Introduction
The global population is growing older and one consequence of this is an increase in the prevalence of dementia, which is now the greatest global challenge for health and social care [1]. Meanwhile, older people are at a high risk of oral problems (such as periodontal disease, orofacial pain and the loss of teeth) [2]. The prevalence of edentulism (complete tooth loss) is 4.1% and peaks at around 25% in adults 75 to 79 years old [3].
Several studies have shown that tooth loss is associated with steeper global cognitive decline [4, 5], an increased risk for cognitive impairment [6–8] and dementia [9], as well as brain atrophy in cognitively normal older individuals [10]. One cross-sectional study found that jaw mobility, bite strength and complaints about masticatory function were associated with variation in episodic memory and executive function [11]. Moreover, emerging evidence suggests that the mechanism behind the association between tooth loss and cognitive decline is linked to reduced mechanical sensory input from poor mastication resulting from the loss of teeth [4, 10].
Mastication can be assessed objectively in terms of bite force, jaw mobility, and number and pattern of occlusal contacts (contact of opposite teeth in upper and lower jaw), and may be closely related to the number and distribution of remaining teeth [15]. Posterior occlusal contact of the remaining dentition has been reported as a marker of poorer masticatory ability [16–18]. The number of posterior occlusal contacts has been associated with cognitive function and a lack of posterior occlusal support influences cognitive decline to a greater extent than the number of teeth alone [19]. So far, only one longitudinal study investigated the relationship between posterior occlusal support and cognitive decline, showing that a lack of posterior occlusal support predicted global cognitive decline [20]. However, no studies have yet investigated the relation of mastication to trajectories in different cognitive domains and dementia risk.
In this study, we aimed to 1) examine the association between poor masticatory ability (reduced posterior occlusal support) and cognitive trajectories in different domains; and 2) investigate whether poor masticatory ability may increase the risk of dementia, using longitudinal data from a population–based study with up to 22 years of follow-up.
Results
Characteristics of the study population
Over the follow-up period, 99 (18.2%) participated in all the waves, 44 (8.1%) participated in at least two follow-ups, 1 (0.2%) participated only at study entry, and 400 participants (73.5%) died. The median follow-up time was 10 years, IQR= 16-3. The number of participants in Eichner category A was 147 (27.0%), in category B 169 (31.1%), and in category C 228 (41.9%). Compared to those in Eichner category A, those in B and C were older and had lower education, while participants in Eichner category C consumed less alcohol, had lower education and childhood SES, and had higher proportions of belonging to the early birth cohort, having heart disease, hypertension, wearing prosthetics, and periodontal disease. Performance in all cognitive domains were lower for those with Eichner category B and C compared to A, except for verbal ability which was only significantly different between category A and C (Table 1). During the study period, 78 (53.1%) died in Eichner category A, 116 (68.6%) in category B, and 206 (90.4%) in category C.
Table 1. Characteristics of the study population at baseline by Eichner Index categories (n= 544).
| Characteristics | Category A, n=147 (27.0%) | Category B, n= 169 (31.1%) | Category C, n= 228 (41.9%) | P value | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age /years | 60.1 (±7.9) | 64.6 (±8.3)a | 69.9 (±7.6)a,b | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Female sex | 80 (54.4) | 98 (58.0) | 136 (59.7) | 0.604 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Education | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Low | 59 (40.7) | 98 (60.1)a | 172 (77.5)a,b | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| High | 86 (59.3) | 65 (39.9)a | 50 (22.5)a,b | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hypertension | 54 (36.7) | 80 (47.3) | 121 (53.1)a | 0.008 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Heart disease | 10 (6.8) | 21 (12.6) | 43 (18.9)a | 0.004 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diabetes | 7 (4.8) | 13 (7.7) | 20 (8.8) | 0.341 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cerebrovascular disease | 0 (0.0) | 3 (1.9) | 4 (2.0= | 0.254 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Any APOE ε4 | 43 (31.6) | 44 (28.6) | 63 (31.3) | 0.812 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BMI (kg/m2) | 25.0 (±3.6) | 25.8 (±4.5) | 25.9 (±3.8) | 0.105 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smokers | 71 (48.0) | 73 (42.0) | 109 (43.8) | 0.631 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Current | 37 (25.9) | 33 (20.0) | 56 (25.3) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Past | 34 (23.8) | 37 (22.4) | 46 (20.8) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Never | 72 (50.4) | 95 (57.6) | 119 (53.9) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alcohol drinkers | 135 (91.8) | 142 (84.0) | 177 (77.6)a | 0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Childhood SES | 0.6 (±2.6) | -0.1 (±2.3) | -0.7 (±2.2)a | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Birth cohort | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Early born 1886-1925 | 46 (31.3) | 91 (53.9)a | 180 (79.0)a,b | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Late born 1926-1958 | 101 (68.7) | 78 (46.2)a | 48 (21.1)a* | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gingivitis | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| No | 113 (76.9) | 132 (78.1) | 192 (85.0) | 0.094 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sometimes/Yes | 34 (23.1) | 37 (21.9) | 34 (15.0) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Periodontitis | 14 (9.7) | 26 (15.5) | 68 (31.9)a,b | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Prosthesis | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| None | 96 (65.3) | 83 (49.1)a | 21 (9.2)a,b | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Half | 0 (0.0) | 0 (0.0) | 185 (81.1)a,b | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Whole | 51 (36.7) | 86 (50.9)a | 22 (9.7)a,b | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cognitive performance | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Verbal ability | 55.0 (±8.7) | 52.5 (±8.3) | 49.2 (±8.7)a,b | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Memory | 56.03 (±9.4) | 51.5 (±9.4)a | 48.8 (±9.9)a | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spatial/fluid abilities | 56.4 (±9.4) | 51.8 (±8.0)a | 47.9 (±9.2)a,b | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Perceptual speed | 57.2 (±9.0) | 52.3 (±8.6)a | 46.5 (±9.2)a,b | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| General cognitive scorec | 57.5 (±9.1) | 52.6 (±7.9)a | 47.2 (±9.0)a,b | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: BMI= Body Mass Index; APOE= apolipoprotein; SES= Socioeconomic status. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Data are n (%) or mean (±SD). | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| aBonferroni pairwise comparison (reference= Eichner Index A). | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| bSignificant difference Eichner Index B vs C. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| cCognitive component score (T-score, mean= 50, standard deviation= 10) from cognitive tests of verbal ability, memory, spatial/fluid abilities, and perceptual speed. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Eichner categories in relation to cognitive decline
In basic-adjusted model (sex, education, birth cohort, and practice effects), compared to the participants in Eichner category A (optimal masticatory ability), those in category B had a lower performance in verbal ability at intercept. Moreover, participants in category B and C had a steeper decline in spatial/fluid abilities after age 65. There was no significant difference between Eichner category A relative to B or C in the intercept or slopes for perceptual speed, memory or the cognitive component score (Table 2 and Figure 1). After further adjustment for hypertension, heart disease, periodontal disease, prosthesis use, childhood SES, and alcohol consumption, the association between Eichner category B and verbal ability intercept remained significant, as did the association between Eichner category B and C with spatial/fluid abilities slope after age 65 (Supplementary Table 1).
