Introduction
The nature of the selection factors underlying the evolution of aging remains controversial [1-3]. Many specialists in evolutionary gerontology support a set of ideas called the “evolutionary theory of aging” [1, 2]. This theory is based on the idea that the selection efficiency decreases with age. It is also assumed that vitality and fertility are high in youth at the cost of reduced fitness at later ages [4, 5]. An alternative view is that programmed aging and death may be favored by some kind of selection [3, 6–16].
A subsequent theoretical experiment called the “Fable about Fox and Hares” was suggested by one of the authors of this paper (VPS) [17]. Two young hares differing “intellectually” have equal chances to escape from a fox since both of hares are running faster than a fox. However, with age, the clever hare acquires some advantage, which becomes of crucial importance when the running speed of hares lowers to that of a fox. Now, the clever hare has a better chance to escape and, hence, to produce clever leverets than the stupid hare. Such an effect becomes possible due to age-dependent lowering of the running speed as a result of the operation of an aging program. This will facilitate the selection for cleverness. Recently, attempts have been made to analyze the fable by computer modeling [18, 19].
The retardation of the operation of chronic aging programs in humans [13, 20–23] and in naked mole rats [13, 24–26] and the acute senile phenoptosis in the nematode Caenorhabditis elegans [12, 14, 15, 27–29] strongly supports the idea that programmed aging plays at present an important role in the abovementioned three animal species.
It is clear that the longevity in highly social mammals, such as H. glaber and humans, is partly due to neoteny, i.e. prolongation of youth and retardation of aging. The aging in both species cannot at present promote evolvability: in H. glaber – due to its hierarchy where only the “queen” and her “husbands” participate in breeding, and in humans – due to the rapid technological progress that in fact replaces the very much slower biological evolution [13]. As to C. elegans, the operation of the aging programs is clearly needed for the production of yolk [29] rather than for the stimulation of natural selection.
This paper is devoted to a comparative analysis of the survival curves of humans and related primates and the possible role of neoteny and other mammalian anti-aging programs in lifespan (LS) prolongation.
Possible mechanisms of changes in the survival curves
Various characteristics of LS have been used for survival curve analysis. One important factor is the heterogeneity of mortality causes, which can be divided into internal factors (intrinsic mortality) and external influences (extrinsic mortality) [30–33].
Terminal-to-average mortality ratio
Jones et al. [33] compared the mortality of Ache Indians, Swedish females (born in 1881), Japanese females (died in 2009), two species of nonhuman primates and 40 other species of animals and plants. The authors analyzed the interval between (i) the onset of sexual maturation and (ii) “terminal” age corresponding to the 95% mortality of the original sexually mature cohort (LS95). Their criterion (mortality at “terminal” age versus the average mortality rate for the entire study period) led to several obviously erroneous conclusions. For instance, the great tit Parus major was assigned to the non-aging species and placed next to hydra. However, P. major just does not have time to grow old owing to a high age-independent mortality. LS95 for the pine Pinus silvestris was as short as 30 years due to the high mortality of the young trees. Tortoise was declared as the most non-aging animal, while crocodile was in the middle of the list. Moreover, long-lived humans (Japanese and Swedes) and animals (southern fulmar) were classified as the species with the most pronounced aging, despite having a long period of a negligibly low mortality rate.
Coefficient of variation of lifespan (CVLS)
It is assumed that we can expect a high variance in LS (i.e., high CVLS values) in the studied population when the degree of genetic regulation of aging is low (i.e., poorly controlled LS) and the rate of background “extrinsic” mortality is high. On the other hand, the LS variability in the population should be low when LS is stringently regulated by the genotype. In this case, it can be assumed that the “true” value of the species LS will be the value in the case of a smaller relative variation of LS. Upon great variation, it can be assumed that the external causes (predators, hunger, etc.) have a higher impact on mortality, while the genome-encoded mechanisms of longevity assurance play a secondary role.
N. Gavrilova, L. Gavrilova, F. Severin and V. Skulachev [34] compared the coefficient of variation (CV) of the parameter determined by the female H. sapiens development program (puberty age) and the aging-related parameters (menopause and death age). The data from the National Survey of the Midlife Development in the U.S. (MIDUS) were used. It has been found that the CV = 8–13% for the age of the onset of puberty, CV = 7-11% for the age of the onset of menopause, and CV = 16–21% for the age of death. Two results of these calculations are noteworthy. (A) The CV values for puberty and menopause coincide, which is predicted by the programmed aging concept since both events should be controlled by an ontogenetic clock. The alternative concept considering aging as a result of stochastic accumulation of damage cannot explain the above coincidences of CVs for puberty and menopause. (B) Variability of the age of death was two times higher than for puberty and menopause. Such relationships are not surprising since, in addition to programmed aging, there are external death factors that increase the variability of death.
Thus, the programmed aging has been demonstrated with a statistical method.
CVLS estimates based on the Jones et al. [33] data from people >10 years old were 16.2% ± 0.1% for the Japanese females, 37.1% ± 0.3% for the Swedish females, and 54.5% ± 0.3% for the Ache Indians. For comparison, CVLS was 59.2% ± 0.2% in >7-year-old chimpanzees [puberty age] and 53.9% ± 0.1% in baboon females in 4 years old [35]. At the same time, the LS95 was 24 years for the baboon females, 49 years for the chimpanzee females, 81 years for the Ache Indians, 89 years for the Swedish females and 102 years for the Japanese females [33]. It is important that Ache hunter-gatherers have, as a rule, minimal (near zero) dependence on modern medicine and do not use any commercial products in their diets [36].
These data are consistent with the idea that the observed variation in human LS in less developed societies is determined to a larger extent by unpredictable extrinsic factors, while the genetic component of variation becomes more prominent in more developed societies.
The human LS steadily grows in a series of generations. For instance, Oeppen and Vaupel [37] showed that the human life expectancy at birth (e0) was growing almost linearly from 1840 to 2000 in developed countries, with an increase of approximately 3 months per year. This growth is due to a rapid steady decline in mortality at all ages [38], with the exception of the mortality of centenarians [39].
The analysis of the following characteristics of the human mortality profile is an important task in the study of the evolution of aging. Among these characteristics are the mortality curve shape; the variability of mortality profile across generations; the presence of the postreproductive life period; and the modal age of death (i.e., the age when mortality is maximal).
Technical progress as a factor that modifies the survival curves
The hunter-gatherers have a longer juvenile period of elevated mortality, and a longer adult LS as compared to chimpanzees (Figure 1A).
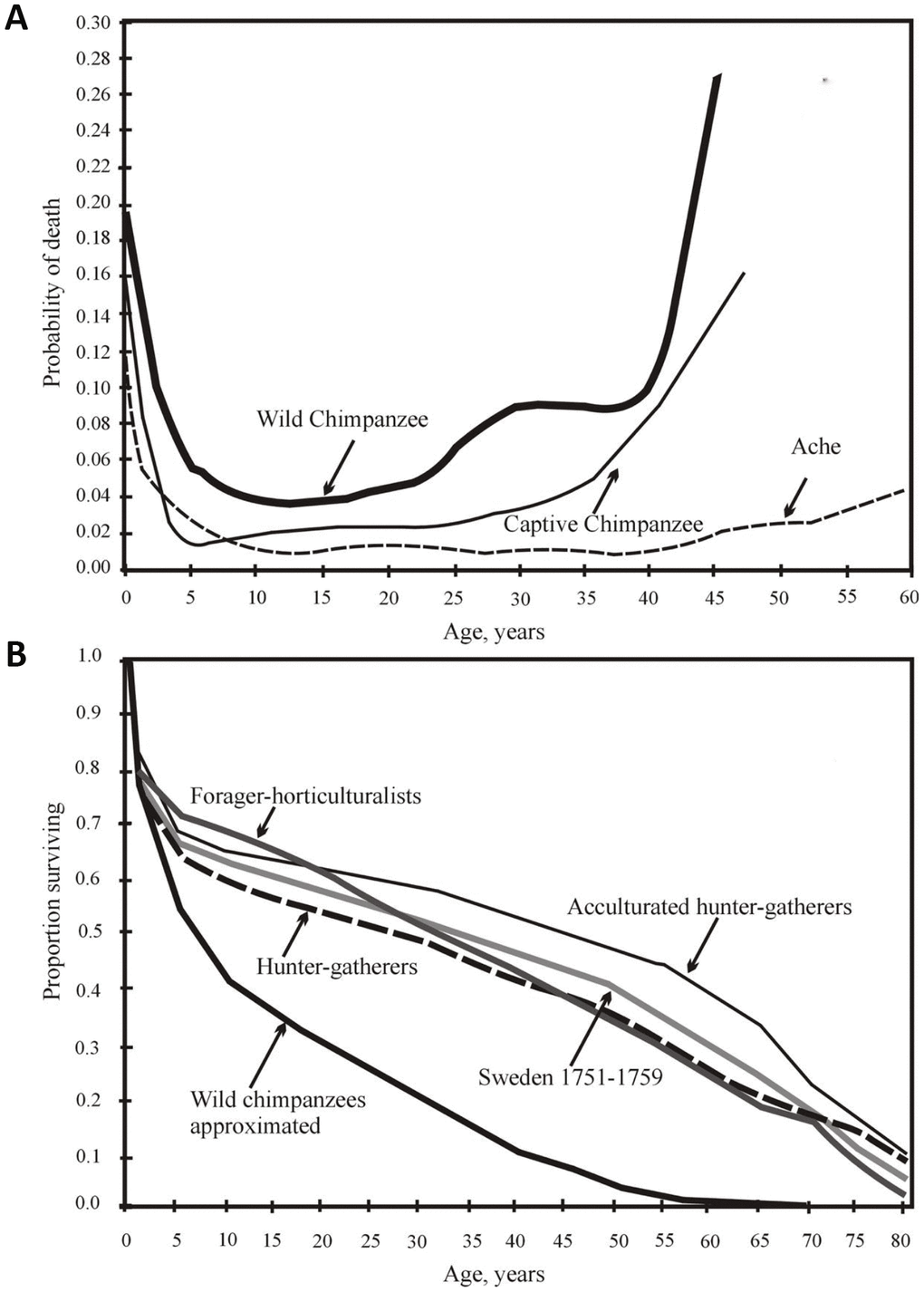
Figure 1. (A) from [44], with minor modifications. Yearly mortality of captive and wild chimpanzees [data from 43] and Ache Indians of Paraguay [32]). (B) from [47]. Survival of chimpanzees in the wild and the survival of various wild tribes of South America, Africa and Asia, and Swedes in 1751-1759.
The mortality rates vary among different human populations and between different time periods, especially regarding the risk of sudden death. However, these differences are small from a comparative cross-species perspective. The mortality profile similarity among different preindustrial populations living in different conditions is evident (Figure 1B).
