Introduction
With advances in medical biology and healthcare technology over recent decades, human lifespans are increasing worldwide, with lifespan expectations of up to 100 years in developed countries by 2025 [1], resulting in a proportionate increase in the aged population. As early as 50 years of age, the frequency of aging-associated cutaneous disorders increases, in parallel with epidermal dysfunction, including compromised permeability homeostasis and reductions in levels of stratum corneum hydration, as well as elevations in skin surface pH, the most prominent features associated with chronic aging. Studies have shown that epidermal dysfunction, in turn, predisposes to the development of a variety of cutaneous abnormalities, including atopic dermatitis, contact dermatitis, pruritus and xerosis, and possibly aging-associated systemic disorders [2–5]. In this review, we discuss aging-associated alterations in epidermal function and their link to cutaneous disorders.
Aging-associated alterations in epidermal function
Compromised epidermal permeability barrier homeostasis
Aging-associated changes in baseline transepidermal water loss (TEWL) rates, an indicator of epidermal permeability barrier, vary greatly with gender, body sites and pigment types. While some studies have shown that baseline TEWL rates on several body sites are lower in the aged than in young skin [6–12], other study demonstrated that TEWL rates on the décolleté region correlated positively with age, but TEWL rates on the neck, forearm and hand were comparable between young and aged women [13]. Moreover, TEWL rates are higher in aged females than in aged males [10]. Yet, in both aged humans and mice, following acute disruption of permeability barrier function, permeability barrier recovery is significantly delayed in comparison to younger age groups [7, 14]. In addition, stratum corneum integrity also decreases in both aged humans and mice [7]. Taken together, aged epidermis displays defects in permeability barrier homeostasis.
Several alterations in the aged skin contribute to a defective permeability barrier function. The epidermal permeability barrier resides in the stratum corneum, the outermost layer of the epidermis. According to the ‘brick and mortar’ model, this permeability barrier is largely determined by quality and quantity of differentiation-related proteins and extracellular lipids in the stratum corneum. Previous studies demonstrated that levels of epidermal growth factor reduced in parallel with a decline in keratinocyte proliferation in the aged epidermis, while keratinocyte apoptosis increased, leading to reductions in the thickness of both the epidermis and the stratum corneum [15–17]. Because high calcium concentration inhibits human keratinocyte proliferation [18], thinning epidermis could also be attributed to an increased calcium gradient in the basal and spinous layers [19], where the keratinocyte proliferation is most active in the epidermis. Moreover, levels of structural proteins for the epidermal permeability barrier, including filaggrin, loricrin and other late cornified envelope proteins, markedly decline in aged skin in comparison to young skin [20–22], perhaps due to reductions in calcium content in the stratum granulosum, leading to defective ‘bricks’ [21]. Deficiencies in these proteins can result in a defective permeability barrier [3].
In addition to such defective ‘bricks,’ reductions in production of the lipid-enriched ‘mortar,’ i.e., the epidermal lipids, are also evident in the aged epidermis. Because formation of a competent epidermal permeability barrier requires an approximately equal molar ratio of cholesterol, free fatty acids and ceramides [23, 24], which are synthesized by epidermal keratinocytes [25, 26], deficiencies in any of these lipids can result in a defective epidermal permeability barrier [25]. Prior studies have shown that the aged stratum corneum displays a >30% reduction in total lipid content in comparison to young stratum corneum [7], due to reduced epidermal lipid synthesis, particularly in cholesterol synthesis, both under basal conditions and after barrier disruption [14]. Notably, aging-associated reduction in ceramide 2 was only observed in females, not in males, although ceramide levels did not differ significantly between aged males and females [27]. In support of evidence that reduced lipid levels contribute to aging-associated dysfunction in epidermal permeability barrier, topical applications of stratum corneum physiologic lipid mixtures can improve epidermal permeability barrier function in aged humans and mice [28]. Thus, these reductions in lipid production and differentiation marker-related protein levels could be causing the compromised epidermal permeability barrier homeostasis in aged skin. The epidermal permeability barrier is also largely made up of extracellular multilamellar bilayers, whose formation requires enzymatic processing of lipid precursors within the extracellular spaces of the stratum corneum [29–31]. The optimal pH for these enzyme activities is ≈5.0 [30, 31]. Yet, aged skin manifests an elevation in skin surface pH in comparison to young skin [32–34]. While topical applications of buffers at neutral pH delay barrier recovery [35], acidification of stratum corneum accelerates barrier recovery in both young and aged murine skin [34, 36, 37]. Hence, the elevated stratum corneum pH of aged skin likely contributes to the delay in permeability barrier recovery.
Chronological aging is accompanied by an increase in glucocorticoid secretion and cortisol content in the skin [15, 38], which can cause epidermal dysfunction. Previous studies have shown that either systemic or topical applications of glucocorticoids compromise epidermal function, including permeability barrier homeostasis and epidermal proliferation [39, 40]. Moreover, glucocorticoid action requires the peripheral conversion of cortisone to cortisol by 11β hydroxysteroid dehydrogenase 1 [41]. In comparison to young skin, aged skin exhibits higher levels and activity of 11β hydroxysteroid dehydrogenase 1 [42], and this epidermal 11β hydroxysteroid dehydrogenase 1 activity correlates negatively with epidermal permeability barrier function [43]. Conversely, inhibition of 11β hydroxysteroid dehydrogenase 1 not only corrects glucocorticoid-induced epidermal functional abnormalities, but also improves aging-associated structural and functional alterations [44, 45]. Thus, the aging-associated increase in epidermal 11β hydroxysteroid dehydrogenase 1 and cortisol content can contribute to defective permeability barrier function in aged skin.
Additionally, other aging-associated changes in the skin can also contribute to altered epidermal function. For example, the aged epidermis displays over 60% reduction in IL-1 receptor antagonist protein in comparison to young epidermis, and a deficiency in IL-1α receptor type 1 delays barrier recovery [46]. Conversely, either upregulation or administration of IL-1α enhances epidermal permeability barrier function in both aged and fetal skin [47, 48]. Similarly, aged skin also exhibits reduced levels of hyaluronic acid [49]. Previous studies have shown that topical applications of hyaluronic acid stimulate keratinocyte differentiation and lipid production, leading to enhancement of epidermal permeability barrier function in both young and aged skin [50, 51]. Finally, aging-associated reductions in epidermal aquaporin 3 expression have also been observed [52–54], while knockout of epidermal aquaporin 3 delays permeability barrier recovery [55]. Conversely, upregulations of epidermal aquaporin 3 expression improve epidermal permeability barrier function [54, 56]. Collectively, aged epidermis displays multiple alterations in keratinocyte function, including altered signaling pathways of calcium, cytokine and hyaluronic acid, stratum corneum acidification, keratinocyte proliferation, differentiation, lipid production, as well as decreased epidermal aquaporin 3 expression, consequently leading to compromised epidermal permeability barrier function (Figure 1).
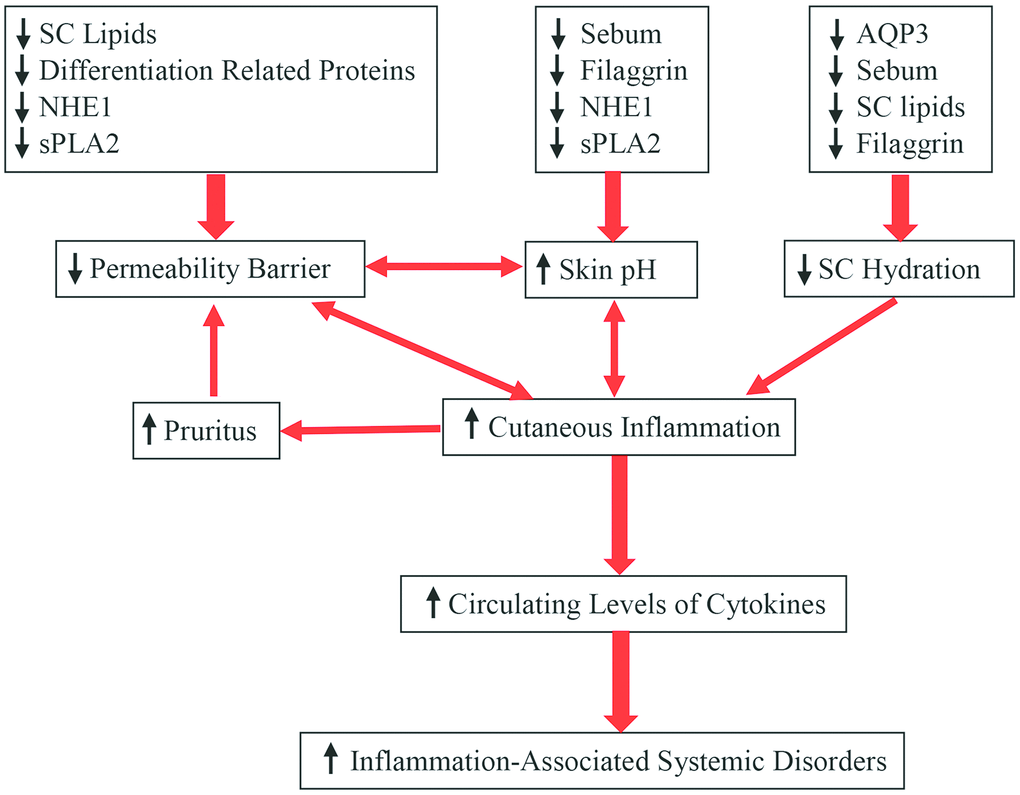
Figure 1. Aging-associated changes in epidermal function and their clinical significance.
