Introduction
Triple negative breast cancer (TNBC) is defined by a lack of estrogen receptor (ER), progesterone receptor (PR) and human epidermal growth factor 2 receptor (HER2) and remains the most challenging breast cancer to treat. Recently, in accordance with ESR1, PGR, and ERBB2 expression and distinct patterns of molecular alterations, TNBC has been further subcategorized into 7 different subtypes: basal-like 1 (BL1), basal-like 2 (BL2), mesenchymal (M), immunomodulatory (IM), luminal androgenic receptor (LAR), mesenchymal stem-like (MSL) [1]. This seven-subtype classification has been shown to independently predict a pathologic complete response (pCR) but not distant metastasis-free or overall survival in a retrospective analysis of TNBC patients treated with neoadjuvant chemotherapy [2]. Clinically, the life-threatening metastatic spread of TNBC preferentially to the lungs and brain usually occurs within 3 years after surgery and leads to a worse disease-specific outcome than other breast cancer subtypes [3]. In the past decade, major efforts have been made to classify TNBC into distinct clinical and molecular subtypes to effectively guide treatment decisions, prevent the development of metastatic disease and ultimately improve survival in this patient population [4]. However, the molecular mechanism underlying TNBC metastasis remains largely unknown.
Gαh is also known as tissue transglutaminase (tTG) or transglutaminase 2 (TG2) because of its transamidation activity when the ratio of the intracellular Ca2+ concentration to the GTP concentration is increased [5]. An increased level of Gαh has been detected in various types of cancer cells and is associated with cancer progression, e.g., therapeutic resistance and metastasis, and poor prognosis [6–11]. Intriguingly, recent reports demonstrated that GTP-binding activity of Gαh, but not transamidation, is required for the metastatic progression of breast cancer [12, 13], although Gαh expression levels are causally correlated with the metastatic potential of other cancers [14, 15]. Our recent report also showed that the coupling of Gαh with phospholipase C-δ1 (PLC-δ1)-related signaling pathway enhances the lung metastasis of TNBC cells [16]. On the other hand, the association between Gαh activity/expression and Akt/mTOR pathway, as well as autophagosome degradation, has been demonstrated in several types of cancer cells [17–22]. Nevertheless, the involvement of the Akt/mTOR pathway and autophagy activity in Gαh/PLC-δ1-driven TNBC metastasis remains unclear.
To this end, in this study, we performed an in silico experiment using gene set enrichment analysis (GSEA) of the transcriptional coexpression status of Gαh in primary tumors derived from ER-negative breast cancer patients defined as having low-level Gαh expression without lung metastasis or high-level Gαh expression with lung metastasis. The GSEA results revealed that the mTORC1-related pathway might be activated in the Gαh-associated lung metastasis of ER-negative breast cancer. We also found that the interruption of the Gαh and PLC-δ1 interaction suppresses the activation of Akt/mTORC1 but promotes the initiation of autophagy, which ultimately inhibits the metastatic progression of TNBC cells in vitro and in vivo. In addition to describing the PPI inhibitor of the Gαh/PLC-δ1 complex, this study suggests another strategy for using a mTORC1 inhibitor, e.g., rapamycin, to combat metastatic TNBC with upregulated Gαh.
Results
The upregulation of Gαh accompanied by mTORC1 activation correlates with an increased risk for lung metastasis in ER(-) breast cancer patients
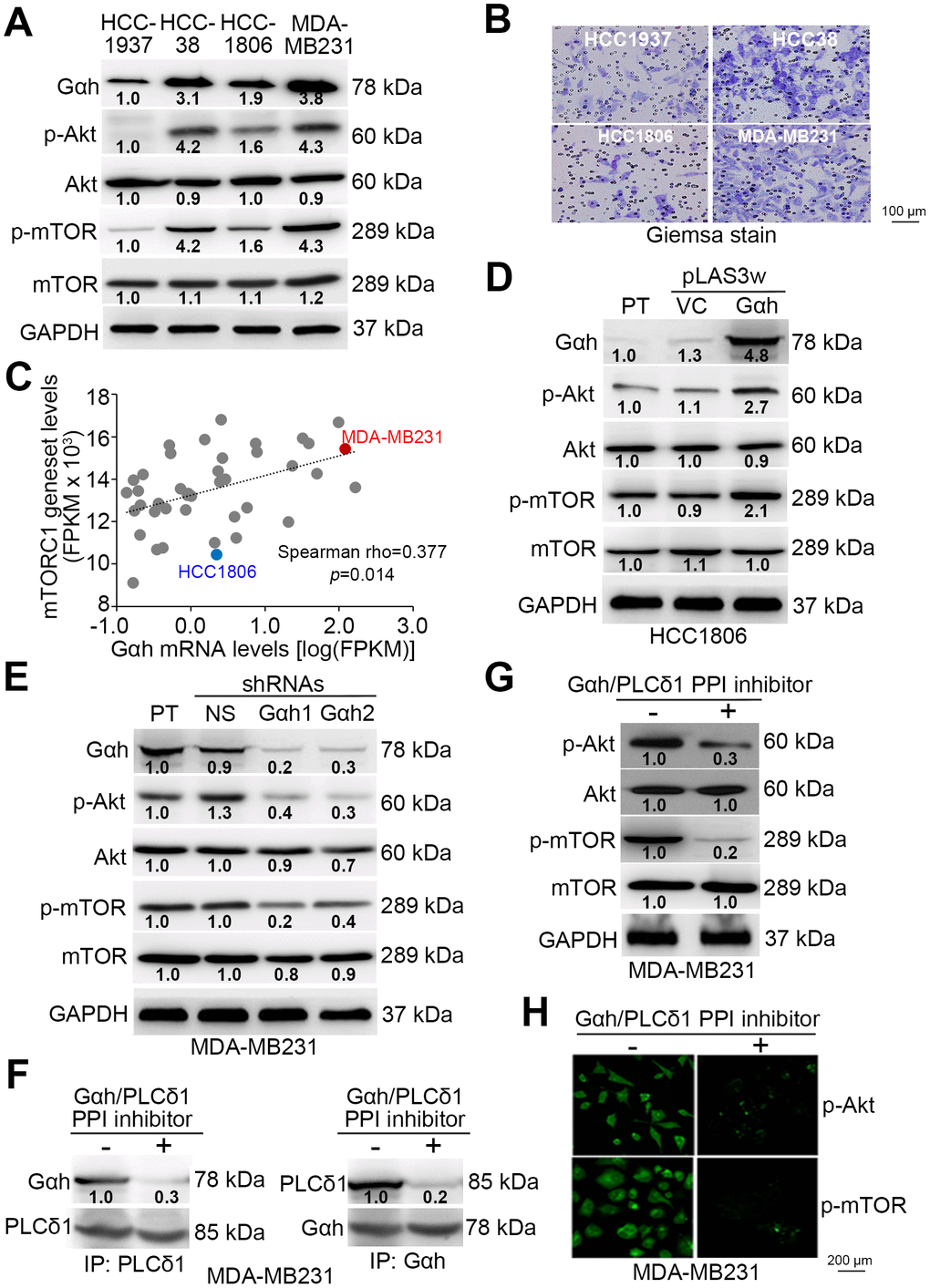
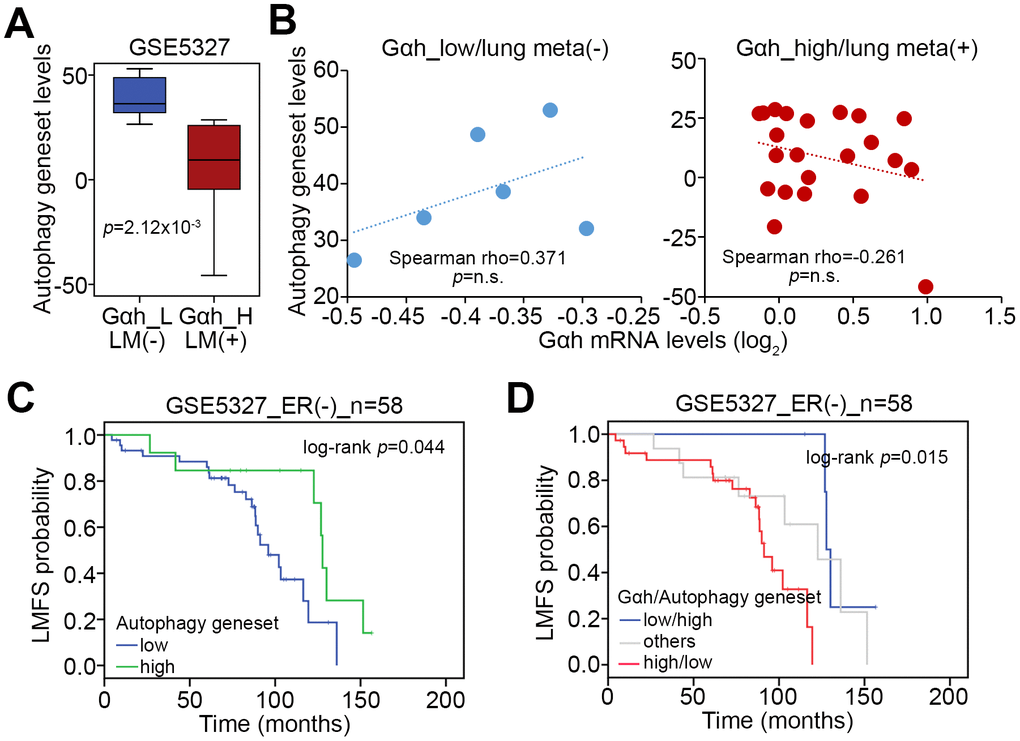