Dental and renal diseases and cerebrovascular pathologies, as well as uterine leiomyomas, are common age-related pathologies of humans and apes. It was previously believed that brain volume reduction, breast and prostate cancer, lung cancer and colorectal cancer, gout and Alzheimer’s disease are characteristic only of aging humans, not of apes [40]. However, the presence of the two classic damaging signs of Alzheimer’s disease (the accumulation of amyloid β and phosphorylation tau proteins with age) were demonstrated in old chimpanzees in 2017, indicating that this pathology is not specific to humans [41]. These signs had been previously described in Oncorhynchus during the spawning period [42].
Life expectancy for humans, wild chimpanzees and captive chimpanzees
Captivity significantly increases the survival of chimpanzee infants and adolescents. The percentage of those who live to 15 years grows from 37% in the wild to 64% in captivity [43]. However, although the proportion of animals surviving to the age of 45 increases sevenfold (from 3% in the wild to 20% in captivity), it is still half as high as that for humans living in primitive conditions [44]. The difference between the chimpanzees and humans over 45 years old is even greater; the additional life expectancy (e45) for the captive chimpanzees is only 7 years, which is about one-third that of the e45 for humans. Thus, chimpanzees age much faster than humans and die earlier, even under external protection [44].
Forty-seven percent of the 15-year-old captive chimpanzees lived to the age of 47 years old, the maximal observed age for wild chimpanzees [44]. Thus, the aging (i.e., an increase in death probability with age) of a chimpanzee under wild conditions is largely determined by external causes and to a much lesser extent by internal causes. H. sapiens are generally characterized by a sharp decline in mortality from infancy to adolescence; mortality then remains very low up to the age of 40 years, after which it steadily increases (Figure 1A).
All the survival curves of Native American populations of H. sapiens proved to be at a considerable distance from the survival curve of wild chimpanzees. Additionally, despite all the differences (lifestyle, food, etc.), all human survival curves are extremely similar, intersecting at 80 years, that is, at the point corresponding to LS95 (Figure 1B).
This again demonstrates that LS95 is well suited for use in the analysis of the survival curves of aging populations (where the probability of the death of an individual increases with age): mice, rats, fruit flies, and nematodes [45]. However, LS95 is less convenient for the analysis of non-aging populations (by this criterion), for example, for H. glaber or higher plant populations [46].
The life expectancies at birth (e0) among different aboriginal tribes are quite similar even across the continents. The values of e0, in ascending order, are as follows: Hiwi (Venezuela) - 32 years, Hadza (Tanzania) – 33 years, !Kung (the Kalahari desert between Botswana and Namibia) and the XVIII century Swedish individuals – 35 years, Ache (Paraguay) – 38 years, Agta (Philippines) and Yanomamo (Venezuela) – 41 years, and Tsimane (Bolivia) – 42 years [47].
The probability of living to the age of 15 is also similar in all preagricultural tribes (from 55% in Hadza to 71% in Ache). The average value of this probability for all hunter-gatherers is 60%, while the average value for various groups of captive chimpanzees is 35% [36]. The 15-year-old hunter-gatherers will live, on average, 39 years more, whereas 15-year-old chimpanzees will live only 15 years more. Fifteen percent of all hunter-gatherers will live to 70 years and 5% to 80 years (i.e., up to the LS95 age) [36]. The maximal human LS is 122.5 years [48], almost twice the value for the chimpanzees (62 years in captivity [43]), which may partially be due to very different sample sizes for humans (many millions of individuals) and chimpanzees (thousands of individuals).
The shape of the distribution of lifespans can be assessed in other ways, including the coefficient of variation of lifespan and Keyfitz’s entropy [49]. In 2012-2017 [34, 35], we suggested that the coefficient of variation can serve as an additional characteristic of the stability of mortality dynamics [35]. Similarly, [49], an equality indicator, based on the measuring of Keyfitz’s entropy, was proposed by Colchero et al. In 2016 [49]. Keyfitz’s entropy is given by the ratio e†/e0, where e† measures life expectancy lost due to the death: e†
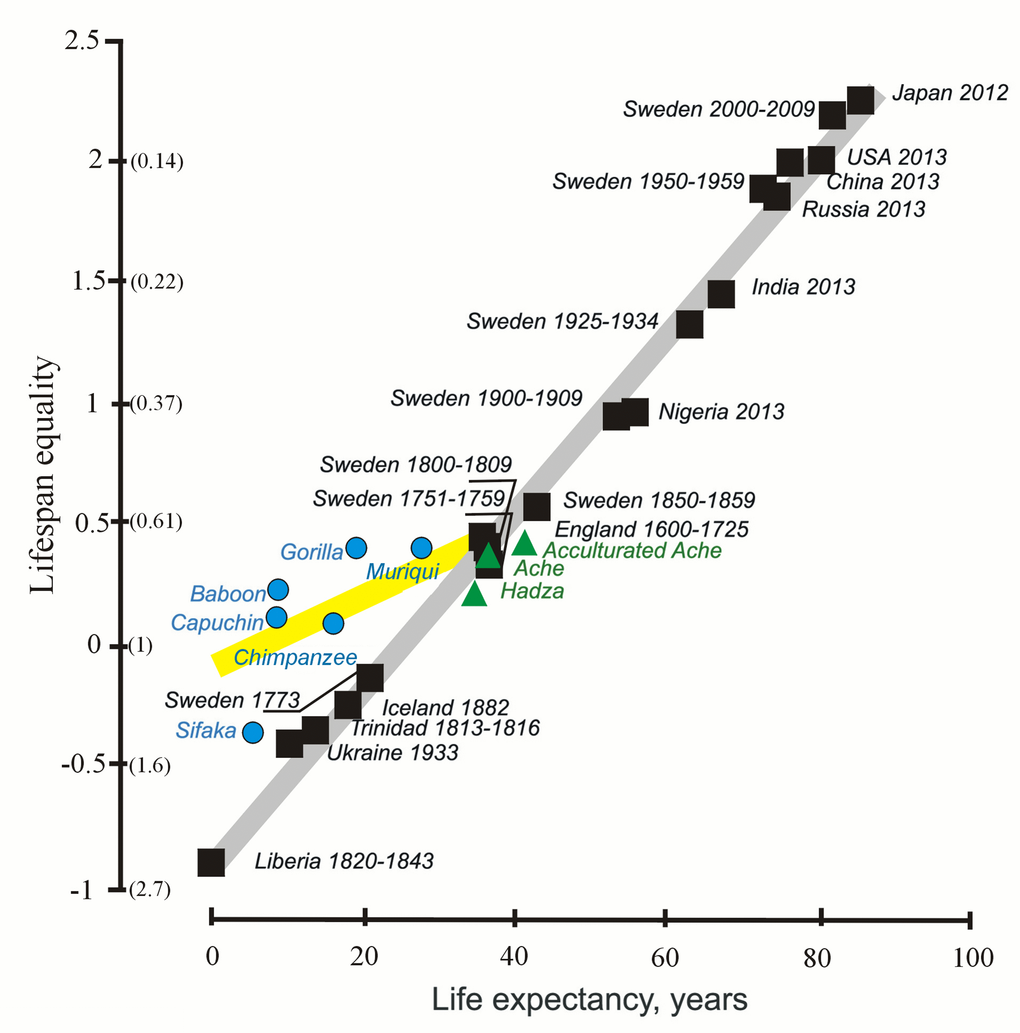
Figure 2. The lifespan equality and life expectancy in humans (black and green) and non-human primates (blue) (from [49], with minor modifications). The y axis shows lifespan equality, the log of the inverse of the Keyfitz’s entropy; corresponding values of the Keyftiz’s entropy are given in parentheses on the y axis.
Mortality rate doubling time (MRDT)
The rate at which the mortality rate doubles is the aging rate measure. It was reported that the mortality rate doubled every 7-8 years for a number of human populations with a very wide range of total mortality [47]. MRDT for the studied preagricultural populations is also mainly within the range of 6-10 years (for example, 6 years for Hadza, 7 years for Ache and 9 years for !Kung) [47].
Dependence on the initial mortality rate is an obvious disadvantage of this parameter. For instance, if the initial mortality rate is 0.01% per year, then it will soon double, while the mortality rate of up to 20% per year may not double throughout the lifetime of the cohort.
Therefore, it is hardly surprising that the MRDTs of various preagricultural tribes are so different (only 2.8 years for Hiwi and 12–18 years for Agta) [47].
The age-dependent dynamics of the probability of death are surprisingly similar among different aboriginal tribes [47]. The mortality rate slows down to 1% per year at the age of 10 years, and then the mortality remains low to approximately 40 years old despite the obvious mortality rate acceleration with age [47]. The results obtained in the groups of hunter-gatherers (Figure 1B and 3A) are similar to the results from Sweden in 1751 (the post-Charles XII period). For example, the life expectancy at birth (e0) was 34 years, and e45 promised an additional 20 years. It turned out that at least one-fourth of the population would live for 15–20 years more after 45 years (without access to modern medical care, public sanitation, immunization, and a predictable food supply) [47].
