Introduction
Lung cancer is one of the leading causes of cancer-related deaths worldwide; non-small cell lung cancer or NSCLC accounts for over 85% of lung cancer cases [1, 2]. The long term outcomes for metastatic NSCLC patients are poor despite significant therapeutic advances including the availability of small molecule inhibitors against epidermal growth factor receptor tyrosine kinases (EGFR-TK), anaplastic lymphoma kinase (ALK) and c-ros oncogene 1 receptor tyrosine kinase (ROS1) because majority of patients report drug resistance [3, 4]. The emergence of immune checkpoint inhibitors (ICI), which includes targeted antibodies against programmed cell death protein 1 (PD-1), programmed cell death-ligand 1 (PD-L1) and cytotoxic T-lymphocyte-associated protein 4 (CTLA4) has increased the survival of metastatic NSCLC patients, but, only a small number of metastatic patients achieve long-term survival [5].
PD-L1 expression and tumor mutation burden (TMB) have been used as clinical biomarkers to predict the response to PD-1/PD-L1 antibody therapy [5, 6], but, the prediction efficacy is sub-optimal when the PD-L1 expression is less than 50% in EGFR+/ALK+ NSCLC patients and non-squamous lung cancer patients [7]. Moreover, there is no consensus regarding the use of TMB as a selection criteria for PD-1/PD-L1 antibody therapy because it would require whole exome sequencing or large scale sequencing of several target genes in the tumor samples of NSCLC patients [6, 8]. A recent study showed that mutations in POLE and POLD1 are potential biomarkers that can effectively predict treatment outcomes of immunotherapies in several cancers, including lung cancer [9].
Several studies suggest that TP53 gene mutations are potential prognostic biomarkers for cancer patients that undergo immunotherapies, such as head and neck squamous cell cancer, lung adenocarcinoma [10, 11]. However, the association between TP53 mutations and the efficacy of immunotherapy remains ambiguous. Therefore, in this study, we investigated the association between TP53 mutations and immunotherapy outcomes of NSCLC patients in a cohort of 350 metastatic or unresectable NSCLC patients who were treated with immunotherapies. This cohort included patients with TP53 non-truncating mutations as well as those with putative truncating mutations because of frameshift, nonsense or splice-site mutations that reduce TP53 protein expression and function [12–14]. We also tested the prognostic prediction efficacy of a survival model that includes TP53 mutation status as a parameter.
Results
NSCLC patients with TP53 truncating mutations in the MSK-IMPACT show lower overall survival than those with wild-type TP53
The flow chart of the study strategy is shown in Figure 1. The clinicopathological characteristics of the 350 metastatic or unresectable NSCLC patients from the Memorial Sloan Kettering Cancer Center Integrated Mutation Profiling of Actionable Cancer Targets (MSK-IMPACT) cohort who received anti PD-1/PD-L1 monotherapy (n=329) or a combination of anti-CTLA-4 and anti-PD-1/PD-L1 immunotherapies (n=21) is shown in Table 1. Kaplan-Meier survival curve analysis showed that the median overall survival (OS) of NSCLC patients with TP53 mutations (n=217) was 10 months compared to 14 months for patients with the wildtype TP53 (n = 133), but, the differences were not statistically significant [hazard ratio (HR) = 1.13, confidence interval (CI) = 0.93-1.37 and P = 0.209; Figure 2A]. Furthermore, the median OS of patients with TP53 truncating mutations (n=67) was significantly lower at 9 months compared to 14 months for patients with wildtype TP53 (n=133) as shown in Figure 2B (HR = 1.36, CI = 1.05-1.76; P = 0.019).
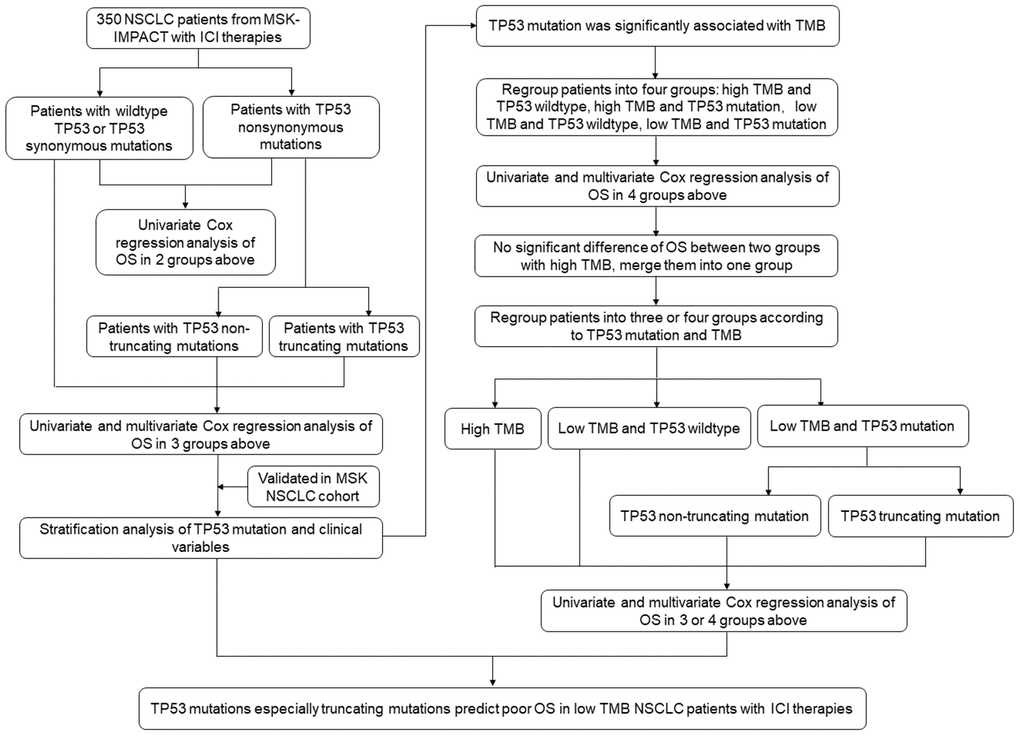
Figure 1. Flowchart of the study strategy.