We selected the top 10% of the upregulated and downregulated genes derived from the non-lung metastatic and lung metastatic ER(-) breast cancer tissues with low- and high-levels of Gαh as previously defined with a Kaplan-Meier analysis [16] to perform an in silico gene set enrichment analysis (GSEA) (Figure 1A). GSEA results demonstrated that the MTORC1 signaling pathway is significantly predicted to be inhibited in non-lung metastatic ER(-) breast cancer tissues with low levels of Gαh expression (p<0.01) but activated in lung metastatic ER(-) breast cancer tissues with high levels of Gαh expression (Figure 1B). Accordingly, the number of transcript for the mTORC1 gene set of lung metastatic ER(-) breast cancer tissues with high Gαh levels was prominently higher than the number of the mTORC1 gene sets for non-lung metastatic ER(-) breast cancer tissues with low Gαh levels (Figure 1C). Whereas the mRNA levels of the mTORC1 gene set and Gαh appeared to be negatively correlated in the non-lung metastatic ER(-) breast cancer tissues, their expression levels were significantly and positively correlated in the lung metastatic ER(-) breast cancer tissues with high Gαh levels (p<0.0001) (Figure 1D). The results from the Kaplan-Meier analysis revealed that higher mRNA levels of the mTORC1 gene set correlated with a poor lung metastasis-free survival probability in ER(-) breast cancer patients of the GSE5327 data set (Figure 1E). Moreover, the signature of the combined high-level mTORC1 gene set and Gαh significantly predicted a shortened period for lung metastasis in ER(-) breast cancer patients of the GSE5327 dataset (p=0.00091) (Figure 1F).
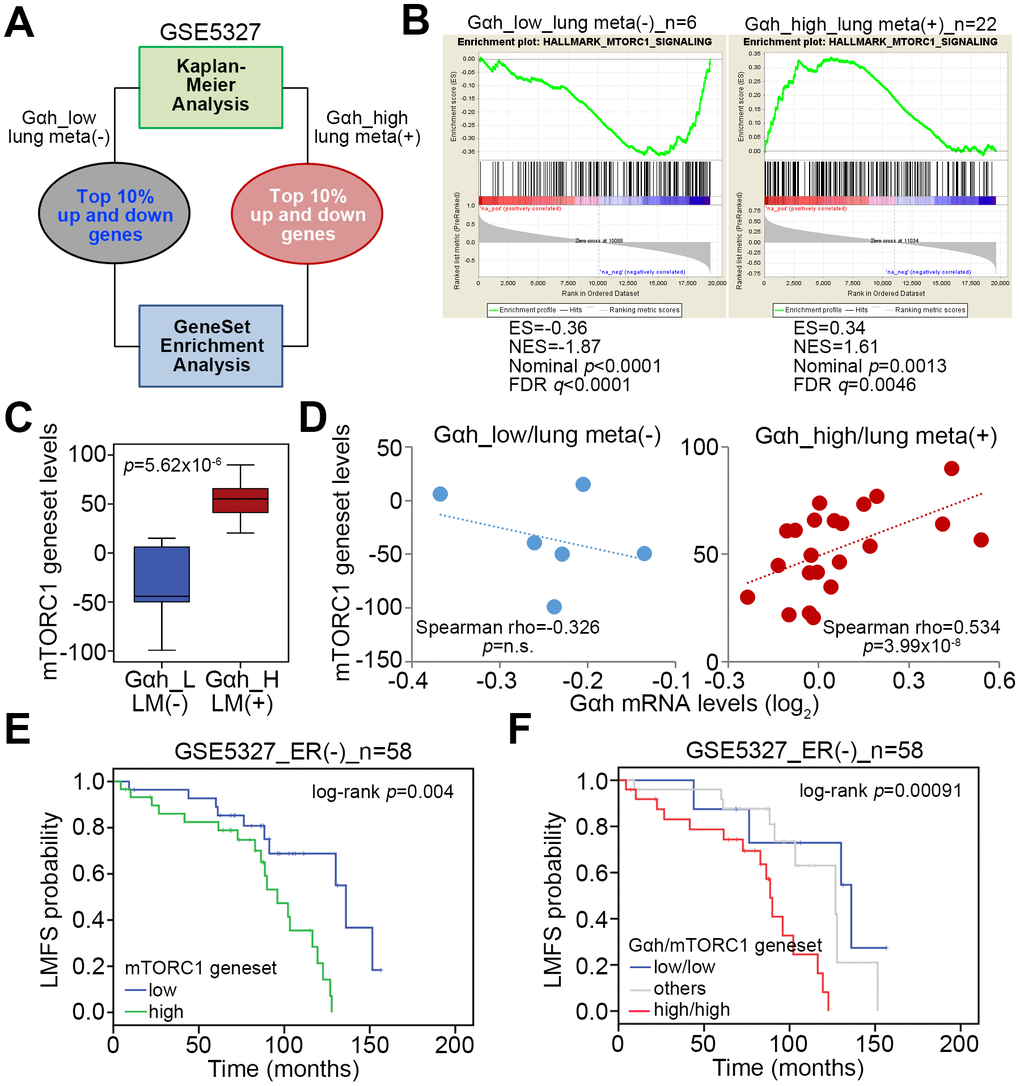
Figure 1. The mTORC1-related pathway is putatively activated in ER(-) breast cancer with high-level Gαh expression and lung metastasis. (A) Flowchart of the gene enrichment analysis (GSEA) using the transcription profiling of the top 10% upregulated and downregulated genes in ER(-) breast cancer tissues that were defined with low-level Gαh expression without lung metastasis or high-level Gαh expression with lung metastasis in a Kaplan-Meier analysis based on the GSE5327 data set. (B) The enrichment score (ES) derived from the correlation between the MTORC1 gene set and the queried gene signatures was plotted as the green curve. The parameters including the normalized enrichment score (NES), nominal p values and false discovery rates (FDRs) are shown as inserts. (C) Transcriptional profiling of the MTORC1 gene set in the groups is shown in A. The statistical significance was analyzed by Student’s t-test. (D) Correlation of the expression of Gαh mRNA levels and MTORC1 gene in the groups is shown in A. (E and F) Results from the Kaplan-Meier analyses of the transcriptional levels of the mTORC1 gene set alone (E) or combined with the mRNA levels of Gαh (F) against ER(-) breast cancer patients from the GSE5327 data set.
The coupling of Gαh/PLC-δ1 with Akt/mTORC1 promotes autophagosome degradation to promote the metastatic potential of TNBC cells
Since the Akt/mTORC1 signaling axis has been shown to promote autophagosome degradation [20], we examined whether autophagosome assembly is involved in Gαh/PLC-δ1-modulated metastatic progression in TNBC. Results from the Western blot analysis revealed that the endogenous levels of LC3-II, which is a phosphatidylethanolamine-conjugated LC3-I and thought to be involved in autophagosome membrane expansion and fusion events, in the poorly invasive HCC1806 cells were higher than they were in the highly invasive MDA-MB231 cells (Figure 3A). Similar view was also found in the endogenous levels of p62, one of autophagy-specific substrate (Figure 3A). Moreover, the mRNA levels between the autophagy-related gene set that was generated to estimate the autophagy activity and Gαh were negatively correlated in a panel of TNBC cell lines (Figure 3B). Whereas the highly invasive MDA-MB231 cells had a signature of a high level of Gαh expression and a low level of autophagy-related gene set expression, the poorly invasive HCC1806 cells had a signature of low-level Gαh expression but a high level of autophagy-related gene set expression (Figure 3B). The overexpression of the exogenous Gαh gene in the poorly invasive HCC1806 cells reduced the intracellular LC3-II and p62 protein levels (Figure 3C). In contrast, knocking down Gαh increased the intracellular LC3-II and p62 protein levels (Figure 3D). The addition of a PPI inhibitor against the Gαh/PLC-δ1 complex promoted the formation of LC3-II and increased the p62 protein levels in the highly invasive MDA-MB231 cells (Figure 3E).