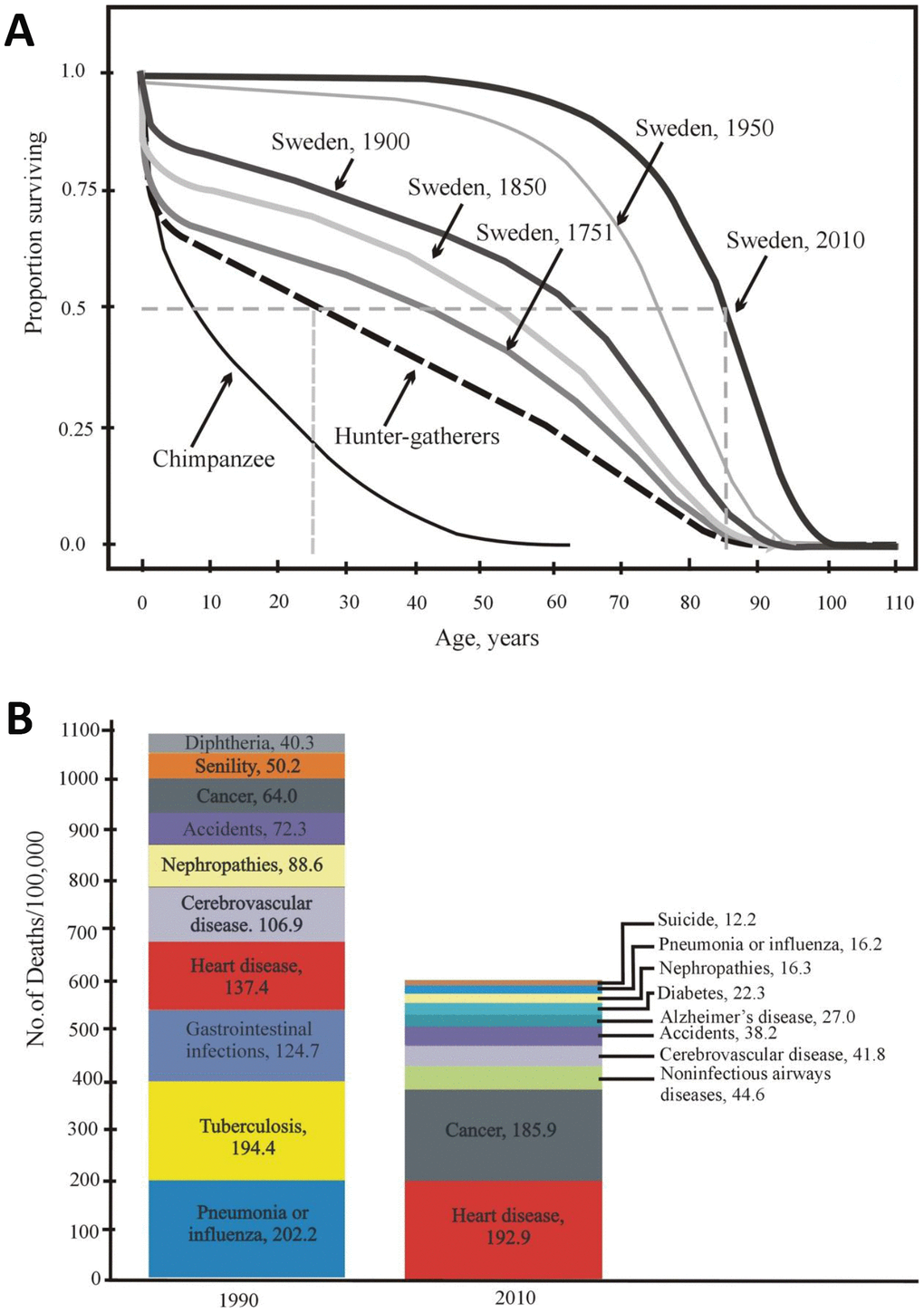
Figure 3. (A) Age-dependent survival of Swedish individuals (in 1751, 1850, 1900, 1950, and 2010) and of Ache Indians and chimpanzees (according to [50, 51] with modifications). (B) Mortality structure in the USA (10 main causes of death in 1900 and 2010) [52].
Currently, Swedish and Japanese individuals practically show negligible mortality until 50-60 years old. Not more than 10% die over 55 years of life, and not more than 70% of the original population dies within 55-80 years of life. Over the last century, life expectancy at birth (e0) has increased in most countries and has exceeded 80 years in several countries [50]. Therefore, it is obvious that mortality in humans in developed countries has decreased so greatly at present that the difference between the hunter-gatherer mortality rate and the current mortality rate in developed countries today is much greater than the difference between hunter-gatherers and wild chimpanzees (Figure 3A).
Moreover, the reduction in human mortality is at the same level or exceeds that in other species, even in animals in captivity or subjected to life-extending mutations as well as laboratory selection for longer life. O. Burger notes that the observed plasticity of the age-related death risk contradicts the generally accepted theories of aging [50, 51], but it can be explained by the “theory of aging as part of a general program of ontogenesis” (see below).
The main part of the pronounced decline in mortality occurred in developed countries from 1900 to 2010 (approximately 4 human generations). Figure 3B shows the structure of mortality in the United States and its change over the aforementioned period from 1900 to 2010. Both earlier and now, diseases of the cardiovascular system contribute to the structure of mortality, taking first place in the number of deaths caused (32.2% in 2010 vs 12.5% in 1900) [52]. In addition, the role of neurodegenerative diseases deserves special attention. These pathologies are important, although they do not have an equally strong influence on the structure of mortality. For example, Alzheimer’s disease takes now sixth place on the list, i.e. 4.5% in 2010. However, neurodegenerative diseases have a very negative effect on the quality of life, socialization and self-sustainment ability of a human being. In fact, they greatly reduce the health span.
The change in survival curves of humans compared to chimpanzees occurs for two reasons: neoteny and very rapid technical progress. An analysis of time scales and survival curves allows us to separate these two causes. Thus, the evolution of neoteny requiring much more time may be responsible for the difference in the mortality curves of chimpanzees and hunter-gatherers, while technical progress is responsible for the great differences in the mortality curves of hunter-gatherers and Swedish individuals in the 20th century.
Aging subprograms as a part of ontogenetic program
The aging of an organism is manifested in an increase in the frequency of age-related diseases and, consequently, in an increasing probability of death [34]. The evolution of the aging phenomenon in Metazoa proceeded from potentially immortal forms. Potential immortality is an ancestral feature gradually lost in the course of evolution. At the same time, aging and death resulting from aging are facultative (rather than obligatory) features of Metazoa. The main principle of the evolution of aging is as follows: it is the substitution of external factors of death of non-aging Metazoa with internal ones programmed in the genome [17, 53]. If the probability of the death of an organism depended entirely on the degree of its age-related “wear”, then the mortality rate of multicellular organisms would increase with age regardless of the species position in the evolutionary tree. This has not been confirmed since the discovery of large differences in the mortality dynamics in different species (ascending, constant, descending, convex and concave mortality trajectories, both in the long-living and short-living species) [32, 33]. M.V. Skulachev and V.P. Skulachev [54, 55] pointed to this contradiction by proposing the acute phenoptosis concept. According to this concept, chronic phenoptosis (slow aging) can be finally substituted by a fast programmed death.
This concept originates from the assumption that septic shock is an example of pathology with signs of acute phenoptosis. Quick death (due to the massive release of special regulators to the blood) is apparently caused by the infected organism itself, presumably to remove the sick individual from the population in order to prevent an epidemic advance [17, 54, 60, 61]. Now it is suggested that aging (slow phenoptosis) results in such a crucial damage of physiological function that reproduction become impossible and the old organism is quickly killed by his own mechanism of acute phenoptosis to eliminate individual that cannot be involved in natural selection [54, 55]2.
Anti-aging programs. Transcription factor Nrf2
A transcription factor called “Nuclear factor erythroid 2-related factor 2 (Nrf2)” seems to be a component of an anti-aging program [62], as well as the repair enzymes [63]. Nrf2 is thought to be a guardian of the health span and a gatekeeper of species longevity [62]. It induces the expression of genes encoding ~200 repairing and detoxifying enzymes, including the most powerful natural antioxidants [64]. Nrf2 signaling activity is positively correlated with LS [62]. The level of Nrf2 decreases in old mice [65–68].
Protein antagonists of Nrf2 (β-TrCP, KEAP1, Bach1 and c-Myc) [64, 69, 70] and mitochondrial ROS generation can be components of aging programs. The first three proteins are inhibited by ROS, which makes the situation even more complicated than the simple competition of anti-aging and aging programs.
Keratin and heavy chain of myosin 16
The degradation of certain genes is one of the most important and “radical” evolutionary changes in the genome [71]. The human-specific loss of the heavy chain of myosin 16 (MYH16) gene occurred presumably due to a change in diet, which, in turn, reduced the dependence on powerful masticatory skeletal muscles. The loss (~2.4 million years ago) of MYH16 was probably a favorable adaptive change that released constraints on the increase in cranial volume and brain size [72]. In addition, the disappearance of the myosin heavy chain weakened the skeletal muscles of the limbs and reduced the overall weight of the body, although it also weakened the physical strength.
The transformation of a gene from a cluster of type I keratin genes (responsible for hair emergence during ontogenesis) into a nonfunctional pseudogene φhHaA (KRTHAP1) in humans was another relatively recent evolutionary event. Orthologs of this gene are functional in gorillas and chimpanzees[73].
Neoteny in some invertebrates
Neoteny has been described in many invertebrates, such as insects (termites [74], mayflies, cicadas [75, 76], beetles [77–79]) and isopod crustaceans [80]. The Turritopsis jellyfish, one of the representatives of the cnidarians, has a unique opportunity torevert, in response to mechanical damage or other adverse changes in external conditions, to an earlier state (polyp) from the stage of jellyfish that reproduce sexually [81]. As a result, the rejuvenation of this organism occurs.
The issue of the presence of neoteny in ants (Hymenoptera, Formicidae) is disputable. A comparison of queens and gamergates3 may help in the search for ant neoteny [82]. Neoteny is formally absent in the worker ants in the majority of species since they never reach sexual maturity. At the same time, it is possible that the very appearance of the gamergates and intercastes in ants in the course of evolution proceeded along a neoteny path [83–85]. The intercastes include the individuals that morphologically differ from the typical winged members of their sex and resemble workers in some respects (the so-called ergatoid females and males). In particular, their wing muscles are not developed. The intercaste females are heterogeneous in their reproductive function: some are involved in egg laying, and others perform the functions of workers [86]. The ergatoid males breed but are not capable of flying. Such males mate in the nest [87].
The gamergates are found in different subfamilies of Formicidae and are especially common in the most ancient Ponerinae subfamily [88–91]. The gamergates have no distinctive external features compared to workers and reproductive females. The gamergates develop from the same young individuals as the workers, and their ability to lay eggs is determined by interactions with other individuals [92, 93].
It is also possible that the caste of workers has been formed not through the development of neoteny but as a separate ontogenetic trajectory since many morphophysiological differences between the individuals in the families of ants are caused by phenotypic plasticity rather than by hereditary variation. In this regard, it is interesting that the size of the brain in the egg-laying gamergates can be reduced compared to the working females. In Harpegnathossaltator, the brain volume of gamergates is only 74% that of workers [94]. It is noteworthy that the Harpegnathos workers who stand out among the ants with very large eyes have extremely developed optical brain lobes, whereas just this part of the brain undergoes a strong reduction in gamergates that spend their whole life in the nest. The authors suggest that the nutrient-rich brain material is expended for egg production. A similar process caused by the lysis of the wing muscles in the fertilized ant females was described by Janet [95].
Neoteny in vertebrates
Among vertebrates, the Mexican salamander (Ambystoma mexicanum) is a classic example of neoteny; this animal can reproduce at the larval stage (axolotl). Its LS is more than 32 years. The transformation of an axolotl into an adult Ambystoma can be achieved by the addition of the hormone thyroxin. Moreover, A. tigrinum can transformed to adult Ambystoma even spontaneously under conditions which can be suspected to cause general disturbance in the central nervous system, e.g., when animals are caught and taken into the laboratory, or during periods of high temperature [96]. Among the caudate amphibians, there are species, for example, the European proteus (Proteus anguinus), whose adult form has not been described (obligate neoteny). Their maximal reported lifespan (in captivity) is 69 years [97], while the LS estimated value reaches 103 years [98]. A salamander resembling the axolotl but having no adult state (Necturus maculosus) lives for more than 29 years and survives only in water [99]. Neoteny has also been found in ray-finned fish [100, 101], lobe-finned lungfish (closely related to the Tetrapoda) [102] and some Passeriformes [103].