Table 1. Clinicopathological characteristics of NSCLC patients in the MSK-IMPACT cohort that underwent ICI treatment.
| Characteristics | No. of Cases (%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| All subjects | 350(100) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age in years | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 31-50 | 34 (9.7) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 50-60 | 75 (21.4) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 61-70 | 119 (34.0) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| >71 | 122 (34.9) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sex | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Female | 180 (51.4) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Male | 170 (48.6) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ICI regime | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PD-1/PD-L1 | 329 (94.0) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Combo | 21 (6.0) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mean of TMB (/Mb) | 9.87 (10.03) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathological type | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Adenocarcinoma | 271 (77.4) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Squamous carcinoma | 45 (12.9) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other NSCLC types | 34 (9.7) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MSK-IMPACT: Memorial Sloan Kettering Cancer Center Integrated Mutation Profiling of Actionable Cancer Targets; NSCLC: Non-small cell lung cancer; ICI: immune checkpoint inhibitors; Combo: combination of anti-CTLA-4 and anti-PD-1/PD-L1 therapy; TMB: tumor mutation burden; /Mb: per Mega bases. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
NSCLC patients with TP53 truncating mutations in the validation cohort show lower OS compared to those with wild-type TP53
Kaplan Meier survival curve analysis of the validation cohort of 75 NSCLC patients that underwent immunotherapies (MSK-NSCLC 2018) [15] showed that the median progression free survival (PFS) of NSCLC patients with TP53 truncating mutations (n=10) was 2.6 months compared to 7.56 months for patients with wild-type TP53 (n=32), but, the differences were not statistically significant (HR = 1.30, CI = 0.71-2.37, P = 0.402; Figure 2C).
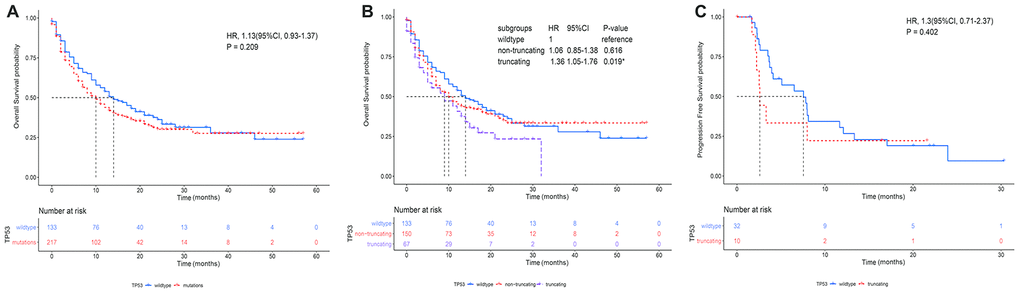
Figure 2. The association between TP53 mutation status and overall survival of NSCLC patients treated with immunotherapies. (A) Kaplan-Meier survival curve analysis shows overall survival of NSCLC patients with TP53 mutations (n=217) and wild-type TP53 (n=133) in the MSK-IMPACT NSCLC cohort. (B) Kaplan-Meier survival curve analysis shows the overall survival of NSCLC patients with wild-type TP53 (n=133), TP53 non-truncating mutations (n=150) and TP53 truncating mutations (n=67) in the MSK-IMPACT NSCLC cohort. (C) Kaplan-Meier survival curve analysis shows the progression free survival (PFS) of patients with TP53 truncating mutations (n=10) and wildtype TP53 (n=32) in the MSK-NSCLC cohort.