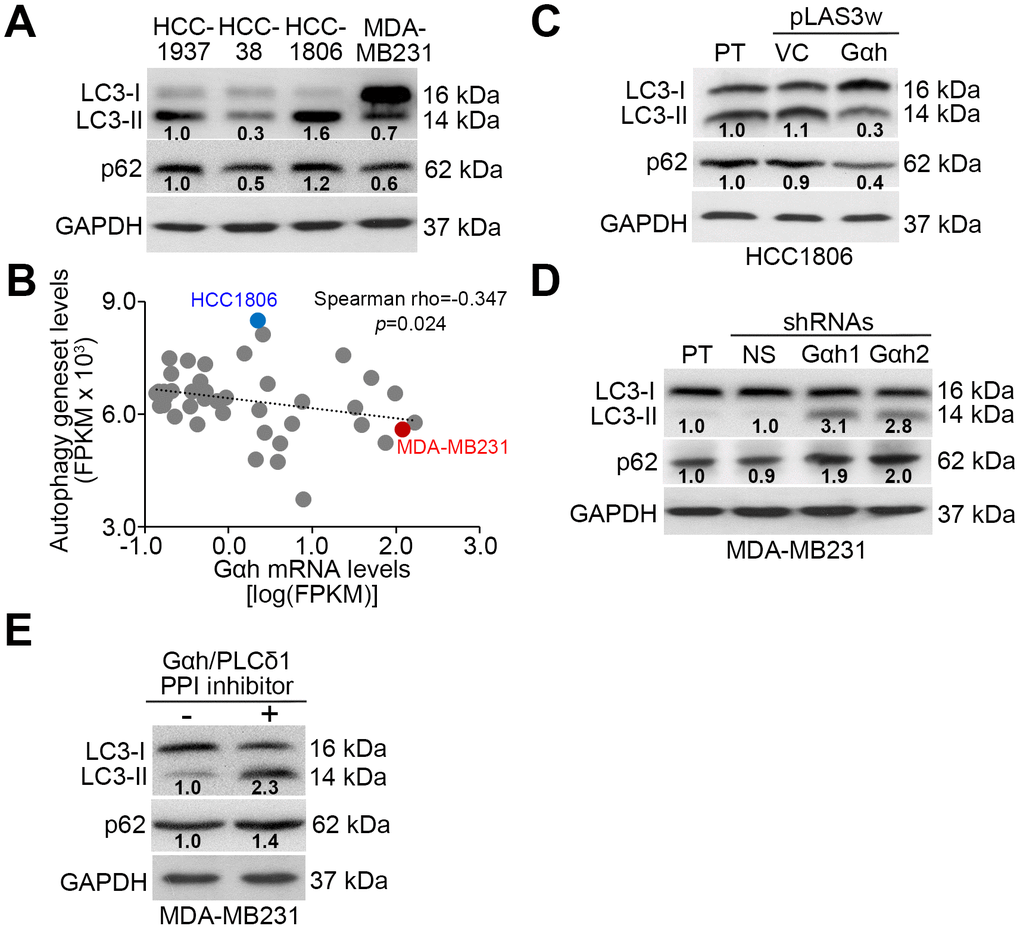
Figure 3. The Gαh-PLC-δ1 axis promotes autophagosome degradation in TNBC cells. (A) Results from the Western blot analysis for LC3-I/II, p62 and GAPDH proteins derived from the indicated TNBC cell lines. (B) Correlation of mRNA expression levels of Gαh and the autophagy-related gene set in a panel of breast cancer cell lines derived from the Cancer Cell Line Encyclopedia (CCLE) database. Spearman’s correlation test was used to estimate the statistical significance. (C–E) Results from the Western blot analysis of the LC3-I/II, p62 and GAPDH proteins derived from the parental (PT) HCC1806 cells without (vector control, VC) or with Gαh overexpression (C) and the parental MDA-MD231 cells without (nonsilenced, NS) or with Gαh knocked down using two independent shRNA clones (D), and MDA-MD231 cells treated without or with 10 μM Gαh/PLC-δ1 protein-protein interaction (PPI) inhibitor for 2 hours (E). In A, C, D, E, GAPDH was used as an internal control of protein loading. The protein intensities of representative blots from three independent experiments were normalized by GAPDH levels and presented as a ratio to the control group.
Autophagosome degradation positively regulates the Gαh-enhanced metastatic potential in TNBC cells
To realize the critical role of autophagosome degradation in Gαh-promoted metastatic progression in TNBC cells, the autophagy inhibitor 3-methyladenine (3-MA) and the mTORC1 inhibitor rapamycin (RAPA) were used to suppress autophagy initiation in Gαh-silenced MDA-MB231 cells and Gαh-overexpressing HCC1806 cells. Treatment with 3-MA suppressed the LC3-II and p62 protein levels enhanced by Gαh knockdown (Figure 4A) and ultimately rescued the invasive ability (Figure 4B and 4C) of the MDA-MB231 cells. Accordingly, the administration of 3-MA into tumor-bearing mice restored the lung colony forming ability in a dose-dependent manner that had been prominently suppressed after Gαh was knocked down in MDA-MB231 cells (Figure 4D and 4E). Conversely, the introduction of RAPA forced LC3-II and p62 protein levels (Figure 5A) and eventually diminished the cellular invasive ability (Figure 5B and 5C) of Gαh-overexpressing HCC1806 cells. Similarly, treatment with RAPA significantly (p<0.01) suppressed the colony formation of Gαh-overexpressing HCC1806 cells in the lungs of tumor-bearing mice (Figure 5D and 5E).