Table 2. β-coefficients and 95% confidence intervals (CI) for age-related differences in mean cognitive performance and decline in different domains (T-scores) in relation to the Eichner Index (n=544).
| Eichner Index | Spatial/fluid abilities | Verbal ability | Memory | Perceptual speed | Component scorea | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| β (95% CI) | β (95% CI) | β (95% CI) | β (95% CI) | β (95% CI) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Intercept | A (Ref) | 68.23 (60.39 to 76.08) | 48.69 (43.79 to 53.59) | 60.45 (51.01 to 69.88) | 68.37 (62.01 to 74.72) | 62.19 (56.88 to 67.51) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| B | -6.90 (-17.40 to 3.60) | -6.94 (-12.48 to -1.40) | -4.59 (-18.49 to 9.31) | -1.79 (-13.67 to 10.08) | -3.23 (-11.56 to 5.10) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| C | -10.55 (-25.53 to 4.43) | -1.95 (-9.73 to 5.83) | -7.48 (-25.81 to 10.83) | -6.09 (-16.49 to 4.31) | -3.07 (-12.03 to 5.90) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Slope (linear age up to 65)b | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| A (Ref) | -0.19 (-0.31 to -0.06) | 0.09 (0.04 to 0.15) | -0.14 (-0.29 to 0.01) | -0.24 (-0.34 to -0.14) | -0.14 (-0.22 to -0.06) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| B | 0.07 (-0.11 to 0.24) | 0.10 (0.02 to 0.18) | 0.01 (-0.22 to 0.23) | -0.01 (-0.20 to 0.19) | 0.02 (-0.11 to 0.16) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| C | 0.10 (-0.13 to 0.34) | -0.00 (-0.11 to 0.11) | 0.07 (-0.22 to 0.36) | 0.03 (-0.14 to 0.20) | -0.01 (-0.15 to 0.13) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Slope (linear age from 65)b | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| A (Ref) | -0.30 (-0.39 to -0.21) | -0.18 (-0.26 to -0.11) | -0.21 (-0.31 to -0.10) | -0.56 (-0.65 to -0.46) | -0.36 (-0.43 to -0.28) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| B | -0.16 (-0.30 to -0.03) | -0.08 (-0.19 to 0.05) | -0.09 (-0.24 to 0.05) | -0.04 (-0.18 to 0.09) | -0.04 (-0.17 to 0.10) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| C | -0.15 (-0.28 to -0.02) | -0.10 (-0.21 to 0.01) | -0.12 (-0.26 to 0.03) | -0.14 (-0.27 to 0.00) | -0.10 (-0.27 to 0.04) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Model with age as timescale, adjusted for sex, education, birth cohort, and practice effect. The reference group was Eichner Index A. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| aCognitive component of tests for spatial/fluid abilities, verbal fluency, memory, and perceptual speed. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| bA knot was placed at age 65 for spatial/fluid, memory, and perceptual speed and at age 70 for verbal ability. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
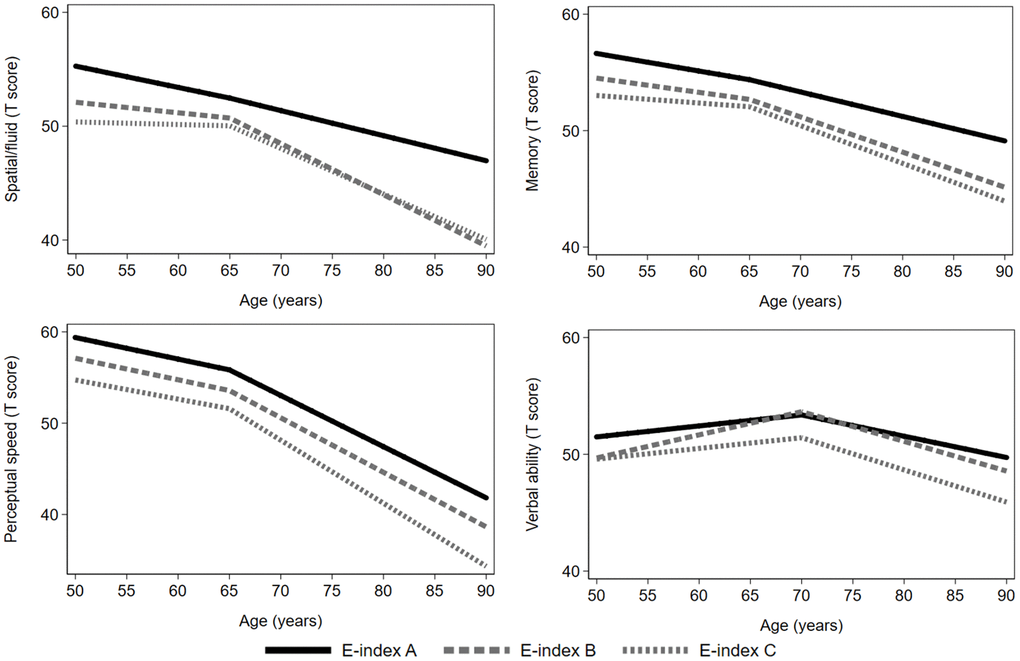
Figure 1. Age–related cognitive trajectories in different domains by Eichner categories. Model with age as timescale, adjusted for sex, education, birth cohort, and practice effects (n=544). The reference group was Eichner category A. A knot was placed at age 65 (spatial/fluid abilities, memory, and perceptual speed) or at age 70 (verbal ability).