Reduction in stratum corneum hydration
In humans, stratum corneum hydration over a lifetime increases to a peak level at age 40 years, followed by a decline, especially on the face and neck in males [10, 32, 57, 58]. The age-dependent differences in hydration are most prominent at a depth of 10-30 μm (on the forearm) in the stratum corneum [59]. It also appears that age-dependent changes in stratum corneum hydration vary with ethnicity. For example, the skin dryness index on the forearm markedly increases in aged African-American and Caucasian skin, but not in aged Chinese and Mexican skin, in comparison to young people of the same ethnicity [60]. The mechanisms underlying reduced stratum corneum hydration in the aged skin can be ascribed to the reduced content of natural humectants in the skin. Firstly, lipid content decreases in the stratum corneum of aged skin [7, 14, 61, 62]. Among these stratum corneum lipids, ceramides exhibit water-holding properties [63]. Prior studies have demonstrated that either oral or topical administration of ceramides can increase stratum corneum hydration [64, 65]. Secondly, aged epidermis exhibits reduced levels of filaggrin [22] and its metabolites, including trans-urocanic acid and pyrrolidone carboxylic acid, which are natural moisturizers in the skin [66]. Thirdly, both sebum and glycerol contents are reduced in aged versus young skin [32, 67]. Deficiency in either sebum or glycerol decreases stratum corneum hydration [68, 69], while topical applications of glycerol improve stratum corneum hydration [56, 69–71]. Finally, levels of aquaporin 3 decrease in aged versus young epidermis [53–55], leading to reduction in stratum corneum hydration. Aquaporin 3 deficiency-induced reduction in stratum corneum hydration is likely due to decreased glycerol content in the stratum corneum [70, 71]. Accordingly, upregulation of epidermal aquaporin 3 expression or topical glycerol improves stratum corneum hydration in aquaporin 3-deficient mice [71, 72]. Thus, aging-associated reductions in stratum corneum hydration can be attributed, in large part, to a reduced content of natural moisturizers in the epidermis (Figure 1).
Elevation in skin surface pH
In humans, skin surface pH is generally higher in the first 2 weeks of life, followed by a decline by 5-6 weeks old [73]. Skin surface pH begins to increase at 55 years of age [32, 34]. Marked elevations in skin surface pH occur in aged humans, particularly in those over 70 years old [32, 34, 74–76]. Human skin surface pH varies with gender and body site. For example, skin surface pH on the upper eyelid is 5.13 ± 0.49 (mean ± SD), and 5.75 ± 43 (mean ± SD) on the forearm in subjects aged 66-83 years [76]. Similarly, the skin surface of the abdomen displays a higher pH than that of the upper back [8]. In males [but not females], the highest skin surface pH was found on the forehead and the forearm in subjects over 70 years of age [32]. Moreover, skin surface pH, at least on the forehead, forearm, cheek and hand, is higher in aged females than in aged males [10, 32]. However, skin surface pH is comparable between males and females on both the axillary vault and fossa [76].
In terms of etiology, at least four factors can contribute to the aging-associated elevation in skin surface pH. One is the sebum content which declines in aged skin [13, 32], resulting in reduced triglycerides in the stratum corneum. Degradation of triglycerides yields free fatty acids, which can acidify stratum corneum [77]. Likewise, generation of free fatty acids from phospholipids by secretory phospholipase 2 [sPLA2] can also acidify the stratum corneum [78]. Expression levels of sPLA2 markedly decreased in aged skin [79]. Thus, aging-associated reduction in sebum and sPLA2 levels can contribute, at least in part, to the elevated skin surface pH in aged skin. Sodium-hydrogen exchanger 1 (NHE1) is another contributor to elevated skin surface pH in aged skin. Prior studies demonstrated that NHE1 deficiency increased skin surface pH in mice [80], while aged skin, at least in mice, exhibits significantly lower expression levels of NHE1 in comparison to young skin [79]. Hence, elevated skin surface pH in aged skin can be due to reduction in epidermal NHE1 expression as well. In addition, aged epidermis displays low expression levels of filaggrin [21], which can be degraded to trans-urocanic acid via a filaggrin-histidine-urocanic acid pathway [81]. Urocanic acid content in the stratum corneum correlates positively with skin acidity [82]. Collectively, reductions in sebum content and levels of NHE1, sPLA2 and filaggrin can contribute to aging-associated elevation in skin surface pH (Figure 1).
Consequences of aging-associated alteration in epidermal function
Reduced stratum corneum hydration
Reductions in stratum corneum hydration have been implicated in the pathogenesis of senile xerosis and aging-associated pruritus [83, 84]. Previous studies have demonstrated that reductions in SC hydration increase inflammatory infiltration, mast cell density, mast cell degranulation, and histamine content in mouse dermis [85, 86]. Increased cytokines and histamine in the skin can provoke scratching due to pruritus, resulting in disruption of epidermal permeability barrier, consequently leading to a further increase in cutaneous inflammation. Because the epidermal permeability barrier homeostasis in aged skin is compromised [7, 14], it cannot be rapidly normalized, leading to a sustained increase in cutaneous inflammation and exacerbation of preexisting inflammatory conditions, such as atopic dermatitis and eczema. Moreover, the sustained increase in cutaneous inflammation could eventually cause systemic inflammation, possibly leading to the development of inflammaging-associated disorders [5, 87]. Additionally, nocturnal pruritus can cause insomnia, resulting in exacerbation of other disorders, such as cardiovascular and Parkinson diseases [88, 89]. Thus, reduced stratum corneum hydration can lead to the development of both cutaneous and extracutaneous disorders.
Dysfunction in epidermal permeability barrier
As mentioned above, permeability barrier recovery is delayed in aged skin, although the baseline permeability barrier is comparable to young subjects. Disruption of epidermal permeability barrier alone not only increases expression levels of cutaneous cytokines [90], but also increases inflammatory infiltration in the skin [91–94], leading to the development of cutaneous inflammation and pruritus. Moreover, when the permeability barrier is disrupted by scratching or other forms of insults, the epidermal ‘window’ would keep opening long enough to let harmful substances penetrate the skin, because of delayed barrier recovery in aged skin. Consequently, aged skin becomes vulnerable to the development of atopic dermatitis and contact dermatitis [95, 96]. Again, sustained cutaneous inflammation can provoke systemic inflammation. Finally, a defective permeability barrier favors bacterial colonization in the skin [97, 98]. Thus, aging-associated dysfunction in epidermal permeability barrier can contribute to the development of cutaneous infections, pruritus, dermatitis, and possible systemic inflammation.
Elevated skin surface pH
Elevated skin pH can impact several aspects of cutaneous function. First, lamellar membrane bilayers in the stratum corneum are the critical structures of the epidermal permeability barrier. Formation of mature membrane bilayers requires processing of lipid precursors by several enzymes, including beta-glucocerebrosidase, acidic sphingomyelinase, and acidic secretory phospholipase A2, with an optimal pH range of 4.5 to 5.2 [30, 31]. Hence, elevated skin pH can abrogate the maturation of membrane bilayers, resulting in a compromised epidermal permeability barrier. Secondly, the antimicrobial properties of the skin are pH-dependent [99]. Certain pathogens, such as Staphylococcus aureus and fungi, favor a neutral pH, while an acidic pH decreases survival ability of Staphylococcus aureus [100–102]. Thus, the increased skin surface pH can contribute to a high prevalence of cutaneous infections in the elderly. Thirdly, the epidermis is rich in proteases with either optimal basic or acidic pH. For example, stratum corneum cathepsin-like protease, with an optimal acidic pH, degrades corneodesmosin, a component of corneodesmosomes [103]. Elevated stratum corneum pH can decrease the activity of cathepsin-like protease, leading to abnormal desquamation. On the other hand, an elevated pH favors other proteases, such as kallikrein-related peptidases 5 and 7, which are both expressed in the epidermis [104–107]. Elevation in skin surface pH can activate kallikrein-related peptidase 5, leading to the development of atopic dermatitis-like lesions in mice via proteinase-activated receptor-2 dependent and independent pathways [108–110]. Moreover, kallikrein-related peptidase 7 can activate IL-1β [reviewed in 111]. Overexpression of epidermal kallikrein-related peptidase 7 results in the development of cutaneous inflammation [112]. Taken together, the elevated skin surface pH can contribute to the development of inflammation, infections and compromised permeability barrier homeostasis in aged skin.
Approaches to emprove epidermal function in aged skin
Because of the substantial impact of epidermal dysfunctions on cutaneous and extracutaneous function, great efforts have been made to develop regimens to improve epidermal functions in chronologically-aged skin. Several approaches have been proven to benefit epidermal functions in aged mice and/or humans.