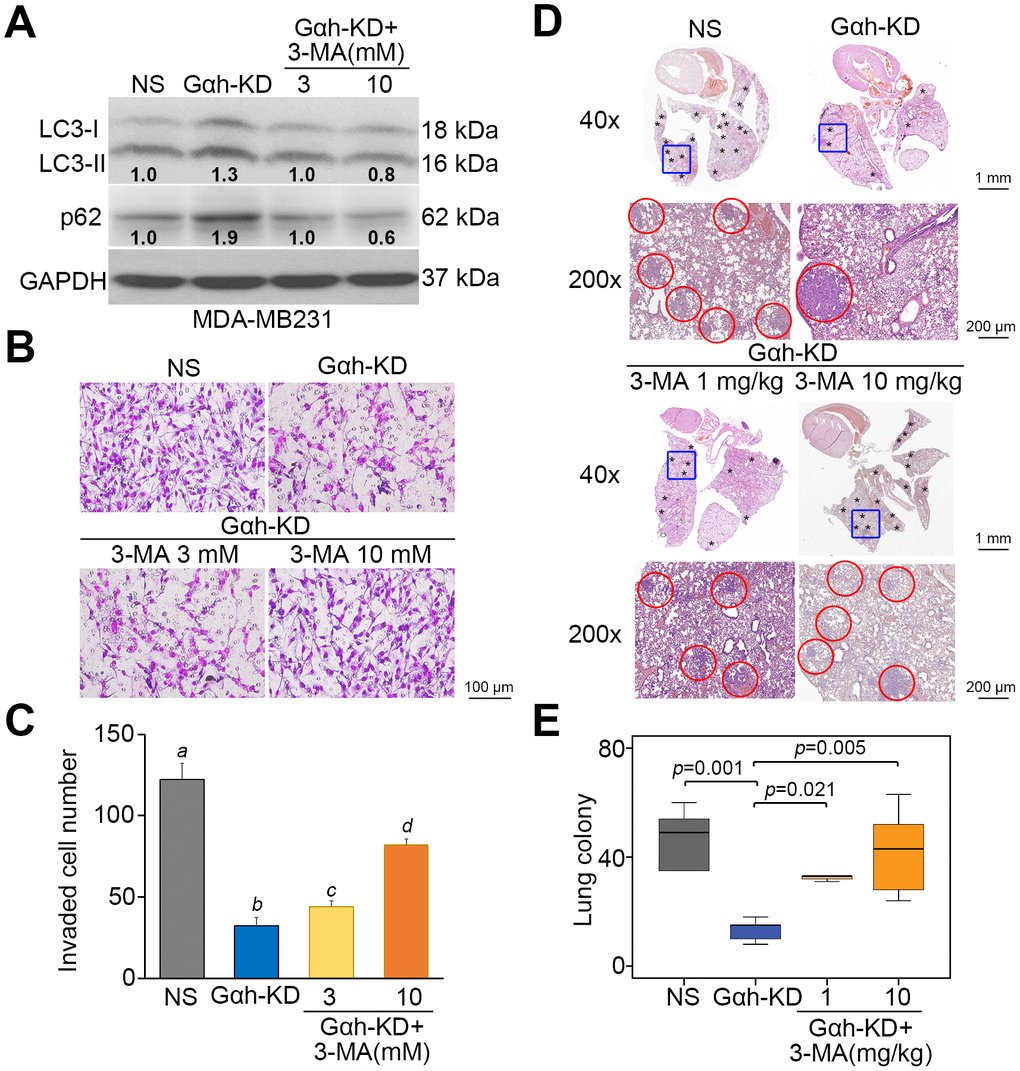
Figure 4. The inhibition of autophagy initiation by 3-MA rescues the metastatic potential of the Gαh-silenced MDA-MB231 cells in vitro and in vivo. (A) The results from the Western blot analysis for the LC3-I/II, p62 and GAPDH proteins derived from MDA-MD231 cells without (NS) or with Gαh knocked down (KD) in the absence or presence of the autophagy inhibitor 3-MA (3 or 10 mM). GAPDH was used as an internal control of protein loading. The protein intensities of representative blots from three independent experiments were normalized by GAPDH levels and presented as a ratio to the control group. (B–C) Giemsa staining (B) and cell number (C) of the invaded MDA-MD231 cell variants shown in A. Data obtained from three independent experiments are presented as the mean ± SEM. Letters indicate the significant differences at p<0.01 analyzed by nonparametric Friedman test. (D and E) H&E stained lung tissues (D) and the number of lung tumor colonies tumors (E) derived from the mice (n=5) transplanted with MDA-MD231 cell variants, shown in A, through tail vein injection for 4 weeks. Tumors are shown in red circles. Statistical significance was determined by nonparametric Mann-Whitney U test.
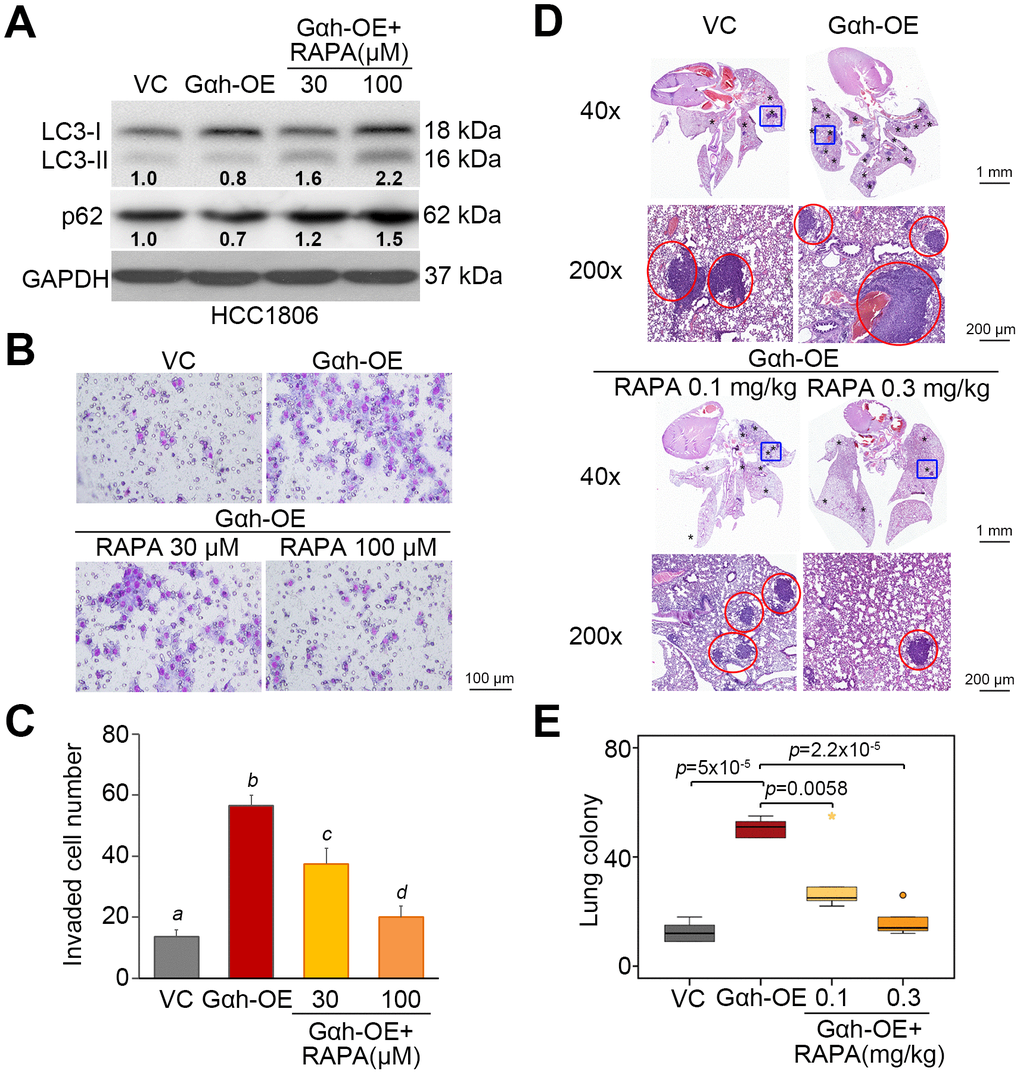
Figure 5. The inhibition of mTORC1 activity by rapamycin restores autophagy function but compromises the cellular invasion and lung metastatic abilities of Gαh-overexpressing HCC1806 cells. (A) Results from the Western blot analysis of the LC3-I/II, p62 and GAPDH proteins derived from HCC1806 cells without (VC) or overexpression Gαh (OE) in the absence or presence of the mTOR inhibitor rapamycin (RAPA) (30 or 100 μM). GAPDH was used as an internal control of protein loading. The protein intensities of representative blots from three independent experiments were normalized by GAPDH levels and presented as a ratio to the control group. (B–C) Giemsa staining (B) and cell number (C) for the invaded HCC1806 cell variants shown in A. Data obtained from three independent experiments are presented as the mean ± SEM. Letters indicate the significant differences at p<0.01 analyzed by nonparametric Friedman test. (D and E) H&E staining of lung tissues (D) and the number of lung tumor colonies (E) derived from mice (n=5) transplanted with the HCC1806 cell variants, shown in A, through tail vein injection for 4 weeks. Tumors are shown in red circles. Statistical significance was analyzed by nonparametric Mann-Whitney U test.
Discussion
Recent reports have demonstrated that the activation of the Akt/mTOR pathway induced by the long noncoding RNAs OECC [23] and MetaLnc9 [24] and the transmembrane 7 superfamily member 4 [25] promotes metastatic progression; conversely, the suppression of the Akt/mTOR pathway in the presence of the ferulic acid derivative FXS-3 [26], cardamonin [27] and microRNA-520a-3p [28] inhibits the metastatic potential of lung cancer cells. Accordingly, the association of the Akt/mTOR pathway with metastatic progression has been reported in other cancer types, including colorectal cancer [29–31], hepatocellular carcinoma [32–34], endometrial cancer [35, 36], ovarian cancer [37], gastric cancer [38–40], melanoma [41], glioma [42, 43], pancreatic ductal adenocarcinoma [44], nasopharyngeal carcinoma [45, 46], osteosarcoma [47–50], renal cell carcinoma [51–53] and prostate cancer [54–56]. In breast cancer, synaptopodin-2 [57] and caveolin-1 [58] have been shown to modulate Akt/mTOR-regulated metastatic progression. Particularly in TNBC, the inclusion of the Rhizoma Amorphophalli appeared to inhibit cell migration, invasion and metastasis by suppressing the Akt/mTOR pathway [59]. Here, we also found that the Akt/mTOR pathway mediates Gαh-promoted TNBC metastatic progression. However, further experiments are still needed to explore a comprehensive scenario in which the Gαh-PLC-δ1 interaction directly activates the Akt/mTOR pathway in TNBC cells.