Eichner categories in relation to dementia risk
During follow-up time (median=10 years, IQR= 16-3), accounting for a total of 8638 person-years, 52 out of the 544 (9.6%) participants developed dementia (8.6 cases per 1000 person-years). In crude and adjusted Cox regression models, estimates did not indicate higher dementia risk for participants in Eichner categories B and C compared to Eichner A (Table 3). Figure 2 shows the cumulative incidence of dementia according to Eichner categories accounting for the competing risk of death.
Table 3. Incidence rates (IR) per 1000 person-years and hazard ratios (HR) with 95% confidence intervals (95% CI) of all-cause dementia (n= 52) over 22-year follow-up by Eichner categories.
| Eichner Index | No. events/ person-years | IR (95% CI) | HR (95% CI)a | Adjusted HR (95% CI)b | Multi-adjusted HR (95% CI)c | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| A | 13/2496 | 5.20 (3.02 to 8.97) | 1.00 (reference) | 1.00 (reference) | 1.00 (reference) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| B | 17/2420 | 6.91 (4.29 to 11.11) | 0.90 (0.43 to 1.86) | 0.83 (0.39 to 1.76) | 1.03 (0.43 to 2.44) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| C | 22/2957 | 7.44 (4.90 to 11.30) | 0.73 (0.35 to 1.50) | 0.63 (0.30 to 1.29) | 0.79 (0.31 to 2.03) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| aCrude model. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| bModel adjusted for baseline sex and education. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| cAdditionally adjusted for birth cohort, hypertension, heart disease, periodontal disease, childhood SES, prosthesis use, diabetes, cerebrovascular disease, and alcohol consumption. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
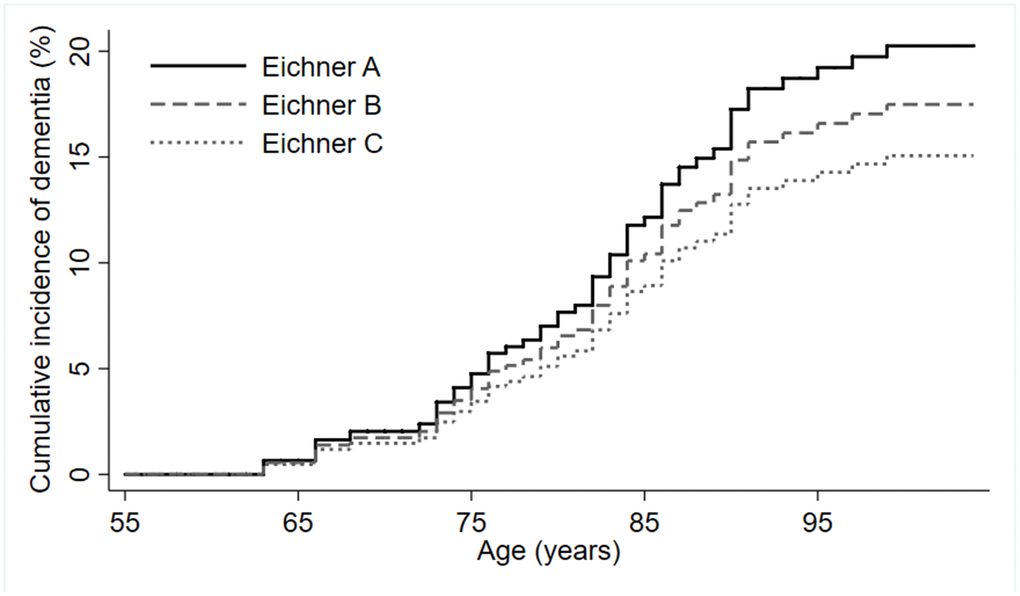
Figure 2. Cumulative incidence of dementia by Eichner categories (n=544).
Discussion
In this population–based longitudinal study with up to 22 years of follow-up, we found that having less posterior occlusal support, a marker of poorer masticatory ability, was related to the level of verbal ability and faster rate of decline in spatial/fluid abilities after the age of 65. We did not find an association between Eichner category and the risk of dementia.
In recent years, several studies have investigated the link between oral health and cognition in older age [21]. Tooth loss was used as a marker of oral health in most of these studies. A majority showed an association between tooth loss and cognitive decline [4, 5]. However, it is possible that some long-term adaptation to the edentulousness/tooth-loss condition in mastication is possible. Therefore, although being practical and reliable, the number of teeth lost per se may not fully reflect masticatory function. Using self-reported masticatory ability as an exposure of interest, a cross-sectional Swedish study of older community dwelling people showed that participants with difficulty in chewing hard food such as apples had a lower cognitive performance [22]. The authors also found that whether or not the dentition was natural or prosthetic had no significant influence on the observed association, suggesting that in mastication, the posterior teeth, more importantly, the contact of these teeth plays the most important role in maintaining cognition. This was indeed supported by our findings, whereby having more posterior contact was associated with better cognitive abilities in older Swedish adults over up to 22 years.
Previous research conducted on the relationship between mastication and cognition has been limited to cross-sectional studies, therefore the temporality for the observed associations is unclear. Some studies have focused on occlusal contacts as a measure of masticatory ability, which has been shown to correspond to self-reported and functional assessments of mastication [23]. In cognitively normal older Dutch persons, mastication was assessed by maximum mouth opening, jaw movement, bite force and the number of occluding pairs. The study revealed that 19% of the variation in episodic memory function could be predicted by jaw mobility and bite strength and that 22% of the variation in executive function was related to self-reported complaints about masticatory function [11]. Another study showed that word recall, verbal fluency, and numeracy was significantly better in people with good chewing ability, functionally measured with a two-color gum mixing ability test [24]. Two cross-sectional studies showed an association between masticatory ability and verbal fluency [24, 25].
