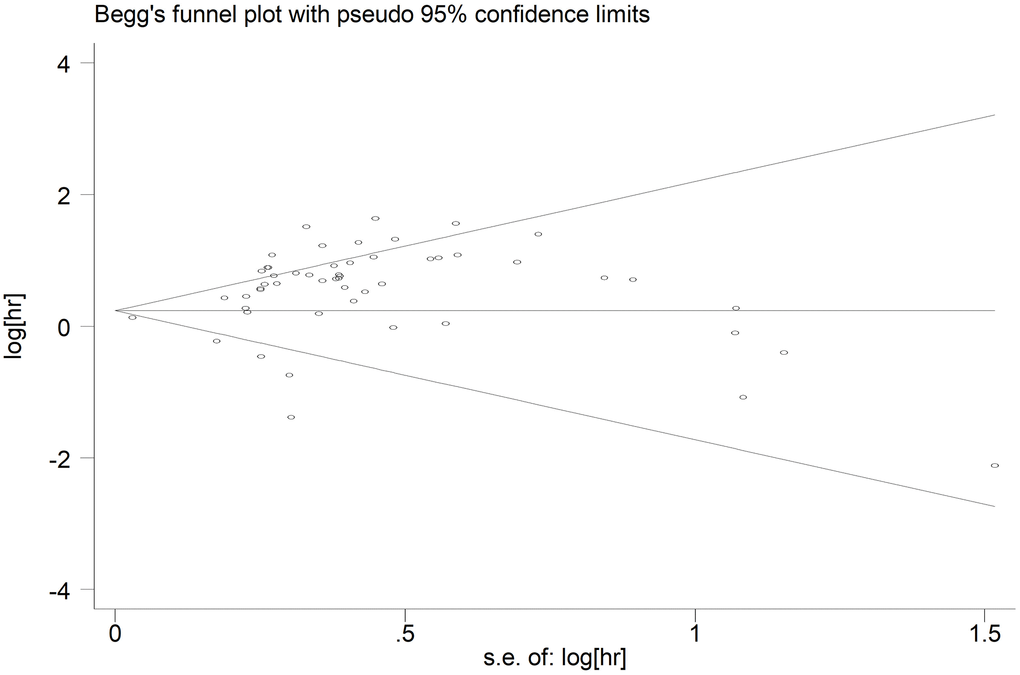
Figure 5. Begg’s funnel plot of publication bias for included studies reporting OS. Abbreviations: HR hazard ratio; OS overall analysis; SE standard error.
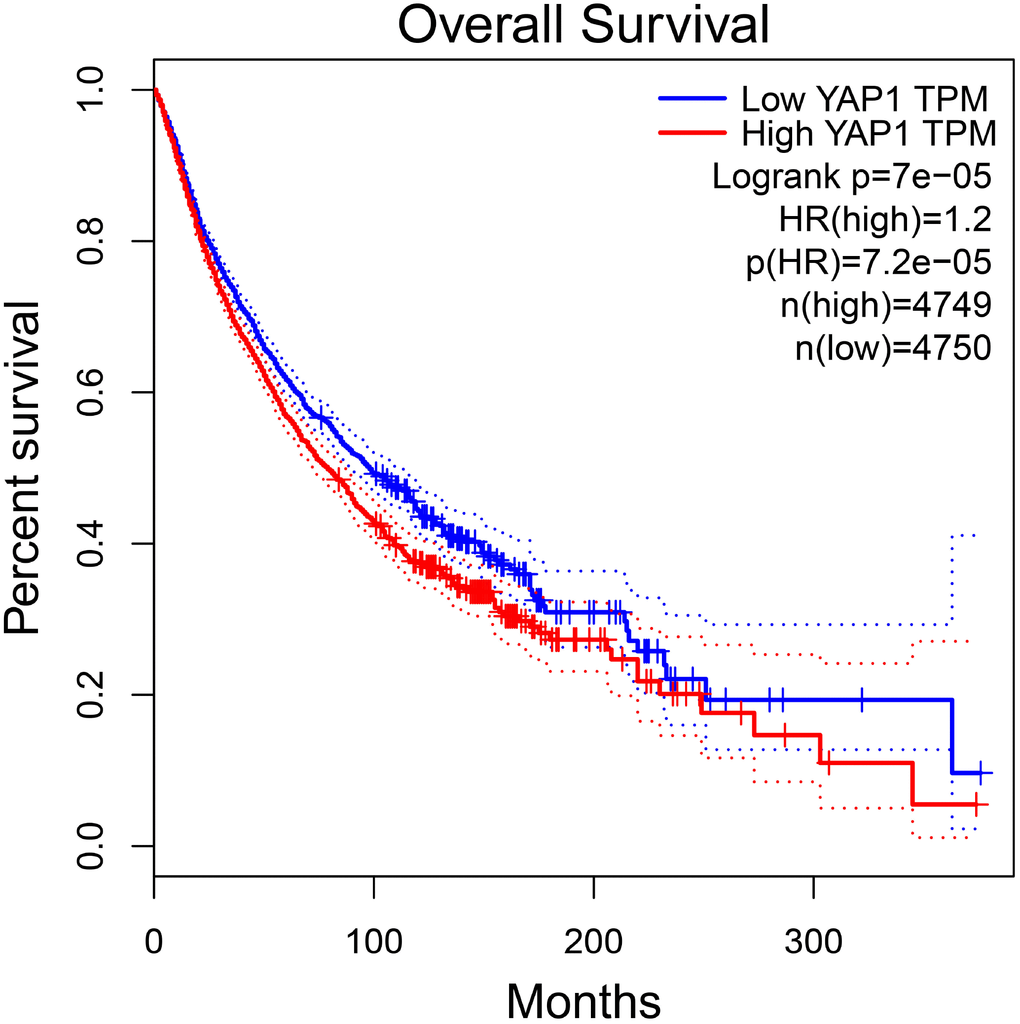
Figure 6. The prognostic value of YAP1 in total patients with tumors in GEPIA. Abbreviations: HR hazard ratio.
Prognostic analysis in GEPIA
To further investigate the association between YAP1 expression and patients’ prognosis in various cancers, we performed the survival analysis using GEPIA. The results showed there was a significant relation between YAP1 overexpression and worse OS in total patients with tumors (HR=1.2, p<0.001) (Figure 6). However, no significance was observed between YAP1 overexpression and DFS in total patients with tumors. Furthermore, we found YAP1 overexpression might indicate unfavorable OS and DFS in patients with glioma, as well as worse OS in esophageal carcinoma patients and better DFS in bladder urothelial carcinoma patients. The results were described in Table 2.