Neoteny in mammals, in addition to humans and H. glaber, was hypothesized for baleen whales [104] and toothed whales (dolphins) [105] as well as for dogs (“behavioral neoteny”) [106].
Naked mole rat neoteny
The H. glaber, a rodent species with an LS unusually high for its weight, has pronounced neotenic traits (for reviews, see [13, 26]). Senescence-related traits, such as muscular and fatty weight reduction, lipofuscin deposition in tissues, and cataracts, start to develop only by approximately 30 years of age in H. glaber [107, 108].
The neoteny of the lungs in H. glaber does not manifest itself as decisively as the neoteny of the hair: the hair completely disappears, and the lungs remain underdeveloped.
The DNA fingerprinting method demonstrated that H. glaber individuals within colonies were genetically almost monomorphic (coefficients of band sharing estimated from DNA fingerprints range from 0.93 to 0.99) [109]. H. glaber has a low level of heterozygosity (1.87 million heterozygous single-nucleotide polymorphisms per diploid genome or ~0.7 heterozygous sites per thousand nucleotides). Similarly, humans have significantly lower heterozygosity than mice and rats [110]. The morphological differences between individuals are probably formed mostly by epigenetic mechanisms. Other peculiar features of the H. glaber genome have also been demonstrated. For example, at least one of the two mammalian characteristic genes, Mtnr1a and Mtnr1b, is nonfunctional [110]. Mtnr1a and Mtnr1b are receptors of melatonin – a hormone responsible for biorhythm [110, 111]. Despite the presence of a corresponding gene, the expression of the last enzyme of the melatonin synthesis pathway (N-acetyl serotonin-O-methyltransferase) is suppressed in naked mole rats, at least in the brain, liver and kidneys [110, 112].
Moreover, the epiphysis has not been found in H. glaber ([113], single-animal study, confirmed by V. Manskikh in our group). H. glaber does not respond significantly to the increased Fos gene expression level in the suprachiasmatic nucleus of the hypothalamus in response to increased illumination, which is different from the response of other visually impaired fossorial species, such as the common mole rat Cryptomys hottentotus [114, 115].
L. Bolk’s hypothesis: pro and contra
As noted above, two factors may account for the LS increase: 1) improved living conditions and 2) the retardation of switching on an aging program, e.g., the disappearance with age of mitochondrial mild depolarization [116]. Humans are traditionally compared to chimpanzees, although these two species have undergone separate evolution under different conditions for a long time. As a result, the modern structure of the human body is a mosaic with many characteristics that develop more slowly in humans than in chimpanzees, while other features, on the contrary, develop faster [117]. For example, the high growth rate of the brain during ontogenesis in human embryos persists until birth, while in chimpanzees, the growth rate of the brain begins to decline at the prenatal stage of ontogenesis [118, 119]. The brain of H. sapiens grows for approximately 15 years after birth, reaches 95% of the adult size between 7 and 11 years, and only then completes the final 5% of growth [120]. The chimpanzee brain volume reaches that of an adult by the age of 5 years [121]. Similarly, the postnatal brain maturation of H. glaber takes four times longer than that of a mouse, despite a more mature brain at birth [122].
In 1871, Darwin pointed to the similarity of the embryos of monkeys and humans, while their adults are much less similar [123]. In the 1920s, L. Bolk was the first to develop in detail the idea of the presence of numerous neotenic (pedomorphic) features in adult humans. He compiled a detailed list of the neotenic traits of humans, which was subsequently supplemented by other authors. In addition to a long period of infantile dependence and growth, greater LS and larger brain weight, there are other numerous neotenic differences between adult humans and other primates. Among them are the features of the skull [brachycephaly, orthognathia (the absence of a protruding muzzle)]; the absence of skull crests; skull bone thinness; a small amount of change in the skull structure from birth to old age; the absence of superciliary arches; certain variations in the structure of the teeth and cranial sutures; the position of the eye sockets below the cranial cavity; the central position of the occipital foramen (migrates backwards during ontogenesis in most primates); small teeth; later teething; some other infantile features of the skeleton (e.g., pelvis shape, longer legs compared to the arms, short limbs compared to the body size, inability for full thumb rotation, the absence of the baculum); the shape of the outer ear, epicanthic fold (skin fold at the upper eyelid), and labia; and a ventrally directed position of the genital canal in women.
Some traits almost disappeared in humans (e.g., body hair and skin pigmentation loss in some populations) [20, 21, 124–128]. The same is true for some skull structures [129]. Humans have sacrificed part of their physical strength in exchange for complex of brain activity (for example, a gorilla is 15 times stronger than a human being; [117]). Such unique human features as bipedality and the reduction in the size of canine teeth appeared before the big brain and stone tool making, presumably due to the transition to monogamy and the increasing contribution of fathers to the care of their offspring [130–132].
S. Gould [124] and, later, J. Verhulst [127] revised Bolk’s theory without refuting it. Verhulst indicated that, in addition to neotenic traits, a number of human morphological features, which are usually considered specializations caused by natural selection, are examples of hypermorphosis (i.e., changes in body proportions caused by the simple prolongation of ontogenesis [124, 127]). As the examples of this, the author mentioned the flattened chest of a human being and some other proportions of his or her physique. As an example of a particularly important hypermorphosis, Verhulst mentioned the structure of the larynx being different in adult humans from the larynx structure of other primates and infants [127]. This hypermorphosis is assumed to have been very important for the formation and socialization of a human being [132].
Strictly speaking, hypermorphosis can be attributed to a special case of neoteny. The situation is rather complicated due to the differences in neoteny levels of different traits and mosaic ontogenesis. In this case, one can rely on the number of characters, the development of which requires significantly more time in the neotenic species than in related non-neotenic species. If this number is much greater than the number of traits that develop faster than usual, then this is neoteny. However, a more attractive criterion of neoteny might be a functional analysis of each trait. For example, a strong argument in favor of neoteny can be a situation where a trait is directly involved to higher life span. For example, in our group, it was recently discovered that mild mitochondrial depolarization, which prevents the formation of mitochondrial ROS, in mice disappears by the age of 2.5 years, and in naked mole rats, depolarization persists for decades [116]. Strong retardation of depolarization and ROS production in naked mole rat prevents age-related protein carbonylation in these animals and, hence, increases their LS.
The study of the transcriptome in the prefrontal cortex of humans, chimpanzees and rhesus monkeys has revealed that it undergoes heavy reconstruction in the postnatal period, which proceeds slower in humans than in apes. In particular, the maximal expression of the genes responsible for synapse formation in the prefrontal cortex in chimpanzees and macaques has been observed at the age of one year and at the age of five years in humans [36]. The period of pregnancy in humans takes more time [22, 133], and the weight gain rate during the first five years after birth is also higher (2.6 kg/year vs 1.6 kg/year for chimpanzees), although this parameter turns almost the opposite over the next 5 years [36].
In general, the difficulty in the interpretation of the role of LS-affecting genes is because the ratio of the contribution of genes and nongenetic components to the variability of the characteristics of each species is not a constant value but rather depends on circumstances. It is obvious that the same allele can reduce the LS of animals in adverse conditions but may not affect the LS in good conditions. The LS-affecting genes in the hunter-gatherers may have no effect on the LS of the urban Japanese population. For example, a gene that affects the joints or any other important organs involved in heavy physical work will not affect the observed aging rate in a society carrying out a small amount of such work.
Conclusion
There is still no agreement among gerontologists as to the main aging-related issue: whether it is an accidental accumulation of damage in the organism or a result of the operation of a specially evolved program. In other words, it is still not clear whether aging is an inevitable phenomenon that is uncontrollable by organisms or whether it is a facultative adaptation that enhances the adaptive ability of species, i.e., their evolvability.
The undoubted neoteny of H. glaber has been described in the last three years: up to now, 58 neotenic traits were found in this species [13, 24–26, 134]. The slowing down of such a large number of developmental processes clearly shows that the longevity of H. glaber is a result of a general slowdown of late ontogenesis.
In 2017-18, the increase in acute phenoptosis was directly demonstrated in the nematode C. elegans. Phenoptosis starts as early as the first day of adult life due to the initiation of two special programs. The first program prevents the organism from being protected against oxidative, thermal, toxic and other stress types, and the second is a program of the autolysis of the intestine. Compounds and energy saved by these two programs are used for the biosynthesis of the yolk of eggs laid by the worm. The first program is activated by blocking specific histone demethylation [12, 27, 28], and the second is activated through insulin stimulation of autophagosomes that attack intestinal cells [29].
The evolutionary changes in humans compared to other primates have the following distinguishing characteristics: large brain, exceptionally large LS, high paternal investment in offspring, and the role of older individuals as helpers in upbringing the children [47]. The large brain is associated with a change in psychological characteristics: enhanced learning and cognition. Even human sleep is shorter, deeper, and has more rapid eye movement phases than that in other primates. Supposedly, the selection pressure in the direction of the reduction in sleep duration and its “quality” improvement were activated in the early stages of human evolution due to the change in the ecological niche and the development of overnight stays on the ground and not in the tree branches [135].
The evolution of these life history characteristics and extremely high intelligence was probably related to some degree to the dietary transition to high-quality, solid and hard-to-get food resources. Improvements in living conditions were due to technical progress (improved quality of food, medical services, industrial goods, etc.) and other evolutionary adaptations: cephalization index, sociality, and postreproductive LS have important effects on the survival curves in humans. A similar effect with respect to the reproductive individuals of H. glaber is caused by the fact that a single breeding female and one or two breeding males can be protected by hundreds of subordinates. Apparently, such protection also enhanced the selection against harmful mutations with late effects, which leads to a slowdown in aging, that is, to the activation of anti-aging programs [63, 116, 136].
In humans, technical progress leads to a sharp decrease in infant mortality and an increase in life expectancy, especially in comparison to wild chimpanzees [51]. Despite the huge variation in the LS of various human populations, starting with preagricultural tribes and ending with the urban population in the developed countries, the differences between their survival curves are still smaller than those between the preagricultural human populations and the chimpanzees living in the wild. This relationship can be explained by the fact that neoteny prolongs LS and health span.