TP53 truncating mutations are an independent predictor of immunotherapeutic outcomes in NSCLC patients
Next, we focused on the correlation between TP53 truncating mutations and the survival of NSCLC patients undergoing immunotherapies. Supplementary Table 1 shows the 55 truncating mutation sites in the TP53 gene from these patient samples. Multivariate Cox regression analysis showed that TP53 truncating mutations were an independent predictor of immunotherapeutic outcomes after the data was adjusted by age, sex, ICI regime and TMB (Table 2). Moreover, the stratification analysis results showed positive correlation between TP53 mutation status and TMB (P <0.001; Table 3). The mean TMB was 5.79, 12.14, and 12.92 for patients with wild-type TP53, non-truncating TP53 mutations, and truncating TP53 mutations, respectively. While high TMB favors survival [6, 8], TP53 mutations, especially truncating mutations reduce survival rates. Therefore, analysis of the association between OS and TP53 mutation status in low or high TMB NSCLC patient subgroups showed that patients with high TMB are associated with longer OS irrespective of the TP53 mutation status, however, the OS of patients with low TMB was significantly reduced by the TP53 mutation status (log-rank P < 0.0001; Figure 3A). We did not observe any differences in the OS of high TMB patients with or without TP53 mutation (log-rank P = 0.96; Supplementary Figure 1). Therefore, we merged the two subgroups of high TMB patients with or without TP53 mutations into one group and compared their survival status with the remaining two low TMB subgroups (with and without TP53 mutations). We observed that patients with low TMB and TP53 mutations showed significantly shorter median OS of 7 months compared to a median OS of 13 months for those with low TMB and wild-type TP53 (log-rank test, P < 0.0001; Figure 3B). Further stratification analysis demonstrated that the median OS for patients with truncating TP53 mutations and low TMB was significantly shorter compared to patients with TP53 non-truncating mutations and low TMB (5 months vs. 8 months; log-rank test, P < 0.0001; Figure 3C). Multivariate Cox regression analysis adjusted for parameters such as age, sex, and ICI regimes showed that TP53 mutations were an independent factor that was associated with shorter OS (HR = 1.41, CI = 1.05-1.89 and P = 0.023; Table 4) in patients with low TMB, especially those with truncating TP53 mutations (HR = 1.40, CI = 1.13-1.73 and P = 0.002; Table 4).
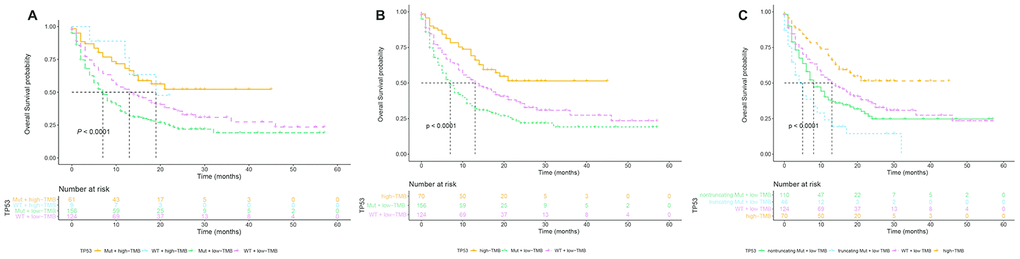
Figure 3. Stratification subgroup analysis of the relationship between overall survival and TP53 mutation status in immunotherapy-treated NSCLC patients of the MSK-IMPACT cohort with low or high TMB. (A) Kaplan-Meier survival curve analysis shows the OS of NSCLC patients in the MSK-IMPACT cohort divided into four subgroups, namely TP53 mutations and high TMB (n=61), TP53 wildtype and high TMB (n=9), TP53 mutations and low TMB (n=156), and TP53 wildtype and low TMB (n=124). (B) Kaplan Meier survival curve analysis shows the OS of NSCLC patients with TP53 mutations and low TMB (median OS=7 months) compared to NSCLC patients with wild-type TP53 and low TMB (median OS=13 months). (C) Kaplan Meier survival curve analysis shows the OS of NSCLC patients with high TMB irrespective of TP53 mutation, TP53 truncating mutations plus low TMB, TP53 non-truncating mutations plus low TMB, and wildtype TP53 plus low TMB. As shown, NSCLC patients with TP53 truncating mutations plus low TMB shows the lowest overall survival compared to other groups.
Table 2. Univariate and multivariate analysis of the association between TP53 mutations and overall survival of NSCLC patients that underwent ICI treatment.
| Variables | Univariate analysis | Multivariate analysis | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HR | 95%CI | P value | HR | 95%CI | P value | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age | 1.01 | 0.74-1.38 | 0.948 | 1.04 | 0.91-1.19 | 0.584 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sex | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Female | 1.00 | reference | 1.00 | reference | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Male | 1.12 | 0.93-1.35 | 0.234 | 1.15 | 0.88-1.51 | 0.309 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ICI regime | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PD-1/PD-L1 | 1.00 | reference | 1.00 | reference | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Combo | 0.53 | 0.33-0.85 | 0.009* | 0.41 | 0.21-0.81 | 0.011* | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TMB (/Mb) | 0.97 | 0.96-0.99 | 0.001* | 0.96 | 0.94-0.98 | 0.000* | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TP53 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Wildtype | 1.00 | reference | 1.00 | reference | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-truncating | 1.06 | 0.85-1.3 | 0.616 | 1.36 | 0.99-1.87 | 0.061 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Truncating | 1.36 | 1.05-1.76 | 0.019* | 1.37 | 1.1-1.7 | 0.005* | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| NSCLC: non-small cell lung cancer; ICI: immune checkpoint inhibitors; HR: hazard ratios; CI: confidence interval; TMB: tumor mutation burden; Combo: combination of anti CTLA-4 and anti PD-1/PD-L1 therapies; /Mb: per Mega bases; * denotes P<0.05. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 3. Stratification analysis of patients with different TP53 gene mutation status in the MSK-IMPACT NSCLC cohort.