The role of autophagy during cancer metastasis is still controversial. A recent review article indicated that autophagy is upregulated during cancer metastasis [60]. In contrast, several lines of evidence have illustrated that autophagy is suppressed during the metastatic progression of some cancer types [61–64]. In TNBC cells, e.g., MDA-MB-231 cells, the induction of autophagy by the selenopurine molecule SLLN-15 has been shown to suppress the metastatic potential in vitro and in vivo by inhibiting Akt-mTORC1 signaling [65]. Accordingly, treatment with parthenolide, a sesquiterpene lactone found in Tanacetum parthenium, appeared to generate autophagy and ultimately suppress the lung metastasis of MDA-MB231 cells in an orthotopic mouse model of breast cancer [66]. Conversely, in an orthotopic mouse model of breast cancer, the suppression of autophagy via hypoxia-induced expression of the kinase-dead unc-51-like autophagy-activating kinase (ULK1) mutant K46N was found to increase the lung metastasis capacity of MDA-MB-231 cells [67]. Here, we also found that the induction of autophagy by rapamycin treatment suppresses the metastatic potential of Gαh-overexpressing HCC1806 cells, whereas the inhibition of autophagy by 3-MA treatment restores the metastatic capacity of Gαh-silenced MDA-MB-231 cells. These findings may elucidate a negative role of autophagy in regulating TNBC metastasis.
Because the Akt/mTOR pathway is one of the important pathways involved in TNBC progression, several Akt/mTOR inhibitors used as monotherapy or in combination therapy for TNBC patients are currently in phase I/II clinical trials [68]. Moreover, the therapeutic targeting of autophagy activity has also been thought to be another promising anticancer strategy. Therefore, our results provide a new strategy to combat the metastatic progression of TNBC due to Gαh upregulation via inhibiting Akt/mTOR activity or preventing autophagosome degradation.
Materials and Methods
Cell lines and cell culture conditions
MDA-MB231 cells were cultured in Leibovitz’s (L-15) medium (Invitrogen) supplemented with 10% fetal bovine serum (FBS, Invitrogen). HCC1937, HCC1806, and HCC38 cells were cultured in RPMI 1640 medium (Invitrogen) with 10% FBS. 293T cells were cultured in DMEM medium with 10% FBS. All cell lines were obtained from American Type Culture Collection (ATCC). All cells were incubated at 37°C with 5% CO2 and routinely authenticated on the basis of short tandem repeat (STR) analysis, morphologic and growth characteristics of the cells and mycoplasma detection.
Microarray data processing
Microarray data and related clinical data from the Gene Expression Omnibus (GEO) GSE5327 data set were downloaded from the NCBI website. Affymetrix DAT files were processed using the Affymetrix Gene Chip Operating System (GCOS) to generate .CEL files. Raw intensities in the .CEL files were normalized by robust multichip analysis (RMA), and fold-change analysis was performed using GeneSpring GX11 (Agilent Technologies). Relative mRNA expression levels were normalized by their median and presented as log2 values. The gene set of autophagy was obtained from Molecular Signatures Database (https://www.gsea-msigdb.org/gsea/msigdb). The sum derived from the expression levels of the gene set was used to represent the autophagy activity.
Immunoprecipitation and Western blot assay
Whole cell lysates (1 mg) were pre-incubated with non-immunized serum and protein A/G-conjugated agarose (Santa Cruz) for 1 hour at 4°C with a gentle rotation. After the centrifugation, the supernatants were further incubated with Gαh or PLC-δ1 (Gentex) antibodies and protein A/G-conjugated agarose overnight at 4°C with a gentle rotation. After several washes, the immunoprecipitates were resuspended in 20 μl of SDS-PAGE protein loading dye and boiled at 95°C. After the centrifugation, the supernatants were subjected to Western blot analysis.
Total protein (100 μg) from the designed experiments was separated by SDS-PAGE and then transferred to PVDF membranes. The membranes were sequentially incubated with blocking buffer (5% nonfat milk in TBS containing 0.1% Tween-20) for 2 hours at room temperature, primary antibodies against Gαh, PLC-δ1 (Gentex), phosphorylated Akt, Akt, phosphorylated mTOR, mTOR, LC3-I/II and p62 (Cell Signaling) or GAPDH (AbFrontier) overnight at 4 °C, and peroxidase-labeled secondary antibodies for 1 hour at room temperature. At each step, the cells were extensively washed. Finally, immunoreactive bands were visualized by an enhanced chemiluminescence system (Amersham Bioscience).
Immunofluorescent staining
MDA-MB231 cells (1 x 105/ml) cultivated in the absence or presence of Gαh/PLC-δ1 PPI inhibitor and grown on cover slides (22 mm in diameter and 0.17 mm in thickness) were fixed in 4% formaldehyde for 15 min at RT. After washing cells two times with PBS, the cells were treated with 95% EtOH/5% CH3COOH at-20°C for 15 min. Before blocking with 2% BSA/0.1% Triton X-100 for 2 hours at room temperature (RT), the cells were washed two times with PBS. Subsequently, the cells were incubated with p-Akt or p-mTOR antibody overnight at 4°C. After washing the cells three times with PBS, the cells were incubated with biotin-conjugated secondary antibody (DAKO) for 1 hour at RT. The cells were washed three times with PBS and incubated with fluorescein-conjugated avidin complex (Vector Laboratories) for 30 min at RT. After mounting the cells were analyzed using a FluoView confocal microscope system (Olympus).
Invasion assay
Cell invasion ability was measured by Boyden Chambers (Neuro Probe) according to the manufacturer’s protocol. Briefly, a polycarbonate membrane (8 μm pore size, 25 × 80 mm, Neuro Probe) was precoated with 10 μg human fibronectin (Sigma) on the lower side and Matrigel (BD Biosciences) on the upper side. The cells (1.5 x 104) obtained from the designed experiments were plated in the top chamber with 50 μl serum-free medium. After 16 hours, stationary cells from the top side of the membrane were removed, whereas the invaded cells in the bottom side of the membrane were fixed in 100% methanol and stained with 10% Giemsa solution (Merck) for 1 hour. The number of invaded cells was counted under a light microscope (400×, ten random fields from each well). All experiments were performed in triplicate.
Animal experiments
NOD/SCID mice were obtained from the National Laboratory Animal Center in Taiwan and maintained in compliance with institutional policy. All animal procedures were approved by the Institutional Animal Care and Use Committee at Taipei Medical University. For the in vivo lung metastatic colonization assay, 1x106 cells in 100 μl PBS were implanted into the mice through tail vein injection. The mice were sacrificed, and the lungs were obtained for histological analysis. Metastatic lung nodules were quantified after staining with H&E using a dissecting microscope.
Statistical analyses
SPSS 17.0 software (Informer Technologies, Roseau, Dominica) was used to analyze statistical significance. Nonparametric Mann-Whitney U tests were utilized to compare mTORC1 and the autophagy-related gene set expression in breast cancer patients. Spearman’s test was performed to estimate the association among Gαh, mTORC1 and autophagy-related gene set expression levels in breast cancer tissues and in the panel of the TNBC cell lines. Survival probabilities were determined by Kaplan-Meier analysis and log-rank tests. Nonparametric Mann-Whitney U and Friedman tests were used to analyze data from 2 independent samples and 3 or more related samples, respectively. P values <0.05 in all analyses were considered to be statistically significant.
Acknowledgments
The authors would like to thank Dr. Michael Hsiao from Academia Sinica in Taiwan for kindly providing TNBC cell lines.
Conflicts of Interest
The authors have declared no potential conflicts of interest.
Funding
This study was supported by the Ministry of Science and Technology, Taiwan to Yuan-Feng Lin (MOST 108-2320-B-038-017-MY3) and Hui-Yu Lin (MOST 108-2314-B-567-002), and Cardinal Tien Hospital, Xindian District, New Taipei City, Taiwan (CTH108A-2A02) to Hui-Yu Lin.