In our study, we found baseline associations between Eichner categories and verbal ability, however, we found no effects of masticatory ability on trajectories of change in verbal ability. This could be because the long-term effects of masticatory problems have different effects as opposed to short term. Verbal ability at any one moment could be affected by mobility problems of the jaw as well as pronunciation difficulties due to more recent tooth loss, rather than a manifestation of neural abnormalities related to poor oral health [26]. Nevertheless, the association between Eichner categories and performance in any cognitive domain at intercept should be interpreted with caution, as there were very few data points available at age 50 in this sample. Similarly, we did not find an association between mastication and memory, as two previous cross-sectional studies have shown [11, 27]. The reason for this could be reverse causality due to pre-clinical cognitive impairment or dementia [28], which can lead to poorer oral health care. When examining the cognitive domains in relation to posterior occlusal support in our study, we removed participants with CIND at baseline, and examined the effects of mastication on normal aging.
Thus far, only two studies examined the relationship between masticatory and cognitive function or dementia longitudinally. One study reported a steeper decline in those with fewer posterior occlusal pairs [20]. However, cognitive function was measured using the Japanese version of the Montreal Cognitive Assessment, and therefore there was no indication of the longitudinal effects of mastication on specific cognitive domains, which could elucidate potential mechanisms involved. In the present study, we found an association between occlusal support and spatial/fluid ability. This is in line with a cross-sectional study showing that poorer mastication is related to worse executive functioning as well as reduced cerebral blood flow to the pre-frontal cortex, responsible for higher order cognitive processes [14]. There are several plausible mechanisms explaining the association between occlusal support and accelerated cognitive decline. A loss of posterior occlusal support encompasses a reduction in afferent nerve stimulation, which may cause sensory and motor cortical reorganization [29–31], affect cerebral functional streams toward multisensory hubs [32, 33], and result in memory and cognitive impairment [20]. Reduced masticatory stimulation might lead to cognitive decline through decreases in cerebral blood flow, decreases in activation of the cortical area and blood oxygen levels, particularly to the frontal cortex [12, 13].
It is important to note the selective survival in this study. Those who died had higher proportions of belonging to Eichner categories B and C, had lower baseline cognitive function and were older and in overall worse health. This means that our results may have been underestimated due to selective survival. Nevertheless, this study has a very long follow-up of maximum 22 years, and the mean age at baseline was 63, therefore, it is expected that a high proportion will die during such a long follow-up. As the results showed that Eichner B and C categories were associated with lower cognitive ability, it is possible that if the participants who died had been included, the association would have been even stronger.
To the best of our knowledge, this is the first prospective study that has investigated the association between an objective measure of mastication and the risk of dementia. We did not find poorer masticatory ability to be associated with a higher risk of dementia. This is in line with another study showing no increased risk of dementia over 4 years in participants with poorer self-reported mastication [34]. However, on average, participants with Eichner C were 10 years older than those in category A, and were overall in worse health and had the highest proportions of death during the study period. Therefore, while poor masticatory function could accelerate cognitive decline, due to the likely competing risk of death, those with the poorest masticatory function may not live long enough to develop clinical manifestations of dementia. Nevertheless, due to the low number of dementia incidence cases, the analysis of the association between Eichner categories and dementia risk may have been underpowered.
The major strengths of our study are the population–based cohort design, the long follow–up time, and the repeated cognitive testing. Furthermore, using composite scores of cognitive domains reduces ceiling and floor effects and measurement error variance common in single cognitive tests. However, some limitations need to be pointed out. First, the statistical power to detect group differences in stratified analyses regarding relevant population characteristics such as birth cohort, education and socioeconomic status was insufficient due to limited sample sizes. Second, we could not account for other functionally important factors involved in mastication such as pain, salivation or jaw mobility, which could have led to an underestimation of the current results.
In conclusion, this study provides evidence that poorer posterior occlusal support as a measure of masticatory ability is associated with a faster age-related decline in in spatial/fluid abilities. Further longitudinal studies with larger sample sizes exploring the association between mastication and cognitive health are warranted.
Materials and Methods
Study population
The Swedish Adoption/Twin Study of Aging (SATSA) is a population–based longitudinal study consisting of a subset of participants from the Swedish Twin Registry (STR) [35]. The study design of SATSA has been reported in detail elsewhere [36]. Briefly, from the base population of SATSA, all twin pairs who were aged ≥50 and participated in a mailed questionnaire in 1984 were invited to undergo clinical examinations and cognitive assessments by trained nurses starting in 1986 (first in–person testing, IPT1; n=759). Subsequently, the participants were followed-up every three years from 1986 until 2012. Throughout the study period, nine waves of examinations (IPT1 to IPT9) were carried out. Information on dental status was collected during IPT2 (1989-1991), therefore, only the participants who were assessed at IPT2 were included in this study (n=595), henceforth referred to as the baseline. After excluding participants with missing information on dental status (n = 5), with dementia (n = 8) or cognitive impairment no dementia (CIND) at baseline (n=38), 544 dementia-free participants remained for the current study.
Informed consent was obtained from all participants. SATSA was approved by the Regional Ethics Board at Karolinska Institutet, Stockholm, Sweden. Confidentiality and anonymity were guaranteed as part of the informed consent. Participants were informed that their involvement in the study was voluntary and that they were free to withdraw from the study at any point in time.
Data collection
From the original questionnaire in 1984, information was collected on demographics (i.e., education) and at each wave, on lifestyle factors (i.e., smoking, alcohol consumption, and physical exercise). Nurses measured blood pressure, weight, and height at baseline and at each follow–up examination. Information on medical conditions (e.g. hypertension, heart diseases), and medication use was obtained through self–report at baseline and each follow-up examination. Specifically, hypertension was defined as resting blood pressure ≥140/90 mmHg, and/or self-reported use of antihypertensive medication [37]. Heart diseases (heart failure, coronary heart diseases, and heart attack), and stroke, and were assessed based on self-reported medical history at baseline. Diabetes was ascertained at baseline and each follow-up based on self-reported medical history, use of hypoglycaemic medications (oral hypoglycemic agents or insulin), or FBG ≥7.0 mmol/L, or nonfasting blood glucose (noFBG) ≥11 mmol/L. Blood samples were taken at study entry and the apolipoprotein E (APOE) gene was genotyped utilizing high-throughput sequencing and dichotomized as any ε4 carriers or ε4 noncarriers.