| Variables | Patients with different TP53 gene mutational status | P value | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Wildtype | Non-truncating mutation | Truncating mutation | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| All patients | 133 (%) | 150 (%) | 67 (%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age in years | 0.695 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 31-50 | 10 (7.5) | 14 (9.3) | 10 (14.9) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 50-60 | 26 (19.5) | 35 (23.3) | 14 (20.9) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 61-70 | 49 (36.8) | 48 (32.0) | 22 (32.8) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| >71 | 48 (36.1) | 53 (35.3) | 21 (31.3) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sex | 0.178 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Female | 74 (55.6) | 78 (52.0) | 28 (41.8) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Male | 59 (44.4) | 72 (48.0) | 39 (58.2) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ICI regime | 0.060 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PD-1/PD-L1 | 122 (91.7) | 140 (93.3) | 67 (100.0) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Combo | 11 (8.3) | 10 (6.7) | 0 (0.0) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TMB ± SD (/Mb) | 5.76 ± 4.33 | 12.14 ±12.09 | 12.92 ±10.43 | <0.001* | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathological type | 0.053 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Adenocarcinoma | 114 (85.7) | 110 (73.3) | 47 (70.1) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Squamous | 11 (8.3) | 24 (16.0) | 10 (14.9) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| other NSCLC types | 8 (6.0) | 16 (10.7) | 10 (14.9) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MSK-IMPACT: Memorial Sloan Kettering Cancer Center Integrated Mutation Profiling of Actionable Cancer Targets; NSCLC: Non-cell lung cancer; ICI: immune checkpoint inhibitors; Combo: combination of anti-CTLA-4 and anti PD-1/PD-L1 therapies; TMB, tumor mutation burden; /Mb, per Mega bases; SD, standard deviation; * denotes P<0.05. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 4. The association between OS and TP53 mutation status in low or high TMB NSCLC patient subgroups from the MSK-IMPACT cohort that underwent immunotherapy.
| TMB < 13.8/Mb | TMB ≥ 13.8/Mb | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TP53 status | Cases | Death (%) | HR (95%CI) | P value | Cases | Death (%) | HR (95%CI) | P value | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2 subgroups | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| wildtype | 124 | 78 (62.90) | 1.00 | 9 | 4 (44.44) | 1.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| mutation | 156 | 111 (71.15) | 1.41 (1.05-1.89) | 0.023* | 61 | 26 (42.62) | 1.29 (0.44-3.28) | 0.645 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3 subgroups | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| wildtype | 124 | 78 (62.90) | 1.00 | 9 | 4 (44.44) | 1.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| non-truncating mutations | 110 | 74 (67.27) | 1.25 (0.91-1.73) | 0.168 | 40 | 16 (40.00) | 1.49 (0.47-4.71) | 0.500 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| truncating mutations | 46 | 37 (80.43) | 1.40 (1.13-1.73) | 0.002* | 21 | 10 (47.62) | 1.06 (0.57-1.96) | 0.855 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OS: overall survival; MSK-IMPACT: Memorial Sloan Kettering Cancer Center Integrated Mutation Profiling of Actionable Cancer Targets; NSCLC: non-small cell lung cancer; HR: hazard ratios; CI: confidence interval; TMB: tumor mutation burden; /Mb: per Mega bases; * denotes P<0.05. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
TP53 mutations were associated with significantly higher infiltration of specific immune cell subsets
Tumor Immune Estimation Resource (TIMER) database analysis of lung adenocarcinoma samples showed that TP53 mutations were associated with significantly higher infiltration of CD8+ T cells, neutrophils and dendritic cells (Figure 4).
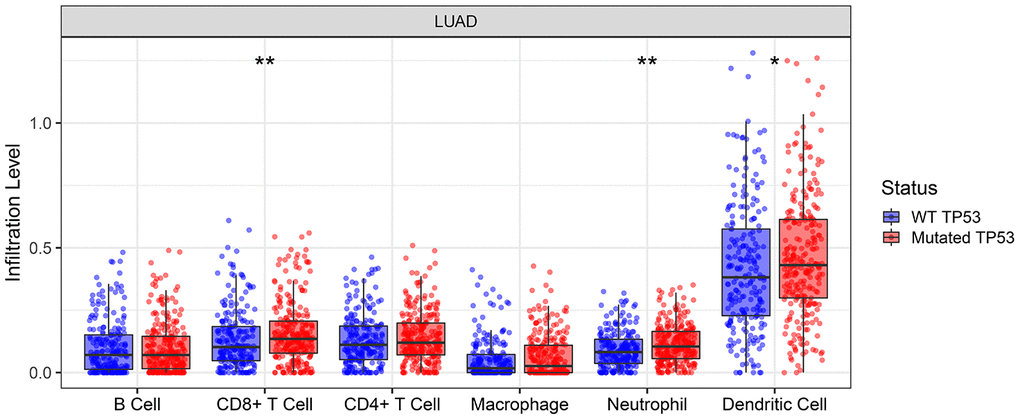
Figure 4. TIMER database analysis shows the abundance of six tumor-infiltrating immune cell types in TP53 wild-type or mutated lung adenocarcinoma tumor samples. * denotes P < 0.05; ** denotes P < 0.01.