Table 2. Association between YAP1 and cancer patients’ prognosis in GEPIA.
| Tumor type | No of YAP1 expression | OS | DFS |
| High | Low | p | HR(high) | p(HR) | p | HR(high) | p(HR) |
| Total tumor | 4746 | 4746 | 0.00007 | 1.2 | 0.000072 | 0.21 | 1 | 0.21 |
| Liver cancer | 199 | 199 | 0.2 | 1.2 | 0.2 | 0.67 | 1.1 | 0.67 |
| Lung cancer | 481 | 481 | 0.39 | 1.1 | 0.39 | 0.67 | 1.1 | 0.67 |
| Esophageal carcinoma | 91 | 91 | 0.012 | 0.56 | 0.013 | 0.96 | 1 | 0.95 |
| Stomach adenocarcinoma | 192 | 192 | 0.92 | 0.98 | 0.92 | 0.35 | 0.83 | 0.34 |
| Colorectal cancer | 181 | 180 | 0.095 | 0.69 | 0.096 | 0.79 | 1.1 | 0.79 |
| Bladder Urothelial Carcinoma | 201 | 201 | 0.14 | 1.2 | 0.14 | 0.036 | 1.4 | 0.037 |
| Renal cancer | 432 | 432 | 0.69 | 0.95 | 0.69 | 0.94 | 0.99 | 0.94 |
| Breast invasive carcinoma | 534 | 535 | 0.17 | 1.3 | 0.17 | 0.48 | 0.88 | 0.49 |
| Uterine cancer | 261 | 261 | 0.31 | 1.2 | 0.3 | 0.92 | 0.66 | 0.65 |
| Ovarian serous cystadenocarcinoma | 212 | 212 | 0.56 | 1.1 | 0.56 | 0.62 | 1.1 | 0.61 |
| Glioma | 338 | 338 | 0.000002 | 1.8 | 0.0000027 | 0.00016 | 1.6 | 0.00018 |
| Others | 1624 | 1624 | 0.0012 | 1.3 | 0.0012 | 0.069 | 1.1 | 0.069 |
| Abbreviations: DFS disease-free survival; HR hazard ratio; OS overall survival. |
Discussion
The mechanism involved in the development of cancer is complicated and is generally thought to be related to the dysfunction of cellular signaling pathways. Previous studies suggested the cellular signaling pathways could regulate the growth factors and expression of some vital genes that are associated with cell proliferation, migration, metabolism and junction between cells and so forth [93–95]. Hippo/YAP1 pathway was firstly discovered through gene screen of tumor suppressors in Drosophila. However, with the identifications of Hippo pathway components, it was demonstrated that Hippo/YAP1 pathway could control the organ size by regulating the balance between cell proliferation and apoptosis in mammalian [96]. Therefore, the dysfunction of Hippo/YAP1 pathway could imbalance the regulation, which could cause the cancer initiation. YAP1, as the main effector of Hippo pathway, plays a critical role in the development of cancers, standing at the cross point of different signaling pathways, mediated by upstream effectors and then regulating targets in collaboration with other transcription factors. Several important signaling pathways are known to participate in the activity of the Hippo/YAP1 pathway, these include TGF-β/SMAD, Wnt-β-catenin, Jak/Stat, Notch signaling pathway [97–100]. When crosstalking with the Sonic Hedgehog pathway, YAP1 can behave like an oncogene promoting cell proliferation and inhibiting cell differentiation [101]. However, in other studies, where p73-mediated cell apoptosis was recognized, YAP1 functioned as a tumor suppressor in response to DNA damage [102, 103].
In our study, overall and subgroup analyses were conducted to provide evidence of the association between the overexpression of YAP1 and patients’ prognosis. The results showed that overexpression of YAP1 predicted poorer OS, DFS, RFS, and DSS. The sensitivity analysis showed that no single study changed the pooled results significantly, which indicated the results of sensitivity analysis were stable. Subgroup analysis was conducted by ethnicity, tumor type, method, staining location, and HR estimation analysis type. According to the results of the subgroup analysis, there were no clear relations between YAP1 with OS in non-Asians, colorectal cancer, lung cancer, renal cancer, and ovarian cancer.
To clarify it further, we investigated the association between YAP1 expression and cancer patients’ prognosis in the online database, GEPIA. The results showed YAP1 expression was observed to be significantly related with worse OS, but not with DFS. Additionally, the same correlations were found between YAP1 expression and unfavorable OS and DFS in patients with glioma, as well as shorter OS in esophageal carcinoma patients and better DFS in bladder urothelial carcinoma patients. The differences between this meta-analysis and the online dataset might come from the following reasons. First of all, the detecting methods are thought to be the main reason for the differences. In most of the studies included in our meta-analysis, IHC was used to detect the expression of YAP1 protein, whereas the data of the online dataset were from the results of RNA sequencing. What’s more, in some specific cancer types, the number of patients that included in meta-analysis was much larger than the dataset. For example, although we have summed up the data of cholangio carcinoma and liver hepatocellular carcinoma, the total number of patients with liver cancer studied in the meta-analysis (1629), far exceed the number of the liver cancer patients represented in the online dataset (398). In addition, the cancer types are different. For instance, the breast cancer patients included in our meta-analysis represented various breast cancer subtypes, while the data in GEPIA included data for breast invasive carcinoma (BRCA) only. Therefore, while GEPIA provides valuable insights into gene-specific cancer survival outcomes, it is still being developed, and results obtained from the robust meta-analysis remain more convincing.
In the present study, we identified 14 studies that reported the relationship between prognosis of patients with liver cancer and YAP1 overexpression. Our results suggested that YAP1 overexpression predicted poorer OS and DFS. YAP1 was reported to be amplified and overexpressed in many cancers, and patients were suggested to receive further treatment, such as chemotherapy and hepatectomy. Although tumors can be removed by hepatectomy, rapid repair of the liver is crucial after partial hepatectomy, in which YAP1 increases and participates in the promotion of cell proliferation [104, 105]. The hedgehog pathway might be involved in this process. After blocking the hedgehog pathway in hepatic stellate cells, nuclear accumulation of YAP1 in neighboring hepatocytes did not occur [106]. Combining all these results of other studies and ours, we believe that YAP1 might induce unfavorable prognosis and be an important target in the treatment of liver cancer.
It is widely accepted that YAP1 is phosphorylated and accumulates in the cytoplasm such that it loses its ability to activate genes that promote cell invasion and proliferation and suppresses cell apoptosis once the Hippo pathway is activated [17, 107]. Therefore, to clarify the mechanisms of YAP1 functioning in cancer, we explored the association between staining location of YAP1 and cancer patients’ prognosis. We found total YAP1 expression and nuclear YAP1 expression indicated the unfavorable prognosis, both with OS and DFS, whereas the YAP1 expressed in cytoplasm was not related with cancer patients’ prognosis. Meanwhile, lots of studies reported that YAP1 expressed stronger in nuclei, while it was weaker in cytoplasm. The results might suggest nuclear YAP1 plays an important role in cancer, but it still needs more studies to be demonstrated furthermore.