Acidification of the stratum corneum
Studies have demonstrated that acidification of the stratum corneum alone can improve epidermal structure and permeability barrier homeostasis in aged skin. For example, acidification of aged mouse skin with topical lactobionic acid normalized permeability barrier homeostasis and the structure of corneodesmosomes in the stratum corneum [36]. Likewise, topical applications of an emollient at pH 4.0 for 29 days markedly improve stratum corneum hydration and lamellar bilayer structure, along with increased resistance to challenges from topical sodium dodecyl sulphate, in aged humans [113]. Similarly, compared to a pH 5.8 emollient, a topical pH 4.0 emollient accelerates permeability barrier recovery following acute disruption, and significantly improves stratum corneum integrity after 28-day treatments on aged humans [114]. Although acidifying the stratum corneum could prevent and alleviate atopic dermatitis-like skin lesions in young mice [115–118], whether the same benefits could be achieved in aged humans remains to be determined. Nonetheless, acidification of the stratum corneum can improve epidermal structure and function in chronologically-aged humans.
Topical applications of stratum corneum lipids
Reductions in stratum corneum lipid content can largely contribute to the delayed permeability barrier recovery in aged skin [7, 14]. Accordingly, topical applications of a lipid mixture containing three key stratum corneum lipids; i.e., cholesterol, free fatty acids and ceramides, accelerate permeability barrier recovery in both aged mice and humans [27]. A recent study demonstrated that topical applications of an emollient containing stratum corneum lipids not only improved epidermal permeability barrier, stratum corneum hydration and skin surface pH, but also lowered circulating levels of proinflammatory cytokines in aged humans [5]. There are at least two possible mechanisms by which the topical lipid mixture improves epidermal function. One is that topical lipids penetrate into keratinocytes in the stratum granulosum, where they are packaged in lamellar bodies, then secreted into the stratum corneum, where lamellar bilayers are formed [23, 119]. The other mechanism is that this lipid mixture contains fatty acids, which can activate peroxisome proliferator-activated receptors (PPAR) [120]. Activation of PPARs stimulates epidermal lipid production and keratinocyte differentiation, resulting in improvement in epidermal permeability barrier function and inhibition of cutaneous inflammation [121–123]. Thus, topical applications of stratum corneum lipid mixtures not only provide lipids for membrane bilayer formation, but also upregulate keratinocyte function, leading to improvements in epidermal function. However, whether topical PPAR ligands alone can improve epidermal functions in the elderly has not been sufficiently proved. Haratake et al. [124] showed that topical application of either cholesterol or mevalonic acid, a precursor of cholesterol, improved the stratum corneum integrity and epidermal permeability barrier recovery in mice. Whether topical cholesterol or mevalonic acid alone can improve epidermal permeability barrier in the aged humans remains to be determined.
Natural ingredients
Studies have shown that several natural ingredients can improve epidermal function in aged humans and mice. For example, oral administration of either vitamin C or linoleic acid improved senile xerosis [125]. Likewise, orally-taken wheat extract oil increased stratum corneum hydration [126, 127], while orally-taken milk, containing green tea extract, borage oil and vitamin E, improved epidermal permeability barrier [128]. Similarly, oral supplements of flaxseed and borage oil increased stratum corneum hydration, while decreasing transepidermal water loss rates in humans [129]. In addition, estrogen replacement can improve multiple epidermal functions, including permeability barrier homeostasis, stratum corneum hydration and stratum corneum integrity, in both mice and humans [130, 131]. Besides oral administration, topical applications of some natural ingredients can also improve epidermal functions in chronologically-aged skin. Man et al. [79] reported that topical applications of hesperidin improved epidermal functions, including reductions in skin surface pH, acceleration of permeability barrier recovery and stimulation of keratinocyte differentiation, in aged mice. Other natural ingredients, such as petrolatum, glycerol, coconut oil and sunflower oil, can also improve stratum corneum hydration and epidermal permeability barrier [132–136]. Interestingly, bathing with soybean oil could also decrease transepidermal water loss, while increasing stratum corneum hydration, in comparison to bathing without soybean oil [137]. In addition to improving epidermal function, topical petrolatum and glycerol could lower circulating levels of cytokines in aged mice, too [87]. Taken together, either oral or topical administration of certain natural ingredients can improve epidermal function in aged skin.
It appears that the influence of some natural ingredients on epidermal functions is due to upregulation of keratinocyte function. For instance, topical hesperidin can upregulate expression levels of mRNA related to epidermal differentiation, lipid production and acidification in aged skin [79]. Likewise, topical applications of a mixture of several plant oils increase ceramide content, along with improvements in stratum corneum hydration and epidermal permeability barrier function in aged skin [138]. Similarly, topical applications of bacterial sphingomyelinase from Streptococcus thermophiles also increase stratum corneum hydration and stratum corneum ceramide content in aged humans [139]. Moreover, glycerol and petrolatum, which are traditionally viewed as inert ingredients, can also stimulate keratinocyte differentiation [140, 141]. Of course, water holding and occlusive properties of glycerol and petrolatum also contribute to enhanced epidermal permeability barrier and stratum corneum hydration.
In summary, chronologically-aged skin displays multiple alterations in epidermal functions, which can contribute to the development of a number of cutaneous and extracutaneous disorders. Accordingly, improvements in epidermal function can be a valuable alternative to prevent and ameliorate disorders, which are linked to epidermal dysfunction in the elderly. A wide range of ingredients can improve epidermal function. However, it is worth noting that a substantial portion of emollients on the market are harmful to epidermal function, although some ingredients in these products may benefit epidermal function [142–145]. The harmful emollients often contain some ingredient, such as eicosadienoic acid-enriched oils, stearic acid, ceteareth 20, PEG-40 castor oil and PEG-100 stearate, which all can induce cutaneous inflammation and/or disrupt epidermal permeability barrier [145–148]. Use of harmful emollients, especially in the long-term, could compromise epidermal function, leading to the development and exacerbation of some cutaneous and extracutaneous disorders associated with epidermal dysfunction. Therefore, caution should be taken when choosing emollients.
Acknowledgments
The authors are grateful to Ms. Joan Wakefield for her superior editing work.
Conflicts of Interest
All authors declare no conflicts of interest.
Funding
This work was supported, in part, by the National Institute of Arthritis, Musculoskeletal and Skin Diseases of the National Institutes of Health (R01 AR061106), administered by the Northern California Institute for Research and Education, with additional resources provided by the Veterans Affairs Medical Center, San Francisco, CA. This content is solely the responsibility of the authors and does not necessarily represent the official views of either the National Institutes of Health or the Department of Veterans Affairs.