References
- 1. Lehmann BD, Bauer JA, Chen X, Sanders ME, Chakravarthy AB, Shyr Y, Pietenpol JA. Identification of human triple-negative breast cancer subtypes and preclinical models for selection of targeted therapies. J Clin Invest. 2011; 121:2750–67. https://doi.org/10.1172/JCI45014 [PubMed]
- 2. Masuda H, Baggerly KA, Wang Y, Zhang Y, Gonzalez-Angulo AM, Meric-Bernstam F, Valero V, Lehmann BD, Pietenpol JA, Hortobagyi GN, Symmans WF, Ueno NT. Differential response to neoadjuvant chemotherapy among 7 triple-negative breast cancer molecular subtypes. Clin Cancer Res. 2013; 19:5533–40. https://doi.org/10.1158/1078-0432.CCR-13-0799 [PubMed]
- 3. Dent R, Hanna WM, Trudeau M, Rawlinson E, Sun P, Narod SA. Pattern of metastatic spread in triple-negative breast cancer. Breast Cancer Res Treat. 2009; 115:423–28. https://doi.org/10.1007/s10549-008-0086-2 [PubMed]
- 4. Garrido-Castro AC, Lin NU, Polyak K. Insights into molecular classifications of triple-negative breast cancer: improving patient selection for treatment. Cancer Discov. 2019; 9:176–98. https://doi.org/10.1158/2159-8290.CD-18-1177 [PubMed]
- 5. Nakaoka H, Perez DM, Baek KJ, Das T, Husain A, Misono K, Im MJ, Graham RM. Gh: a GTP-binding protein with transglutaminase activity and receptor signaling function. Science. 1994; 264:1593–96. https://doi.org/10.1126/science.7911253 [PubMed]
- 6. Bagatur Y, Ilter Akulke AZ, Bihorac A, Erdem M, Telci D. Tissue transglutaminase expression is necessary for adhesion, metastatic potential and cancer stemness of renal cell carcinoma. Cell Adh Migr. 2018; 12:138–51. https://doi.org/10.1080/19336918.2017.1322255 [PubMed]
- 7. Yamaguchi H, Kuroda K, Sugitani M, Takayama T, Hasegawa K, Esumi M. Transglutaminase 2 is upregulated in primary hepatocellular carcinoma with early recurrence as determined by proteomic profiles. Int J Oncol. 2017; 50:1749–59. https://doi.org/10.3892/ijo.2017.3917 [PubMed]
- 8. Fisher ML, Keillor JW, Xu W, Eckert RL, Kerr C. Transglutaminase is required for epidermal squamous cell carcinoma stem cell survival. Mol Cancer Res. 2015; 13:1083–94. https://doi.org/10.1158/1541-7786.MCR-14-0685-T [PubMed]
- 9. Caffarel MM, Chattopadhyay A, Araujo AM, Bauer J, Scarpini CG, Coleman N. Tissue transglutaminase mediates the pro-malignant effects of oncostatin M receptor over-expression in cervical squamous cell carcinoma. J Pathol. 2013; 231:168–79. https://doi.org/10.1002/path.4222 [PubMed]
- 10. Delaine-Smith R, Wright N, Hanley C, Hanwell R, Bhome R, Bullock M, Drifka C, Eliceiri K, Thomas G, Knight M, Mirnezami A, Peake N. Transglutaminase-2 mediates the biomechanical properties of the colorectal cancer tissue microenvironment that contribute to disease progression. Cancers (Basel). 2019; 11:701. https://doi.org/10.3390/cancers11050701 [PubMed]
- 11. Lee HT, Huang CH, Chen WC, Tsai CS, Chao YL, Liu SH, Chen JH, Wu YY, Lee YJ. Transglutaminase 2 promotes migration and invasion of lung cancer cells. Oncol Res. 2018; 26:1175–82. https://doi.org/10.3727/096504018X15149761920868 [PubMed]
- 12. Kumar A, Xu J, Sung B, Kumar S, Yu D, Aggarwal BB, Mehta K. Evidence that GTP-binding domain but not catalytic domain of transglutaminase 2 is essential for epithelial-to-mesenchymal transition in mammary epithelial cells. Breast Cancer Res. 2012; 14:R4. https://doi.org/10.1186/bcr3085 [PubMed]
- 13. Seo S, Moon Y, Choi J, Yoon S, Jung KH, Cheon J, Kim W, Kim D, Lee CH, Kim SW, Park KS, Lee DH. The GTP binding activity of transglutaminase 2 promotes bone metastasis of breast cancer cells by downregulating microRNA-205. Am J Cancer Res. 2019; 9:597–607. [PubMed]
- 14. Oh K, Moon HG, Lee DS, Yoo YB. Tissue transglutaminase-interleukin-6 axis facilitates peritoneal tumor spreading and metastasis of human ovarian cancer cells. Lab Anim Res. 2015; 31:188–97. https://doi.org/10.5625/lar.2015.31.4.188 [PubMed]
- 15. Lin CY, Tsai PH, Kandaswami CC, Chang GD, Cheng CH, Huang CJ, Lee PP, Hwang JJ, Lee MT. Role of tissue transglutaminase 2 in the acquisition of a mesenchymal-like phenotype in highly invasive A431 tumor cells. Mol Cancer. 2011; 10:87. https://doi.org/10.1186/1476-4598-10-87 [PubMed]
- 16. Huang SP, Liu PY, Kuo CJ, Chen CL, Lee WJ, Tsai YH, Lin YF. The gαh-PLCδ1 signaling axis drives metastatic progression in triple-negative breast cancer. J Hematol Oncol. 2017; 10:114. https://doi.org/10.1186/s13045-017-0481-4 [PubMed]
- 17. Kang JH, Lee SH, Cheong H, Lee CH, Kim SY. Transglutaminase 2 promotes autophagy by LC3 induction through p53 depletion in cancer cell. Biomol Ther (Seoul). 2019; 27:34–40. https://doi.org/10.4062/biomolther.2018.140 [PubMed]
- 18. Kang JH, Lee JS, Hong D, Lee SH, Kim N, Lee WK, Sung TW, Gong YD, Kim SY. Renal cell carcinoma escapes death by p53 depletion through transglutaminase 2-chaperoned autophagy. Cell Death Dis. 2016; 7:e2163. https://doi.org/10.1038/cddis.2016.14 [PubMed]
- 19. Verhaar R, Drukarch B, Bol JG, Jongenelen CA, Wilhelmus MM. Tissue transglutaminase cross-links beclin 1 and regulates autophagy in MPP+-treated human SH-SY5Y cells. Neurochem Int. 2013; 62:486–91. https://doi.org/10.1016/j.neuint.2013.01.024 [PubMed]
- 20. Akar U, Ozpolat B, Mehta K, Fok J, Kondo Y, Lopez-Berestein G. Tissue transglutaminase inhibits autophagy in pancreatic cancer cells. Mol Cancer Res. 2007; 5:241–49. https://doi.org/10.1158/1541-7786.MCR-06-0229 [PubMed]
- 21. Ding Y, Zhang J, Wang R. Inhibition of tissue transglutaminase attenuates lipopolysaccharide-induced inflammation in glial cells through AKT/mTOR signal pathway. Biomed Pharmacother. 2017; 89:1310–19. https://doi.org/10.1016/j.biopha.2017.03.027 [PubMed]
- 22. Cao J, Huang W. Compensatory increase of transglutaminase 2 is responsible for resistance to mTOR inhibitor treatment. PLoS One. 2016; 11:e0149388. https://doi.org/10.1371/journal.pone.0149388 [PubMed]
- 23. Zou Y, Zhang B, Mao Y, Zhang H, Hong W. Long non-coding RNA OECC promotes cell proliferation and metastasis through the PI3K/Akt/mTOR signaling pathway in human lung cancer. Oncol Lett. 2019; 18:3017–24. https://doi.org/10.3892/ol.2019.10644 [PubMed]
- 24. Yu T, Zhao Y, Hu Z, Li J, Chu D, Zhang J, Li Z, Chen B, Zhang X, Pan H, Li S, Lin H, Liu L, et al. MetaLnc9 facilitates lung cancer metastasis via a PGK1-activated AKT/mTOR pathway. Cancer Res. 2017; 77:5782–94. https://doi.org/10.1158/0008-5472.CAN-17-0671 [PubMed]