Previously, Sun et al. [22] performed a meta-analysis, including 21 studies with 2983 patients, and reported that overexpression of YAP1 was associated with worse OS and DFS. Same results were demonstrated in the previous meta-analysis by Zhang et al. in gastrointestinal cancer. Compared with these studies, our study had some further research results. Firstly, our meta-analysis included a larger number of studies which were the latest. Secondly, in this study, not only OS and DFS, but RFS, PFS, and DSS were analyzed to understand the issue more comprehensively. Thirdly, having benefited from more studies, our study further explored the prognostic value of YAP1 expression in different specific cancers. Fourthly, we tried our best to illustrate the potential mechanism of YAP1 functioned in cancer development through subgroup analysis of YAP1 staining location.
Despite our efforts to make an accurate and comprehensive analysis, a few limitations inevitably existed in our study. First of all, some studies did not report clinical data; we had to extract the HR from the Kaplan-Meier survival curves indirectly, which might influence the accuracy of the original data. Secondly, publication bias was observed in our study among the studies reporting OS and we thought it was associated with the studies we included. Most of the studies on the relation between YAP1 and prognosis were based on Asian patients. Meanwhile, the bias might also be due to the non-publishing of studies with negative results. More studies are needed to resolve this limitation. Thirdly, significant heterogeneity between studies with OS, PFS, and RFS were detected. However, we failed to identify the source of heterogeneity by subgroup analysis. Then we minimized the effect by applying a random-effects model. Possibly, it was due to the differences in the methods of surgery and treatment, and baseline YAP1 expression level. Additionally, owing to the lack of clinical data, we could not confirm their contribution to heterogeneity.
Conclusions
Overall, our study provides evidence that overexpression of YAP1 is significantly associated with the progression and recurrence of tumors and it might be a useful prognosticator in various cancers. However, future prospective studies with more complete and available clinical information are expected and required to confirm the results.
Methods
Literature search strategy
Studies related to YAP1 were searched through PubMed, EMBASE, Web of Science, WANFANG, and CNKI. The main keywords used in the search were as follows: “Yes-associated protein 1”, “YAP1”, “Hippo”, “Neoplasms”, “Neoplasia”, “Tumor”, “Cancer”, “Malignancy”, “Prognosis”, “Survival” and “Metastasis”. The search strategy for PubMed is shown in Supplementary Table 1.
Study selection criteria
Studies were included when they met the following criteria: (1) studies explored the association between YAP1 expression and patients’ prognosis; (2) studies declared the number and information of patients; (3) studies measured positive or high YAP1 expression in patients with any carcinoma by immunohistochemistry and polymerase chain reaction; (4) the endpoints were listed which are OS, RFS, DFS, PFS, and DSS; (5) the HRs with their 95% CIs or Kaplan-Meier survival curves were provided; (6) as for the overlapping data, the latest and most complete studies were included.
Studies were excluded if they: (1) were published meta-analyses, letters, abstracts, reviews or case reports; (2) did not provide survival information; (3) the data were not related to humans; (4) used data from public databases; or (5) were questionable data or the full text was not found.
Data extraction
The information extracted from the included studies were: first author, publication year, patient source, number of patients, tumor type, specimen, methods, YAP1 expression, staining location, median follow-up time, prognostic outcome, HR estimation (when both univariate and multivariate analysis were provided, we included the outcomes of the multivariate analysis). We applied the HR with its 95% CI from the text directly or calculated the HR using Engauge Digitizer 4.0 by Tierney et al. through Kaplan-Meier survival curve analysis. The literature and data were selected and abstracted by two authors, and divergence views were resolved in a group meeting.
Quality assessment
The NOS was used to assess the quality of the literature. Two authors of our group scored all eligible studies independently, and all discrepancies were discussed to arrive at a conclusion. The scores of studies included ranged from 6 to 8, which implied high quality.
Prognostic analysis in GEPIA
Gene Expression Profiling Interactive Analysis (GEPIA) is a new online dataset, including 9736 tumors and 8587 normal samples from The Cancer Genome Atlas (TCGA) and Genotype-expression (GTEx) projects. In this study, for further verifying the association between YAP1 and patients’ prognosis with tumors, the GEPIA was used to perform the survival analysis.
Statistical analysis
HRs and their 95% CIs were used to assess the relation between YAP1 expression and prognosis of patients with carcinoma. We evaluated the heterogeneity using the Q test and I2 test. Significant heterogeneity was defined by a p value <0.1 or I2 value >50%, based on which we applied a random-effects model; if no significant heterogeneity was found, we used a fixed-effects model. Subgroup analysis was conducted according to ethnicity, tumor type, staining location, and analysis type. In the sensitivity analysis, to estimate the impact of each study on our meta-analysis, we omitted individual studies consecutively. Begg’s test and Egger’s test were performed to assess the potential publication bias. If the funnel plots showed the points distributed asymmetrically on both sides of the axis, the publish bias was regarded to be significant. All statistical tests were bi-directional and a p-value less than 0.05 indicated statistical significance. Data analysis was performed using STATA 12.0 (Stata Corporation, College Station, Texas, USA).
Abbreviations
CI: confidence interval;
DFS: disease-free survival;
DSS: disease-specific survival;
HR: hazard ratio;
IHC: immunohistochemistry;
NOS: Newcastle-Ottawa Scale;
OS: overall survival;
PCR: polymerase chain reaction;
PFS: progression-free survival;
RFS: recurrence-free survival;
YAP1: yes-associated protein 1.
Author Contributions
YW, YS H and ZJ D conceived and designed the study. XP, YS H, DYJ and HQ took responsibility for preparation of data and manuscript. TT, LH Z and NL analyzed the data. MW, KL and CD contributed materials and analysis tools. ZJ D was involved in manuscript revision. All authors read and approved the final manuscript.
Acknowledgments
We would like to thank to all researchers and participants for their contributions.
Conflicts of Interest
The authors declare that they have no conflicts of interests to disclose.
Funding
This work was supported by the National Natural Science Foundation of China (No.81471670), and the Key research and development plan, Shaanxi Province, China (2017ZDXM-SF-066), Science and technology branch project of Xinjiang Uygur Autonomous Region, People’s Republic of China (2017E0262).
References
-
1.
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015; 65:87–108. https://doi.org/10.3322/caac.21262 [PubMed]
-
2.
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018; 68:394–424. https://doi.org/10.3322/caac.21492 [PubMed]
-
3.
Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011; 144:646–74. https://doi.org/10.1016/j.cell.2011.02.013 [PubMed]
-
4.