References
- 1. Christensen K, Doblhammer G, Rau R, Vaupel JW. Ageing populations: the challenges ahead. Lancet. 2009; 374:1196–208. https://doi.org/10.1016/S0140-6736(09)61460-4 [PubMed]
- 2. Kim BE, Leung DY. Significance of Skin Barrier Dysfunction in Atopic Dermatitis. Allergy Asthma Immunol Res. 2018; 10:207–15. https://doi.org/10.4168/aair.2018.10.3.207 [PubMed]
- 3. Scharschmidt TC, Man MQ, Hatano Y, Crumrine D, Gunathilake R, Sundberg JP, Silva KA, Mauro TM, Hupe M, Cho S, Wu Y, Celli A, Schmuth M, et al. Filaggrin deficiency confers a paracellular barrier abnormality that reduces inflammatory thresholds to irritants and haptens. J Allergy Clin Immunol. 2009; 124:496–506, 506.e1–6. https://doi.org/10.1016/j.jaci.2009.06.046 [PubMed]
- 4. Man MQ, Elias PM. Stratum corneum hydration regulates key epidermal function and serves as an indicator and contributor to other conditions. J Eur Acad Dermatol Venereol. 2019; 33:15–16. https://doi.org/10.1111/jdv.15374 [PubMed]
- 5. Ye L, Mauro TM, Dang E, Wang G, Hu LZ, Yu C, Jeong S, Feingold K, Elias PM, Lv CZ, Man MQ. Topical applications of an emollient reduce circulating pro-inflammatory cytokine levels in chronically aged humans: a pilot clinical study. J Eur Acad Dermatol Venereol. 2019; 33:2197–201. https://doi.org/10.1111/jdv.15540 [PubMed]
- 6. Boireau-Adamezyk E, Baillet-Guffroy A, Stamatas GN. Age-dependent changes in stratum corneum barrier function. Skin Res Technol. 2014; 20:409–15. https://doi.org/10.1111/srt.12132 [PubMed]
- 7. Ghadially R, Brown BE, Sequeira-Martin SM, Feingold KR, Elias PM. The aged epidermal permeability barrier. Structural, functional, and lipid biochemical abnormalities in humans and a senescent murine model. J Clin Invest. 1995; 95:2281–90. https://doi.org/10.1172/JCI117919 [PubMed]
- 8. Wilhelm KP, Cua AB, Maibach HI. Skin aging. Effect on transepidermal water loss, stratum corneum hydration, skin surface pH, and casual sebum content. Arch Dermatol. 1991; 127:1806–09. https://doi.org/10.1001/archderm.1991.04520010052006 [PubMed]
- 9. Galzote C, Estanislao R, Suero MO, Khaiat A, Mangubat MI, Moideen R, Tagami H, Wang X. Characterization of facial skin of various Asian populations through visual and non-invasive instrumental evaluations: influence of age and skincare habits. Skin Res Technol. 2013; 19:454–65. https://doi.org/10.1111/srt.12069 [PubMed]
- 10. Luebberding S, Krueger N, Kerscher M. Skin physiology in men and women: in vivo evaluation of 300 people including TEWL, SC hydration, sebum content and skin surface pH. Int J Cosmet Sci. 2013; 35:477–83. https://doi.org/10.1111/ics.12068 [PubMed]
- 11. Mehta HH, Nikam VV, Jaiswal CR, Mehta HB. A cross-sectional study of variations in the biophysical parameters of skin among healthy volunteers. Indian J Dermatol Venereol Leprol. 2018; 84:521. https://doi.org/10.4103/ijdvl.IJDVL_1151_15 [PubMed]
- 12. Machková L, Švadlák D, Dolečková I. A comprehensive in vivo study of Caucasian facial skin parameters on 442 women. Arch Dermatol Res. 2018; 310:691–99. https://doi.org/10.1007/s00403-018-1860-6 [PubMed]
- 13. Luebberding S, Krueger N, Kerscher M. Age-related changes in skin barrier function - quantitative evaluation of 150 female subjects. Int J Cosmet Sci. 2013; 35:183–90. https://doi.org/10.1111/ics.12024 [PubMed]
- 14. Ghadially R, Brown BE, Hanley K, Reed JT, Feingold KR, Elias PM. Decreased epidermal lipid synthesis accounts for altered barrier function in aged mice. J Invest Dermatol. 1996; 106:1064–69. https://doi.org/10.1111/1523-1747.ep12338692 [PubMed]
- 15. Kinn PM, Holdren GO, Westermeyer BA, Abuissa M, Fischer CL, Fairley JA, Brogden KA, Brogden NK. Age-dependent variation in cytokines, chemokines, and biologic analytes rinsed from the surface of healthy human skin. Sci Rep. 2015; 5:10472. https://doi.org/10.1038/srep10472 [PubMed]
- 16. Gilhar A, Ullmann Y, Karry R, Shalaginov R, Assy B, Serafimovich S, Kalish RS. Aging of human epidermis: reversal of aging changes correlates with reversal of keratinocyte fas expression and apoptosis. J Gerontol A Biol Sci Med Sci. 2004; 59:411–15. https://doi.org/10.1093/gerona/59.5.B411 [PubMed]
- 17. Thomas JR. Effects of age and diet on rat skin histology. Laryngoscope. 2005; 115:405–11. https://doi.org/10.1097/01.mlg.0000157845.86154.48 [PubMed]
- 18. Micallef L, Belaubre F, Pinon A, Jayat-Vignoles C, Delage C, Charveron M, Simon A. Effects of extracellular calcium on the growth-differentiation switch in immortalized keratinocyte HaCaT cells compared with normal human keratinocytes. Exp Dermatol. 2009; 18:143–51. https://doi.org/10.1111/j.1600-0625.2008.00775.x [PubMed]
- 19. Denda M, Tomitaka A, Akamatsu H, Matsunaga K. Altered distribution of calcium in facial epidermis of aged adults. J Invest Dermatol. 2003; 121:1557–58. https://doi.org/10.1111/j.1523-1747.2003.12619.x [PubMed]
- 20. Tezuka T, Qing J, Saheki M, Kusuda S, Takahashi M. Terminal differentiation of facial epidermis of the aged: immunohistochemical studies. Dermatology. 1994; 188:21–24. https://doi.org/10.1159/000247079 [PubMed]
- 21. Rinnerthaler M, Duschl J, Steinbacher P, Salzmann M, Bischof J, Schuller M, Wimmer H, Peer T, Bauer JW, Richter K. Age-related changes in the composition of the cornified envelope in human skin. Exp Dermatol. 2013; 22:329–35. https://doi.org/10.1111/exd.12135 [PubMed]
- 22. Takahashi M, Tezuka T. The content of free amino acids in the stratum corneum is increased in senile xerosis. Arch Dermatol Res. 2004; 295:448–52. https://doi.org/10.1007/s00403-003-0448-x [PubMed]
- 23. Man MQ, Feingold KR, Elias PM. Exogenous lipids influence permeability barrier recovery in acetone-treated murine skin. Arch Dermatol. 1993; 129:728–38. https://doi.org/10.1001/archderm.1993.01680270066008 [PubMed]
- 24. Man MQ M, Feingold KR, Thornfeldt CR, Elias PM. Optimization of physiological lipid mixtures for barrier repair. J Invest Dermatol. 1996; 106:1096–101. https://doi.org/10.1111/1523-1747.ep12340135 [PubMed]
- 25. Feingold KR, Elias PM. Role of lipids in the formation and maintenance of the cutaneous permeability barrier. Biochim Biophys Acta. 2014; 1841:280–94. https://doi.org/10.1016/j.bbalip.2013.11.007 [PubMed]
- 26. Mao-Qiang M, Elias PM, Feingold KR. Fatty acids are required for epidermal permeability barrier function. J Clin Invest. 1993; 92:791–98. https://doi.org/10.1172/JCI116652 [PubMed]
- 27. Denda M, Koyama J, Hori J, Horii I, Takahashi M, Hara M, Tagami H. Age- and sex-dependent change in stratum corneum sphingolipids. Arch Dermatol Res. 1993; 285:415–17. https://doi.org/10.1007/BF00372135 [PubMed]
- 28. Zettersten EM, Ghadially R, Feingold KR, Crumrine D, Elias PM. Optimal ratios of topical stratum corneum lipids improve barrier recovery in chronologically aged skin. J Am Acad Dermatol. 1997; 37:403–08. https://doi.org/10.1016/S0190-9622(97)70140-3 [PubMed]
- 29. Mao-Qiang M, Feingold KR, Jain M, Elias PM. Extracellular processing of phospholipids is required for permeability barrier homeostasis. J Lipid Res. 1995; 36:1925–35. [PubMed]
- 30. Holleran WM, Takagi Y, Menon GK, Legler G, Feingold KR, Elias PM. Processing of epidermal glucosylceramides is required for optimal mammalian cutaneous permeability barrier function. J Clin Invest. 1993; 91:1656–64. https://doi.org/10.1172/JCI116374 [PubMed]
- 31. Takagi Y, Kriehuber E, Imokawa G, Elias PM, Holleran WM. Beta-glucocerebrosidase activity in mammalian stratum corneum. J Lipid Res. 1999; 40:861–69. [PubMed]
- 32. Man MQ, Xin SJ, Song SP, Cho SY, Zhang XJ, Tu CX, Feingold KR, Elias PM. Variation of skin surface pH, sebum content and stratum corneum hydration with age and gender in a large Chinese population. Skin Pharmacol Physiol. 2009; 22:190–99. https://doi.org/10.1159/000231524 [PubMed]