Educational level was dichotomized as low (elementary or vocational, ≤ 9 years) and high (high school or above, >9 years). Body mass index (BMI) was calculated as weight in kilograms divided by squared height in meters (kg/m2) [38]. Smoking status was categorized as non‒smoker (participants who had never smoked), past smoker and current smoker. Alcohol consumption was dichotomized as never-drinker (never drink alcohol) and drinker (former and current drinker). Socioeconomic status (SES) in childhood (rearing home) was measured from a scale including three components: material resources within the household, highest education of the parents, and highest occupational status of the parents. This scale is based on factor analyses. Variables were standardized to a mean of 0 and a standard deviation of 1 before summing. A higher score on the scale reflects higher SES level [39].
Assessment of dental status
During IPT2 trained nurses examined and recorded the presence/absence of each tooth and type of filling if present. The nurses also collected information using a questionnaire about whether the participants used prostheses, categorized as none, half-prosthesis or whole prosthesis. Additionally, the participants were asked if they have problems with gingivitis (bleeding gums; no/yes or sometimes) or periodontal disease (yes/no).
Each participant was categorized according to the Eichner Index [18]. In the Eichner classification, each posterior contact, including both the premolar and molar regions, are counted as one zone, yielding a total of four supporting zones [40]. The Eichner Index describes the existing posterior occlusal support zones by dividing the occlusal status into three main groups (A, B and C). Individuals classified in Group A have occlusal contacts in all four posterior support zones (indicating optimal masticatory ability), those in group B have 1-3 occlusal contacts (indicating moderate masticatory ability) and those in group C have no posterior occlusal contact at all (indicating poor masticatory ability). The categorization was checked for errors independently by I.W. (DDS, specialist in Orofacial medicine) for a random 10% of the sample.
Assessment of cognitive domains, CIND, and dementia
The cognitive battery included 12 tests assessing four cognitive domains: verbal abilities (information, synonyms, and analogies), spatial/fluid (Figure logic, Kohs Block Design, and Card rotations), memory (Digit span forwards and backward, Thurstone’s pictures memory, Name and faces immediate and delayed recall), and perceptual speed (Symbol digit, and Figure identification) [41]. These domains were identified by principal–component analysis (PCA) [42]. Briefly, cognitive assessments at each wave were standardized using the means and variances observed at baseline. For each wave, a factor representing each cognitive domain was generated by combining the standardized cognitive scores using the factor weights derived from the PCA at baseline. A cognitive component was created based on the first principal component of nine cognitive tests (Information, Synonyms, Analogies, Koh's Block Design, Card Rotations, Thurstone's Picture Memory, Digit Span forwards and backward, Symbol Digit, and Figure Identification). All component scores were rescaled as t-scores by adding a constant of 50 and multiplying by 10 [43].
Cognitive impairment–no dementia (CIND) was considered as the condition where the observed cognitive deficits were not severe enough to meet the criteria for dementia diagnosis. A person was categorized has having CIND if the person’s Mini–Mental State Examination (MMSE) at study entry was at least 1 SD or 2 SDs below the age- and education-specific mean MMSE in people aged 50-75 years or ≥75 years, respectively [44]. Dementia was diagnosed at follow–up examinations according to criteria from the Diagnostic and Statistical Manual of Mental Disorders, Third or Fourth Editions (DSM–III or DSM–IV) [42, 45]. Clinical diagnosis of dementia was determined during a consensus meeting, in which performance on cognitive tests, health, daily functioning, and medical records were reviewed [46].
Statistical analyses
Differences in characteristics of the participants by Eichner categories (A, B, and C) at study entry were assessed using chi-square (χ2) or two–tailed one-way ANOVA with post hoc group comparisons with Bonferroni correction. Piecewise linear mixed-effects models were used to estimate the association of Eichner categories with intercept and annual rate of change in each cognitive domain using age as the time scale. Previous studies on the SATSA cohort have shown that age-related decline in memory, spatial ability, and perceptual speed starts around age 65, therefore a knot was positioned at age 65 for these domains, whereas for verbal ability decline starts around 70 hence, the knot was positioned at age 70 [47]. The fixed effects included baseline Eichner category (category A vs category B or C), linear age, and their interaction (Eichner category × age). All models included a random intercept and two random slopes (splines) for age before and after the knot, allowing individual differences at intercept and over time (age). The random effects accounted for both the repeated measures for each person and the presence of twin pairs by using a person–specific identifier and a common twin–pair identifier. The follow–up was censored when dementia occurred. An unstructured variance–covariance matrix was employed in all models with robust standard errors. Likelihood-ratio tests were used to determine which parameters should be included in the final model.
Sex, education, and birth cohort (defined as early birth Cohort 1: born 1886-1925 and late birth Cohort 2: born 1926-1958) were included as covariates in the main analysis. Birth cohort was adjusted for as the participants had a wide age-range at study entry, which may give rise to cohort effects due to the long follow-up period. To account for the possibility of practice effects for the cognitive testing, we also included a time-varying retest covariate (“First cognitive assessment” vs “Follow-up assessment”). In additional analyses, we further adjusted for demographic factors, baseline lifestyle factors and medical conditions such as alcohol consumption, hypertension, heart disease, prosthesis use, periodontal disease, cerebrovascular disease, diabetes, and childhood socioeconomic status.
Incidence rates (IRs) and 95% confidence intervals (CIs) of dementia per 1000 person-years were calculated for participants as the number of events during the follow-up period divided by person-years of follow-up. Cox proportional hazards regression models were used to estimate the cause-specific hazard ratios (HRs) and 95% CIs of dementia related to Eichner category A, B or C. Follow-up time was calculated as age from study entry until dementia diagnosis, otherwise until death or last in-person testing. Age was used as the time scale. The proportional hazard assumption was tested for the predictor and covariates, using Schoenfeld’s residuals regressed against follow-up time (age). No violation of proportionality was observed. Crude, basic-adjusted (sex and education), and multi-adjusted models (additionally adjusted for birth cohort, childhood socioeconomic status, periodontal disease, hypertension, heart disease, alcohol use and prosthesis use) were computed. A robust standard error estimator was used to adjust for the potential dependencies within twin pairs [48]. All statistical analyses were performed with Stata SE 15.0 (StataCorp LP., College Station, Texas, USA) and RStudio v. 1.2.5001 (RStudio: Integrated Development for R. RStudio, Inc., Boston).