The improvement in living conditions by new medicinal products, such as mitochondria-targeted antioxidants, could be effective in prolonging the average lifespan so that they can be considered not just as age-related disease treatment drugs but also as “true” anti-aging drugs. Such products may suppress chronic and acute phenoptosis processes. The example of such medicine is a mitochondria-addressed antioxidant SkQ developed at the Belozersky Institute, Moscow State University [55, 137–145]. Rapamycin, which seems to cause aging retardation and an increase in LS, appears to act in the same direction [146–149] due to the suppression of chronic phenoptosis. Metformin also causes a significant increase in LS and the retardation of aging, not only due to its properties as an antidiabetic drug [150–152] but also as an inhibitor of the aging program. It is essential that both SkQ and metformin specifically inhibit ROS generation in mitochondria at the beginning of the respiratory chain, where SkQ is effective at a thousand times lower concentration than metformin.
The retardation of the aging process in the human body with special drugs is a promising approach to extend the health span. Such an approach appears probable because aging retardation in some mammals is already achieved through neoteny, a natural physiological phenomenon.
Author Contributions
VPS and GS wrote the manuscript. TP and NP prepared the graphic materials. VPS, GS, MS, TP, AM and VAS analyzed and interpreted the data. All authors critically reviewed and approved the final manuscript.
Conflicts of Interest
MVS is the director and VPS is a board member of Mitotech LLC, a biotech company developing SkQ1-based pharmaceutical products.
Funding
The reported study was funded by the Russian Foundation for Basic Research (project No. 18-29-13037).
Footnotes
Phenoptosis – programmed death of an organism [3].
It is often assumed that the traits appearing in the postreproductive period are not subject to natural selection [1, 4, 56]. However, in certain cases the skills useful for the population and acquired in the postreproductive period by some animals (especially social ones) are transferred, via social learning, to the individuals still capable of reproduction, thus increasing their fitness and, in general, increasing the resilience of the population and hence adaptations to changes of the external environment [57–59]. If longevity is favored by selection, the special protective and repair systems might evolve that slow down chronic phenoptosis. Since LS is a stable species-specific characteristic, similar to body size or fecundity, the life duration should somehow be programmed in the genome [3, 8, 42].
Gamergates – worker ants able to reproduce sexually.
References
- 1. Hamilton WD. The moulding of senescence by natural selection. J Theor Biol. 1966; 12:12–45. https://doi.org/10.1016/0022-5193(66)90184-6 [PubMed]
- 2. Rose MR. Evolutionary biology of aging. New York: Oxford University Press; 1991.
- 3. Skulachev VP. Aging is a specific biological function rather than the result of a disorder in complex living systems: biochemical evidence in support of Weismann’s hypothesis. Biochemistry (Mosc). 1997; 62:1191–95. [PubMed]
- 4. Medawar PB. An unsolved problem of biology. 1952. H.K. Lewis, London.
- 5. Williams GC. Pleiotropy, natural selection and the evolution of senescence. Evolution. 1957; 11:398–411. https://doi.org/10.1111/j.1558-5646.1957.tb02911.x
- 6. Weismann A. Essays upon heredity and kindred biological problems. Oxford: Clarendon press; 1889. https://doi.org/10.5962/bhl.title.101564
- 7. Comfort A. The biology of senescence. New York: Elsevier; 1979.
- 8. Skulachev VP. Phenoptosis: programmed death of an organism. Biochemistry (Mosc). 1999; 64:1418–26. [PubMed]
- 9. Longo VD, Mitteldorf J, Skulachev VP. Programmed and altruistic ageing. Nat Rev Genet. 2005; 6:866–72. https://doi.org/10.1038/nrg1706 [PubMed]
- 10. Goldsmith TC. Aging, evolvability, and the individual benefit requirement; medical implications of aging theory controversies. J Theor Biol. 2008; 252:764–68. https://doi.org/10.1016/j.jtbi.2008.02.035 [PubMed]
- 11. Libertini G. Non-programmed versus programmed aging paradigm. Curr Aging Sci. 2015; 8:56–68. https://doi.org/10.2174/1874609808666150422111623 [PubMed]
- 12. Li J, Labbadia J, Morimoto RI. Rethinking HSF1 in stress, development, and organismal health. Trends Cell Biol. 2017; 27:895–905. https://doi.org/10.1016/j.tcb.2017.08.002 [PubMed]
- 13. Skulachev VP, Holtze S, Vyssokikh MY, Bakeeva LE, Skulachev MV, Markov AV, Hildebrandt TB, Sadovnichii VA. Neoteny, prolongation of youth: from naked mole rats to “naked apes” (humans). Physiol Rev. 2017; 97:699–720. https://doi.org/10.1152/physrev.00040.2015 [PubMed]
- 14. Galimov ER, Lohr JN, Gems D. When and how can death be an adaptation? Biochemistry (Mosc). 2019; 84:1433–37. https://doi.org/10.1134/S0006297919120010 [PubMed]
- 15. Lohr JN, Galimov ER, Gems D. Does senescence promote fitness in Caenorhabditis elegans by causing death? Ageing Res Rev. 2019; 50:58–71. https://doi.org/10.1016/j.arr.2019.01.008 [PubMed]
- 16. Skulachev VP. Phenoptosis as a phenomenon widespread among many groups of living organisms including mammals (Commentary to the paper by E. R. Galimov, J. N. Lohr, and D. Gems (2019) Biochemistry (Moscow), 84, 1433-1437). Biochemistry (Mosc). 2019; 84:1438–41. https://doi.org/10.1134/S0006297919120022 [PubMed]
- 17. Skulachev VP. Aging and the programmed death phenomena. Topics Curr Genet, Model Systems in Aging, ed Nystrom T, Osiewacz, H. D. Springer, Berlin, Heidelberg. 2003; 3:192–237. https://doi.org/10.1007/978-3-540-37005-5_8
- 18. Chistyakov VA, Denisenko YV, Bren AB. Presence of old individuals in a population accelerates and optimizes the process of selection: in silico experiments. Biochemistry (Mosc). 2018; 83:159–67. https://doi.org/10.1134/S0006297918020086 [PubMed]
- 19. Markov AV, Barg MA, Yakovleva EY. Can aging develop as an adaptation to optimize natural selection? (Application of computer modeling for searching conditions, when the «Fable about hares» can explain the evolution of aging). Biochemistry (Mosc). 2018; 83:1504–16. https://doi.org/10.1134/S0006297918120088 [PubMed]
- 20. Bolk L. The problem of human development. Jena: Gustav Fischer; 1926.
- 21. Bolk L. On the origin of human races. P K Akad Wet-Amsterd. 1927; 30:320–328.
- 22. Liu X, Somel M, Tang L, Yan Z, Jiang X, Guo S, Yuan Y, He L, Oleksiak A, Zhang Y, Li N, Hu Y, Chen W, et al. Extension of cortical synaptic development distinguishes humans from chimpanzees and macaques. Genome Res. 2012; 22:611–22. https://doi.org/10.1101/gr.127324.111 [PubMed]
- 23. Skulachev MV, Severin FF, Skulachev VP. Aging as an evolvability-increasing program which can be switched off by organism to mobilize additional resources for survival. Curr Aging Sci. 2015; 8:95–109. https://doi.org/10.2174/1874609808666150422122401 [PubMed]
- 24. Penz OK, Fuzik J, Kurek AB, Romanov R, Larson J, Park TJ, Harkany T, Keimpema E. Protracted brain development in a rodent model of extreme longevity. Sci Rep. 2015; 5:11592. https://doi.org/10.1038/srep11592 [PubMed]
- 25. Skulachev VP. Moscow news: two more representatives of sodium motive force generators (Na+-cbb3 oxidase and Na+-bacteriorhodopsin); natural delay of the aging program (neoteny) in mammals, namely in naked mole rat and “naked ape” (human). XI Mitochondrial Physiol. Conf. (MIP), p. 64–66, 2015, Luční Bouda, Pec pod Snezkou, Czech Republic., www.mitophysiology.org.
- 26. Popov NA, Skulachev VP. Neotenic traits in Heterocephalus glaber and Homo sapiens. Biochemistry (Mosc). 2019; 84:1484–89. https://doi.org/10.1134/S0006297919120071 [PubMed]
- 27. Labbadia J, Morimoto RI. Proteostasis and longevity: when does aging really begin? F1000Prime Rep. 2014; 6:7. https://doi.org/10.12703/P6-07 [PubMed]
- 28. Labbadia J, Morimoto RI. The biology of proteostasis in aging and disease. Annu Rev Biochem. 2015; 84:435–64. https://doi.org/10.1146/annurev-biochem-060614-033955 [PubMed]
- 29. Ezcurra M, Benedetto A, Sornda T, Gilliat AF, Au C, Zhang Q, van Schelt S, Petrache AL, Wang H, de la Guardia Y, Bar-Nun S, Tyler E, Wakelam MJ, Gems D. C. elegans eats its own intestine to make yolk leading to multiple senescent pathologies. Curr Biol. 2018; 28:2544–2556.e5. https://doi.org/10.1016/j.cub.2018.06.035 [PubMed]
- 30. Gompertz B. On the nature of the function expressive of the law of human mortality and on a new mode of determining life contingencies. Philos Trans R Soc Lond A. 1825; 115:513–85. https://doi.org/10.1098/rstl.1825.0026
- 31. Gavrilov LA, Gavrilova NS. The biology of life span: a quantitative approach. New York: Harwood Academic Publisher; 1991.
- 32. Hill KH, Hurtado AM. Ache life history: The ecology and demography of a foraging people. New York: Aldine de Gruyter; 1996.