- 33. Schreml S, Zeller V, Meier RJ, Korting HC, Behm B, Landthaler M, Babilas P. Impact of age and body site on adult female skin surface pH. Dermatology. 2012; 224:66–71. https://doi.org/10.1159/000337029 [PubMed]
- 34. Choi EH, Man MQ, Xu P, Xin S, Liu Z, Crumrine DA, Jiang YJ, Fluhr JW, Feingold KR, Elias PM, Mauro TM. Stratum corneum acidification is impaired in moderately aged human and murine skin. J Invest Dermatol. 2007; 127:2847–56. https://doi.org/10.1038/sj.jid.5700913 [PubMed]
- 35. Mauro T, Holleran WM, Grayson S, Gao WN, Man MQ, Kriehuber E, Behne M, Feingold KR, Elias PM. Barrier recovery is impeded at neutral pH, independent of ionic effects: implications for extracellular lipid processing. Arch Dermatol Res. 1998; 290:215–22. https://doi.org/10.1007/s004030050293 [PubMed]
- 36. Hachem JP, Roelandt T, Schürer N, Pu X, Fluhr J, Giddelo C, Man MQ, Crumrine D, Roseeuw D, Feingold KR, Mauro T, Elias PM. Acute acidification of stratum corneum membrane domains using polyhydroxyl acids improves lipid processing and inhibits degradation of corneodesmosomes. J Invest Dermatol. 2010; 130:500–10. https://doi.org/10.1038/jid.2009.249 [PubMed]
- 37. Hachem JP, Crumrine D, Fluhr J, Brown BE, Feingold KR, Elias PM. pH directly regulates epidermal permeability barrier homeostasis, and stratum corneum integrity/cohesion. J Invest Dermatol. 2003; 121:345–53. https://doi.org/10.1046/j.1523-1747.2003.12365.x [PubMed]
- 38. Yiallouris A, Tsioutis C, Agapidaki E, Zafeiri M, Agouridis AP, Ntourakis D, Johnson EO. Adrenal Aging and Its Implications on Stress Responsiveness in Humans. Front Endocrinol (Lausanne). 2019; 10:54. Lausanne https://doi.org/10.3389/fendo.2019.00054 [PubMed]
- 39. Kao JS, Fluhr JW, Man MQ, Fowler AJ, Hachem JP, Crumrine D, Ahn SK, Brown BE, Elias PM, Feingold KR. Short-term glucocorticoid treatment compromises both permeability barrier homeostasis and stratum corneum integrity: inhibition of epidermal lipid synthesis accounts for functional abnormalities. J Invest Dermatol. 2003; 120:456–64. https://doi.org/10.1046/j.1523-1747.2003.12053.x [PubMed]
- 40. Demerjian M, Choi EH, Man MQ, Chang S, Elias PM, Feingold KR. Activators of PPARs and LXR decrease the adverse effects of exogenous glucocorticoids on the epidermis. Exp Dermatol. 2009; 18:643–49. https://doi.org/10.1111/j.1600-0625.2009.00841.x [PubMed]
- 41. Tomlinson JW, Walker EA, Bujalska IJ, Draper N, Lavery GG, Cooper MS, Hewison M, Stewart PM. 11beta-hydroxysteroid dehydrogenase type 1: a tissue-specific regulator of glucocorticoid response. Endocr Rev. 2004; 25:831–66. https://doi.org/10.1210/er.2003-0031 [PubMed]
- 42. Tiganescu A, Walker EA, Hardy RS, Mayes AE, Stewart PM. Localization, age- and site-dependent expression, and regulation of 11β-hydroxysteroid dehydrogenase type 1 in skin. J Invest Dermatol. 2011; 131:30–36. https://doi.org/10.1038/jid.2010.257 [PubMed]
- 43. Choe SJ, Kim D, Kim EJ, Ahn JS, Choi EJ, Son ED, Lee TR, Choi EH. Psychological Stress Deteriorates Skin Barrier Function by Activating 11β-Hydroxysteroid Dehydrogenase 1 and the HPA Axis. Sci Rep. 2018; 8:6334. https://doi.org/10.1038/s41598-018-24653-z [PubMed]
- 44. Tiganescu A, Hupe M, Uchida Y, Mauro T, Elias PM, Holleran WM. Topical 11β-Hydroxysteroid Dehydrogenase Type 1 Inhibition Corrects Cutaneous Features of Systemic Glucocorticoid Excess in Female Mice. Endocrinology. 2018; 159:547–56. https://doi.org/10.1210/en.2017-00607 [PubMed]
- 45. Tiganescu A, Tahrani AA, Morgan SA, Otranto M, Desmoulière A, Abrahams L, Hassan-Smith Z, Walker EA, Rabbitt EH, Cooper MS, Amrein K, Lavery GG, Stewart PM. 11β-Hydroxysteroid dehydrogenase blockade prevents age-induced skin structure and function defects. J Clin Invest. 2013; 123:3051–60. https://doi.org/10.1172/JCI64162 [PubMed]
- 46. Ye J, Garg A, Calhoun C, Feingold KR, Elias PM, Ghadially R. Alterations in cytokine regulation in aged epidermis: implications for permeability barrier homeostasis and inflammation. I. IL-1 gene family. Exp Dermatol. 2002; 11:209–16. https://doi.org/10.1034/j.1600-0625.2002.110303.x [PubMed]
- 47. Barland CO, Zettersten E, Brown BS, Ye J, Elias PM, Ghadially R. Imiquimod-induced interleukin-1 alpha stimulation improves barrier homeostasis in aged murine epidermis. J Invest Dermatol. 2004; 122:330–36. https://doi.org/10.1046/j.0022-202X.2004.22203.x [PubMed]
- 48. Jiang YJ, Lu B, Crumrine D, Man MQ, Elias PM, Feingold KR. IL-1alpha accelerates stratum corneum formation and improves permeability barrier homeostasis during murine fetal development. J Dermatol Sci. 2009; 54:88–98. https://doi.org/10.1016/j.jdermsci.2009.01.001 [PubMed]
- 49. Oh JH, Kim YK, Jung JY, Shin JE, Chung JH. Changes in glycosaminoglycans and related proteoglycans in intrinsically aged human skin in vivo. Exp Dermatol. 2011; 20:454–56. https://doi.org/10.1111/j.1600-0625.2011.01258.x [PubMed]
- 50. Bourguignon LY, Ramez M, Gilad E, Singleton PA, Man MQ, Crumrine DA, Elias PM, Feingold KR. Hyaluronan-CD44 interaction stimulates keratinocyte differentiation, lamellar body formation/secretion, and permeability barrier homeostasis. J Invest Dermatol. 2006; 126:1356–65. https://doi.org/10.1038/sj.jid.5700260 [PubMed]
- 51. Bourguignon LY, Wong G, Xia W, Man MQ, Holleran WM, Elias PM. Selective matrix (hyaluronan) interaction with CD44 and RhoGTPase signaling promotes keratinocyte functions and overcomes age-related epidermal dysfunction. J Dermatol Sci. 2013; 72:32–44. https://doi.org/10.1016/j.jdermsci.2013.05.003 [PubMed]
- 52. Seleit I, Bakry OA, El Rebey HS, El-Akabawy G, Hamza G. Is Aquaporin-3 a Determinant Factor of Intrinsic and Extrinsic Aging? An Immunohistochemical and Morphometric Study. Appl Immunohistochem Mol Morphol. 2017; 25:49–57. https://doi.org/10.1097/PAI.0000000000000265 [PubMed]
- 53. Li J, Tang H, Hu X, Chen M, Xie H. Aquaporin-3 gene and protein expression in sun-protected human skin decreases with skin ageing. Australas J Dermatol. 2010; 51:106–12. https://doi.org/10.1111/j.1440-0960.2010.00629.x [PubMed]
- 54. Ikarashi N, Kon R, Kaneko M, Mizukami N, Kusunoki Y, Sugiyama K. Relationship between Aging-Related Skin Dryness and Aquaporins. Int J Mol Sci. 2017; 18:E1559. https://doi.org/10.3390/ijms18071559 [PubMed]
- 55. Hara M, Ma T, Verkman AS. Selectively reduced glycerol in skin of aquaporin-3-deficient mice may account for impaired skin hydration, elasticity, and barrier recovery. J Biol Chem. 2002; 277:46616–21. https://doi.org/10.1074/jbc.M209003200 [PubMed]
- 56. Schrader A, Siefken W, Kueper T, Breitenbach U, Gatermann C, Sperling G, Biernoth T, Scherner C, Stäb F, Wenck H, Wittern KP, Blatt T. Effects of glyceryl glucoside on AQP3 expression, barrier function and hydration of human skin. Skin Pharmacol Physiol. 2012; 25:192–99. https://doi.org/10.1159/000338190 [PubMed]
- 57. Luebberding S, Krueger N, Kerscher M. Age-related changes in male skin: quantitative evaluation of one hundred and fifty male subjects. Skin Pharmacol Physiol. 2014; 27:9–17. https://doi.org/10.1159/000351349 [PubMed]
- 58. Sato N, Kitahara T, Fujimura T. Age-related changes of stratum corneum functions of skin on the trunk and the limbs. Skin Pharmacol Physiol. 2014; 27:181. https://doi.org/10.1159/000353912 [PubMed]
- 59. Egawa M, Tagami H. Comparison of the depth profiles of water and water-binding substances in the stratum corneum determined in vivo by Raman spectroscopy between the cheek and volar forearm skin: effects of age, seasonal changes and artificial forced hydration. Br J Dermatol. 2008; 158:251–60. https://doi.org/10.1111/j.1365-2133.2007.08311.x [PubMed]