Supplementary Materials
Author Contributions
Name Location Role Contribution Christina S. Dintica Karolinska Institutet, Stockholm, Sweden Author, Corresponding author Design and conceptualized study; analyzed the data; drafted the manuscript for intellectual content Anna Marseglia, PhD Karolinska Institutet, Stockholm, Sweden Author Drafting/revising manuscript for content; analysis interpretation Inger Wårdh Karolinska Institutet, Stockholm, Sweden Author Drafting/revising manuscript for content; checked the categorisation of the exposure (Eichner Index) Debora Rizzuto, PhD Karolinska Institutet, Stockholm, Sweden Author Drafting/revising manuscript for content; analysis interpretation Ying Shang, MSc Karolinska Institutet, Stockholm, Sweden Author Statistical advice and analysis interpretation Nancy L. Pedersen, PhD Karolinska Institutet, Stockholm, Sweden Author Acquisition of data; Drafting/revising manuscript for content; study supervision Weili Xu, MD, PhD Karolinska Institutet, Stockholm, Sweden Author Drafting/revising manuscript for content; Design and conceptualized study; study supervision
Acknowledgments
The authors would like to express their gratitude to the participants and staff involved in the data collection and management in the SATSA.
Conflicts of Interest
Christina S. Dintica, Anna Marseglia, Inger Wårdh, Debora Rizzuto, Ying Shang, Weili Xu, and Nancy L. Pedersen report no conflicts of interest.
Funding
This work was supported by grants from the National Institutes of Health (grant numbers AG 04563, AG10175), the MacArthur Research Network on Successful Aging, and the Swedish Research Council. WX received grants from the Swedish Research Council (grant number 2017-00981), the National Natural Science Foundation of China (grant number 81771519), Demensfonden, the Konung Gustaf V:s och Drottning Victorias Frimurare Foundation (grant number 2016-2017) and Alzeimerfonden (grant number 2017-2018). CSD is supported in part by Karolinska Institutet Doctoral Training grant, SWEAH, and Stiftelsen För Gamla Tjänarinnor. This project is part of CoSTREAM (http://www.costream.eu/) and received funding from the European Union’s Horizon 2020 research and innovation programme (grant number 667375).
References
- 1. Livingston G, Sommerlad A, Orgeta V, Costafreda SG, Huntley J, Ames D, Ballard C, Banerjee S, Burns A, Cohen-Mansfield J, Cooper C, Fox N, Gitlin LN, et al. Dementia prevention, intervention, and care. Lancet. 2017; 390:2673–734. https://doi.org/10.1016/S0140-6736(17)31363-6 [PubMed]
- 2. Delwel S, Binnekade TT, Perez RS, Hertogh CM, Scherder EJ, Lobbezoo F. Oral health and orofacial pain in older people with dementia: a systematic review with focus on dental hard tissues. Clin Oral Investig. 2017; 21:17–32. https://doi.org/10.1007/s00784-016-1934-9 [PubMed]
- 3. Kassebaum NJ, Smith AG, Bernabé E, Fleming TD, Reynolds AE, Vos T, Murray CJ, Marcenes W, Abyu GY, Alsharif U, Asayesh H, Benzian H, Dandona L, et al, and GBD 2015 Oral Health Collaborators. Global, Regional, and National Prevalence, Incidence, and Disability-Adjusted Life Years for Oral Conditions for 195 Countries, 1990-2015: A Systematic Analysis for the Global Burden of Diseases, Injuries, and Risk Factors. J Dent Res. 2017; 96:380–87. https://doi.org/10.1177/0022034517693566 [PubMed]
- 4. Cerutti-Kopplin D, Feine J, Padilha DM, de Souza RF, Ahmadi M, Rompré P, Booij L, Emami E. Tooth Loss Increases the Risk of Diminished Cognitive Function: A Systematic Review and Meta-analysis. JDR Clin Trans Res. 2016; 1:10–19. https://doi.org/10.1177/2380084416633102 [PubMed]
- 5. Li J, Xu H, Pan W, Wu B. Association between tooth loss and cognitive decline: A 13-year longitudinal study of Chinese older adults. PLoS One. 2017; 12:e0171404. https://doi.org/10.1371/journal.pone.0171404 [PubMed]
- 6. Kaye EK, Valencia A, Baba N, Spiro A
3rd , Dietrich T, Garcia RI. Tooth loss and periodontal disease predict poor cognitive function in older men. J Am Geriatr Soc. 2010; 58:713–18. https://doi.org/10.1111/j.1532-5415.2010.02788.x [PubMed] - 7. Okamoto N, Morikawa M, Amano N, Yanagi M, Takasawa S, Kurumatani N. Effects of Tooth Loss and the Apolipoprotein E ɛ4 Allele on Mild Memory Impairment in the Fujiwara-kyo Study of Japan: A Nested Case-Control Study. J Alzheimers Dis. 2017; 55:575–83. https://doi.org/10.3233/JAD-160638 [PubMed]
- 8. Kato H, Takahashi Y, Iseki C, Igari R, Sato H, Sato H, Koyama S, Tobita M, Kawanami T, Iino M, Ishizawa K, Kato T. Tooth Loss-associated Cognitive Impairment in the Elderly: A Community-based Study in Japan. Intern Med. 2019; 58:1411–16. https://doi.org/10.2169/internalmedicine.1896-18 [PubMed]
- 9. Chen J, Ren CJ, Wu L, Xia LY, Shao J, Leng WD, Zeng XT. Tooth Loss Is Associated With Increased Risk of Dementia and With a Dose-Response Relationship. Front Aging Neurosci. 2018; 10:415. https://doi.org/10.3389/fnagi.2018.00415 [PubMed]