Prognostic model with TP53 mutation status shows better survival prediction of NSCLC patients that underwent immunotherapy
Next, we performed receiver operating characteristic (ROC) curve analysis to determine the prognostic prediction efficiency of TP53 mutational status (wild-type, non-truncating mutations or truncating mutations). As shown in Figure 5, the area under curve (AUC) value for the prognostic model with a combination of age, sex, ICI regime, TMB and TP53 mutational status was higher but statistically insignificant compared to the prognostic model that excluded TP53 mutational status at one year (AUC: 64.9% vs. 60.2%; P = 0.052) and two years (AUC: 70.9% vs. 66.1%; P = 0.098) after immunotherapy (Figure 5A–5B). The time-dependent AUC of the prognostic model with TP53 mutational status was higher than the model without TP53 mutational status (Figure 5C).
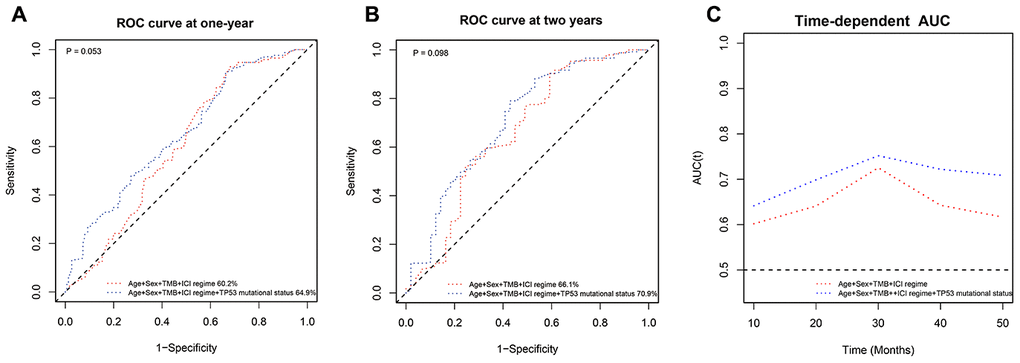
Figure 5. ROC curve analysis of the prognostic prediction models with or without TP53 mutation status. (A, B) The ROC curves show the comparative prognostic prediction efficiency of models with age, sex, ICI regime, and TMB or age, sex, ICI regime, TMB, and TP53 mutation status (wild-type, non-truncating or truncating mutations) as parameters at one year and two years after the 350 NSCLC patients in the MSK-IMPACT cohort received immunotherapies. (C) Time-dependent AUC of the prognostic prediction model with age, sex, ICI regime, TMB and TP53 mutation status was higher than the time-dependent AUC of the model with age, sex, ICI regime, and TMB.
Discussion
Previous reports show that TP53 is an important tumor suppressor gene that determines cancer initiation, growth and progression, and is mutated in nearly 50% of all NSCLC patients [16]. Our analysis also shows that the TP53 gene is mutated in over 50% NSCLC patients, with truncating mutations in the TP53 gene accounting for 19.1% and 13.5% among the patients in the MSK-IMPACT- and MSK-NSCLC cohorts, respectively. A couple of studies have demonstrated that TP53 mutations are positively associated with immunotherapeutic outcomes in NSCLC patients [17, 18], but, these results have not been validated. Our analysis shows that TP53 mutations, particularly the TP53 truncating mutations, are negatively associated with immunotherapeutic outcomes in NSCLC patients, especially in the low TMB subgroup. Some of non-synonymous mutations such as missense mutations result in a single amino acid change that may not dramatically change TP53 protein expression and function [19]. However, truncating mutations may cause nonsense-mediated decay of the premature mRNAs resulting in significantly lower levels of the TP53 protein as well as loss of function [12, 20, 21]. Our study shows that the OS rates are not significantly different for NSCLC patients with wild-type TP53 and those with non-synonymous TP53 mutations. However, the overall survival is significantly lower in NSCLC patients with truncating TP53 mutations compared to those with wild-type TP53.
Previous studies suggest that tumor-infiltrating immune cells play an important role in the survival outcomes of patients treated with immunotherapy for various types of cancers [22, 23]. Our research shows that the infiltration status of three tumor-infiltrating immune cell types, CD8+ T cells, neutrophils, and dendritic cells, is significantly associated with the mutational status of the TP53 gene and may impact survival outcomes of immunotherapy. Moreover, a previous study showed that the tumor-killing efficiency of cytotoxic T-lymphocytes is reduced when the cancer cells express mutant TP53 protein compared to those expressing the wildtype TP53 [24]. This suggests adverse immunotherapy outcomes for patients with mutant TP53 tumors. The results of our study are consistent with these findings, but, rigorous large-scale studies are necessary to confirm.