Zhao B, Tumaneng K, Guan KL. The Hippo pathway in organ size control, tissue regeneration and stem cell self-renewal. Nat Cell Biol. 2011; 13:877–83. https://doi.org/10.1038/ncb2303 [PubMed]
-
5.
Pan D. Hippo signaling in organ size control. Genes Dev. 2007; 21:886–97. https://doi.org/10.1101/gad.1536007 [PubMed]
-
6.
Harvey KF, Pfleger CM, Hariharan IK. The Drosophila Mst ortholog, hippo, restricts growth and cell proliferation and promotes apoptosis. Cell. 2003; 114:457–67. https://doi.org/10.1016/S0092-8674(03)00557-9 [PubMed]
-
7.
Jia J, Zhang W, Wang B, Trinko R, Jiang J. The Drosophila Ste20 family kinase dMST functions as a tumor suppressor by restricting cell proliferation and promoting apoptosis. Genes Dev. 2003; 17:2514–19. https://doi.org/10.1101/gad.1134003 [PubMed]
-
8.
Justice RW, Zilian O, Woods DF, Noll M, Bryant PJ. The Drosophila tumor suppressor gene warts encodes a homolog of human myotonic dystrophy kinase and is required for the control of cell shape and proliferation. Genes Dev. 1995; 9:534–46. https://doi.org/10.1101/gad.9.5.534 [PubMed]
-
9.
Kango-Singh M, Nolo R, Tao C, Verstreken P, Hiesinger PR, Bellen HJ, Halder G. Shar-pei mediates cell proliferation arrest during imaginal disc growth in Drosophila. Development. 2002; 129:5719–30. https://doi.org/10.1242/dev.00168 [PubMed]
-
10.
Pantalacci S, Tapon N, Léopold P. The Salvador partner Hippo promotes apoptosis and cell-cycle exit in Drosophila. Nat Cell Biol. 2003; 5:921–27. https://doi.org/10.1038/ncb1051 [PubMed]
-
11.
Praskova M, Xia F, Avruch J. MOBKL1A/MOBKL1B phosphorylation by MST1 and MST2 inhibits cell proliferation. Curr Biol. 2008; 18:311–21. https://doi.org/10.1016/j.cub.2008.02.006 [PubMed]
-
12.
Tapon N, Harvey KF, Bell DW, Wahrer DC, Schiripo TA, Haber D, Hariharan IK. salvador Promotes both cell cycle exit and apoptosis in Drosophila and is mutated in human cancer cell lines. Cell. 2002; 110:467–78. https://doi.org/10.1016/S0092-8674(02)00824-3 [PubMed]
-
13.
Udan RS, Kango-Singh M, Nolo R, Tao C, Halder G. Hippo promotes proliferation arrest and apoptosis in the Salvador/Warts pathway. Nat Cell Biol. 2003; 5:914–20. https://doi.org/10.1038/ncb1050 [PubMed]
-
14.
Wu S, Huang J, Dong J, Pan D. hippo encodes a Ste-20 family protein kinase that restricts cell proliferation and promotes apoptosis in conjunction with salvador and warts. Cell. 2003; 114:445–56. https://doi.org/10.1016/S0092-8674(03)00549-X [PubMed]
-
15.
Xu T, Wang W, Zhang S, Stewart RA, Yu W. Identifying tumor suppressors in genetic mosaics: the Drosophila lats gene encodes a putative protein kinase. Development. 1995; 121:1053–63. [PubMed]
-
16.
Huang J, Wu S, Barrera J, Matthews K, Pan D. The Hippo signaling pathway coordinately regulates cell proliferation and apoptosis by inactivating Yorkie, the Drosophila Homolog of YAP. Cell. 2005; 122:421–34. https://doi.org/10.1016/j.cell.2005.06.007 [PubMed]
-
17.
Dong J, Feldmann G, Huang J, Wu S, Zhang N, Comerford SA, Gayyed MF, Anders RA, Maitra A, Pan D. Elucidation of a universal size-control mechanism in Drosophila and mammals. Cell. 2007; 130:1120–33. https://doi.org/10.1016/j.cell.2007.07.019 [PubMed]
-
18.
Muramatsu T, Imoto I, Matsui T, Kozaki K, Haruki S, Sudol M, Shimada Y, Tsuda H, Kawano T, Inazawa J. YAP is a candidate oncogene for esophageal squamous cell carcinoma. Carcinogenesis. 2011; 32:389–98. https://doi.org/10.1093/carcin/bgq254 [PubMed]
-
19.
Shao DD, Xue W, Krall EB, Bhutkar A, Piccioni F, Wang X, Schinzel AC, Sood S, Rosenbluh J, Kim JW, Zwang Y, Roberts TM, Root DE, et al. KRAS and YAP1 converge to regulate EMT and tumor survival. Cell. 2014; 158:171–84. https://doi.org/10.1016/j.cell.2014.06.004 [PubMed]
-
20.
Matallanas D, Romano D, Yee K, Meissl K, Kucerova L, Piazzolla D, Baccarini M, Vass JK, Kolch W, O’neill E. RASSF1A elicits apoptosis through an MST2 pathway directing proapoptotic transcription by the p73 tumor suppressor protein. Mol Cell. 2007; 27:962–75. https://doi.org/10.1016/j.molcel.2007.08.008 [PubMed]
-
21.
Sun PL, Kim JE, Yoo SB, Kim H, Jin Y, Jheon S, Kim K, Lee CT, Chung JH. Cytoplasmic YAP expression is associated with prolonged survival in patients with lung adenocarcinomas and epidermal growth factor receptor tyrosine kinase inhibitor treatment. Ann Surg Oncol. 2014 (Suppl 4); 21:S610–18. https://doi.org/10.1245/s10434-014-3715-5 [PubMed]
-
22.
Sun Z, Xu R, Li X, Ren W, Ou C, Wang Q, Zhang H, Zhang X, Ma J, Wang H, Li G. Prognostic Value of Yes-Associated Protein 1 (YAP1) in Various Cancers: A Meta-Analysis. PLoS One. 2015; 10:e0135119. https://doi.org/10.1371/journal.pone.0135119 [PubMed]
-
23.
Fan Y, Gao Y, Rao J, Wang K, Zhang F, Zhang C. YAP-1 Promotes Tregs Differentiation in Hepatocellular Carcinoma by Enhancing TGFBR2 Transcription. Cell Physiol Biochem. 2017; 41:1189–1198. https://doi.org/10.1159/000464380 [PubMed]
-
24.
Han SX, Bai E, Jin GH, He CC, Guo XJ, Wang LJ, Li M, Ying X, Zhu Q. Expression and clinical significance of YAP, TAZ, and AREG in hepatocellular carcinoma. J Immunol Res. 2014; 2014:261365. https://doi.org/10.1155/2014/261365 [PubMed]
-
25.