- 25. Yang X, Song X, Wang X, Liu X, Peng Z. Downregulation of TM7SF4 inhibits cell proliferation and metastasis of A549 cells through regulating the PI3K/AKT/mTOR signaling pathway. Mol Med Rep. 2017; 16:6122–27. https://doi.org/10.3892/mmr.2017.7324 [PubMed]
- 26. Yue SJ, Zhang PX, Zhu Y, Li NG, Chen YY, Li JJ, Zhang S, Jin RY, Yan H, Shi XQ, Tang YP, Duan JA. A ferulic acid derivative FXS-3 inhibits proliferation and metastasis of human lung cancer A549 cells via positive JNK signaling pathway and negative ERK/p38, AKT/mTOR and MEK/ERK signaling pathways. Molecules. 2019; 24:2165. https://doi.org/10.3390/molecules24112165 [PubMed]
- 27. Zhou X, Zhou R, Li Q, Jie X, Hong J, Zong Y, Dong X, Zhang S, Li Z, Wu G. Cardamonin inhibits the proliferation and metastasis of non-small-cell lung cancer cells by suppressing the PI3K/Akt/mTOR pathway. Anticancer Drugs. 2019; 30:241–50. https://doi.org/10.1097/CAD.0000000000000709 [PubMed]
- 28. Lv X, Li CY, Han P, Xu XY. MicroRNA-520a-3p inhibits cell growth and metastasis of non-small cell lung cancer through PI3K/AKT/mTOR signaling pathway. Eur Rev Med Pharmacol Sci. 2018; 22:2321–27. https://doi.org/10.26355/eurrev_201804_14822 [PubMed]
- 29. Meng S, Jian Z, Yan X, Li J, Zhang R. LncRNA SNHG6 inhibits cell proliferation and metastasis by targeting ETS1 via the PI3K/AKT/mTOR pathway in colorectal cancer. Mol Med Rep. 2019; 20:2541–48. https://doi.org/10.3892/mmr.2019.10510 [PubMed]
- 30. Mao HY, Liu SP, Kong GM, Xu Y, Xu YC, Sun GZ, Wang Z, Han F, Tong JD, Bo P. FBLN3 inhibited the invasion and metastasis of colorectal cancer through the AKT/mTOR pathway. Neoplasma. 2019; 66:336–42. https://doi.org/10.4149/neo_2018_180703N441 [PubMed]
- 31. Yang J, Li TZ, Xu GH, Luo BB, Chen YX, Zhang T. Low-concentration capsaicin promotes colorectal cancer metastasis by triggering ROS production and modulating Akt/mTOR and STAT-3 pathways. Neoplasma. 2013; 60:364–72. https://doi.org/10.4149/neo_2013_048 [PubMed]
- 32. Zhang Y, Zhang C, Zhao Q, Wei W, Dong Z, Shao L, Li J, Wu W, Zhang H, Huang H, Liu F, Jin S. The miR-873/NDFIP1 axis promotes hepatocellular carcinoma growth and metastasis through the AKT/mTOR-mediated warburg effect. Am J Cancer Res. 2019; 9:927–44. [PubMed]
- 33. Hua H, Zhu Y, Song YH. Ruscogenin suppressed the hepatocellular carcinoma metastasis via PI3K/Akt/mTOR signaling pathway. Biomed Pharmacother. 2018; 101:115–22. https://doi.org/10.1016/j.biopha.2018.02.031 [PubMed]
- 34. Wang H, Zhang C, Xu L, Zang K, Ning Z, Jiang F, Chi H, Zhu X, Meng Z. Bufalin suppresses hepatocellular carcinoma invasion and metastasis by targeting HIF-1α via the PI3K/AKT/mTOR pathway. Oncotarget. 2016; 7:20193–208. https://doi.org/10.18632/oncotarget.7935 [PubMed]
- 35. Lin Q, Chen H, Zhang M, Xiong H, Jiang Q. Knocking down FAM83B inhibits endometrial cancer cell proliferation and metastasis by silencing the PI3K/AKT/mTOR pathway. Biomed Pharmacother. 2019; 115:108939. https://doi.org/10.1016/j.biopha.2019.108939 [PubMed]
- 36. Xu X, Kong X, Liu T, Zhou L, Wu J, Fu J, Wang Y, Zhu M, Yao S, Ding Y, Ding L, Li R, Zhu X, et al. Metastasis-associated protein 1, modulated by miR-30c, promotes endometrial cancer progression through AKT/mTOR/4E-BP1 pathway. Gynecol Oncol. 2019; 154:207–17. https://doi.org/10.1016/j.ygyno.2019.04.005 [PubMed]
- 37. Qiao J, Wang WJ, Zhang Y. Aclidinium inhibits proliferation and metastasis of ovarian cancer SKOV3 cells via downregulating PI3K/AKT/mTOR signaling pathway. Oncol Lett. 2018; 16:6417–22. https://doi.org/10.3892/ol.2018.9460 [PubMed]
- 38. Qian Y, Yan Y, Lu H, Zhou T, Lv M, Fang C, Hou J, Li W, Chen X, Sun H, Li Y, Wang Z, Zhao N, et al. Celastrus orbiculatus extracts inhibit the metastasis through attenuating PI3K/Akt/mTOR signaling pathway in human gastric cancer. Anticancer Agents Med Chem. 2019; 19:1754–61. https://doi.org/10.2174/1871520619666190731162722 [PubMed]
- 39. Zhang XR, Wang SY, Sun W, Wei C. Isoliquiritigenin inhibits proliferation and metastasis of MKN28 gastric cancer cells by suppressing the PI3K/AKT/mTOR signaling pathway. Mol Med Rep. 2018; 18:3429–36. https://doi.org/10.3892/mmr.2018.9318 [PubMed]
- 40. Xing X, Zhang L, Wen X, Wang X, Cheng X, Du H, Hu Y, Li L, Dong B, Li Z, Ji J. PP242 suppresses cell proliferation, metastasis, and angiogenesis of gastric cancer through inhibition of the PI3K/AKT/mTOR pathway. Anticancer Drugs. 2014; 25:1129–40. https://doi.org/10.1097/CAD.0000000000000148 [PubMed]
- 41. Yang TY, Wu ML, Chang CI, Liu CI, Cheng TC, Wu YJ. Bornyl cis-4-hydroxycinnamate suppresses cell metastasis of melanoma through FAK/PI3K/Akt/mTOR and MAPK signaling pathways and inhibition of the epithelial-to-mesenchymal transition. Int J Mol Sci. 2018; 19:2152. https://doi.org/10.3390/ijms19082152 [PubMed]
- 42. Zhao KH, Zhang C, Bai Y, Li Y, Kang X, Chen JX, Yao K, Jiang T, Zhong XS, Li WB. Antiglioma effects of cytarabine on leptomeningeal metastasis of high-grade glioma by targeting the PI3K/Akt/mTOR pathway. Drug Des Devel Ther. 2017; 11:1905–15. https://doi.org/10.2147/DDDT.S135711 [PubMed]
- 43. Tu M, Wange W, Cai L, Zhu P, Gao Z, Zheng W. IL-13 receptor α2 stimulates human glioma cell growth and metastasis through the src/PI3K/Akt/mTOR signaling pathway. Tumour Biol. 2016; 37:14701–09. https://doi.org/10.1007/s13277-016-5346-x [PubMed]
- 44. Liu X, Tan X, Liu P, Wu Y, Qian S, Zhang X. Phosphoglycerate mutase 1 (PGAM1) promotes pancreatic ductal adenocarcinoma (PDAC) metastasis by acting as a novel downstream target of the PI3K/ Akt/mTOR pathway. Oncol Res. 2018; 26:1123–31. https://doi.org/10.3727/096504018X15166223632406 [PubMed]
- 45. Wang MH, Sun R, Zhou XM, Zhang MY, Lu JB, Yang Y, Zeng LS, Yang XZ, Shi L, Xiao RW, Wang HY, Mai SJ. Epithelial cell adhesion molecule overexpression regulates epithelial-mesenchymal transition, stemness and metastasis of nasopharyngeal carcinoma cells via the PTEN/AKT/mTOR pathway. Cell Death Dis. 2018; 9:2. https://doi.org/10.1038/s41419-017-0013-8 [PubMed]
- 46. Zhou XM, Sun R, Luo DH, Sun J, Zhang MY, Wang MH, Yang Y, Wang HY, Mai SJ. Upregulated TRIM29 promotes proliferation and metastasis of nasopharyngeal carcinoma via PTEN/AKT/mTOR signal pathway. Oncotarget. 2016; 7:13634–50. https://doi.org/10.18632/oncotarget.7215 [PubMed]