- 60. Diridollou S, de Rigal J, Querleux B, Leroy F, Holloway Barbosa V. Comparative study of the hydration of the stratum corneum between four ethnic groups: influence of age. Int J Dermatol. 2007 (Suppl 1); 46:11–14. https://doi.org/10.1111/j.1365-4632.2007.03455.x [PubMed]
- 61. Rogers J, Harding C, Mayo A, Banks J, Rawlings A. Stratum corneum lipids: the effect of ageing and the seasons. Arch Dermatol Res. 1996; 288:765–70. https://doi.org/10.1007/BF02505294 [PubMed]
- 62. Akimoto K, Yoshikawa N, Higaki Y, Kawashima M, Imokawa G. Quantitative analysis of stratum corneum lipids in xerosis and asteatotic eczema. J Dermatol. 1993; 20:1–6. https://doi.org/10.1111/j.1346-8138.1993.tb03820.x [PubMed]
- 63. Imokawa G, Abe A, Jin K, Higaki Y, Kawashima M, Hidano A. Decreased level of ceramides in stratum corneum of atopic dermatitis: an etiologic factor in atopic dry skin? J Invest Dermatol. 1991; 96:523–26. https://doi.org/10.1111/1523-1747.ep12470233 [PubMed]
- 64. Bizot V, Cestone E, Michelotti A, Nobile V. Improving Skin Hydration and Age-related Symptoms by Oral Administration of Wheat Glucosylceramides and Digalactosyl Diglycerides: A Human Clinical Study. Cosmetics. 2017; 4:37. https://doi.org/10.3390/cosmetics4040037
- 65. Huang HC, Chang TM. Ceramide 1 and ceramide 3 act synergistically on skin hydration and the transepidermal water loss of sodium lauryl sulfate-irritated skin. Int J Dermatol. 2008; 47:812–19. https://doi.org/10.1111/j.1365-4632.2008.03687.x [PubMed]
- 66. McAleer MA, Irvine AD. The multifunctional role of filaggrin in allergic skin disease. J Allergy Clin Immunol. 2013; 131:280–91. https://doi.org/10.1016/j.jaci.2012.12.668 [PubMed]
- 67. Choi EH, Man MQ, Wang F, Zhang X, Brown BE, Feingold KR, Elias PM. Is endogenous glycerol a determinant of stratum corneum hydration in humans? J Invest Dermatol. 2005; 125:288–93. https://doi.org/10.1111/j.0022-202X.2005.23799.x [PubMed]
- 68. Fluhr JW, Mao-Qiang M, Brown BE, Wertz PW, Crumrine D, Sundberg JP, Feingold KR, Elias PM. Glycerol regulates stratum corneum hydration in sebaceous gland deficient (asebia) mice. J Invest Dermatol. 2003; 120:728–37. https://doi.org/10.1046/j.1523-1747.2003.12134.x [PubMed]
- 69. Ma T, Hara M, Sougrat R, Verbavatz JM, Verkman AS. Impaired stratum corneum hydration in mice lacking epidermal water channel aquaporin-3. J Biol Chem. 2002; 277:17147–53. https://doi.org/10.1074/jbc.M200925200 [PubMed]
- 70. Hara M, Verkman AS. Glycerol replacement corrects defective skin hydration, elasticity, and barrier function in aquaporin-3-deficient mice. Proc Natl Acad Sci USA. 2003; 100:7360–65. https://doi.org/10.1073/pnas.1230416100 [PubMed]
- 71. Dumas M, Sadick NS, Noblesse E, Juan M, Lachmann-Weber N, Boury-Jamot M, Sougrat R, Verbavatz JM, Schnebert S, Bonté F. Hydrating skin by stimulating biosynthesis of aquaporins. J Drugs Dermatol. 2007 (Suppl ); 6:s20–24. [PubMed]
- 72. Chaudhuri RK, Bojanowski K. Improvement of hydration and epidermal barrier function in human skin by a novel compound isosorbide dicaprylate. Int J Cosmet Sci. 2017; 39:518–26. https://doi.org/10.1111/ics.12405 [PubMed]
- 73. Fluhr JW, Darlenski R, Lachmann N, Baudouin C, Msika P, De Belilovsky C, Hachem JP. Infant epidermal skin physiology: adaptation after birth. Br J Dermatol. 2012; 166:483–90. https://doi.org/10.1111/j.1365-2133.2011.10659.x [PubMed]
- 74. Zlotogorski A. Distribution of skin surface pH on the forehead and cheek of adults. Arch Dermatol Res. 1987; 279:398–401. https://doi.org/10.1007/BF00412626 [PubMed]
- 75. Marrakchi S, Maibach HI. Biophysical parameters of skin: map of human face, regional, and age-related differences. Contact Dermat. 2007; 57:28–34. https://doi.org/10.1111/j.1600-0536.2007.01138.x [PubMed]
- 76. Burry JS, Coulson HF, Esser I, Marti V, Melling SJ, Rawlings AV, Roberts G, Mills AK. Erroneous gender differences in axillary skin surface/sweat pH. Int J Cosmet Sci. 2001; 23:99–107. https://doi.org/10.1046/j.1467-2494.2001.00085.x [PubMed]
- 77. Yamamoto A, Serizawa S, Ito M, Sato Y. Effect of aging on sebaceous gland activity and on the fatty acid composition of wax esters. J Invest Dermatol. 1987; 89:507–12. https://doi.org/10.1111/1523-1747.ep12461009 [PubMed]
- 78. Fluhr JW, Kao J, Jain M, Ahn SK, Feingold KR, Elias PM. Generation of free fatty acids from phospholipids regulates stratum corneum acidification and integrity. J Invest Dermatol. 2001; 117:44–51. https://doi.org/10.1046/j.0022-202x.2001.01399.x [PubMed]
- 79. Man G, Mauro TM, Zhai Y, Kim PL, Cheung C, Hupe M, Crumrine D, Elias PM, Man MQ. Topical hesperidin enhances epidermal function in an aged murine model. J Invest Dermatol. 2015; 135:1184–87. https://doi.org/10.1038/jid.2014.486 [PubMed]
- 80. Behne MJ, Meyer JW, Hanson KM, Barry NP, Murata S, Crumrine D, Clegg RW, Gratton E, Holleran WM, Elias PM, Mauro TM. NHE1 regulates the stratum corneum permeability barrier homeostasis. Microenvironment acidification assessed with fluorescence lifetime imaging. J Biol Chem. 2002; 277:47399–406. https://doi.org/10.1074/jbc.M204759200 [PubMed]
- 81. Vávrová K, Henkes D, Strüver K, Sochorová M, Školová B, Witting MY, Friess W, Schreml S, Meier RJ, Schäfer-Korting M, Fluhr JW, Küchler S. Filaggrin deficiency leads to impaired lipid profile and altered acidification pathways in a 3D skin construct. J Invest Dermatol. 2014; 134:746–53. https://doi.org/10.1038/jid.2013.402 [PubMed]
- 82. Krien PM, Kermici M. Evidence for the existence of a self-regulated enzymatic process within the human stratum corneum -an unexpected role for urocanic acid. J Invest Dermatol. 2000; 115:414–20. https://doi.org/10.1046/j.1523-1747.2000.00083.x [PubMed]
- 83. Norman RA. Xerosis and pruritus in the elderly: recognition and management. Dermatol Ther. 2003; 16:254–59. https://doi.org/10.1046/j.1529-8019.2003.01635.x [PubMed]
- 84. Valdes-Rodriguez R, Stull C, Yosipovitch G. Chronic pruritus in the elderly: pathophysiology, diagnosis and management. Drugs Aging. 2015; 32:201–15. https://doi.org/10.1007/s40266-015-0246-0 [PubMed]
- 85. Ashida Y, Denda M. Dry environment increases mast cell number and histamine content in dermis in hairless mice. Br J Dermatol. 2003; 149:240–47. https://doi.org/10.1046/j.1365-2133.2003.05408.x [PubMed]
- 86. Denda M, Sato J, Tsuchiya T, Elias PM, Feingold KR. Low humidity stimulates epidermal DNA synthesis and amplifies the hyperproliferative response to barrier disruption: implication for seasonal exacerbations of inflammatory dermatoses. J Invest Dermatol. 1998; 111:873–78. https://doi.org/10.1046/j.1523-1747.1998.00364.x [PubMed]
- 87. Hu L, Mauro TM, Dang E, Man G, Zhang J, Lee D, Wang G, Feingold KR, Elias PM, Man MQ. Epidermal Dysfunction Leads to an Age-Associated Increase in Levels of Serum Inflammatory Cytokines. J Invest Dermatol. 2017; 137:1277–85. https://doi.org/10.1016/j.jid.2017.01.007 [PubMed]
- 88. Bollu PC, Sahota P. Sleep and Parkinson Disease. Mo Med. 2017; 114:381–86. [PubMed]
- 89. Hepburn M, Bollu PC, French B, Sahota P. Sleep Medicine: stroke and Sleep. Mo Med. 2018; 115:527–32. [PubMed]
- 90. Elias PM. Primary role of barrier dysfunction in the pathogenesis of atopic dermatitis. Exp Dermatol. 2018; 27:847–51. https://doi.org/10.1111/exd.13693 [PubMed]
- 91. Proksch E, Brasch J, Sterry W. Integrity of the permeability barrier regulates epidermal Langerhans cell density. Br J Dermatol. 1996; 134:630–38. https://doi.org/10.1111/j.1365-2133.1996.tb06961.x [PubMed]
- 92. Lin TK, Man MQ, Santiago JL, Park K, Roelandt T, Oda Y, Hupe M, Crumrine D, Lee HJ, Gschwandtner M, Thyssen JP, Trullas C, Tschachler E, et al. Topical antihistamines display potent anti-inflammatory activity linked in part to enhanced permeability barrier function. J Invest Dermatol. 2013; 133:469–78. https://doi.org/10.1038/jid.2012.335 [PubMed]