The survival outcomes are significantly higher for NSCLC patients with higher TMB after receiving immune checkpoint inhibitors therapy [6, 8]. However, stratification analysis has not been performed to identify prognostic biomarkers for immunotherapy outcomes in NSCLC patients with low TMB. Moreover, the cutoff value for TMB was defined as 10 or 13.8 per mega-bases in previous studies [6, 8]. Therefore, majority of NSCLC patients were classified in the low TMB group. In our analysis, 80% of the NSCLC patients were classified in the low TMB group. Effective prognostic prediction biomarkers are required to identify NSCLC patients that are suitable to undergo immunotherapy. Our study demonstrates that TP53 truncating mutations are a negative independent predictive biomarker for NSCLC patients. These findings demonstrate the potential of TP53 mutations as a prognostic biomarker for NSCLC patients. However, our findings need to be validated by larger clinical trials.
Materials and Methods
NSCLC patient datasets
We downloaded two independent datasets from the publicly available cBioPortal database, namely, the MSK-IMPACT dataset [25], which includes 350 NSCLC patients that received immunotherapy, and the MSK-NSCLC dataset (MSK, Cancer Cell 2018) [15], which includes 75 NSCLC patients that had received immunotherapy.
Survival analysis of MSK-IMPACT NSCLC dataset
We divided the 350 NSCLC patients from the MSK-IMPACT dataset into 2 groups based on the nonsynonymous mutation status of the TP53 gene, namely, TP53 wildtype and TP53 mutant groups. The NSCLC patients with synonymous mutations were included in the wild-type group because the mutations did not change the protein length, structure, and expression significantly. We then plotted the Kaplan-Meier survival curves and performed univariate Cox proportional hazards regression analysis of the two groups to evaluate the association between TP53 mutation status and OS of NSCLC patients. Since truncating mutations such as frameshift insertions or deletions, nonsense, and splice-site mutations can alter the function of the proteins significantly, we then divided the NSCLC patients into 3 groups, namely, those with wild-type TP53 (n=133), truncating TP53 mutations (n=67), and TP53 non-truncating mutations (n=150) and performed univariate and multivariate Cox proportional hazards regression analysis with the data adjusted for age, sex, ICI regime, and TMB. TMB was defined as the total number of nonsynonymous somatic mutations per mega-base (Mb) of the genome. We calculated the TMB in the MSK-IMPACT cohort by normalizing the total number of nonsynonymous somatic mutations to the total number of mega-bases sequenced. We used the sequencing data from a 469 genes panel [6]. We performed Kaplan-Meier survival curve analysis for the TP53 wild-type and TP53 truncating mutation groups of NSCLC patients. We also analyzed the truncating mutation sites in the TP53 gene to identify any hotspot mutations using R language.
Survival analysis of the MSK-NSCLC validation dataset
Next, we analyzed another cohort of NSCLC patients that received immunotherapy, namely, the MSK-NSCLC dataset containing 75 metastatic or unresectable NSCLC patients, and compared the PFS data to validate the association between TP53 truncating mutations and survival outcomes of NSCLC patients undergoing immunotherapy. We compared Kaplan-Meier survival curves for patients with wild-type TP53 (n=32) and those with truncated TP53 mutations (n=10) using univariate Cox proportional hazards regression analysis. We also compared the PFS between the two groups of NSCLC patients.
Stratification analysis
Next, we performed correlation analysis of the MSK-IMPACT dataset to determine the relationship between TP53 mutations and clinicopathological variables. We observed significant correlation between TP53 mutations and TMB status of NSCLC patients. Hence, we performed stratification analysis of OS and TP53 status in low or high TMB subgroups. We determined the cutoff value for classifying patients into low or high TMB groups as 13.8/Mb, based on previously published data using the same dataset [6]. We then performed Kaplan-Meier survival curve analysis including the log-rank test and multivariate Cox regression analysis adjusted by age, sex, and ICI regimes for these subgroups.
Tumor-infiltrating immune cell analysis
We used the TIMER database (https://cistrome.shinyapps.io/timer/) to determine the abundance of six tumor-infiltrating immune subsets, namely, B cells, CD4+ T cells, CD8+ T cells, neutrophils, macrophages, and dendritic cells in NSCLC tumor samples from patients with the wild-type TP53 and those with TP53 mutations. The results were derived from the sequencing data obtained from 10897 tumor samples belonging to 32 cancer types at The Cancer Genome Atlas (TCGA) database [26]. Specifically, we analyzed six subsets of tumor infiltrating immune cells in 544 TCGA lung adenocarcinoma samples using TIMER online tools.
ROC curve analysis of the prognostic prediction models with or without TP53 mutation status
We investigated the prognostic prediction efficiency of the TP53 mutation status by constructing two prognostic models: (1) age, sex, ICI regime, and TMB; (2) age, sex, ICI regime, TMB and TP53 mutation status. Then, we generated ROC curves based on the one-year and 2-year survival data and also evaluated the time-dependent dynamic AUC of the two models.
Statistical analysis
All the statistical analysis was performed using R language (version 3.5.1). The Cox proportional hazards regression analysis was used to perform univariate and multivariate analysis of the clinical variables. Stratification analysis was used to estimate the true association between clinical variables and survival outcomes by analyzing the variables in subgroups. The groups were compared using two-sided t-test and P< 0.05 was considered statistically significant.