Wang YM. The expression and clinical significance of YAP gene in esophageal squamous cell carcinoma. 2014.
-
26.
Salcedo Allende MT, Zeron-Medina J, Hernandez J, Macarulla T, Balsells J, Merino X, Allende H, Tabernero J, Ramon Y Cajal S. Overexpression of Yes Associated Protein 1, an Independent Prognostic Marker in Patients With Pancreatic Ductal Adenocarcinoma, Correlated With Liver Metastasis and Poor Prognosis. Pancreas. 2017; 46:913–20. https://doi.org/10.1097/MPA.0000000000000867 [PubMed]
-
27.
Xu MZ, Yao TJ, Lee NP, Ng IO, Chan YT, Zender L, Lowe SW, Poon RT, Luk JM. Yes-associated protein is an independent prognostic marker in hepatocellular carcinoma. Cancer. 2009; 115:4576–85. https://doi.org/10.1002/cncr.24495 [PubMed]
-
28.
Hall CA, Wang R, Miao J, Oliva E, Shen X, Wheeler T, Hilsenbeck SG, Orsulic S, Goode S. Hippo pathway effector Yap is an ovarian cancer oncogene. Cancer Res. 2010; 70:8517–25. https://doi.org/10.1158/0008-5472.CAN-10-1242 [PubMed]
-
29.
Wang JJ. Clinicopathologic significance and relationship between the expression of YAP and Ki-67 in gastric carcinoma. NanKai University. Master; 2010.
-
30.
Wang Y, Dong Q, Zhang Q, Li Z, Wang E, Qiu X. Overexpression of yes-associated protein contributes to progression and poor prognosis of non-small-cell lung cancer. Cancer Sci. 2010; 101:1279–85. https://doi.org/10.1111/j.1349-7006.2010.01511.x [PubMed]
-
31.
Kang W, Tong JH, Chan AW, Lee TL, Lung RW, Leung PP, So KK, Wu K, Fan D, Yu J, Sung JJ, To KF. Yes-associated protein 1 exhibits oncogenic property in gastric cancer and its nuclear accumulation associates with poor prognosis. Clin Cancer Res. 2011; 17:2130–2139. https://doi.org/10.1158/1078-0432.CCR-10-2467 [PubMed]
-
32.
Song M, Cheong JH, Kim H, Noh SH, Kim H. Nuclear expression of Yes-associated protein 1 correlates with poor prognosis in intestinal type gastric cancer. Anticancer Res. 2012; 32:3827–34. [PubMed]
-
33.
Yeo MK, Kim SH, Kim JM, Huang SM, Kim MR, Song KS, Kim KH. Correlation of expression of phosphorylated and non-phosphorylated Yes-associated protein with clinicopathological parameters in esophageal squamous cell carcinoma in a Korean population. Anticancer Res. 2012; 32:3835–40. [PubMed]
-
34.
Kim DH, Kim SH, Lee OJ, Huang SM, Kwon JL, Kim JM, Kim JY, Seong IO, Song KS, Kim KH. Differential expression of Yes-associated protein and phosphorylated Yes-associated protein is correlated with expression of Ki-67 and phospho-ERK in colorectal adenocarcinoma. Histol Histopathol. 2013; 28:1483–90. https://doi.org/10.14670/HH-28.1483 [PubMed]
-
35.
Kim GJ, Kim H, Park YN. Increased expression of Yes-associated protein 1 in hepatocellular carcinoma with stemness and combined hepatocellular-cholangiocarcinoma. PLoS One. 2013; 8:e75449. https://doi.org/10.1371/journal.pone.0075449 [PubMed]
-
36.
Liu JY, Li YH, Lin HX, Liao YJ, Mai SJ, Liu ZW, Zhang ZL, Jiang LJ, Zhang JX, Kung HF, Zeng YX, Zhou FJ, Xie D. Overexpression of YAP 1 contributes to progressive features and poor prognosis of human urothelial carcinoma of the bladder. BMC Cancer. 2013; 13:349. https://doi.org/10.1186/1471-2407-13-349 [PubMed]
-
37.
Liu T, Liu Y, Gao H, Meng F, Yang S, Lou G. Clinical significance of yes-associated protein overexpression in cervical carcinoma: the differential effects based on histotypes. Int J Gynecol Cancer. 2013; 23:735–742. https://doi.org/10.1097/IGC.0b013e31828c8619 [PubMed]
-
38.
Wang L, Shi S, Guo Z, Zhang X, Han S, Yang A, Wen W, Zhu Q. Overexpression of YAP and TAZ is an independent predictor of prognosis in colorectal cancer and related to the proliferation and metastasis of colon cancer cells. PLoS One. 2013; 8:e65539. https://doi.org/10.1371/journal.pone.0065539 [PubMed]
-
39.
Wang Y, Xie C, Li Q, Xu K, Wang E. Clinical and prognostic significance of Yes-associated protein in colorectal cancer. Tumour Biol. 2013; 34:2169–74. https://doi.org/10.1007/s13277-013-0751-x [PubMed]
-
40.
Xu B, Li SH, Zheng R, Gao SB, Ding LH, Yin ZY, Lin X, Feng ZJ, Zhang S, Wang XM, Jin GH. Menin promotes hepatocellular carcinogenesis and epigenetically up-regulates Yap1 transcription. Proc Natl Acad Sci USA. 2013; 110:17480–85. https://doi.org/10.1073/pnas.1312022110 [PubMed]
-
41.
Xu XN. The clinical significance of YAP expression in hepatocellular carcinoma and its relationship with hepatic stellate cells. Fudan University. Doctor; 2013.
-
42.
Hu X, Xin Y, Xiao Y, Zhao J. Overexpression of YAP1 is correlated with progression, metastasis and poor prognosis in patients with gastric carcinoma. Pathol Oncol Res. 2014; 20:805–11. https://doi.org/10.1007/s12253-014-9757-y [PubMed]
-
43.
Kim SK, Jung WH, Koo JS. Expression of Yes-associated protein (YAP) in breast phyllodes tumor. Int J Clin Exp Pathol. 2014; 7:5997–6005. [PubMed]
-
44.
Kim SK, Jung WH, Koo JS. Yes-associated protein (YAP) is differentially expressed in tumor and stroma according to the molecular subtype of breast cancer. Int J Clin Exp Pathol. 2014; 7:3224–34. [PubMed]
-
45.
Li H, Wang S, Wang G, Zhang Z, Wu X, Zhang T, Fu B, Chen G. Yes-associated protein expression is a predictive marker for recurrence of hepatocellular carcinoma after liver transplantation. Dig Surg. 2014; 31:468–78. https://doi.org/10.1159/000370252 [PubMed]
-
46.