- 33. Jones OR, Scheuerlein A, Salguero-Gómez R, Camarda CG, Schaible R, Casper BB, Dahlgren JP, Ehrlén J, García MB, Menges ES, Quintana-Ascencio PF, Caswell H, Baudisch A, Vaupel JW. Diversity of ageing across the tree of life. Nature. 2014; 505:169–73. https://doi.org/10.1038/nature12789 [PubMed]
- 34. Gavrilova NS, Gavrilov LA, Severin FF, Skulachev VP. Testing predictions of the programmed and stochastic theories of aging: comparison of variation in age at death, menopause, and sexual maturation. Biochemistry (Mosc). 2012; 77:754–60. https://doi.org/10.1134/S0006297912070085 [PubMed]
- 35. Shilovsky GA, Putyatina TS, Ashapkin VV, Luchkina OS, Markov AV. Coefficient of variation of lifespan across the tree of life: is it a signature of programmed aging? Biochemistry (Mosc). 2017; 82:1480–92. https://doi.org/10.1134/S0006297917120070 [PubMed]
- 36. Kaplan H, Hill K, Lancaster J, Hurtado A. A theory of human life history evolution: Diet, intelligence, and longevity. Evol Anthropol. 2000; 9:156–85. https://doi.org/10.1002/1520-6505(2000)9:4<156::AID-EVAN5>3.0.CO;2-7
- 37. Oeppen J, Vaupel JW. Demography. Broken limits to life expectancy. Science. 2002; 296:1029–31. https://doi.org/10.1126/science.1069675 [PubMed]
- 38. Tuljapurkar S, Li N, Boe C. A universal pattern of mortality decline in the G7 countries. Nature. 2000; 405:789–92. https://doi.org/10.1038/35015561 [PubMed]
- 39. Gavrilov LA, Krut’ko VN, Gavrilova NS. The future of human longevity. Gerontology. 2017; 63:524–26. https://doi.org/10.1159/000477965 [PubMed]
- 40. Lowenstine LJ, McManamon R, Terio KA. Comparative pathology of aging great apes: bonobos, chimpanzees, gorillas, and orangutans. Vet Pathol. 2016; 53:250–76. https://doi.org/10.1177/0300985815612154 [PubMed]
- 41. Edler MK, Sherwood CC, Meindl RS, Hopkins WD, Ely JJ, Erwin JM, Mufson EJ, Hof PR, Raghanti MA. Aged chimpanzees exhibit pathologic hallmarks of Alzheimer’s disease. Neurobiol Aging. 2017; 59:107–20. https://doi.org/10.1016/j.neurobiolaging.2017.07.006 [PubMed]
- 42. Skulachev VP, Fenyuk BA, Skulachev MV. Life without aging. Moscow: EXMO; 2014.[(in Russian)].
- 43. Dyke B, Gage TB, Alford PL, Swenson B, Williams-Blangero S. Model life table for captive chimpanzees. Am J Primatol. 1995; 37:25–37. https://doi.org/10.1002/ajp.1350370104 [PubMed]
- 44. Hill K, Boesch C, Goodall J, Pusey A, Williams J, Wrangham R. Mortality rates among wild chimpanzees. J Hum Evol. 2001; 40:437–50. https://doi.org/10.1006/jhev.2001.0469 [PubMed]
- 45. Shilovsky GA, Putyatina TS, Lysenkov SN, Ashapkin VV, Luchkina OS, Markov AV, Skulachev VP. Is it possible to prove the existence of an aging program by quantitative analysis of mortality dynamics? Biochemistry (Mosc). 2016; 81:1461–76. https://doi.org/10.1134/S0006297916120075 [PubMed]
- 46. Ruby JG, Smith M, Buffenstein R. Naked Mole-Rat mortality rates defy gompertzian laws by not increasing with age. eLife. 2018; 7:e31157. https://doi.org/10.7554/eLife.31157 [PubMed]
- 47. Gurven M, Kaplan H. Longevity among hunter-gatherers: a cross-cultural examination. Popul Dev Rev. 2007; 33:321–65. https://doi.org/10.1111/j.1728-4457.2007.00171.x
- 48. Allard M. Jeanne Calment: From Van Goghs time to ours: 122 extraordinary years. New York: W.H. Freeman; 1998.
- 49. Colchero F, Rau R, Jones OR, Barthold JA, Conde DA, Lenart A, Nemeth L, Scheuerlein A, Schoeley J, Torres C, Zarulli V, Altmann J, Brockman DK, et al. The emergence of longevous populations. Proc Natl Acad Sci USA. 2016; 113:E7681–90. https://doi.org/10.1073/pnas.1612191113 [PubMed]
- 50. Burger O, Baudisch A, Vaupel JW. Human mortality improvement in evolutionary context. Proc Natl Acad Sci USA. 2012; 109:18210–14. https://doi.org/10.1073/pnas.1215627109 [PubMed]
- 51. Burger O. Evolutionary demography of the human mortality profile. In: Shefferson P, Jones OR, Salgnero-Gomez R, editors. The evolution of senescence in the tree of life. Volume R. Cambridge: Cambridge Univ Press; 2017. https://doi.org/10.1017/9781139939867.006
- 52. Jones DS, Podolsky SH, Greene JA. The burden of disease and the changing task of medicine. N Engl J Med. 2012; 366:2333–38. https://doi.org/10.1056/NEJMp1113569 [PubMed]
- 53. Boĭko AG, Labas IA, Gordeeva AV. [An outline on the phylogenetic history of metazoa aging phenomenon (to the study of creating a common metazoa aging theory)]. Adv Gerontol. 2010; 23:21–29. [PubMed]
- 54. Skulachev MV, Skulachev VP. New data on programmed aging - slow phenoptosis. Biochemistry (Mosc). 2014; 79:977–93. https://doi.org/10.1134/S0006297914100010 [PubMed]
- 55. Skulachev MV, Skulachev VP. Programmed aging of mammals: proof of concept and prospects of biochemical approaches for anti-aging therapy. Biochemistry (Mosc). 2017; 82:1403–22. https://doi.org/10.1134/S000629791712001X [PubMed]
- 56. Fisher RA. The genetical theory of natural selection. Oxford: Clarendon Press; 1930. https://doi.org/10.5962/bhl.title.27468
- 57. Nusbaum NJ. What good is it to get old? Med Hypotheses. 1996; 47:77–79. https://doi.org/10.1016/S0306-9877(96)90441-7 [PubMed]
- 58. Putyatina TS. Effect of recreational pressure on ant communities of open biocenoses in Moscow. Moscow Univ Biol Sci Bull. 2011; 66:42–45. https://doi.org/10.3103/S0096392511010081
- 59. Brent LJ, Franks DW, Foster EA, Balcomb KC, Cant MA, Croft DP. Ecological knowledge, leadership, and the evolution of menopause in killer whales. Curr Biol. 2015; 25:746–50. https://doi.org/10.1016/j.cub.2015.01.037 [PubMed]
- 60. Skulachev VP. Programmed death phenomena: from organelle to organism. Ann N Y Acad Sci. 2002; 959:214–37. https://doi.org/10.1111/j.1749-6632.2002.tb02095.x [PubMed]
- 61. Skulachev VP. Aging as a particular case of phenoptosis, the programmed death of an organism (a response to Kirkwood and Melov “On the programmed/non-programmed nature of ageing within the life history”). Aging (Albany NY). 2011; 3:1120–23. https://doi.org/10.18632/aging.100403 [PubMed]
- 62. Lewis KN, Mele J, Hayes JD, Buffenstein R. Nrf2, a guardian of healthspan and gatekeeper of species longevity. Integr Comp Biol. 2010; 50:829–43. https://doi.org/10.1093/icb/icq034 [PubMed]
- 63. Frolkis VV. Aging and life-prolonging processes. Wien, New York: Springer Verlag; 1982. https://doi.org/10.1007/978-3-7091-8649-7
- 64. Lewis KN, Wason E, Edrey YH, Kristan DM, Nevo E, Buffenstein R. Regulation of Nrf2 signaling and longevity in naturally long-lived rodents. Proc Natl Acad Sci USA. 2015; 112:3722–27. https://doi.org/10.1073/pnas.1417566112 [PubMed]
- 65. Miller CJ, Gounder SS, Kannan S, Goutam K, Muthusamy VR, Firpo MA, Symons JD, Paine R
3rd , Hoidal JR, Rajasekaran NS. Disruption of Nrf2/ARE signaling impairs antioxidant mechanisms and promotes cell degradation pathways in aged skeletal muscle. Biochim Biophys Acta. 2012; 1822:1038–50. https://doi.org/10.1016/j.bbadis.2012.02.007 [PubMed] - 66. Li N, Muthusamy S, Liang R, Sarojini H, Wang E. Increased expression of miR-34a and miR-93 in rat liver during aging, and their impact on the expression of Mgst1 and Sirt1. Mech Ageing Dev. 2011; 132:75–85. https://doi.org/10.1016/j.mad.2010.12.004 [PubMed]
- 67. Suh JH, Shenvi SV, Dixon BM, Liu H, Jaiswal AK, Liu RM, Hagen TM. Decline in transcriptional activity of Nrf2 causes age-related loss of glutathione synthesis, which is reversible with lipoic acid. Proc Natl Acad Sci USA. 2004; 101:3381–86. https://doi.org/10.1073/pnas.0400282101 [PubMed]
- 68. Duan W, Zhang R, Guo Y, Jiang Y, Huang Y, Jiang H, Li C. Nrf2 activity is lost in the spinal cord and its astrocytes of aged mice. In Vitro Cell Dev Biol Anim. 2009; 45:388–97. https://doi.org/10.1007/s11626-009-9194-5 [PubMed]
- 69. Zhou Y, Wu H, Zhao M, Chang C, Lu Q. The Bach family of transcription factors: a comprehensive review. Clin Rev Allergy Immunol. 2016; 50:345–56. https://doi.org/10.1007/s12016-016-8538-7 [PubMed]
- 70. Tonelli C, Chio II, Tuveson DA. Transcriptional regulation by Nrf2. Antioxid Redox Signal. 2018; 29:1727–45. https://doi.org/10.1089/ars.2017.7342 [PubMed]
- 71. Albalat R, Cañestro C. Evolution by gene loss. Nat Rev Genet. 2016; 17:379–91. https://doi.org/10.1038/nrg.2016.39 [PubMed]
- 72. Stedman HH, Kozyak BW, Nelson A, Thesier DM, Su LT, Low DW, Bridges CR, Shrager JB, Minugh-Purvis N, Mitchell MA. Myosin gene mutation correlates with anatomical changes in the human lineage. Nature. 2004; 428:415–18. https://doi.org/10.1038/nature02358 [PubMed]
- 73. Winter H, Langbein L, Krawczak M, Cooper DN, Jave-Suarez LF, Rogers MA, Praetzel S, Heidt PJ, Schweizer J. Human type I hair keratin pseudogene phihHaA has functional orthologs in the chimpanzee and gorilla: evidence for recent inactivation of the human gene after the Pan-Homo divergence. Hum Genet. 2001; 108:37–42. https://doi.org/10.1007/s004390000439 [PubMed]
- 74. Soltani-Mazouni N, Bordereau C. Changes in the cuticle, ovaries and colleterial glands during the pseudergate and neotenic molt in Kalotermes flavicollis (Fabr) (Isoptera, Kalotermitidae). Int J Insect Morphol. 1987; 16:221–35. https://doi.org/10.1016/0020-7322(87)90022-5
- 75. Harrison R. Dispersal polymorphisms in insects. Annu Rev Ecol Syst. 1980; 11:95–118. https://doi.org/10.1146/annurev.es.11.110180.000523
- 76. Soldán T, Godunko RJ. Description of larva, redescription of adults and biology of Mortogenesia mesopotamica (Morton, 1921) (Ephemeroptera: palingeniidae). Zootaxa. 2013; 3741:265–78. https://doi.org/10.11646/zootaxa.3741.2.5 [PubMed]
- 77. Jordal BH, Beaver RA, Normark BB, Farrell BD. Extraordinary sex ratios and the evolution of male neoteny in sib-mating Ozopemon beetles. Biol J Linn Soc Lond. 2002; 75:353–60. https://doi.org/10.1111/j.1095-8312.2002.tb02076.x
- 78. Bocak L, Bocakova M, Hunt T, Vogler AP. Multiple ancient origins of neoteny in Lycidae(Coleoptera): consequences for ecology and macroevolution. Proc Biol Sci. 2008; 275:2015–23. https://doi.org/10.1098/rspb.2008.0476 [PubMed]
- 79. Erezyilmaz DF, Hayward A, Huang Y, Paps J, Acs Z, Delgado JA, Collantes F, Kathirithamby J. Expression of the pupal determinant broad during metamorphic and neotenic development of the strepsipteran Xenos vesparum Rossi. PLoS One. 2014; 9:e93614. https://doi.org/10.1371/journal.pone.0093614 [PubMed]
- 80. Brokeland W, Brandt A. Two new species of Ischnomesidae (Crustacea: Isopoda) from the Southern Ocean, displaying neoteny. Deep Sea Res Part II Top Stud Oceanogr. 2004; 51:1769–85. https://doi.org/10.1016/j.dsr2.2004.06.034
- 81. Piraino S, Boero F, Aeschbach B, Schmid V. Reversing the life cycle: medusae transforming into polyps and cell transdifferentiation in Turritopsis nutricula (Cnidaria, Hydrozoa). Biol Bull. 1996; 190:302–12. https://doi.org/10.2307/1543022 [PubMed]
- 82. Shilovsky GA, Putyatina TS, Ashapkin VV, Rozina AA, Lyubetsky VA, Minina EP, Bychkovskaia IB, Markov AV, Skulachev VP. Ants as object of gerontological research. Biochemistry (Mosc). 2018; 83:1489–503. https://doi.org/10.1134/S0006297918120076 [PubMed]
- 83. Heinze J, Buschinger A. Queen polymorphism in a non-parasitic Leptothorax species (Hymenoptera, Formicidae). Insectes Soc. 1987; 34:28–43. https://doi.org/10.1007/BF02224205
- 84. Liebig J. Eusociality, female caste specialization and regulation of reproduction in the ponerine ant Harpegnathos saltator Jerdon. Berlin: Wissenschaft & Technik; 1998.