Author Contributions
Linqin Zhao, Xiaofei Qu and Xiaowei Zhang conceived and designed the study; Linqin Zhao, Zhenhua Wu and Yuehua Li wrote the manuscript; Xiaofei Qu analyzed the statistical data; Weijian Guo made revisions; Xiaowei Zhang and Weijian Guo approved the final version. All authors consented to the final manuscript.
Conflicts of Interest
The authors state that there are no conflicts of interest.
Funding
This work was financially supported by the Natural Science Foundation of China (Grant No. 81871948).
References
- 1. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018; 68:394–424. https://doi.org/10.3322/caac.21492 [PubMed]
- 2. Travis WD, Brambilla E, Nicholson AG, Yatabe Y, Austin JH, Beasley MB, Chirieac LR, Dacic S, Duhig E, Flieder DB, Geisinger K, Hirsch FR, Ishikawa Y, et al, and WHO Panel. The 2015 world health organization classification of lung tumors: impact of genetic, clinical and radiologic advances since the 2004 classification. J Thorac Oncol. 2015; 10:1243–60. https://doi.org/10.1097/JTO.0000000000000630 [PubMed]
- 3. Stella GM, Luisetti M, Pozzi E, Comoglio PM. Oncogenes in non-small-cell lung cancer: emerging connections and novel therapeutic dynamics. Lancet Respir Med. 2013; 1:251–61. https://doi.org/10.1016/S2213-2600(13)70009-2 [PubMed]
- 4. Westover D, Zugazagoitia J, Cho BC, Lovly CM, Paz-Ares L. Mechanisms of acquired resistance to first- and second-generation EGFR tyrosine kinase inhibitors. Ann Oncol. 2018; 29:i10–19. https://doi.org/10.1093/annonc/mdx703 [PubMed]
- 5. Hirsch FR, Scagliotti GV, Mulshine JL, Kwon R, Curran WJ
Jr , Wu YL, Paz-Ares L. Lung cancer: current therapies and new targeted treatments. Lancet. 2017; 389:299–311. https://doi.org/10.1016/S0140-6736(16)30958-8 [PubMed] - 6. Samstein RM, Lee CH, Shoushtari AN, Hellmann MD, Shen R, Janjigian YY, Barron DA, Zehir A, Jordan EJ, Omuro A, Kaley TJ, Kendall SM, Motzer RJ, et al. Tumor mutational load predicts survival after immunotherapy across multiple cancer types. Nat Genet. 2019; 51:202–06. https://doi.org/10.1038/s41588-018-0312-8 [PubMed]
- 7. Borghaei H, Paz-Ares L, Horn L, Spigel DR, Steins M, Ready NE, Chow LQ, Vokes EE, Felip E, Holgado E, Barlesi F, Kohlhäufl M, Arrieta O, et al. Nivolumab versus docetaxel in advanced nonsquamous non-small-cell lung cancer. N Engl J Med. 2015; 373:1627–39. https://doi.org/10.1056/NEJMoa1507643 [PubMed]
- 8. Ready N, Hellmann MD, Awad MM, Otterson GA, Gutierrez M, Gainor JF, Borghaei H, Jolivet J, Horn L, Mates M, Brahmer J, Rabinowitz I, Reddy PS, et al. First-line nivolumab plus ipilimumab in advanced non-small-cell lung cancer (CheckMate 568): outcomes by programmed death ligand 1 and tumor mutational burden as biomarkers. J Clin Oncol. 2019; 37:992–1000. https://doi.org/10.1200/JCO.18.01042 [PubMed]
- 9. Wang F, Zhao Q, Wang YN, Jin Y, He MM, Liu ZX, Xu RH. Evaluation of POLE and POLD1 mutations as biomarkers for immunotherapy outcomes across multiple cancer types. JAMA Oncol. 2019; 5:1504–06. https://doi.org/10.1001/jamaoncol.2019.2963 [PubMed]
- 10. Lyu H, Li M, Jiang Z, Liu Z, Wang X. Correlate the TP53 mutation and the HRAS mutation with immune signatures in head and neck squamous cell cancer. Comput Struct Biotechnol J. 2019; 17:1020–30. https://doi.org/10.1016/j.csbj.2019.07.009 [PubMed]
- 11. Dong ZY, Zhong WZ, Zhang XC, Su J, Xie Z, Liu SY, Tu HY, Chen HJ, Sun YL, Zhou Q, Yang JJ, Yang XN, Lin JX, et al. Potential predictive value of TP53 and KRAS mutation status for response to PD-1 blockade immunotherapy in lung adenocarcinoma. Clin Cancer Res. 2017; 23:3012–24. https://doi.org/10.1158/1078-0432.CCR-16-2554 [PubMed]
- 12. Hug N, Longman D, Cáceres JF. Mechanism and regulation of the nonsense-mediated decay pathway. Nucleic Acids Res. 2016; 44:1483–95. https://doi.org/10.1093/nar/gkw010 [PubMed]