- 10. Dintica CS, Rizzuto D, Marseglia A, Kalpouzos G, Welmer AK, Wårdh I, Bäckman L, Xu W. Tooth loss is associated with accelerated cognitive decline and volumetric brain differences: a population-based study. Neurobiol Aging. 2018; 67:23–30. https://doi.org/10.1016/j.neurobiolaging.2018.03.003 [PubMed]
- 11. Scherder E, Posthuma W, Bakker T, Vuijk PJ, Lobbezoo F. Functional status of masticatory system, executive function and episodic memory in older persons. J Oral Rehabil. 2008; 35:324–36. https://doi.org/10.1111/j.1365-2842.2007.01842.x [PubMed]
- 12. Onozuka M, Fujita M, Watanabe K, Hirano Y, Niwa M, Nishiyama K, Saito S. Age-related changes in brain regional activity during chewing: a functional magnetic resonance imaging study. J Dent Res. 2003; 82:657–60. https://doi.org/10.1177/154405910308200817 [PubMed]
- 13. Otsuka T, Yamasaki R, Shimazaki T, Yoshino F, Sasaguri K, Kawata T. Effects of mandibular retrusive deviation on prefrontal cortex activation: a functional near-infrared spectroscopy study. Biomed Res Int. 2015; 2015:373769. https://doi.org/10.1155/2015/373769 [PubMed]
- 14. Hirano Y, Obata T, Takahashi H, Tachibana A, Kuroiwa D, Takahashi T, Ikehira H, Onozuka M. Effects of chewing on cognitive processing speed. Brain Cogn. 2013; 81:376–81. https://doi.org/10.1016/j.bandc.2012.12.002 [PubMed]
- 15. Naka O, Anastassiadou V, Pissiotis A. Association between functional tooth units and chewing ability in older adults: a systematic review. Gerodontology. 2014; 31:166–77. https://doi.org/10.1111/ger.12016 [PubMed]
- 16. Fontijn-Tekamp FA, Slagter AP, Van Der Bilt A, Van 'T Hof MA, Witter DJ, Kalk W, Jansen JA. Biting and chewing in overdentures, full dentures, and natural dentitions. J Dent Res. 2000; 79:1519–24. https://doi.org/10.1177/00220345000790071501 [PubMed]
- 17. Ikebe K, Matsuda K, Morii K, Furuya-Yoshinaka M, Nokubi T, Renner RP. Association of masticatory performance with age, posterior occlusal contacts, occlusal force, and salivary flow in older adults. Int J Prosthodont. 2006; 19:475–81. [PubMed]
- 18. Ikebe K, Matsuda K, Kagawa R, Enoki K, Okada T, Yoshida M, Maeda Y. Masticatory performance in older subjects with varying degrees of tooth loss. J Dent. 2012; 40:71–6. https://doi.org/10.1016/j.jdent.2011.10.007 [PubMed]
- 19. Takeuchi K, Ohara T, Furuta M, Takeshita T, Shibata Y, Hata J, Yoshida D, Yamashita Y, Ninomiya T. Tooth Loss and Risk of Dementia in the Community: the Hisayama Study. J Am Geriatr Soc. 2017; 65:e95–100. https://doi.org/10.1111/jgs.14791 [PubMed]
- 20. Hatta K, Ikebe K, Gondo Y, Kamide K, Masui Y, Inagaki H, Nakagawa T, Matsuda KI, Ogawa T, Inomata C, Takeshita H, Mihara Y, Fukutake M, et al. Influence of lack of posterior occlusal support on cognitive decline among 80-year-old Japanese people in a 3-year prospective study. Geriatr Gerontol Int. 2018; 18:1439–46. https://doi.org/10.1111/ggi.13508 [PubMed]
- 21. Wu B, Fillenbaum GG, Plassman BL, Guo L. Association Between Oral Health and Cognitive Status: A Systematic Review. J Am Geriatr Soc. 2016; 64:739–51. https://doi.org/10.1111/jgs.14036 [PubMed]
- 22. Lexomboon D, Trulsson M, Wårdh I, Parker MG. Chewing ability and tooth loss: association with cognitive impairment in an elderly population study. J Am Geriatr Soc. 2012; 60:1951–56. https://doi.org/10.1111/j.1532-5415.2012.04154.x [PubMed]
- 23. Osterberg T, Mellström D, Sundh V. Dental health and functional ageing. A study of 70-year-old people. Community Dent Oral Epidemiol. 1990; 18:313–18. https://doi.org/10.1111/j.1600-0528.1990.tb00087.x [PubMed]
- 24. Listl S. Oral health conditions and cognitive functioning in middle and later adulthood. BMC Oral Health. 2014; 14:70. https://doi.org/10.1186/1472-6831-14-70 [PubMed]
- 25. Weijenberg RA, Lobbezoo F, Visscher CM, Scherder EJ. Oral mixing ability and cognition in elderly persons with dementia: a cross-sectional study. J Oral Rehabil. 2015; 42:481–86. https://doi.org/10.1111/joor.12283 [PubMed]
- 26. Mefferd AS, Corder EE. Assessing articulatory speed performance as a potential factor of slowed speech in older adults. J Speech Lang Hear Res. 2014; 57:347–60. https://doi.org/10.1044/2014_JSLHR-S-12-0261 [PubMed]
- 27. Hansson P, Sunnegårdh-Grönberg K, Bergdahl J, Bergdahl M, Nyberg L, Nilsson LG. Relationship between natural teeth and memory in a healthy elderly population. Eur J Oral Sci. 2013; 121:333–40. https://doi.org/10.1111/eos.12060 [PubMed]
- 28. Jack CR
Jr , Knopman DS, Jagust WJ, Petersen RC, Weiner MW, Aisen PS, Shaw LM, Vemuri P, Wiste HJ, Weigand SD, Lesnick TG, Pankratz VS, Donohue MC, Trojanowski JQ. Tracking pathophysiological processes in Alzheimer’s disease: an updated hypothetical model of dynamic biomarkers. Lancet Neurol. 2013; 12:207–16. https://doi.org/10.1016/S1474-4422(12)70291-0 [PubMed] - 29. Henry EC, Marasco PD, Catania KC. Plasticity of the cortical dentition representation after tooth extraction in naked mole-rats. J Comp Neurol. 2005; 485:64–74. https://doi.org/10.1002/cne.20511 [PubMed]