- 93. Proksch E, Brasch J. Influence of epidermal permeability barrier disruption and Langerhans’ cell density on allergic contact dermatitis. Acta Derm Venereol. 1997; 77:102–04. [PubMed]
- 94. Katoh N, Hirano S, Kishimoto S, Yasuno H. Acute cutaneous barrier perturbation induces maturation of Langerhans’ cells in hairless mice. Acta Derm Venereol. 1997; 77:365–69. [PubMed]
- 95. Tanei R, Hasegawa Y. Atopic dermatitis in older adults: A viewpoint from geriatric dermatology. Geriatr Gerontol Int. 2016 (Suppl 1); 16:75–86. https://doi.org/10.1111/ggi.12771 [PubMed]
- 96. Prakash AV, Davis MD. Contact dermatitis in older adults: a review of the literature. Am J Clin Dermatol. 2010; 11:373–81. https://doi.org/10.2165/11319290-000000000-00000 [PubMed]
- 97. Jinnestål CL, Belfrage E, Bäck O, Schmidtchen A, Sonesson A. Skin barrier impairment correlates with cutaneous Staphylococcus aureus colonization and sensitization to skin-associated microbial antigens in adult patients with atopic dermatitis. Int J Dermatol. 2014; 53:27–33. https://doi.org/10.1111/ijd.12198 [PubMed]
- 98. Wanke I, Skabytska Y, Kraft B, Peschel A, Biedermann T, Schittek B. Staphylococcus aureus skin colonization is promoted by barrier disruption and leads to local inflammation. Exp Dermatol. 2013; 22:153–55. https://doi.org/10.1111/exd.12083 [PubMed]
- 99. Korting HC, Hübner K, Greiner K, Hamm G, Braun-Falco O. Differences in the skin surface pH and bacterial microflora due to the long-term application of synthetic detergent preparations of pH 5.5 and pH 7.0. Results of a crossover trial in healthy volunteers. Acta Derm Venereol. 1990; 70:429–31. [PubMed]
- 100. Rippke F, Berardesca E, Weber TM. pH and Microbial Infections. Curr Probl Dermatol. 2018; 54:87–94. https://doi.org/10.1159/000489522 [PubMed]
- 101. Chirife J, Herszage L, Joseph A, Kohn ES. Effect of potassium sorbate and pH on the survival of Staphylococcus aureus in saturated sugar solutions. Rev Argent Microbiol. 1993; 25:80–87. [PubMed]
- 102. Giannuzzi L, Contreras E, Zaritzky N. Modeling the aerobic growth and decline of Staphylococcus aureus as affected by pH and potassium sorbate concentration. J Food Prot. 1999; 62:356–62. https://doi.org/10.4315/0362-028X-62.4.356 [PubMed]
- 103. Bernard D, Méhul B, Thomas-Collignon A, Simonetti L, Remy V, Bernard MA, Schmidt R. Analysis of proteins with caseinolytic activity in a human stratum corneum extract revealed a yet unidentified cysteine protease and identified the so-called “stratum corneum thiol protease” as cathepsin l2. J Invest Dermatol. 2003; 120:592–600. https://doi.org/10.1046/j.1523-1747.2003.12086.x [PubMed]
- 104. Sakabe J, Yamamoto M, Hirakawa S, Motoyama A, Ohta I, Tatsuno K, Ito T, Kabashima K, Hibino T, Tokura Y. Kallikrein-related peptidase 5 functions in proteolytic processing of profilaggrin in cultured human keratinocytes. J Biol Chem. 2013; 288:17179–89. https://doi.org/10.1074/jbc.M113.476820 [PubMed]
- 105. Egelrud T, Brattsand M, Kreutzmann P, Walden M, Vitzithum K, Marx UC, Forssmann WG, Mägert HJ. hK5 and hK7, two serine proteinases abundant in human skin, are inhibited by LEKTI domain 6. Br J Dermatol. 2005; 153:1200–03. https://doi.org/10.1111/j.1365-2133.2005.06834.x [PubMed]
- 106. Voegeli R, Rawlings AV, Breternitz M, Doppler S, Schreier T, Fluhr JW. Increased stratum corneum serine protease activity in acute eczematous atopic skin. Br J Dermatol. 2009; 161:70–77. https://doi.org/10.1111/j.1365-2133.2009.09142.x [PubMed]
- 107. Komatsu N, Saijoh K, Kuk C, Liu AC, Khan S, Shirasaki F, Takehara K, Diamandis EP. Human tissue kallikrein expression in the stratum corneum and serum of atopic dermatitis patients. Exp Dermatol. 2007; 16:513–19. https://doi.org/10.1111/j.1600-0625.2007.00562.x [PubMed]
- 108. Jang H, Matsuda A, Jung K, Karasawa K, Matsuda K, Oida K, Ishizaka S, Ahn G, Amagai Y, Moon C, Kim SH, Arkwright PD, Takamori K, et al. Skin pH Is the Master Switch of Kallikrein 5-Mediated Skin Barrier Destruction in a Murine Atopic Dermatitis Model. J Invest Dermatol. 2016; 136:127–35. https://doi.org/10.1038/JID.2015.363 [PubMed]
- 109. Zhu Y, Underwood J, Macmillan D, Shariff L, O’Shaughnessy R, Harper JI, Pickard C, Friedmann PS, Healy E, Di WL. Persistent kallikrein 5 activation induces atopic dermatitis-like skin architecture independent of PAR2 activity. J Allergy Clin Immunol. 2017; 140:1310–1322.e5. https://doi.org/10.1016/j.jaci.2017.01.025 [PubMed]
- 110. Stefansson K, Brattsand M, Roosterman D, Kempkes C, Bocheva G, Steinhoff M, Egelrud T. Activation of proteinase-activated receptor-2 by human kallikrein-related peptidases. J Invest Dermatol. 2008; 128:18–25. https://doi.org/10.1038/sj.jid.5700965 [PubMed]
- 111. Kalinska M, Meyer-Hoffert U, Kantyka T, Potempa J. Kallikreins - The melting pot of activity and function. Biochimie. 2016; 122:270–82. https://doi.org/10.1016/j.biochi.2015.09.023 [PubMed]
- 112. Hansson L, Bäckman A, Ny A, Edlund M, Ekholm E, Ekstrand Hammarström B, Törnell J, Wallbrandt P, Wennbo H, Egelrud T. Epidermal overexpression of stratum corneum chymotryptic enzyme in mice: a model for chronic itchy dermatitis. J Invest Dermatol. 2002; 118:444–49. https://doi.org/10.1046/j.0022-202x.2001.01684.x [PubMed]
- 113. Kilic A, Masur C, Reich H, Knie U, Dähnhardt D, Dähnhardt-Pfeiffer S, Abels C. Skin acidification with a water-in-oil emulsion (pH 4) restores disrupted epidermal barrier and improves structure of lipid lamellae in the elderly. J Dermatol. 2019; 46:457–65. https://doi.org/10.1111/1346-8138.14891 [PubMed]
- 114. Angelova-Fischer I, Fischer TW, Abels C, Zillikens D. Accelerated barrier recovery and enhancement of the barrier integrity and properties by topical application of a pH 4 vs. a pH 5·8 water-in-oil emulsion in aged skin. Br J Dermatol. 2018; 179:471–77. https://doi.org/10.1111/bjd.16591 [PubMed]
- 115. Lee NR, Lee HJ, Yoon NY, Kim D, Jung M, Choi EH. Application of Topical Acids Improves Atopic Dermatitis in Murine Model by Enhancement of Skin Barrier Functions Regardless of the Origin of Acids. Ann Dermatol. 2016; 28:690–96. https://doi.org/10.5021/ad.2016.28.6.690 [PubMed]
- 116. Lee HJ, Lee NR, Kim BK, Jung M, Kim DH, Moniaga CS, Kabashima K, Choi EH. Acidification of stratum corneum prevents the progression from atopic dermatitis to respiratory allergy. Exp Dermatol. 2017; 26:66–72. https://doi.org/10.1111/exd.13144 [PubMed]
- 117. Lee HJ, Yoon NY, Lee NR, Jung M, Kim DH, Choi EH. Topical acidic cream prevents the development of atopic dermatitis- and asthma-like lesions in murine model. Exp Dermatol. 2014; 23:736–41. https://doi.org/10.1111/exd.12525 [PubMed]
- 118. Hatano Y, Man MQ, Uchida Y, Crumrine D, Scharschmidt TC, Kim EG, Mauro TM, Feingold KR, Elias PM, Holleran WM. Maintenance of an acidic stratum corneum prevents emergence of murine atopic dermatitis. J Invest Dermatol. 2009; 129:1824–35. https://doi.org/10.1038/jid.2008.444 [PubMed]
- 119. Mao-Qiang M, Brown BE, Wu-Pong S, Feingold KR, Elias PM. Exogenous nonphysiologic vs physiologic lipids. Divergent mechanisms for correction of permeability barrier dysfunction. Arch Dermatol. 1995; 131:809–16. https://doi.org/10.1001/archderm.1995.01690190063012 [PubMed]
- 120. Grygiel-Górniak B. Peroxisome proliferator-activated receptors and their ligands: nutritional and clinical implications—a review. Nutr J. 2014; 13:17. https://doi.org/10.1186/1475-2891-13-17 [PubMed]