- 13. Miao D, Margolis CA, Gao W, Voss MH, Li W, Martini DJ, Norton C, Bossé D, Wankowicz SM, Cullen D, Horak C, Wind-Rotolo M, Tracy A, et al. Genomic correlates of response to immune checkpoint therapies in clear cell renal cell carcinoma. Science. 2018; 359:801–06. https://doi.org/10.1126/science.aan5951 [PubMed]
- 14. Williams DS, Bird MJ, Jorissen RN, Yu YL, Walker F, Zhang HH, Nice EC, Burgess AW. Nonsense mediated decay resistant mutations are a source of expressed mutant proteins in colon cancer cell lines with microsatellite instability. PLoS One. 2010; 5:e16012. https://doi.org/10.1371/journal.pone.0016012 [PubMed]
- 15. Hellmann MD, Nathanson T, Rizvi H, Creelan BC, Sanchez-Vega F, Ahuja A, Ni A, Novik JB, Mangarin LM, Abu-Akeel M, Liu C, Sauter JL, Rekhtman N, et al. Genomic features of response to combination immunotherapy in patients with advanced non-small-cell lung cancer. Cancer Cell. 2018; 33:843–52.e4. https://doi.org/10.1016/j.ccell.2018.03.018 [PubMed]
- 16. Scoccianti C, Vesin A, Martel G, Olivier M, Brambilla E, Timsit JF, Tavecchio L, Brambilla C, Field JK, Hainaut P, and European Early Lung Cancer Consortium. Prognostic value of TP53, KRAS and EGFR mutations in nonsmall cell lung cancer: the EUELC cohort. Eur Respir J. 2012; 40:177–84. https://doi.org/10.1183/09031936.00097311 [PubMed]
- 17. Biton J, Mansuet-Lupo A, Pécuchet N, Alifano M, Ouakrim H, Arrondeau J, Boudou-Rouquette P, Goldwasser F, Leroy K, Goc J, Wislez M, Germain C, Laurent-Puig P, et al. TP53, STK11, and EGFR mutations predict tumor immune profile and the response to anti-PD-1 in lung adenocarcinoma. Clin Cancer Res. 2018; 24:5710–23. https://doi.org/10.1158/1078-0432.CCR-18-0163 [PubMed]
- 18. Assoun S, Theou-Anton N, Nguenang M, Cazes A, Danel C, Abbar B, Pluvy J, Gounant V, Khalil A, Namour C, Brosseau S, Zalcman G. Association of TP53 mutations with response and longer survival under immune checkpoint inhibitors in advanced non-small-cell lung cancer. Lung Cancer. 2019; 132:65–71. https://doi.org/10.1016/j.lungcan.2019.04.005 [PubMed]
- 19. Hainaut P, Pfeifer GP. Somatic TP53 mutations in the era of genome sequencing. Cold Spring Harb Perspect Med. 2016; 6:a026179. https://doi.org/10.1101/cshperspect.a026179 [PubMed]
- 20. Bykov VJ, Eriksson SE, Bianchi J, Wiman KG. Targeting mutant p53 for efficient cancer therapy. Nat Rev Cancer. 2018; 18:89–102. https://doi.org/10.1038/nrc.2017.109 [PubMed]
- 21. Lykke-Andersen S, Jensen TH. Nonsense-mediated mRNA decay: an intricate machinery that shapes transcriptomes. Nat Rev Mol Cell Biol. 2015; 16:665–77. https://doi.org/10.1038/nrm4063 [PubMed]
- 22. Gentles AJ, Newman AM, Liu CL, Bratman SV, Feng W, Kim D, Nair VS, Xu Y, Khuong A, Hoang CD, Diehn M, West RB, Plevritis SK, Alizadeh AA. The prognostic landscape of genes and infiltrating immune cells across human cancers. Nat Med. 2015; 21:938–45. https://doi.org/10.1038/nm.3909 [PubMed]
- 23. Li B, Li T, Pignon JC, Wang B, Wang J, Shukla SA, Dou R, Chen Q, Hodi FS, Choueiri TK, Wu C, Hacohen N, Signoretti S, et al. Landscape of tumor-infiltrating T cell repertoire of human cancers. Nat Genet. 2016; 48:725–32. https://doi.org/10.1038/ng.3581 [PubMed]
- 24. Braun MW, Iwakuma T. Regulation of cytotoxic t-cell responses by p53 in cancer. Transl Cancer Res. 2016; 5:692–97. https://doi.org/10.21037/tcr.2016.11.76 [PubMed]
- 25. Cheng DT, Mitchell TN, Zehir A, Shah RH, Benayed R, Syed A, Chandramohan R, Liu ZY, Won HH, Scott SN, Brannon AR, O’Reilly C, Sadowska J, et al. Memorial sloan kettering-integrated mutation profiling of actionable cancer targets (MSK-IMPACT): a hybridization capture-based next-generation sequencing clinical assay for solid tumor molecular oncology. J Mol Diagn. 2015; 17:251–64. https://doi.org/10.1016/j.jmoldx.2014.12.006 [PubMed]
- 26. Li T, Fan J, Wang B, Traugh N, Chen Q, Liu JS, Li B, Liu XS. TIMER: a web server for comprehensive analysis of tumor-infiltrating immune cells. Cancer Res. 2017; 77:e108–10. https://doi.org/10.1158/0008-5472.CAN-17-0307 [PubMed]