- 47. Ma H, Su R, Feng H, Guo Y, Su G. Long noncoding RNA UCA1 promotes osteosarcoma metastasis through CREB1-mediated epithelial-mesenchymal transition and activating PI3K/AKT/mTOR pathway. J Bone Oncol. 2019; 16:100228. https://doi.org/10.1016/j.jbo.2019.100228 [PubMed]
- 48. Jin R, Jin YY, Tang YL, Yang HJ, Zhou XQ, Lei Z. GPNMB silencing suppresses the proliferation and metastasis of osteosarcoma cells by blocking the PI3K/Akt/mTOR signaling pathway. Oncol Rep. 2018; 39:3034–40. https://doi.org/10.3892/or.2018.6346 [PubMed]
- 49. Cai AL, Zeng W, Cai WL, Liu JL, Zheng XW, Liu Y, Yang XC, Long Y, Li J. Peroxiredoxin-1 promotes cell proliferation and metastasis through enhancing Akt/mTOR in human osteosarcoma cells. Oncotarget. 2017; 9:8290–302. https://doi.org/10.18632/oncotarget.23662 [PubMed]
- 50. Li E, Zhao Z, Ma B, Zhang J. Long noncoding RNA HOTAIR promotes the proliferation and metastasis of osteosarcoma cells through the AKT/mTOR signaling pathway. Exp Ther Med. 2017; 14:5321–28. https://doi.org/10.3892/etm.2017.5248 [PubMed]
- 51. Xie J, Lin W, Huang L, Xu N, Xu A, Chen B, Watanabe M, Liu C, Huang P. Bufalin suppresses the proliferation and metastasis of renal cell carcinoma by inhibiting the PI3K/Akt/mTOR signaling pathway. Oncol Lett. 2018; 16:3867–73. https://doi.org/10.3892/ol.2018.9111 [PubMed]
- 52. Miao C, Liang C, Tian Y, Xu A, Zhu J, Zhao K, Zhang J, Hua Y, Liu S, Dong H, Zhang C, Su S, Li P, et al. Overexpression of CAPN2 promotes cell metastasis and proliferation via AKT/mTOR signaling in renal cell carcinoma. Oncotarget. 2017; 8:97811–21. https://doi.org/10.18632/oncotarget.22083 [PubMed]
- 53. Fang Z, Tang Y, Fang J, Zhou Z, Xing Z, Guo Z, Guo X, Wang W, Jiao W, Xu Z, Liu Z. Simvastatin inhibits renal cancer cell growth and metastasis via AKT/mTOR, ERK and JAK2/STAT3 pathway. PLoS One. 2013; 8:e62823. https://doi.org/10.1371/journal.pone.0062823 [PubMed]
- 54. Xu S, Ge J, Zhang Z, Zhou W. MiR-129 inhibits cell proliferation and metastasis by targeting ETS1 via PI3K/AKT/mTOR pathway in prostate cancer. Biomed Pharmacother. 2017; 96:634–41. https://doi.org/10.1016/j.biopha.2017.10.037 [PubMed]
- 55. Chen Y, Zheng L, Liu J, Zhou Z, Cao X, Lv X, Chen F. Shikonin inhibits prostate cancer cells metastasis by reducing matrix metalloproteinase-2/-9 expression via AKT/mTOR and ROS/ERK1/2 pathways. Int Immunopharmacol. 2014; 21:447–55. https://doi.org/10.1016/j.intimp.2014.05.026 [PubMed]
- 56. Ni J, Cozzi P, Hao J, Beretov J, Chang L, Duan W, Shigdar S, Delprado W, Graham P, Bucci J, Kearsley J, Li Y. Epithelial cell adhesion molecule (EpCAM) is associated with prostate cancer metastasis and chemo/ radioresistance via the PI3K/Akt/mTOR signaling pathway. Int J Biochem Cell Biol. 2013; 45:2736–48. https://doi.org/10.1016/j.biocel.2013.09.008 [PubMed]
- 57. Xia E, Zhou X, Bhandari A, Zhang X, Wang O. Synaptopodin-2 plays an important role in the metastasis of breast cancer via PI3K/Akt/mTOR pathway. Cancer Manag Res. 2018; 10:1575–83. https://doi.org/10.2147/CMAR.S162670 [PubMed]
- 58. Yang H, Guan L, Li S, Jiang Y, Xiong N, Li L, Wu C, Zeng H, Liu Y. Mechanosensitive caveolin-1 activation-induced PI3K/Akt/mTOR signaling pathway promotes breast cancer motility, invadopodia formation and metastasis in vivo. Oncotarget. 2016; 7:16227–47. https://doi.org/10.18632/oncotarget.7583 [PubMed]
- 59. Wu C, Qiu S, Liu P, Ge Y, Gao X. Rhizoma amorphophalli inhibits TNBC cell proliferation, migration, invasion and metastasis through the PI3K/Akt/mTOR pathway. J Ethnopharmacol. 2018; 211:89–100. https://doi.org/10.1016/j.jep.2017.09.033 [PubMed]
- 60. Mowers EE, Sharifi MN, Macleod KF. Autophagy in cancer metastasis. Oncogene. 2017; 36:1619–30. https://doi.org/10.1038/onc.2016.333 [PubMed]
- 61. Zhang M, Liu S, Chua MS, Li H, Luo D, Wang S, Zhang S, Han B, Sun C. SOCS5 inhibition induces autophagy to impair metastasis in hepatocellular carcinoma cells via the PI3K/Akt/mTOR pathway. Cell Death Dis. 2019; 10:612. https://doi.org/10.1038/s41419-019-1856-y [PubMed]
- 62. Zhu JF, Huang W, Yi HM, Xiao T, Li JY, Feng J, Yi H, Lu SS, Li XH, Lu RH, He QY, Xiao ZQ. Annexin A1-suppressed autophagy promotes nasopharyngeal carcinoma cell invasion and metastasis by PI3K/AKT signaling activation. Cell Death Dis. 2018; 9:1154. https://doi.org/10.1038/s41419-018-1204-7 [PubMed]
- 63. Zhao GS, Gao ZR, Zhang Q, Tang XF, Lv YF, Zhang ZS, Zhang Y, Tan QL, Peng DB, Jiang DM, Guo QN. TSSC3 promotes autophagy via inactivating the src-mediated PI3K/Akt/mTOR pathway to suppress tumorigenesis and metastasis in osteosarcoma, and predicts a favorable prognosis. J Exp Clin Cancer Res. 2018; 37:188. https://doi.org/10.1186/s13046-018-0856-6 [PubMed]
- 64. Chai D, Shan H, Wang G, Li H, Fang L, Song J, Zhang Q, Bai J, Zheng J. AIM2 is a potential therapeutic target in human renal carcinoma and suppresses its invasion and metastasis via enhancing autophagy induction. Exp Cell Res. 2018; 370:561–70. https://doi.org/10.1016/j.yexcr.2018.07.021 [PubMed]
- 65. Chang CH, Bijian K, Wernic D, Su J, da Silva SD, Yu H, Qiu D, Asslan M, Alaoui-Jamali MA. A novel orally available seleno-purine molecule suppresses triple-negative breast cancer cell proliferation and progression to metastasis by inducing cytostatic autophagy. Autophagy. 2019; 15:1376–90. https://doi.org/10.1080/15548627.2019.1582951 [PubMed]
- 66. D'Anneo A, Carlisi D, Lauricella M, Puleio R, Martinez R, Di Bella S, Di Marco P, Emanuele S, Di Fiore R, Guercio A, Vento R, Tesoriere G. Parthenolide generates reactive oxygen species and autophagy in MDA-MB231 cells. A soluble parthenolide analogue inhibits tumour growth and metastasis in a xenograft model of breast cancer. Cell Death Dis. 2013; 4:e891. https://doi.org/10.1038/cddis.2013.415 [PubMed]
- 67. Dower CM, Bhat N, Wang EW, Wang HG. Selective reversible inhibition of autophagy in hypoxic breast cancer cells promotes pulmonary metastasis. Cancer Res. 2017; 77:646–57. https://doi.org/10.1158/0008-5472.CAN-15-3458 [PubMed]
- 68. Khan MA, Jain VK, Rizwanullah M, Ahmad J, Jain K. PI3K/AKT/mTOR pathway inhibitors in triple-negative breast cancer: a review on drug discovery and future challenges. Drug Discov Today. 2019; 24:2181–91. https://doi.org/10.1016/j.drudis.2019.09.001 [PubMed]