Tsujiura M, Mazack V, Sudol M, Kaspar HG, Nash J, Carey DJ, Gogoi R. Yes-associated protein (YAP) modulates oncogenic features and radiation sensitivity in endometrial cancer. PLoS One. 2014; 9:e100974. https://doi.org/10.1371/journal.pone.0100974 [PubMed]
-
47.
Xia Y, Chang T, Wang Y, Liu Y, Li W, Li M, Fan HY. YAP promotes ovarian cancer cell tumorigenesis and is indicative of a poor prognosis for ovarian cancer patients. PLoS One. 2014; 9:e91770. https://doi.org/10.1371/journal.pone.0091770 [PubMed]
-
48.
Ahmed AA, Abedalthagafi M, Anwar AE, Bui MM. Akt and Hippo Pathways in Ewing’s Sarcoma Tumors and Their Prognostic Significance. J Cancer. 2015; 6:1005–10. https://doi.org/10.7150/jca.12703 [PubMed]
-
49.
Hayashi H, Higashi T, Yokoyama N, Kaida T, Sakamoto K, Fukushima Y, Ishimoto T, Kuroki H, Nitta H, Hashimoto D, Chikamoto A, Oki E, Beppu T, Baba H. An Imbalance in TAZ and YAP Expression in Hepatocellular Carcinoma Confers Cancer Stem Cell-like Behaviors Contributing to Disease Progression. Cancer Res. 2015; 75:4985–97. https://doi.org/10.1158/0008-5472.CAN-15-0291 [PubMed]
-
50.
Kim HM, Jung WH, Koo JS. Expression of Yes-associated protein (YAP) in metastatic breast cancer. Int J Clin Exp Pathol. 2015; 8:11248–57. [PubMed]
-
51.
Kim MH, Kim YK, Shin DH, Lee HJ, Shin N, Kim A, Lee JH, Choi KU, Kim JY, Lee CH, Sol MY. Yes associated protein is a poor prognostic factor in well-differentiated lung adenocarcinoma. Int J Clin Exp Pathol. 2015; 8:15933–39. [PubMed]
-
52.
Liu XL. The underlying effects of DNA damage repair related genes MDC1 and YAP1 in ovarian cancer. Shandong University. Doctor; 2015.
-
53.
Pei T, Li Y, Wang J, Wang H, Liang Y, Shi H, Sun B, Yin D, Sun J, Song R, Pan S, Sun Y, Jiang H, et al. YAP is a critical oncogene in human cholangiocarcinoma. Oncotarget. 2015; 6:17206–20. https://doi.org/10.18632/oncotarget.4043 [PubMed]
-
54.
Suh JH, Won KY, Kim GY, Bae GE, Lim SJ, Sung JY, Park YK, Kim YW, Lee J. Expression of tumoral FOXP3 in gastric adenocarcinoma is associated with favorable clinicopathological variables and related with Hippo pathway. Int J Clin Exp Pathol. 2015; 8:14608–18. [PubMed]
-
55.
Wang YP, Tang DX. Expression of Yes-associated protein in liver cancer and its correlation with clinicopathological features and prognosis of liver cancer patients. Int J Clin Exp Med. 2015; 8:1080–86. [PubMed]
-
56.
Abduch RH, Carolina Bueno A, Leal LF, Cavalcanti MM, Gomes DC, Brandalise SR, Masterallo MJ, Yunes JA, Martinelli CE Jr, Tone LG, Tucci S, Molina CA, Ramalho FS, et al. Unraveling the expression of the oncogene YAP1, a Wnt/beta-catenin target, in adrenocortical tumors and its association with poor outcome in pediatric patients. Oncotarget. 2016; 7:84634–44. https://doi.org/10.18632/oncotarget.12382 [PubMed]
-
57.
Cao LY. Role of Hippo and Wnt signaling pathways in colorectal carcinogenesis and impact of siRNA-mediated silencing YAP and survivin genes on biological behavior of colorectal cancer cells. AnHui Medical University. Doctor; 2016.
-
58.
Li J. The expression and clinical significance of MST1/2, YAP1 protein in colorectal cancer. Nanchang University. Master; 2016.
-
59.
Li P, Sun D, Li X, He Y, Li W, Zhao J, Wang Y, Wang H, Xin Y. Elevated expression of Nodal and YAP1 is associated with poor prognosis of gastric adenocarcinoma. J Cancer Res Clin Oncol. 2016; 142:1765–73. https://doi.org/10.1007/s00432-016-2188-2 [PubMed]
-
60.
Luo X. Expression of YAP and cIAP1 in ovarian serous adenocarcinoma and its clinical significance. Da Lian Medical University. Master; 2016.
-
61.
Ni XG, Yang PF. Expression of YAP1 and SOX2 in epithelial ovarian carcinoma and their clinical significance. Journal of Jiangsu University. 2016; 26:405–09. Medicine Edition.
-
62.
Sun D, Li X, He Y, Li W, Wang Y, Wang H, Jiang S, Xin Y. YAP1 enhances cell proliferation, migration, and invasion of gastric cancer in vitro and in vivo. Oncotarget. 2016; 7:81062–76. https://doi.org/10.18632/oncotarget.13188 [PubMed]
-
63.
Wu H, Liu Y, Jiang XW, Li WF, Guo G, Gong JP, Ding X. Clinicopathological and prognostic significance of Yes-associated protein expression in hepatocellular carcinoma and hepatic cholangiocarcinoma. Tumour Biol. 2016; 37:13499–508. https://doi.org/10.1007/s13277-016-5211-y [PubMed]
-
64.
Zhao C. The Expression of Axl and YAP in Non small cell lung cancer and prognostic evaluation. Zhejiang University School of Medicine. Master; 2016.
-
65.
Cao L, Sun PL, Yao M, Jia M, Gao H. Expression of YES-associated protein (YAP) and its clinical significance in breast cancer tissues. Hum Pathol. 2017; 68:166–74. https://doi.org/10.1016/j.humpath.2017.08.032 [PubMed]
-
66.
Chaib I, Karachaliou N, Pilotto S, Codony Servat J, Cai X, Li X, Drozdowskyj A, Servat CC, Yang J, Hu C, Cardona AF, Vivanco GL, Vergnenegre A, et al. Co-activation of STAT3 and YES-Associated Protein 1 (YAP1) Pathway in EGFR-Mutant NSCLC. J Natl Cancer Inst. 2017; 109. https://doi.org/10.1093/jnci/djx014 [PubMed]
-
67.
Chen XX. The roles and mechanisms of YAP1 in regulating bladder cancer EMT. Guangzhou. 2017; Doctor.
-
68.
Gao JF. Expression and prognostic correlation of MDC1 and YAP1 in ovarian cancer patients. Zhongguo Fuyou Baojian. 2017; 32:2747–50.