- 85. Hartmann A, Wantia J, Heinze J. Facultative sexual reproduction in the parthenogenetic ant Platythyrea punctata. Insectes Soc. 2005; 52:155–62. https://doi.org/10.1007/s00040-004-0786-5
- 86. Buschinger A, Francoeur A. Queen polymorphism and functional monogyny in the ant, Leptothorax sphagnicolus Francoeur. Psyche (Camb, Mass). 1991; 98:119–33. https://doi.org/10.1155/1991/52726
- 87. Heinze J, Kühnholz S, Schilder K, Hölldobler B. Behavior of ergatoid males in the ant, Cardiocondyla nuda. Insectes Soc. 1993; 40:273–82. https://doi.org/10.1007/BF01242363
- 88. Buschinger A, Peeters C, Crosier RH. Life pattern studies on an australian Sphinctomyrmex (Formicidae: Ponerinae; Cerapachyini): functional polygyny, brood periodicity and rayding behavior. Psyche (Camb, Mass). 1989; 96:287–300. https://doi.org/10.1155/1989/13614
- 89. Ito F, Higashi S. A linear dominance hierarchy regulating reproduction and polyethism of the queenless ant Pachycondyla sublaevis. Naturwissenschaften. 1991; 78:80–82. https://doi.org/10.1007/BF01206263
- 90. Peeters C, Crewe R. Worker reproduction in the ponerine ant Ophtalmopone berthoudi: an alternative form of eusocial organization. Behav Ecol Sociobiol. 1985; 18:29–37.
- 91. Tsuji K. Reproduction division of labour related to age in the Japanese queenless ant Pristomyrmex pungens. Anim Behav. 1990; 39:843–49. https://doi.org/10.1016/S0003-3472(05)80948-0
- 92. Peeters C, Higashi S. Reproductive dominance controlled by mutilation in the queenless ant Diacamma australe. Naturwissenschaften. 1989; 76:177–80. https://doi.org/10.1007/BF00366404
- 93. Peeters C, Tsuji K. Reproductive conflicts among ant workers in Diacamma sp. from Japan: doninance and oviposition in the absence of the gamergate. Insectes Soc. 1993; 40:1–18. https://doi.org/10.1007/BF01240701
- 94. Gronenberg W, Liebig J. Smaller brains and optic lobes in reproductive workers of the ant Harpegnathos. Naturwissenschaften. 1999; 86:343–45. https://doi.org/10.1007/s001140050631
- 95. Janet C. Anatomie du corselet et histolyse des muscles vibrateurs, après le vol nuptial, chez la reine de la fourmi (Lasius niger). Limoges: Ducourtieux et Goux; 1907.
- 96. Rosenkilde P, Ussing AP. What mechanisms control neoteny and regulate induced metamorphosis in urodeles? Int J Dev Biol. 1996; 40:665–73. [PubMed]
- 97. Snider AT, Bowler JK. Longevity of reptiles and amphibians in North American collections. ed. Oxford (Ohio): Society for the Study of Amphibians and Reptiles; 1992.
- 98. Voituron Y, de Fraipont M, Issartel J, Guillaume O, Clobert J. Extreme lifespan of the human fish (Proteus anguinus): a challenge for ageing mechanisms. Biol Lett. 2011; 7:105–07. https://doi.org/10.1098/rsbl.2010.0539 [PubMed]
- 99. Bonin J, Desgranges JL, Bishop CA, Rodrigue J, Gendron A, Elliott JE. Comparative-study of contaminants in the mudpuppy (Amphibia) and the common snapping turtle (Reptilia), St-Lawrence-River, Canada. Arch Environ Contam Toxicol. 1995; 28:184–94. https://doi.org/10.1007/BF00217615
- 100. Zelditch ML, Sheets HD, Fink WL. Spatiotemporal reorganization of growth rates in the evolution of ontogeny. Evolution. 2000; 54:1363–71. https://doi.org/10.1111/j.0014-3820.2000.tb00568.x [PubMed]
- 101. Harada Y, Harada S, Kinoshita I, Tanaka M, Tagawa M. Thyroid gland development in a neotenic goby (ice goby, Leucopsarion petersii) and a common goby (ukigori, Gymnogobius urotaenia) during early life stages. Zool Sci. 2003; 20:883–88. https://doi.org/10.2108/zsj.20.883 [PubMed]
- 102. Joss JM. Lungfish evolution and development. Gen Comp Endocrinol. 2006; 148:285–89. https://doi.org/10.1016/j.ygcen.2005.10.010 [PubMed]
- 103. Foster MS. Delayed maturation, neoteny, and social system differences in two manakins of the genus Chiroxiphia. Evolution. 1987; 41:547–58. https://doi.org/10.1111/j.1558-5646.1987.tb05825.x [PubMed]
- 104. Tsai CH, Fordyce RE. Juvenile morphology in baleen whale phylogeny. Naturwissenschaften. 2014; 101:765–69. https://doi.org/10.1007/s00114-014-1216-9 [PubMed]
- 105. Fettuccia DC, da Silva VM, Simões-Lopes PC. Non-metric characters in two species of Sotalia (Gray, 1866) (Cetacea, Delphinidae). Braz J Biol. 2009; 69:907–17. https://doi.org/10.1590/S1519-69842009000400020 [PubMed]
- 106. Bradshaw JW, Pullen AJ, Rooney NJ. Why do adult dogs ‘play’? Behav Processes. 2015; 110:82–87. https://doi.org/10.1016/j.beproc.2014.09.023 [PubMed]
- 107. Buffenstein R. Negligible senescence in the longest living rodent, the naked mole-rat: insights from a successfully aging species. J Comp Physiol B. 2008; 178:439–45. https://doi.org/10.1007/s00360-007-0237-5 [PubMed]
- 108. Lewis KN, Soifer I, Melamud E, Roy M, McIsaac RS, Hibbs M, Buffenstein R. Unraveling the message: insights into comparative genomics of the naked mole-rat. Mamm Genome. 2016; 27:259–78. https://doi.org/10.1007/s00335-016-9648-5 [PubMed]
- 109. Faulkes CG, Abbott DH, O’Brien HP, Lau L, Roy MR, Wayne RK, Bruford MW. Micro- and macrogeographical genetic structure of colonies of naked mole-rats Heterocephalus glaber. Mol Ecol. 1997; 6:615–28. https://doi.org/10.1046/j.1365-294X.1997.00227.x [PubMed]
- 110. Kim EB, Fang X, Fushan AA, Huang Z, Lobanov AV, Han L, Marino SM, Sun X, Turanov AA, Yang P, Yim SH, Zhao X, Kasaikina MV, et al. Genome sequencing reveals insights into physiology and longevity of the naked mole rat. Nature. 2011; 479:223–27. https://doi.org/10.1038/nature10533 [PubMed]
- 111. Dilman VM. Ontogenetic model of ageing and disease formation and mechanisms of natural selection. J Theor Biol. 1986; 118:73–81. https://doi.org/10.1016/S0022-5193(86)80009-1 [PubMed]
- 112. Fang X, Seim I, Huang Z, Gerashchenko MV, Xiong Z, Turanov AA, Zhu Y, Lobanov AV, Fan D, Yim SH, Yao X, Ma S, Yang L, et al. Adaptations to a subterranean environment and longevity revealed by the analysis of mole rat genomes. Cell Rep. 2014; 8:1354–64. https://doi.org/10.1016/j.celrep.2014.07.030 [PubMed]
- 113. Quay WB. Pineal atrophy and other neuroendocrine and circumventricular features of the naked mole-rat, Heterocephalus glaber (Rüppell), a fossorial, equatorial rodent. J Neural Transm (Vienna). 1981; 52:107–15. https://doi.org/10.1007/BF01253102 [PubMed]
- 114. Oosthuizen MK, Bennett NC, Cooper HM. Fos expression in the suprachiasmatic nucleus in response to light stimulation in a solitary and social species of African mole-rat (family Bathyergidae). Neuroscience. 2005; 133:555–60. https://doi.org/10.1016/j.neuroscience.2005.01.017 [PubMed]
- 115. Oosthuizen MK, Bennett NC, Cooper HM. Photic induction of Fos in the suprachiasmatic nucleus of African mole-rats: responses to increasing irradiance. Chronobiol Int. 2010; 27:1532–45. https://doi.org/10.3109/07420528.2010.510227 [PubMed]
- 116. Vyssokikh MY, Holtze S, Averina OA, Lyamzaev KG, Panteleeva AA, Marey MV, Zinovkin RA, Severin FF, Skulachev MV, Fasel N, Hildebrandt TB, Skulachev VP. Mild depolarization of the inner mitochondrial membrane is a crucial component of an anti-aging program. Proc Natl Acad Sci USA. 2020; 117:6491–501. https://doi.org/10.1073/pnas.1916414117 [PubMed]
- 117. Bufill E, Agustí J, Blesa R. Human neoteny revisited: the case of synaptic plasticity. Am J Hum Biol. 2011; 23:729–39. https://doi.org/10.1002/ajhb.21225 [PubMed]
- 118. Hüppi PS, Warfield S, Kikinis R, Barnes PD, Zientara GP, Jolesz FA, Tsuji MK, Volpe JJ. Quantitative magnetic resonance imaging of brain development in premature and mature newborns. Ann Neurol. 1998; 43:224–35. https://doi.org/10.1002/ana.410430213 [PubMed]
- 119. Sakai T, Hirata S, Fuwa K, Sugama K, Kusunoki K, Makishima H, Eguchi T, Yamada S, Ogihara N, Takeshita H. Fetal brain development in chimpanzees versus humans. Curr Biol. 2012; 22:R791–92. https://doi.org/10.1016/j.cub.2012.06.062 [PubMed]
- 120. Caviness VS
Jr , Kennedy DN, Richelme C, Rademacher J, Filipek PA. The human brain age 7-11 years: a volumetric analysis based on magnetic resonance images. Cereb Cortex. 1996; 6:726–36. https://doi.org/10.1093/cercor/6.5.726 [PubMed] - 121. Bogin B, Smith BH. Evolution of the human life cycle. Am J Hum Biol. 1996; 8:703–16. https://doi.org/10.1002/(SICI)1520-6300(1996)8:6<703::AID-AJHB2>3.0.CO;2-U [PubMed]
- 122. Orr ME, Garbarino VR, Salinas A, Buffenstein R. Sustained high levels of neuroprotective, high molecular weight, phosphorylated tau in the longest-lived rodent. Neurobiol Aging. 2015; 36:1496–504. https://doi.org/10.1016/j.neurobiolaging.2014.12.004 [PubMed]
- 123. Darwin C. The descent of man, and selection in relation to sex. London: J. Murray; 1871. https://doi.org/10.5962/bhl.title.2092
- 124. Gould SJ. Ontogeny and phylogeny. Cambridge (Mass.): Belknap Press of Harvard University Press; 1977.