- 30. Avivi-Arber L, Lee JC, Sessle BJ. Effects of incisor extraction on jaw and tongue motor representations within face sensorimotor cortex of adult rats. J Comp Neurol. 2010; 518:1030–45. https://doi.org/10.1002/cne.22261 [PubMed]
- 31. Avivi-Arber L, Lee JC, Sessle BJ. Dental Occlusal Changes Induce Motor Cortex Neuroplasticity. J Dent Res. 2015; 94:1757–64. https://doi.org/10.1177/0022034515602478 [PubMed]
- 32. Sepulcre J, Sabuncu MR, Yeo TB, Liu H, Johnson KA. Stepwise connectivity of the modal cortex reveals the multimodal organization of the human brain. J Neurosci. 2012; 32:10649–61. https://doi.org/10.1523/JNEUROSCI.0759-12.2012 [PubMed]
- 33. Sepulcre J. Functional streams and cortical integration in the human brain. Neuroscientist. 2014; 20:499–508. https://doi.org/10.1177/1073858414531657 [PubMed]
- 34. Yamamoto T, Kondo K, Hirai H, Nakade M, Aida J, Hirata Y. Association between self-reported dental health status and onset of dementia: a 4-year prospective cohort study of older Japanese adults from the Aichi Gerontological Evaluation Study (AGES) Project. Psychosom Med. 2012; 74:241–48. https://doi.org/10.1097/PSY.0b013e318246dffb [PubMed]
- 35. Lichtenstein P, De Faire U, Floderus B, Svartengren M, Svedberg P, Pedersen NL. The Swedish Twin Registry: a unique resource for clinical, epidemiological and genetic studies. J Intern Med. 2002; 252:184–205. https://doi.org/10.1046/j.1365-2796.2002.01032.x [PubMed]
- 36. Finkel D, Pedersen NL. Processing Speed and Longitudinal Trajectories of Change for Cognitive Abilities: The Swedish Adoption/Twin Study of Aging. Neuropsychol Dev Cogn B Aging Neuropsychol Cogn. 2004; 11:325–45. https://doi.org/10.1080/13825580490511152
- 37. Chobanian AV, Bakris GL, Black HR, Cushman WC, Green LA, Izzo JL
Jr , Jones DW, Materson BJ, Oparil S, Wright JTJr , Roccella EJ; National Heart, Lung, and Blood Institute Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure, and National High Blood Pressure Education Program Coordinating Committee. The Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure: the JNC 7 report. JAMA. 2003; 289:2560–72. https://doi.org/10.1001/jama.289.19.2560 [PubMed] - 38. Dahl AK, Hassing LB, Fransson EI, Gatz M, Reynolds CA, Pedersen NL. Body mass index across midlife and cognitive change in late life. Int J Obes. 2013; 37:296–302. https://doi.org/10.1038/ijo.2012.37 [PubMed]
- 39. Ericsson M, Lundholm C, Fors S, Dahl Aslan AK, Zavala C, Reynolds CA, Pedersen NL. Childhood social class and cognitive aging in the Swedish Adoption/Twin Study of Aging. Proc Natl Acad Sci USA. 2017; 114:7001–06. https://doi.org/10.1073/pnas.1620603114 [PubMed]
- 40. Hiltunen K, Vehkalahti MM, Peltola JS, Ainamo A. A 5-year follow-up of occlusal status and radiographic findings in mandibular condyles of the elderly. Int J Prosthodont. 2002; 15:539–43. [PubMed]
- 41. Pedersen NL, Plomin R, Nesselroade JR, McClearn GE. A Quantitative Genetic Analysis of Cognitive Abilities during the Second Half of the Life Span. Psychol Sci. 1992; 3:346–53. https://doi.org/10.1111/j.1467-9280.1992.tb00045.x
- 42. Finkel D, Reynolds CA, McArdle JJ, Pedersen NL. Age changes in processing speed as a leading indicator of cognitive aging. Psychol Aging. 2007; 22:558–68. https://doi.org/10.1037/0882-7974.22.3.558 [PubMed]
- 43. Finkel D, Reynolds CA, McArdle JJ, Pedersen NL. Cohort differences in trajectories of cognitive aging. J Gerontol B Psychol Sci Soc Sci. 2007; 62:286–94. https://doi.org/10.1093/geronb/62.5.P286 [PubMed]
- 44. Marseglia A, Fratiglioni L, Laukka EJ, Santoni G, Pedersen NL, Bäckman L, Xu W. Early cognitive deficits in type 2 diabetes: a population-based study. J Alzheimers Dis. 2016; 53:1069–78. https://doi.org/10.3233/JAD-160266 [PubMed]
- 45. Bokenberger K, Pedersen NL, Gatz M, Dahl AK. The type A behavior pattern and cardiovascular disease as predictors of dementia. Health Psychol. 2014; 33:1593–601. https://doi.org/10.1037/hea0000028 [PubMed]
- 46. Bokenberger K, Ström P, Dahl Aslan AK, Johansson AL, Gatz M, Pedersen NL, Åkerstedt T. Association Between Sleep Characteristics and Incident Dementia Accounting for Baseline Cognitive Status: A Prospective Population-Based Study. J Gerontol A Biol Sci Med Sci. 2017; 72:134–39. https://doi.org/10.1093/gerona/glw127 [PubMed]
- 47. Finkel D, Reynolds CA, McArdle JJ, Gatz M, Pedersen NL. Latent growth curve analyses of accelerating decline in cognitive abilities in late adulthood. Dev Psychol. 2003; 39:535–50. https://doi.org/10.1037/0012-1649.39.3.535 [PubMed]
- 48. Lin DY, Wei LJ. The Robust Inference for the Cox Proportional Hazards Model. J Am Stat Assoc. 1989; 84:1074–78. https://doi.org/10.1080/01621459.1989.10478874