-
69.
Hong SA, Son MW, Cho J, Jang SH, Lee HJ, Lee JH, Cho HD, Oh MH, Lee MS. Low angiomotin-p130 with concomitant high Yes-associated protein 1 expression is associated with adverse prognosis of advanced gastric cancer. APMIS. 2017; 125:996–1006. https://doi.org/10.1111/apm.12750 [PubMed]
-
70.
Huang S, Zhu L, Cao Y, Li L, Xie Y, Deng J, Xiong J. Significant association of YAP1 and HSPC111 proteins with poor prognosis in Chinese gastric cancer patients. Oncotarget. 2017; 8:80303–14. https://doi.org/10.18632/oncotarget.17932 [PubMed]
-
71.
Liu F, Wang G, Wang X, Che Z, Dong W, Guo X, Wang Z, Chen P, Hou D, Zhang Q, Zhang W, Pan Y, Yang D, Liu H. Targeting high Aurora kinases expression as an innovative therapy for hepatocellular carcinoma. Oncotarget. 2017; 8:27953–65. https://doi.org/10.18632/oncotarget.15853 [PubMed]
-
72.
Liu M, Lin Y, Zhang XC, Tan YH, Yao YL, Tan J, Zhang X, Cui YH, Liu X, Wang Y, Bian XW. Phosphorylated mTOR and YAP serve as prognostic markers and therapeutic targets in gliomas. Lab Invest. 2017; 97:1354–63. https://doi.org/10.1038/labinvest.2017.70 [PubMed]
-
73.
Liu Z, Zeng W, Maimaiti Y, Ming J, Guo Y, Liu Y, Liu C, Huang T. High Expression of Yes-activated Protein-1 in Papillary Thyroid Carcinoma Correlates With Poor Prognosis. Appl Immunohistochem Mol Morphol. 2019; 27:59–64. https://doi.org/10.1097/PAI.0000000000000544 [PubMed]
-
74.
Pan C, Du Z, Cai Z, Liu Y, Sun Y, Chen J, Tong T, Chen Q, Zhou L, Bing D, Tao Y, Chu H. Elevated expression of yes-associated protein is associated with the malignant status and prognosis of laryngeal squamous cell carcinoma. Mol Med Rep. 2017; 16:4934–40. https://doi.org/10.3892/mmr.2017.7187 [PubMed]
-
75.
Rybarczyk A, Klacz J, Wronska A, Matuszewski M, Kmiec Z, Wierzbicki PM. Overexpression of the YAP1 oncogene in clear cell renal cell carcinoma is associated with poor outcome. Oncol Rep. 2017; 38:427–39. https://doi.org/10.3892/or.2017.5642 [PubMed]
-
76.
Sugimachi K, Nishio M, Aishima S, Kuroda Y, Iguchi T, Komatsu H, Hirata H, Sakimura S, Eguchi H, Bekki Y, Takenaka K, Maehara Y, Suzuki A, Mimori K. Altered Expression of Hippo Signaling Pathway Molecules in Intrahepatic Cholangiocarcinoma. Oncology. 2017; 93:67–74. https://doi.org/10.1159/000463390 [PubMed]
-
77.
Wei H, Xu Z, Liu F, Wang F, Wang X, Sun X, Li J. Hypoxia induces oncogene yes-associated protein 1 nuclear translocation to promote pancreatic ductal adenocarcinoma invasion via epithelial-mesenchymal transition. Tumour Biol. 2017; 39:1010428317691684. https://doi.org/10.1177/1010428317691684 [PubMed]
-
78.
Wu DW, Lin PL, Wang L, Huang CC, Lee H. The YAP1/SIX2 axis is required for DDX3-mediated tumor aggressiveness and cetuximab resistance in KRAS-wild-type colorectal cancer. Theranostics. 2017; 7:1114–32. https://doi.org/10.7150/thno.18175 [PubMed]
-
79.
Zhang Q. The clinical significance of YAP1, MMP14 and AJUBA in ESCC and the prognostic role of YAP1 in common alimentary system cancers: a Meta-analysis. Hebei Medical University. Master; 2017.
-
80.
Chen Y, Zhou Y, Yin BH, Li HL, Hou WX, Zhou YL, Zhang SQ, Xu AB, Zhang YX. Expression of YAP and FOXPl mRNA in hepatocellular carcinoma and its influence on prognosis of patients with hepatocellular carcinoma. Chinese Journal Of Clinical Medicine. 2018; 25:549–54.
-
81.
Godlewski J, Kiezun J, Krazinski BE, Kozielec Z, Wierzbicki PM, Kmiec Z. The Immunoexpression of YAP1 and LATS1 Proteins in Clear Cell Renal Cell Carcinoma: Impact on Patients’ Survival. BioMed Res Int. 2018; 2018:2653623. https://doi.org/10.1155/2018/2653623 [PubMed]
-
82.
Guichet PO, Masliantsev K, Tachon G, Petropoulos C, Godet J, Larrieu D, Milin S, Wager M, Karayan-Tapon L. Fatal correlation between YAP1 expression and glioma aggressiveness: clinical and molecular evidence. J Pathol. 2018; 246:205–16. https://doi.org/10.1002/path.5133 [PubMed]
-
83.
Hong SA, Jang SH, Oh MH, Kim SJ, Kang JH, Hong SH. Overexpression of YAP1 in EGFR mutant lung adenocarcinoma prior to tyrosine kinase inhibitor therapy is associated with poor survival. Pathol Res Pract. 2018; 214:335–42. https://doi.org/10.1016/j.prp.2018.01.010 [PubMed]
-
84.
Huang XF, Zhang X, Song JX, Wang YD, Tang B. Expression and its clinical significance of Yes-associated protein in gallbladder carcinoma tissues. Guangxi Medical Journal. 2018; 40:2764–2766.
-
85.
Kim MK, Park JY, Kang YN. Tumorigenic role of YAP in hepatocellular carcinogenesis is involved in SHP2 whose function is different in vitro and in vivo. Pathol Res Pract. 2018; 214:1031–39. https://doi.org/10.1016/j.prp.2018.04.010 [PubMed]
-
86.
Luu AK, Schott CR, Jones R, Poon AC, Golding B, Hamed R, Deheshi B, Mutsaers A, Wood GA, Viloria-Petit AM. An evaluation of TAZ and YAP crosstalk with TGFbeta signalling in canine osteosarcoma suggests involvement of hippo signalling in disease progression. BMC Vet Res. 2018; 14:365. https://doi.org/10.1186/s12917-018-1651-5 [PubMed]
-
87.