- 125. Montagu A. Time, morphology and neoteny in the evolution of man. In: Montagu A, editor. Culture and the evolution of man. New York: Oxford University Press; 1962.
- 126. Montagu A. Growing young. 1989. Granby, Mass: Bergin & Garvey Publishers.
- 127. Verhulst J. Louis Bolk revisited. II—Retardation, hypermorphosis and body proportions of humans. Med Hypotheses. 1993; 41:100–14. https://doi.org/10.1016/0306-9877(93)90054-T [PubMed]
- 128. Henke W, Tattersall I, Hardt T. Handbook of paleoanthropology. New York: Springer; 2007. https://doi.org/10.1007/978-3-540-33761-4
- 129. Penin X, Berge C, Baylac M. Ontogenetic study of the skull in modern humans and the common chimpanzees: neotenic hypothesis reconsidered with a tridimensional Procrustes analysis. Am J Phys Anthropol. 2002; 118:50–62. https://doi.org/10.1002/ajpa.10044 [PubMed]
- 130. Lovejoy CO. Reexamining human origins in light of Ardipithecus ramidus. Science. 2009; 326:74e1–8. https://doi.org/10.1126/science.1175834 [PubMed]
- 131. Clark G, Henneberg M. Ardipithecus ramidus and the evolution of language and singing: an early origin for hominin vocal capability. Homo. 2017; 68:101–21. https://doi.org/10.1016/j.jchb.2017.03.001 [PubMed]
- 132. Hammond AS, Almécija S. Lower ilium evolution in apes and hominins. Anat Rec (Hoboken). 2017; 300:828–44. https://doi.org/10.1002/ar.23545 [PubMed]
- 133. Somel M, Franz H, Yan Z, Lorenc A, Guo S, Giger T, Kelso J, Nickel B, Dannemann M, Bahn S, Webster MJ, Weickert CS, Lachmann M, et al. Transcriptional neoteny in the human brain. Proc Natl Acad Sci USA. 2009; 106:5743–48. https://doi.org/10.1073/pnas.0900544106 [PubMed]
- 134. Orr ME, Garbarino VR, Salinas A, Buffenstein R. Extended postnatal brain development in the longest-lived rodent: prolonged maintenance of neotenous traits in the naked mole-rat brain. Front Neurosci. 2016; 10:504. https://doi.org/10.3389/fnins.2016.00504 [PubMed]
- 135. Samson DR, Nunn CL. Sleep intensity and the evolution of human cognition. Evol Anthropol. 2015; 24:225–37. https://doi.org/10.1002/evan.21464 [PubMed]
- 136. Austad SN. Why we age. New York: John Wiley and Sons; 1997.
- 137. Skulachev VP. A biochemical approach to the problem of aging: “megaproject” on membrane-penetrating ions. The first results and prospects. Biochemistry (Mosc). 2007; 72:1385–96. https://doi.org/10.1134/S0006297907120139 [PubMed]
- 138. Obukhova LA, Skulachev VP, Kolosova NG. Mitochondria-targeted antioxidant SkQ1 inhibits age-dependent involution of the thymus in normal and senescence-prone rats. Aging (Albany NY). 2009; 1:389–401. https://doi.org/10.18632/aging.100043 [PubMed]
- 139. Stefanova NA, Muraleva NA, Skulachev VP, Kolosova NG. Alzheimer’s disease-like pathology in senescence-accelerated OXYS rats can be partially retarded with mitochondria-targeted antioxidant SkQ1. J Alzheimers Dis. 2014; 38:681–94. https://doi.org/10.3233/JAD-131034 [PubMed]
- 140. Rokitskaya TI, Sumbatyan NV, Tashlitsky VN, Korshunova GA, Antonenko YN, Skulachev VP. Mitochondria-targeted penetrating cations as carriers of hydrophobic anions through lipid membranes. Biochim Biophys Acta. 2010; 1798:1698–706. https://doi.org/10.1016/j.bbamem.2010.05.018 [PubMed]
- 141. Skulachev VP, Anisimov VN, Antonenko YN, Bakeeva LE, Chernyak BV, Erichev VP, Filenko OF, Kalinina NI, Kapelko VI, Kolosova NG, Kopnin BP, Korshunova GA, Lichinitser MR, et al. An attempt to prevent senescence: a mitochondrial approach. Biochim Biophys Acta. 2009; 1787:437–61. https://doi.org/10.1016/j.bbabio.2008.12.008 [PubMed]
- 142. Skulachev VP, Antonenko YN, Cherepanov DA, Chernyak BV, Izyumov DS, Khailova LS, Klishin SS, Korshunova GA, Lyamzaev KG, Pletjushkina OY, Roginsky VA, Rokitskaya TI, Severin FF, et al. Prevention of cardiolipin oxidation and fatty acid cycling as two antioxidant mechanisms of cationic derivatives of plastoquinone (SkQs). Biochim Biophys Acta. 2010; 1797:878–89. https://doi.org/10.1016/j.bbabio.2010.03.015 [PubMed]
- 143. Skulachev MV, Antonenko YN, Anisimov VN, Chernyak BV, Cherepanov DA, Chistyakov VA, Egorov MV, Kolosova NG, Korshunova GA, Lyamzaev KG, Plotnikov EY, Roginsky VA, Savchenko AY, et al. Mitochondrial-targeted plastoquinone derivatives. Effect on senescence and acute age-related pathologies. Curr Drug Targets. 2011; 12:800–26. https://doi.org/10.2174/138945011795528859 [PubMed]
- 144. Brzheskiy VV, Efimova EL, Vorontsova TN, Alekseev VN, Gusarevich OG, Shaidurova KN, Ryabtseva AA, Andryukhina OM, Kamenskikh TG, Sumarokova ES, Miljudin ES, Egorov EA, Lebedev OI, et al. Results of a multicenter, randomized, double-masked, placebo-controlled clinical study of the efficacy and safety of Visomitin eye drops in patients with dry eye syndrome. Adv Ther. 2015; 32:1263–79. https://doi.org/10.1007/s12325-015-0273-6 [PubMed]
- 145. Demyanenko IA, Popova EN, Zakharova VV, Ilyinskaya OP, Vasilieva TV, Romashchenko VP, Fedorov AV, Manskikh VN, Skulachev MV, Zinovkin RA, Pletjushkina OY, Skulachev VP, Chernyak BV. Mitochondria-targeted antioxidant SkQ1 improves impaired dermal wound healing in old mice. Aging (Albany NY). 2015; 7:475–85. https://doi.org/10.18632/aging.100772 [PubMed]
- 146. Blagosklonny MV. An anti-aging drug today: from senescence-promoting genes to anti-aging pill. Drug Discov Today. 2007; 12:218–24. https://doi.org/10.1016/j.drudis.2007.01.004 [PubMed]
- 147. Blagosklonny MV. Why human lifespan is rapidly increasing: solving “longevity riddle” with “revealed-slow-aging” hypothesis. Aging (Albany NY). 2010; 2:177–82. https://doi.org/10.18632/aging.100139 [PubMed]
- 148. Blagosklonny MV. From rapalogs to anti-aging formula. Oncotarget. 2017; 8:35492–507. https://doi.org/10.18632/oncotarget.18033 [PubMed]
- 149. Aliper A, Jellen L, Cortese F, Artemov A, Karpinsky-Semper D, Moskalev A, Swick AG, Zhavoronkov A. Towards natural mimetics of metformin and rapamycin. Aging (Albany NY). 2017; 9:2245–68. https://doi.org/10.18632/aging.101319 [PubMed]
- 150. Anisimov VN. Metformin: do we finally have an anti-aging drug? Cell Cycle. 2013; 12:3483–89. https://doi.org/10.4161/cc.26928 [PubMed]
- 151. Barzilai N, Crandall JP, Kritchevsky SB, Espeland MA. Metformin as a tool to target aging. Cell Metab. 2016; 23:1060–65. https://doi.org/10.1016/j.cmet.2016.05.011 [PubMed]
- 152. Campbell JM, Bellman SM, Stephenson MD, Lisy K. Metformin reduces all-cause mortality and diseases of ageing independent of its effect on diabetes control: A systematic review and meta-analysis. Ageing Res Rev. 2017; 40:31–44. https://doi.org/10.1016/j.arr.2017.08.003 [PubMed]