- 121. Man MQ, Choi EH, Schmuth M, Crumrine D, Uchida Y, Elias PM, Holleran WM, Feingold KR. Basis for improved permeability barrier homeostasis induced by PPAR and LXR activators: liposensors stimulate lipid synthesis, lamellar body secretion, and post-secretory lipid processing. J Invest Dermatol. 2006; 126:386–92. https://doi.org/10.1038/sj.jid.5700046 [PubMed]
- 122. Sheu MY, Fowler AJ, Kao J, Schmuth M, Schoonjans K, Auwerx J, Fluhr JW, Man MQ, Elias PM, Feingold KR. Topical peroxisome proliferator activated receptor-alpha activators reduce inflammation in irritant and allergic contact dermatitis models. J Invest Dermatol. 2002; 118:94–101. https://doi.org/10.1046/j.0022-202x.2001.01626.x [PubMed]
- 123. Hatano Y, Man MQ, Uchida Y, Crumrine D, Mauro TM, Feingold KR, Elias PM, Holleran WM. Murine atopic dermatitis responds to peroxisome proliferator-activated receptors alpha and beta/delta (but not gamma) and liver X receptor activators. J Allergy Clin Immunol. 2010; 125:160–9.e1–5. https://doi.org/10.1016/j.jaci.2009.06.049 [PubMed]
- 124. Haratake A, Ikenaga K, Katoh N, Uchiwa H, Hirano S, Yasuno H. Topical mevalonic acid stimulates de novo cholesterol synthesis and epidermal permeability barrier homeostasis in aged mice. J Invest Dermatol. 2000; 114:247–52. https://doi.org/10.1046/j.1523-1747.2000.00875.x [PubMed]
- 125. Cosgrove MC, Franco OH, Granger SP, Murray PG, Mayes AE. Dietary nutrient intakes and skin-aging appearance among middle-aged American women. Am J Clin Nutr. 2007; 86:1225–31. https://doi.org/10.1093/ajcn/86.4.1225 [PubMed]
- 126. Guillou S, Ghabri S, Jannot C, Gaillard E, Lamour I, Boisnic S. The moisturizing effect of a wheat extract food supplement on women’s skin: a randomized, double-blind placebo-controlled trial. Int J Cosmet Sci. 2011; 33:138–43. https://doi.org/10.1111/j.1468-2494.2010.00600.x [PubMed]
- 127. Boisnic S, Keophiphath M, Serandour AL, Branchet MC, Le Breton S, Lamour I, Gaillard E. Polar lipids from wheat extract oil improve skin damages induced by aging: evidence from a randomized, placebo-controlled clinical trial in women and an ex vivo study on human skin explant. J Cosmet Dermatol. 2019; 18:2027–36. https://doi.org/10.1111/jocd.12967 [PubMed]
- 128. Puch F, Samson-Villeger S, Guyonnet D, Blachon JL, Rawlings AV, Lassel T. Consumption of functional fermented milk containing borage oil, green tea and vitamin E enhances skin barrier function. Exp Dermatol. 2008; 17:668–74. https://doi.org/10.1111/j.1600-0625.2007.00688.x [PubMed]
- 129. De Spirt S, Stahl W, Tronnier H, Sies H, Bejot M, Maurette JM, Heinrich U. Intervention with flaxseed and borage oil supplements modulates skin condition in women. Br J Nutr. 2009; 101:440–45. https://doi.org/10.1017/S0007114508020321 [PubMed]
- 130. Chen Y, Yokozeki H, Katagiri K. Physiological and functional changes in the stratum corneum restored by oestrogen in an ovariectomized mice model of climacterium. Exp Dermatol. 2017; 26:394–401. https://doi.org/10.1111/exd.13214 [PubMed]
- 131. Thornton MJ. Estrogens and aging skin. Dermatoendocrinol. 2013; 5:264–70. https://doi.org/10.4161/derm.23872 [PubMed]
- 132. Agero AL, Verallo-Rowell VM. A randomized double-blind controlled trial comparing extra virgin coconut oil with mineral oil as a moisturizer for mild to moderate xerosis. Dermatitis. 2004; 15:109–16. https://doi.org/10.2310/6620.2004.04006 [PubMed]
- 133. Danby SG, AlEnezi T, Sultan A, Lavender T, Chittock J, Brown K, Cork MJ. Effect of olive and sunflower seed oil on the adult skin barrier: implications for neonatal skin care. Pediatr Dermatol. 2013; 30:42–50. https://doi.org/10.1111/j.1525-1470.2012.01865.x [PubMed]
- 134. Vaughn AR, Clark AK, Sivamani RK, Shi VY. Natural Oils for Skin-Barrier Repair: Ancient Compounds Now Backed by Modern Science. Am J Clin Dermatol. 2018; 19:103–17. https://doi.org/10.1007/s40257-017-0301-1 [PubMed]
- 135. Lin TK, Zhong L, Santiago JL. Anti-Inflammatory and Skin Barrier Repair Effects of Topical Application of Some Plant Oils. Int J Mol Sci. 2017; 19:E70. https://doi.org/10.3390/ijms19010070 [PubMed]
- 136. Fluhr JW, Gloor M, Lehmann L, Lazzerini S, Distante F, Berardesca E. Glycerol accelerates recovery of barrier function in vivo. Acta Derm Venereol. 1999; 79:418–21. https://doi.org/10.1080/000155599750009825 [PubMed]
- 137. Kottner J, Kanti V, Dobos G, Hahnel E, Lichterfeld-Kottner A, Richter C, Hillmann K, Vogt A, Blume-Peytavi U. The effectiveness of using a bath oil to reduce signs of dry skin: A randomized controlled pragmatic study. Int J Nurs Stud. 2017; 65:17–24. https://doi.org/10.1016/j.ijnurstu.2016.10.010 [PubMed]
- 138. Blaak J, Dähnhardt D, Dähnhardt-Pfeiffer S, Bielfeldt S, Wilhelm KP, Wohlfart R, Staib P. A plant oil-containing pH 4 emulsion improves epidermal barrier structure and enhances ceramide levels in aged skin. Int J Cosmet Sci. 2017; 39:284–91. https://doi.org/10.1111/ics.12374 [PubMed]
- 139. Di Marzio L, Cinque B, Cupelli F, De Simone C, Cifone MG, Giuliani M. Increase of skin-ceramide levels in aged subjects following a short-term topical application of bacterial sphingomyelinase from Streptococcus thermophilus. Int J Immunopathol Pharmacol. 2008; 21:137–43. https://doi.org/10.1177/039463200802100115 [PubMed]
- 140. Czarnowicki T, Malajian D, Khattri S, Correa da Rosa J, Dutt R, Finney R, Dhingra N, Xiangyu P, Xu H, Estrada YD, Zheng X, Gilleaudeau P, Sullivan-Whalen M, et al. Petrolatum: barrier repair and antimicrobial responses underlying this “inert” moisturizer. J Allergy Clin Immunol. 2016; 137:1091–1102.e7. https://doi.org/10.1016/j.jaci.2015.08.013 [PubMed]
- 141. Páyer E, Szabó-Papp J, Ambrus L, Szöllősi AG, Andrási M, Dikstein S, Kemény L, Juhász I, Szegedi A, Bíró T, Oláh A. Beyond the physico-chemical barrier: glycerol and xylitol markedly yet differentially alter gene expression profiles and modify signalling pathways in human epidermal keratinocytes. Exp Dermatol. 2018; 27:280–84. https://doi.org/10.1111/exd.13493 [PubMed]
- 142. Huang LN, Zhong YP, Liu D, Wang XH, Gong CY, Wen S, Elias PM, Yang B, Man MQ. Adverse cutaneous reactions to skin care products on the face vary with age, but not with sex. Contact Dermat. 2018; 79:365–69. https://doi.org/10.1111/cod.13102 [PubMed]
- 143. Liu D, Wen S, Huang LN, Wang X, Gong CY, Li Z, Wang H, Elias PM, Yang B, Man MQ. Comparison of transepidermal water loss rates in subjects with skin patch test positive vs negative to skin care products. J Cosmet Dermatol. 2019. [Epub ahead of print]. https://doi.org/10.1111/jocd.13264 [PubMed]
- 144. Kwa M, Welty LJ, Xu S. Adverse Events Reported to the US Food and Drug Administration for Cosmetics and Personal Care Products. JAMA Intern Med. 2017; 177:1202–04. https://doi.org/10.1001/jamainternmed.2017.2762 [PubMed]
- 145. Li Z, Hu L, Elias PM, Man MQ. Skin care products can aggravate epidermal function: studies in a murine model suggest a pathogenic role in sensitive skin. Contact Dermat. 2018; 78:151–58. https://doi.org/10.1111/cod.12909 [PubMed]
- 146. Huang YS, Huang WC, Li CW, Chuang LT. Eicosadienoic acid differentially modulates production of pro-inflammatory modulators in murine macrophages. Mol Cell Biochem. 2011; 358:85–94. https://doi.org/10.1007/s11010-011-0924-0 [PubMed]
- 147. Miao H, Chen L, Hao L, Zhang X, Chen Y, Ruan Z, Liang H. Stearic acid induces proinflammatory cytokine production partly through activation of lactate-HIF1α pathway in chondrocytes. Sci Rep. 2015; 5:13092. https://doi.org/10.1038/srep13092 [PubMed]
- 148. Anderson EK, Hill AA, Hasty AH. Stearic acid accumulation in macrophages induces toll-like receptor 4/2-independent inflammation leading to endoplasmic reticulum stress-mediated apoptosis. Arterioscler Thromb Vasc Biol. 2012; 32:1687–95. https://doi.org/10.1161/ATVBAHA.112.250142 [PubMed]