Qian F, Xiao J, Gai L, Zhu J. HMGB1-RAGE signaling facilitates Ras-dependent Yap1 expression to drive colorectal cancer stemness and development. Mol Carcinog. 2018. https://doi.org/10.1002/mc.22944 [PubMed]
-
88.
Chen MJ, Wang YC, Wu DW, Chen CY, Lee H. Association of nuclear localization of SHP2 and YAP1 with unfavorable prognosis in non-small cell lung cancer. Pathol Res Pract. 2019; 215:801–06. https://doi.org/10.1016/j.prp.2019.01.027 [PubMed]
-
89.
Ding N, Huang T, Yuan J, Mao J, Duan Y, Liao W, Xiao Z. Yes-associated protein expression in paired primary and local recurrent breast cancer and its clinical significance. Curr Probl Cancer. 2019. [Epub ahead of print]. https://doi.org/10.1016/j.currproblcancer.2018.12.005 [PubMed]
-
90.
Kim E, Ahn B, Oh H, Lee YJ, Lee JH, Lee Y, Kim CH, Chae YS, Kim JY. High Yes-associated protein 1 with concomitant negative LATS1/2 expression is associated with poor prognosis of advanced gastric cancer. Pathology. 2019; 51:261–67. https://doi.org/10.1016/j.pathol.2019.01.001 [PubMed]
-
91.
Van Haele M, Moya IM, Karaman R, Rens G, Snoeck J, Govaere O, Nevens F, Verslype C, Topal B, Monbaliu D, Halder G, Roskams T. YAP and TAZ Heterogeneity in Primary Liver Cancer: An Analysis of Its Prognostic and Diagnostic Role. Int J Mol Sci. 2019; 20. https://doi.org/10.3390/ijms20030638 [PubMed]
-
92.
Zhang S, Wei Q, Yang Y, Qin H, Li X, Cai S, Ma Y. Loss of yes-associated protein represents an aggressive subtype of colorectal cancer. J Cancer. 2019; 10:689–96. https://doi.org/10.7150/jca.28333 [PubMed]
-
93.
Chen M, Wang J. Initiator caspases in apoptosis signaling pathways. Apoptosis. 2002; 7:313–319. [PubMed]
-
94.
Halder G, Johnson RL. Hippo signaling: growth control and beyond. Development. 2011; 138:9–22. https://doi.org/10.1242/dev.045500 [PubMed]
-
95.
Bejsovec A. Wingless/Wnt signaling in Drosophila: the pattern and the pathway. Mol Reprod Dev. 2013; 80:882–94. https://doi.org/10.1002/mrd.22228 [PubMed]
-
96.
Johnson R, Halder G. The two faces of Hippo: targeting the Hippo pathway for regenerative medicine and cancer treatment. Nat Rev Drug Discov. 2014; 13:63–79. https://doi.org/10.1038/nrd4161 [PubMed]
-
97.
Varelas X, Sakuma R, Samavarchi-Tehrani P, Peerani R, Rao BM, Dembowy J, Yaffe MB, Zandstra PW, Wrana JL. TAZ controls Smad nucleocytoplasmic shuttling and regulates human embryonic stem-cell self-renewal. Nat Cell Biol. 2008; 10:837–48. https://doi.org/10.1038/ncb1748 [PubMed]
-
98.
Mauviel A, Nallet-Staub F, Varelas X. Integrating developmental signals: a Hippo in the (path)way. Oncogene. 2012; 31:1743–56. https://doi.org/10.1038/onc.2011.363 [PubMed]
-
99.
Esteves de Lima J, Bonnin MA, Birchmeier C, Duprez D. Muscle contraction is required to maintain the pool of muscle progenitors via YAP and NOTCH during fetal myogenesis. eLife. 2016; 5:e15593. https://doi.org/10.7554/eLife.15593 [PubMed]
-
100.
Totaro A, Castellan M, Battilana G, Zanconato F, Azzolin L, Giulitti S, Cordenonsi M, Piccolo S. YAP/TAZ link cell mechanics to Notch signalling to control epidermal stem cell fate. Nat Commun. 2017; 8:15206. https://doi.org/10.1038/ncomms15206 [PubMed]
-
101.
Fernandez-L A, Northcott PA, Dalton J, Fraga C, Ellison D, Angers S, Taylor MD, Kenney AM. YAP1 is amplified and up-regulated in hedgehog-associated medulloblastomas and mediates Sonic hedgehog-driven neural precursor proliferation. Genes Dev. 2009; 23:2729–41. https://doi.org/10.1101/gad.1824509 [PubMed]
-
102.
Lapi E, Di Agostino S, Donzelli S, Gal H, Domany E, Rechavi G, Pandolfi PP, Givol D, Strano S, Lu X, Blandino G. PML, YAP, and p73 are components of a proapoptotic autoregulatory feedback loop. Mol Cell. 2008; 32:803–14. https://doi.org/10.1016/j.molcel.2008.11.019 [PubMed]
-
103.
Yuan M, Tomlinson V, Lara R, Holliday D, Chelala C, Harada T, Gangeswaran R, Manson-Bishop C, Smith P, Danovi SA, Pardo O, Crook T, Mein CA, et al. Yes-associated protein (YAP) functions as a tumor suppressor in breast. Cell Death Differ. 2008; 15:1752–59. https://doi.org/10.1038/cdd.2008.108 [PubMed]
-
104.
Wang C, Zhang L, He Q, Feng X, Zhu J, Xu Z, Wang X, Chen F, Li X, Dong J. Differences in Yes-associated protein and mRNA levels in regenerating liver and hepatocellular carcinoma. Mol Med Rep. 2012; 5:410–14. https://doi.org/10.3892/mmr.2011.640 [PubMed]
-
105.
Grijalva JL, Huizenga M, Mueller K, Rodriguez S, Brazzo J, Camargo F, Sadri-Vakili G, Vakili K. Dynamic alterations in Hippo signaling pathway and YAP activation during liver regeneration. Am J Physiol Gastrointest Liver Physiol. 2014; 307:G196–204. https://doi.org/10.1152/ajpgi.00077.2014 [PubMed]
-
106.
Swiderska-Syn M, Xie G, Michelotti GA, Jewell ML, Premont RT, Syn WK, Diehl AM. Hedgehog regulates yes-associated protein 1 in regenerating mouse liver. Hepatology. 2016; 64:232–44. https://doi.org/10.1002/hep.28542 [PubMed]
-
107.
Zhao B, Wei X, Li W, Udan RS, Yang Q, Kim J, Xie J, Ikenoue T, Yu J, Li L, Zheng P, Ye K, Chinnaiyan A, et al. Inactivation of YAP oncoprotein by the Hippo pathway is involved in cell contact inhibition and tissue growth control. Genes Dev. 2007; 21:2747–61. https://doi.org/10.1101/gad.1602907 [PubMed]